Bottom-up Synthesis of Porous NiMo Alloy for Hydrogen Evolution Reaction
Abstract
1. Introduction
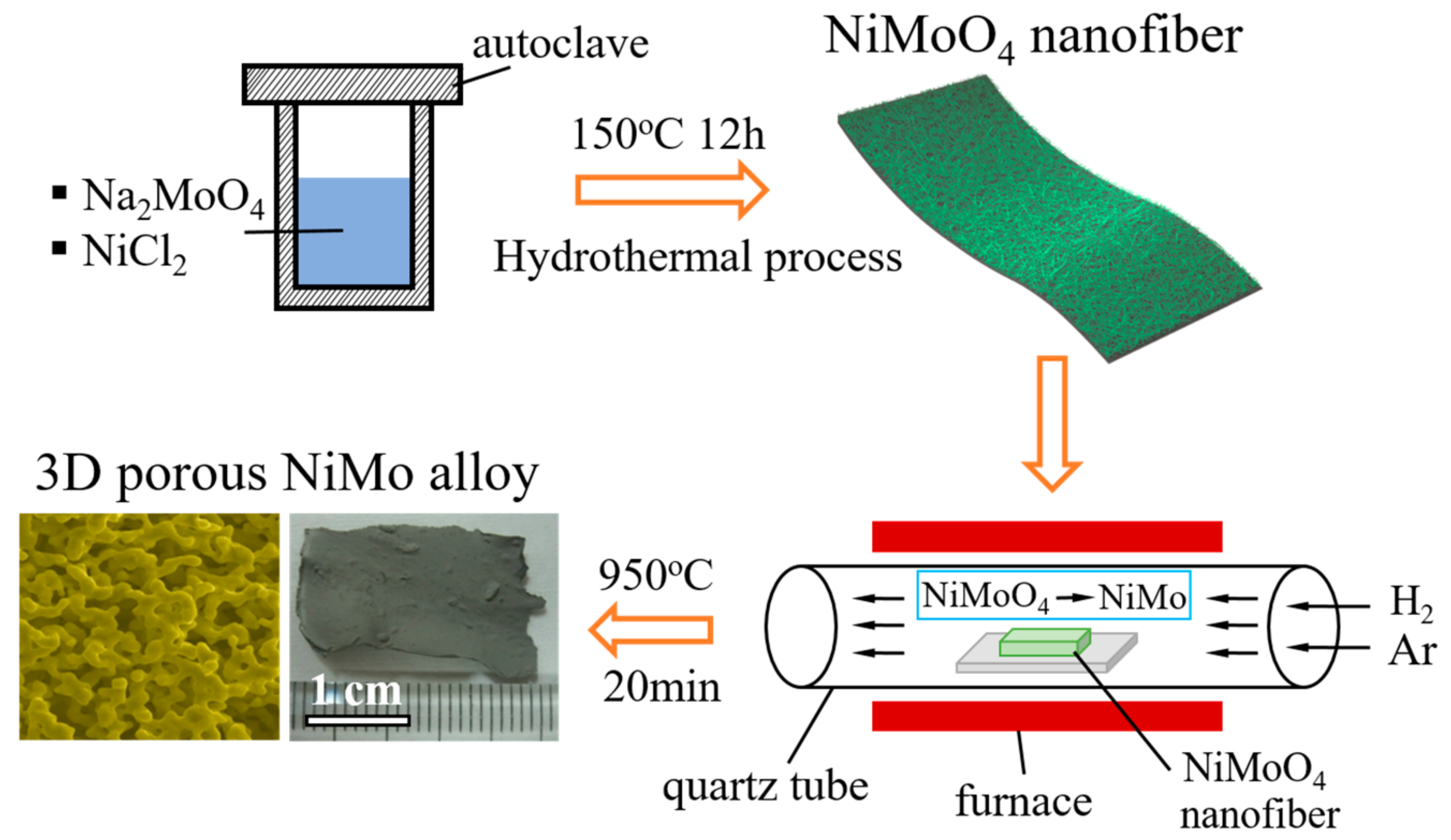
2. Materials and Methods
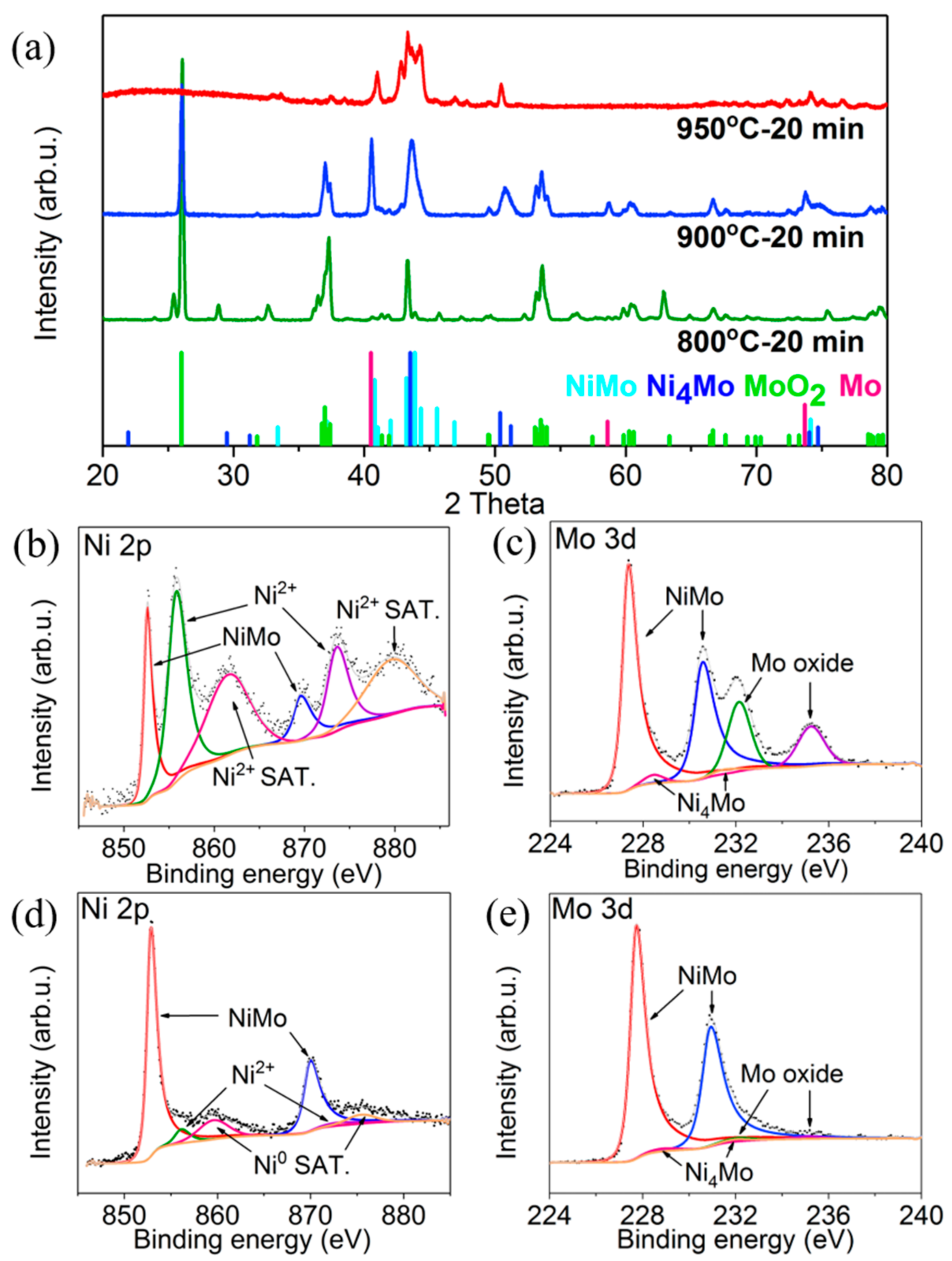
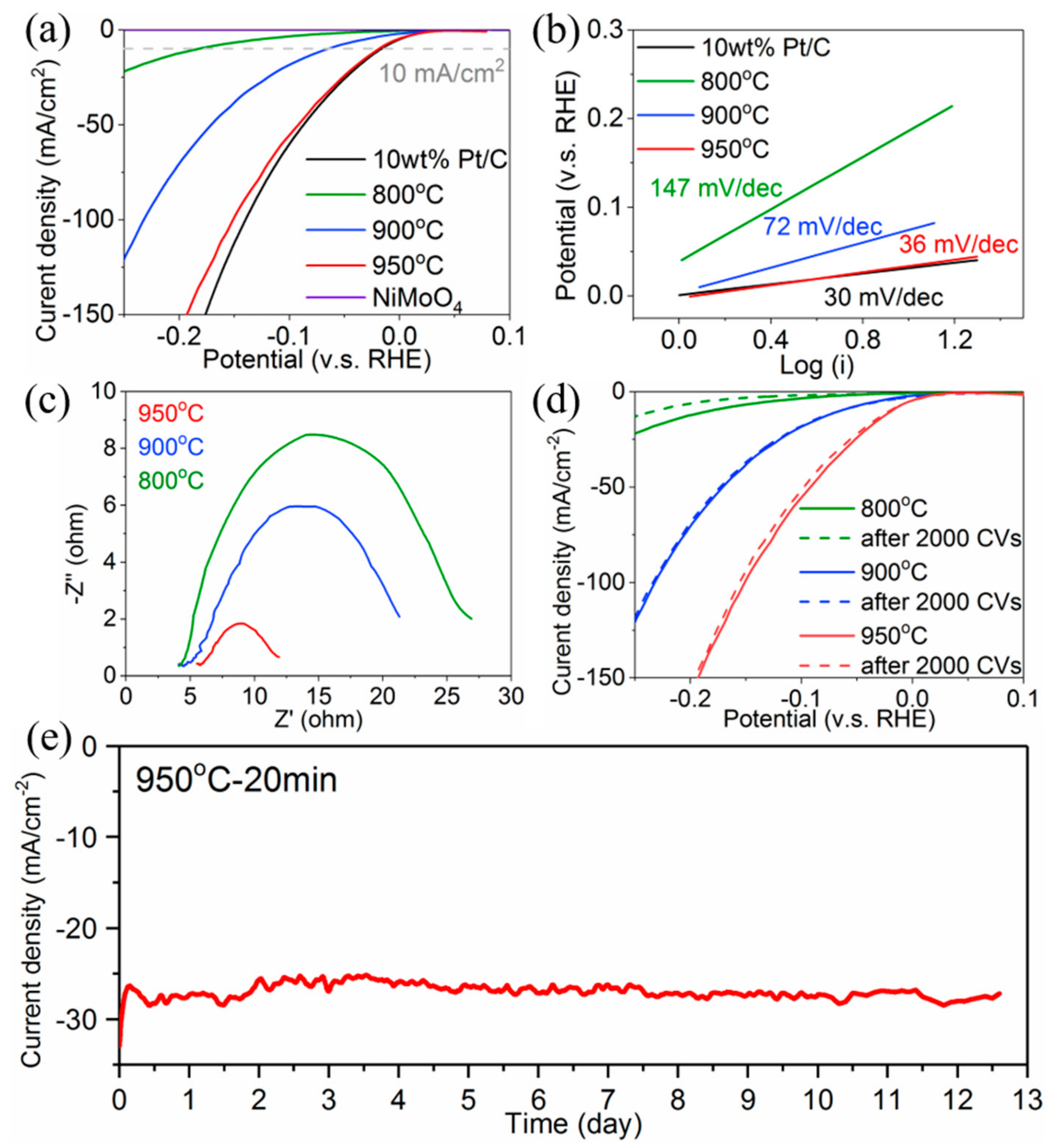
3. Results and Discussion
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ding, Y.; Chen, M.W. Nanoporous metals for catalytic and optical applications. MRS Bull. 2009, 34, 569–576. [Google Scholar] [CrossRef]
- Weissmüller, J.; Newman, R.C.; Jin, H.J.; Hodge, A.M.; Kysar, J.W. Nanoporous metals by alloy corrosion: Formation and mechanical properties. MRS Bull. 2009, 34, 577–586. [Google Scholar] [CrossRef]
- Qiu, H.J.; Ito, Y.; Chen, M.W. Hierarchical nanoporous nickel alloy as three-dimensional electrodes for high-efficiency energy storage. Scr. Mater. 2014, 89, 69–72. [Google Scholar] [CrossRef]
- Qiu, H.J.; Kang, J.L.; Liu, P.; Hirata, A.; Fujita, T.; Chen, M.W. Fabrication of large-scale nanoporous nickel with a tunable pore size for energy storage. J. Power Sources 2014, 247, 896–905. [Google Scholar] [CrossRef]
- Guo, X.W.; Han, J.H.; Liu, P.; Ito, Y.; Hirata, A.; Chen, M.W. Graphene@Nanoporous nickel cathode for Li-O2 batteries. ChemNanoMat 2016, 2, 176–181. [Google Scholar] [CrossRef]
- Guo, X.W.; Liu, P.; Han, J.H.; Ito, Y.; Hirata, A.; Fujita, T.; Chen, M.W. 3D nanoporous nitrogen-doped graphene with encapsulated RuO2 nanoparticles for Li-O2 batteries. Adv. Mater. 2015, 27, 6137–6143. [Google Scholar] [CrossRef] [PubMed]
- Linic, S.; Christopher, P.; Ingram, D.B. Plasmonic-metal nanostructures for efficient conversion of solar to chemical energy. Nat. Mater. 2011, 10, 911–921. [Google Scholar] [CrossRef] [PubMed]
- Artero, V.; Kerlidou, M.C.; Fontecave, M. Splitting water with cobalt. Angew. Chem. Int. Ed. Engl. 2011, 50, 7238–7266. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Izumi, M.; Hojo, D.; Wakisaka, M.; Aida, T.; Adschiri, T. One-step nanoporous structure formation using NiO nanoparticles: Pore size control and pore size dependence of hydrogen evolution reaction. Chem. Lett. 2017, 46, 267–270. [Google Scholar] [CrossRef]
- Hansen, T.W.; DeLaRiva, A.T.; Challa, S.R.; Datye, A.K. Sintering of catalytic nanoparticles: Particle migration or ostwald ripening? Acc. Chem. Res. 2013, 46, 1720–1730. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, T.; Liu, P.; Liao, Z.Q.; Liu, S.H.; Zhuang, X.D.; Chen, M.W.; Zschech, E.; Feng, X.L. Efficient hydrogen production on MoNi4 electrocatalysts with fast water dissociation kinetics. Nat. Commun. 2017, 8, 15437. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Zhang, Y.; Zhang, X.; Tang, T.; Luo, H.; Niu, S.; Dai, Z.H.; Wan, L.J.; Hu, J.S. Self-templated fabrication of MoNi4/MoO3−X nanorod arrays with dual active components for highly efficient hydrogen evolution. Adv. Mater. 2017, 29, 1703311. [Google Scholar] [CrossRef] [PubMed]
- McCrory, C.C.; Jung, S.; Ferrer, I.M.; Chatman, S.M.; Peters, J.C.; Jaramillo, T.F. Benchmarking hydrogen evolving reaction and oxygen evolving reaction electrocatalysts for solar water splitting devices. J. Am. Chem. Soc. 2015, 137, 4347–4357. [Google Scholar] [CrossRef] [PubMed]
- McCrory, C.C.; Jung, S.; Peters, J.C.; Jaramillo, T.F. Benchmarking heterogeneous electrocatalysts for the oxygen evolution reaction. J. Am. Chem. Soc. 2013, 135, 16977–16987. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.S.; Yue, X.; Shu, C.; Huang, S.L.; Shen, P.K. Three-dimensional porous MoNi4 networks constructed by nanosheets as bifunctional electrocatalysts for overall water splitting. J. Mater. Chem. A 2017, 5, 2508–2513. [Google Scholar] [CrossRef]
- McKone, J.R.; Sadtler, B.F.; Werlang, C.L.; Lewis, N.S.; Gray, H.B. Ni–Mo nanopowders for efficient electrochemical hydrogen evolution. ACS Catal. 2013, 3, 166–169. [Google Scholar] [CrossRef]
- Fang, M.; Guo, W.; Dong, G.F.; Xia, Z.M.; Yip, S.P.; Qin, Y.B.; Qu, Y.Q.; Ho, J.C. Hierarchical NiMo-based 3D electrocatalysts for highly-efficient hydrogen evolution in alkaline conditions. Nano Energy 2016, 27, 247–254. [Google Scholar] [CrossRef]
- Jothi, P.R.; Kannan, S.; Velayutham, G. Enhanced methanol electro-oxidation over in-situ carbon and graphene supported one dimensional NiMoO4 nanorods. J. Power Sources 2015, 277, 350–359. [Google Scholar] [CrossRef]
- Kuang, P.Y.; Tong, T.; Fan, K.; Yu, J.G. In situ fabrication of Ni–Mo bimetal sulfide hybrid as an efficient electrocatalyst for hydrogen evolution over a wide pH range. ACS Catal. 2017, 7, 6179–6187. [Google Scholar] [CrossRef]
- Wertheim, G.K.; Wernick, J.H.; Crecelius, G. Surface effects on valence in rare-earth intermetallic compounds. Phys. Rev. B 1978, 18, 875. [Google Scholar] [CrossRef]
- Roustila, A.; Severac, C.; Chêne, J.; Percheron-Guégan, A. Hydrogen effects on the electronic and microstructural properties of Ce, Ni, and CeNi2 intermetallic compound. Surf. Sci. 1994, 311, 33–44. [Google Scholar] [CrossRef]
- Lebugle, A.; Axelsson, U.; Nyholm, R.; Mårtensson, N. Experimental L and M core level binding energies for the metals 22Ti to 30Zn. Phys. Scr. 1981, 23, 825–827. [Google Scholar] [CrossRef]
- Brainard, W.A.; Wheeler, D.R. An XPS study of the adherence of refractory carbide silicide and boride rf-sputtered wear-resistant coatings. J. Vac. Sci. Technol. 1978, 15, 1800–1805. [Google Scholar] [CrossRef]
- Bianchi, C.L.; Cattania, M.G.; Villa, P. XPS characterization of Ni and Mo oxides before and after “in situ” treatments. Appl. Surf. Sci. 1993, 70, 211–216. [Google Scholar] [CrossRef]
- Takano, I.; Isobe, S.; Sasaki, T.A.; Baba, Y. Nitrogenation of various transition metals by N+2-ion implantation. Appl. Surf. Sci. 1989, 37, 25–32. [Google Scholar] [CrossRef]
- Conway, B.E.; Tilak, B.V. Interfacial processes involving electrocatalytic evolution and oxidation of H2, and the role of chemisorbed H. Electrochim. Acta 2002, 47, 3571–3594. [Google Scholar] [CrossRef]
- Zheng, Y.; Jiao, Y.; Zhu, Y.H.; Li, L.H.; Han, Y.; Chen, Y.; Du, A.J.; Jaroniec, M.; Qiao, S.Z. Hydrogen evolution by a metal-free electrocatalyst. Nat. Commun. 2014, 5, 3783. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.X.; Ahn, H.S.; Bard, A.J. A study of the mechanism of the hydrogen evolution reaction on nickel by surface interrogation scanning electrochemical microscopy. J. Am. Chem. Soc. 2017, 139, 4854–4858. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, K.; Jeong, S.; Wakisaka, M.; Fujita, J.-i.; Ito, Y. Bottom-up Synthesis of Porous NiMo Alloy for Hydrogen Evolution Reaction. Metals 2018, 8, 83. https://doi.org/10.3390/met8020083
Hu K, Jeong S, Wakisaka M, Fujita J-i, Ito Y. Bottom-up Synthesis of Porous NiMo Alloy for Hydrogen Evolution Reaction. Metals. 2018; 8(2):83. https://doi.org/10.3390/met8020083
Chicago/Turabian StyleHu, Kailong, Samuel Jeong, Mitsuru Wakisaka, Jun-ichi Fujita, and Yoshikazu Ito. 2018. "Bottom-up Synthesis of Porous NiMo Alloy for Hydrogen Evolution Reaction" Metals 8, no. 2: 83. https://doi.org/10.3390/met8020083
APA StyleHu, K., Jeong, S., Wakisaka, M., Fujita, J.-i., & Ito, Y. (2018). Bottom-up Synthesis of Porous NiMo Alloy for Hydrogen Evolution Reaction. Metals, 8(2), 83. https://doi.org/10.3390/met8020083




