1. Introduction
Titanium and its alloys are widely used as orthopedic implants due to their excellent corrosion resistance, biocompatibility, outstanding fatigue resistance, and lower elastic moduli compared to other metallic biomaterials such as stainless steel and Co-Cr-based alloys. For a long time, the most used alloy for orthopedic applications was the α + β alloy, Ti-6Al-4V. However, studies have shown that the release of small amounts of V and Al into the human body can induce cytotoxic effects and neurological disorders, leading to the development of new β titanium alloys over the past decade [
1]. Additionally, beta titanium alloys, with their lower Young’s moduli, offer significant advantages in biomedical applications.
Corrosion resistance is a critical property for biomaterials, as the fluids within the human body can create an aggressive environment for metals. Titanium exhibits excellent corrosion resistance [
2]. Implants are expected to resist uniform corrosion over the long term, with a corrosion rate of less than 1 μm/year [
3]. The passive film on Ti alloys is predominantly composed of TiO
2. Metastable β-Ti alloys, containing elements such as Nb, Ta, and Zr, form a passive film that incorporates Nb
2O
5, ZrO
2, and Ta
2O
5. When combined with TiO
2, this film offers improved stability and potentially provides superior protection compared to the Ti-6Al-4V alloy [
4,
5,
6,
7]. Various studies have demonstrated the good corrosion resistance of Ti-Nb-Zr [
8,
9,
10] and Ti-Nb-Mo-Zr [
11,
12] alloys.
The electrochemical behavior of metastable β-Ti alloys can also be influenced by the microstructures obtained after thermomechanical treatments: single-phased β, β + α, β + ω, and β+α”. Several authors have reported that single-phase β microstructure provides the best corrosion resistance for these alloys [
13,
14]. The literature indicates the formation of stress-induced α” martensite during cold rolling of metastable β-Ti alloys [
15,
16]. According to Lin J. et al., β phase transforms into an α” phase without diffusion and, for this reason, the corrosion resistance of the alloy tends to be the same as that in the solid solution due to the close thermodynamic properties of α” and β phase [
17]. The aging of metastable β-Ti alloys leads to the formation of ω or α precipitates inside the β matrix, depending on aging temperature and duration [
18]. A study by Li Q. et al. showed that the corrosion resistance of the Ti-24 at% Nb-2 at% Zr alloy decreases upon precipitation of the ω phase, due to the formation of an unstable passive film [
19]. On the other hand, Monteiro et al. indicated that α phase precipitates significantly decreased the corrosion resistance of a Ti-30Nb-7Zr alloy [
20]. For Su B. et al., the fact that β phase is more resistant to corrosion than α phase is related to the higher Nb, Zr, and Mo content of β phase [
21].
Chui et al. [
11] reported results for a Ti-Zr-Nb-Mo alloy, achieving a yield strength of 834 MPa and a hardness of 486 HV. The Young’s modulus ranged from 96 GPa to 105 GPa, which is lower than that of the Ti-6Al-4V alloy. The anodic polarization and EIS revealed an increase in the electrochemical corrosion resistance in simulated body fluid at 37 °C. The best performance in terms of corrosion resistance was expressed by corrosion potential and corrosion current density equal to 0.344 ± 0.005 V and 71.9 ± 5.3 nA/cm
2, respectively. The performance was attributed to the formation of a more protective passive layer due to grain refinement.
This study aims to investigate the electrochemical corrosion resistance of titanium alloy Ti-23.6Nb-5.1Mo-6.7Zr after various thermomechanical treatments: cold-rolling, annealing, or aging at various temperatures following cold rolling. The microstructures obtained for these different processing routes have already been previously studied and reported by our research team [
22,
23]. Electrochemical behaviors were characterized using a combination of anodic polarization, open circuit potential (OCP), and electrochemical impedance spectroscopy (EIS) tests in a synthetic solution (Ringer’s solution) to simulate body fluid. This integrated approach provides a more accurate and consistent assessment of corrosion resistance compared to relying solely on short immersion time anodic polarization curves. The objective of the present work is to demonstrate that all processing conditions applied to the Ti-23.6Nb-5.1Mo-6.7Zr alloy result in lower elastic modulus, a high and comparable hardness-to-Young’s modulus ratio, and excellent passive film stability, ensuring its suitability for long-term use in the human body. Ti-6Al-4V was used as reference material.
2. Materials and Methods
A 6.5 kg cylindrical ingot, with a diameter of 120 mm and a length of 125 mm, was cast in a vacuum arc remelting furnace (VAR) using a titanium bar (grade 2—ASTM B 348) to which high-purity Zr, Mo, and Nb were added. To ensure homogeneity of the resulting alloy, the ingot was remelted three times. The chemical composition of the alloy is presented in
Table 1. The ingot was machined into a rectangular shape and subjected to homogenization treatment at 1000 °C for 24 h in an argon atmosphere (HM), followed by cold rolling until a 90% reduction in thickness was achieved (CR90). After cold rolling, the material was either vacuum-annealed at 950 °C for 1 h followed by water quenching (CR90-ANN) or aged for 4 h at temperatures of 300 °C (CR90-AG300), 400 °C (CR90-AG400), and 500 °C (CR90-AG500), respectively, with all aged samples also water-quenched. A detailed description of the procedure can be found elsewhere [
22].
Young’s moduli (YMs) were obtained using the impulse excitation technique, described in ASTM E 1876 [
24]. Ten measurements were taken for each sample. Vickers microhardness values were measured with a 200 g load and a dwell time of 15 s. The mechanical properties of a Ti-6Al-4V alloy (ASTM F136/ISO 5832-4) were also assessed under the same conditions for comparison [
25,
26].
Electrochemical corrosion resistance was assessed by open circuit potentials measurements, anodic polarization, and electrochemical impedance spectroscopy in the presence of Ringer’s solution (NaCl 8.6g/L; CaCl2 0.33g/L; KCl 0.3g/L) at 37 ± 1 °C, corresponding to the body fluid and corporal temperature, respectively. Long-term tests were used to better assess the stability of the passive condition expected from the alloys and to better mimic in vivo applications.
The main experimental setup was a three-electrode electrochemical cell, attached to a multichannel digital potentiostat Multi Palm Sems 4 (10 channels) (Palmsens B.V, Houten, The Netherlands) controlled by the software MultiTrace 4.5. Double-walled glass cells were used, containing circulating water at 37 °C set by a ultratermostatic bath model Q214M2 (QUIMIS, São Paulo, Brazil). Using the multichannel configuration, six tests were carried out in parallel over 120 h. The working electrodes were manufactured from small segments of the alloys, embedded in resin, having a flat exposed area to the solution of approximately 1 cm2. Platinum wires were used as counter-electrodes, and AgCl was used as reference. Surface preparation of the working electrodes included grinding until 600 mesh, ultrasonic cleaning in isopropyl alcohol, and drying with warm air, followed by storage in a drying chamber before the experiments.
The experimental work was carried out in the following sequence: initial surface characterization using smart digital microscope (Smartzoom 5, Carl Zeiss Microscopy GmbH, Oberkochen, Germany) performed at 100× and 500× magnifications, which included measurements of the exposed area; open circuit potential (OCP) monitoring over 120 h; electrochemical impedance spectroscopy (EIS) performed at intervals of 6 h up to 120 h, in the frequency range of 10
−2 to 10
4 Hz with a 10 mV at OCP; anodic polarization to determine corrosion potential (Ecorr), conducted up to 1.5 V above Ecorr, with a scan rate of 0.167 mV/s (ASTM F2129) and a limit current of 100 μA; evaluation of the initial and final pH of the solution; and final surface characterization using Smartzoom [
27].
Electrochemical corrosion resistance was assessed using a multiparametric approach by integrating results obtained with different electrochemical methods. The following parameters were defined and determined: Delta OCP (∆OCP) (Open circuit potential variation), corresponding to the difference between final and initial potentials, and a positive shift during prolonged exposure to Ringer’s solution at body temperature indicating the evolution of the alloy’s passive state and improved the protective ability of the aged passive film; Ip (Anodic current density), measured at the median value within the passive range, reflecting the rate of dissolution and ion release under oxidizing conditions; and Rp (Polarization resistance), obtained via electrochemical impedance spectroscopy measurements, corresponding to the lower limit of the complex impedance value obtained at the lower frequency of the Nyquist or Bode spectra; it represents the pure resistive component of the passive film formed on the alloy surface and can be correlated with its protective ability. This value was measured at the open circuit potential (OCP) after prolonged exposure to the simulated body fluid.
Composed parameters were calculated from the three parameters described, and the results were cross-correlated to the mechanical properties parameters of the alloys. From this approach, the best configurations among the alloys were selected and compared to similar values corresponding to the Ti-6Al-4V, the reference alloy.
4. Discussion
Several works are available in the scientific literature aiming at the development of alternative Ti alloys. The main objectives have been the suppression of Al and V, due to biocompatibility requirements, and an improved mechanical response, aiming to provide a better compliance with biological tissues and biological structures. Within the frame of this development, it is essential to preserve high corrosion resistance, a mandatory requirement for biomaterials. The scientific literature points out the fact that Zr, Ta, and Nb oxides improve the protection ability of the passive films formed in modified Ti alloys [
5]. Zr increases the corrosion resistance monotonically. The mechanism would be related to grain refinement. This conclusion is based on corrosion current density measurements, improved polarization resistance, and reduced corrosion rate. Some authors concluded that the corrosion morphology changes from uniform corrosion to mixed pitting and uniform corrosion when the Zr content is up to 40 wt% [
21].
In the present work, the adopted approach aimed to integrate the two objectives and perform an in-depth analysis of the electrochemical corrosion resistance of alternative alloys obtained by different processing routes. The physiological solution selected for evaluation was Ringer’s solution, widely used in the literature. The evaluations were carried out under a longer exposure time, 120 h, necessary to reach a stationary condition of interaction between metallic materials and potentially corrosive media. The electrochemical parameters were determined in independent tests for each material, using triplicate working electrodes. The use of integrated indices, determined by mechanical and electrochemical responses, is the most relevant contribution of the adopted approach, besides the definition of the best processing route. The Ti-6Al-4V alloy was used as a reference or control material.
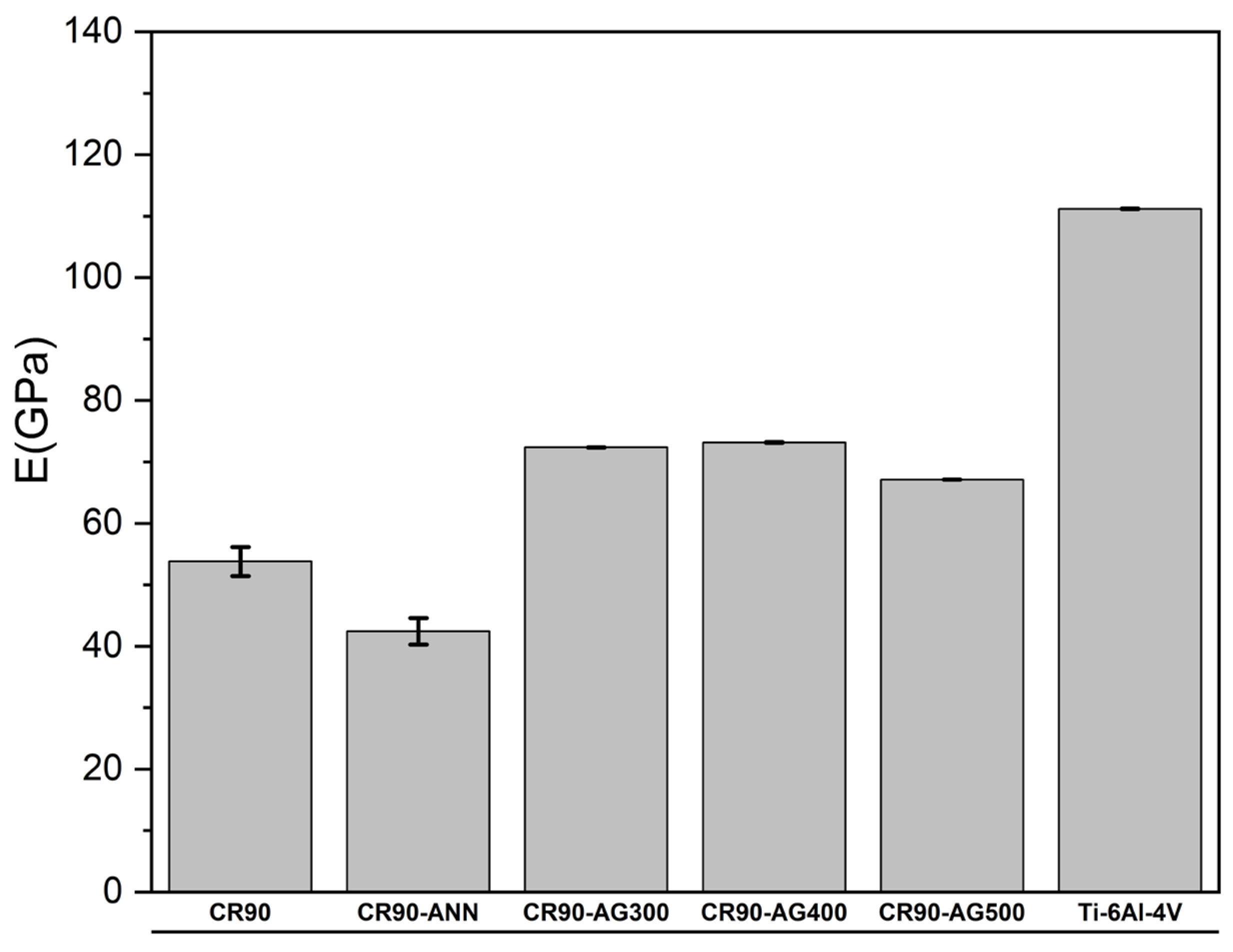
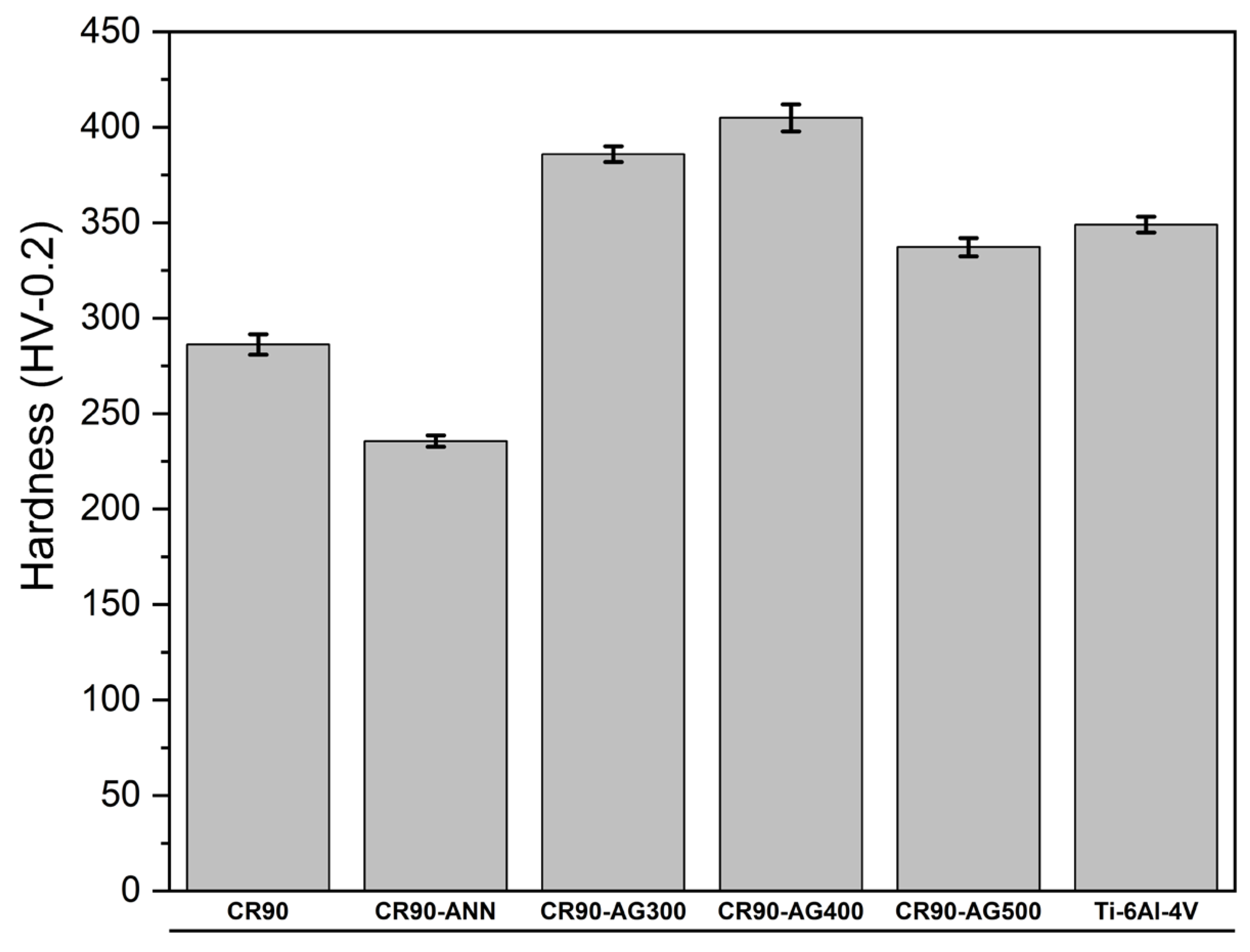
Figure 2 reveals that the Young’s modulus of the five processed alloys is much lower than the Young’s modulus of Ti-6Al-4V. A low Young’s modulus is important for alloys used in orthopedic implants. This requirement is mandatory to prevent the stress shielding that occurs due to the differences in Young’s moduli between the implant and the cortical bone. On the other hand, the hardness of Ti-6Al-4V is intermediate compared to the hardness of the processed alloys, as shown in
Figure 3. Combining these two mechanical properties,
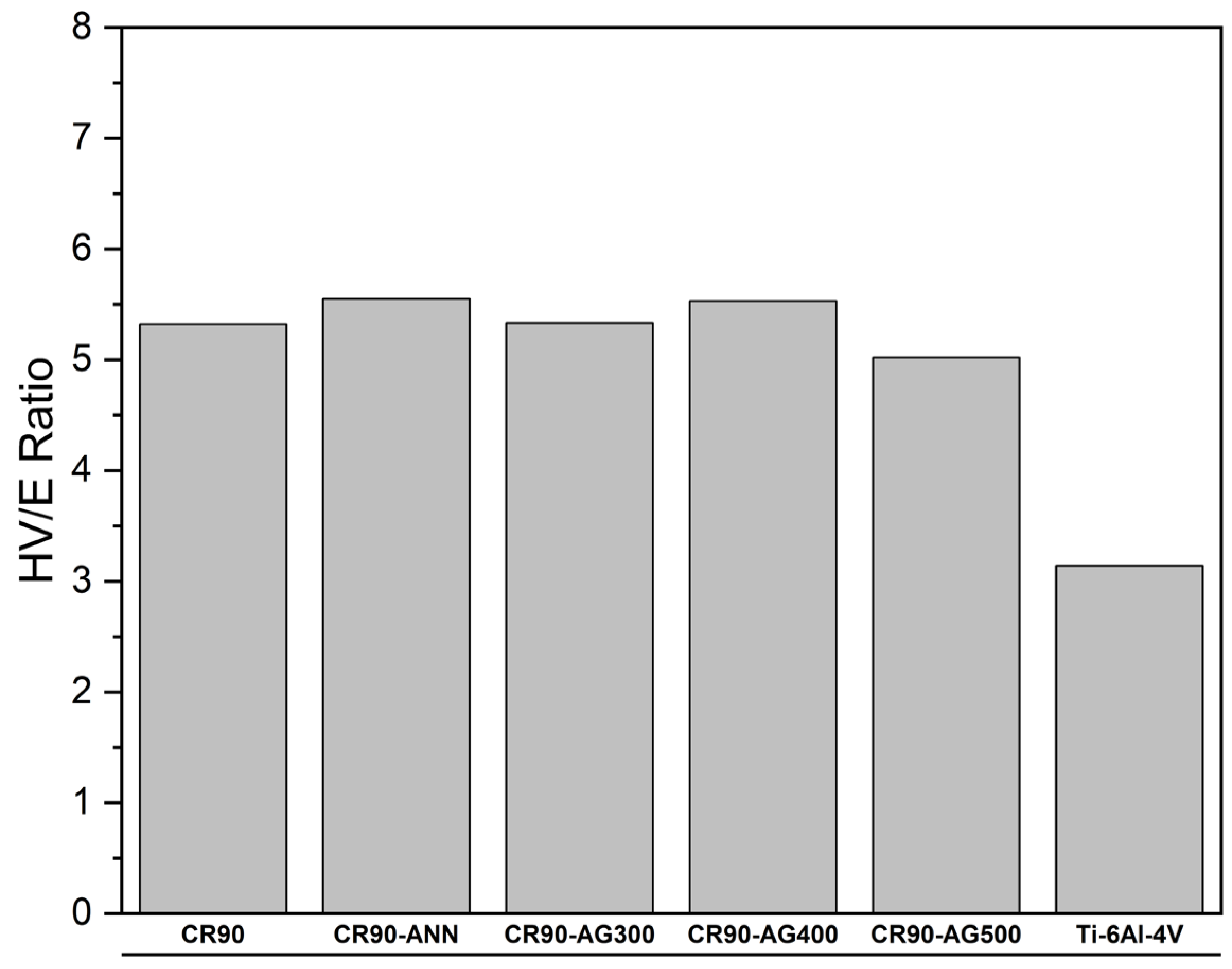
Figure 4 indicates that the HV/E ratio is much higher for all the processed alloys compared to the HV/E ratio of Ti-6Al-4V. These higher HV/E values express a better compliance with biological tissues and structures. Therefore, the five processed alloys have superior mechanical performances than that of Ti-6Al-4V for orthopedic implants.
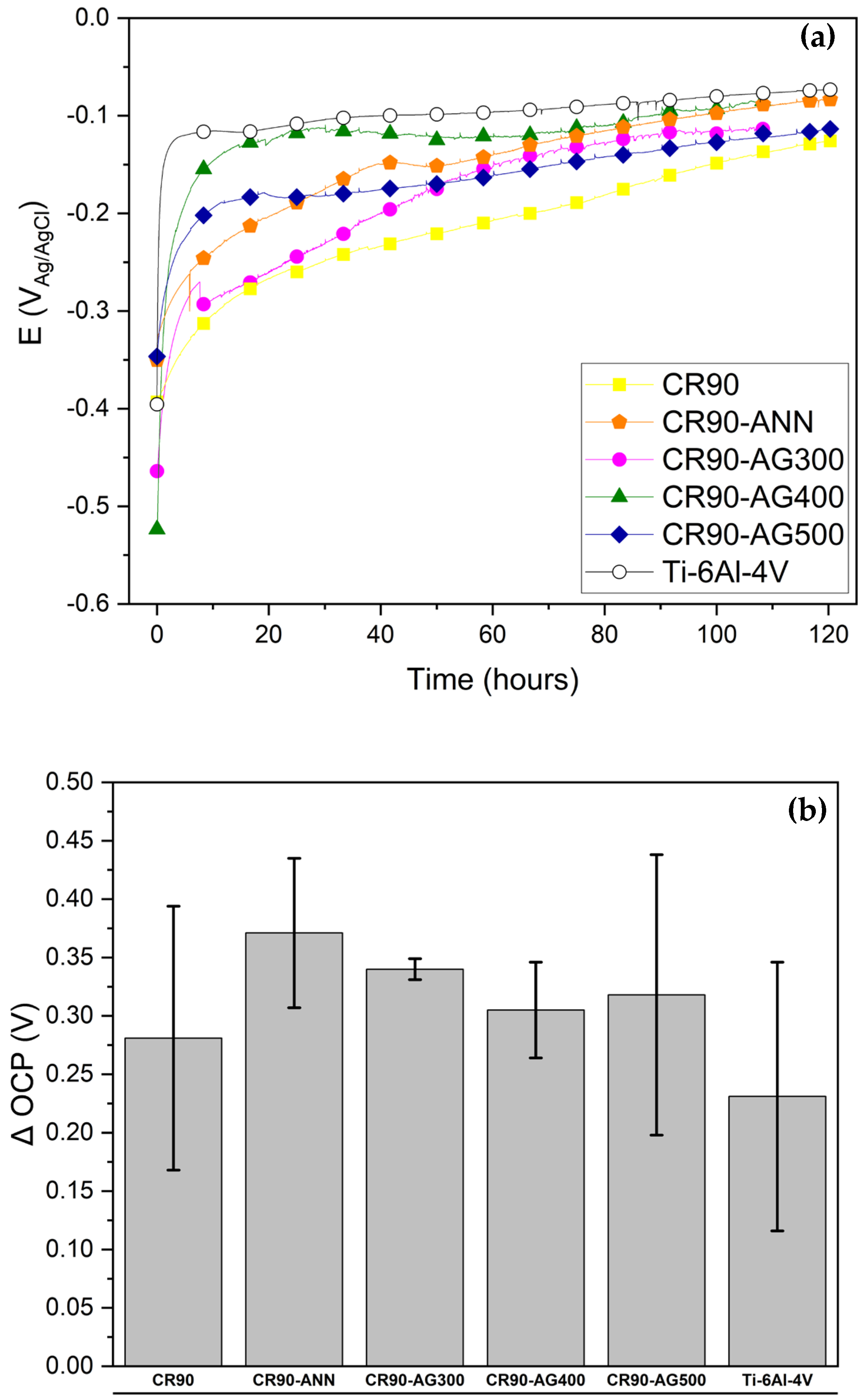
The open circuit potential results, as shown in
Figure 5 for all processing conditions, exhibited in general higher positive variations in OCP than Ti-6Al-4V alloy in the Ringer’s solution. The ranking of the results as described above indicates better performance of (CR90-ANN), followed by the (CR90-AG300), (CR90-AG500), (CR90-AG400), and (CR90) processing conditions. The lowest ∆OCP was registered for the Ti-6Al-4V alloy. This parameter expresses the aging of the passive films with immersion time towards a more protective condition, as expected, and suggests that the modifications introduced are positive in terms of electrochemical corrosion resistance. These data were used to calculate and integrate electrochemical parameters. The trend of a more positive OCP with time in simulated body fluid of Ti and Ti alloys is reported in the literature by [
4,
7,
20,
30,
31].
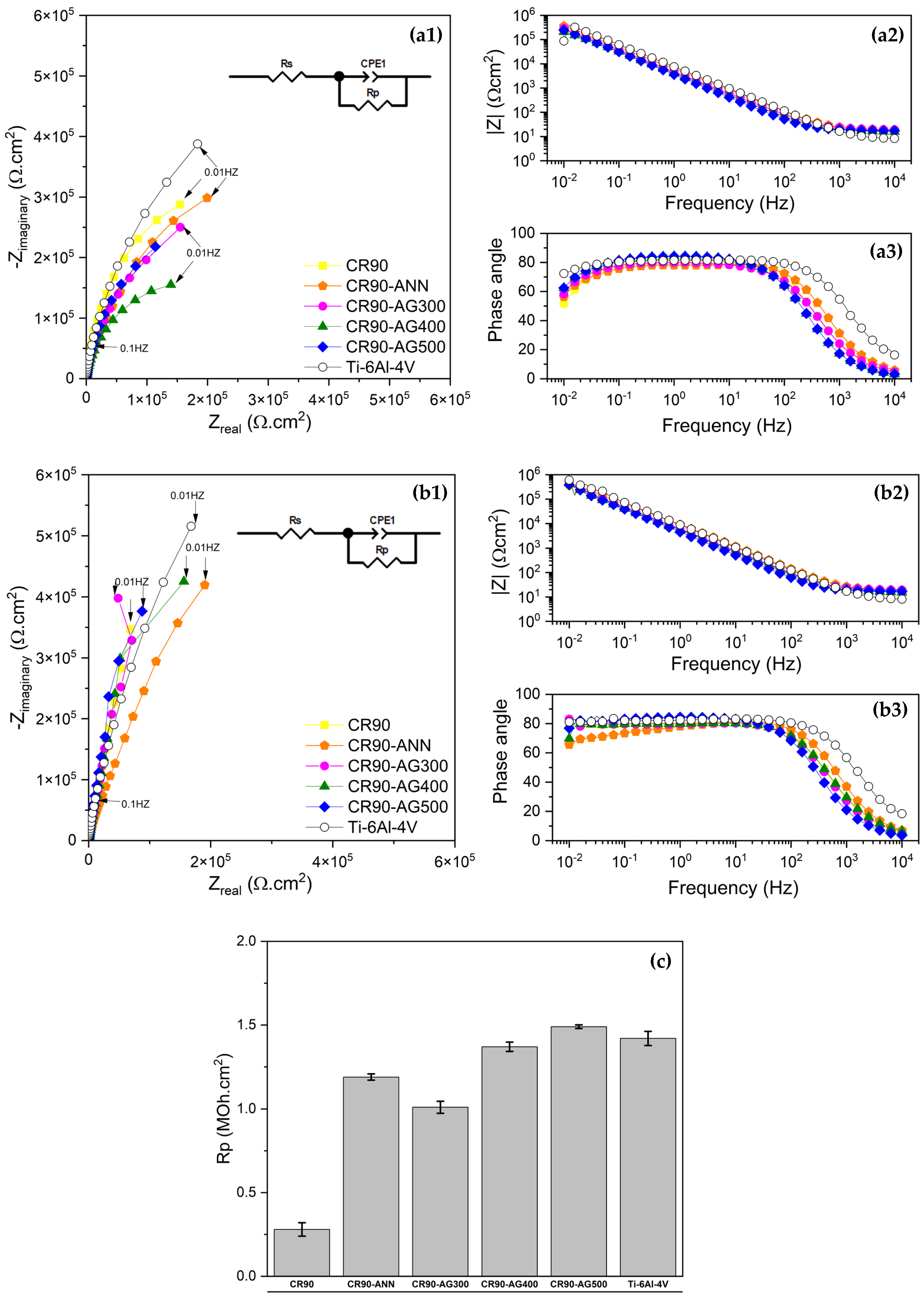
Results obtained by EIS are shown in
Figure 6. All materials exhibited a pure capacitive response containing a single capacitive loop within the frequency range used. It can be observed that an increment in the impedance with time was registered for all conditions. This trend can be associated with the positive evolution of the passive conditions towards higher corrosion resistance. Only the initial branch of the capacitive loop was obtained, as commonly reported for passive materials containing a semiconductor passive film at the metal/solution interface. The extraction of the pure resistive component of impedance at lower frequency, or Rp—polarization resistance—was possible by numerical fit of the data using the Zview software. These values correspond to the polarization resistance of the passive film acting as a semiconductor. Higher Rp values suggest higher protection abilities of the passive films. The results obtained after 6 h and 120 h comparatively show the increment of Rp for all materials with immersion time. Considering the absolute values after 120 h, summarized in
Figure 6c, it is possible to observe that the values of the Ti-6Al-4V are intermediate compared to those of the group of alloys under investigation. The order of magnitude for Ti-6Al-4V alloy and (CR90 AG400), (CR90-AG300), (CR90-ANN) processing conditions was 10
6 Ω·cm
2. Similar values obtained for alloys (CR90-AG500) were significantly lower, having 10
5 as order of magnitude. Capacitive passive layers of dielectric characteristics of high-niobium Ti alloys in saline solution are reported [
32]. The same capacitive response of passive Bo-modified Ti alloys in Hanks Balanced Salt Solution (HBSS). Increment of Z with immersion time was reported, up to 168 h, and the Rp values were in the range 0.5 to 1.0 MΩ·cm
2 [
33]. Regarding the structure and composition of the passive films, new knowledge is obtained by using Mott–Schottky and EIS analysis. Detrimental effects of Nb in Ti-Zr alloys have also been reported. When Nb is higher than 20%, the corrosion resistance increases. The authors mention localized corrosion incidence [
34].
Anodic polarization curves are shown in
Figure 7. All conditions exhibited a fully passive response, without any incidence of localized corrosion. This result is expected for Ti alloys in body fluids, being commonly reported in the literature [
4,
7,
9,
12,
20,
30,
31,
35]. Different profiles in the anodic polarization curves are related to different OCPs and slight variations among the passive current densities. The former values were used to estimate an additional parameter, Ip, the anodic current density registered at a potential corresponding to 50% of the passive range. Ti-6Al-4V exhibited the lowest value, followed by processing conditions (CR90-AG300), (CR90-AG500), (CR90-AG400), (CR90), and (CR90-ANN). Ip is related to corrosion rate, at almost zero, but deserves special attention if long-term exposures can lead to release of adverse ions during in vivo applications [
5,
31,
36,
37]. Anodic polarization results of modified Ti alloys that formed a stable protective passive layer within the tested potential range, with no incidence of localized corrosion, are reported [
12,
34,
35] and used to rank the corrosion resistance.
Surface analysis indicated no traces of localized corrosion attack for all processing conditions after anodic polarization. This result confirms the stable passive condition achieved in the Ringer’s solution after 120 h immersion.
The corrosion test results demonstrated that no direct correlation could be established between the microstructure, with its different phases, and the observed corrosion resistance. However, this correlation is reported by some authors, who advocate higher corrosion resistance for certain microstructural configurations, especially martensitic Ti-6Al-4V extra-low interstitial alloy [
10], and reduction of the resistance promoted by preferential dissolution at α/β phase boundaries during long-term exposure in 5 M HCl, unlike the neutral Ringer’s solution used as physiological environment in the present work [
4].
In order to establish a classification of the corrosion resistance for the different processing conditions, the electrochemical parameters were merged on a unique parameter, Elindex, composed of ΔOCP, Rp, and Ip, calculated through the ratio (Rp x ΔOCP)/Ip [
38]. This formulation is based on the positive and negative effects of increasing values on electrochemical corrosion resistance. The higher the ELindex, the higher the corrosion resistance. Based on the results shown in
Table 5, it can be concluded that the alternative alloy (CR90-AG500) exhibited the best performance, however, slightly below the response obtained with the reference alloy Ti-6Al-4V. The integrated electrochemical index permits a more accurate analysis of the corrosion resistance of the Ti alloys in Ringer’s solution.
As mentioned above, the β-Ti alloy in the CR90-AG500 condition has a much higher HV/E ratio, associated with a lower elastic modulus, compared to the reference Ti-6Al-4V alloy and, therefore, a better compliance with biological tissues and structures. Considering the results obtained in the present work, the β-Ti alloy in the CR90-AG500 condition stands out as a superior alternative to the Ti-6Al-4V alloy, offering comparable corrosion resistance and a markedly improved mechanical response, making it a more advantageous choice for biomedical applications.
Author Contributions
Conceptualization, A.R.V.N., J.A.P.G., and L.H.d.A.; Data curation, A.R.V.N., C.D.d.R.B., G.G.C., P.T.d.S., S.B., and J.D.; Formal analysis, A.R.V.N., C.D.d.R.B., P.T.d.S., S.B., G.G.C., J.D., and J.A.P.G.; Funding acquisition, A.R.V.N., S.B., and L.H.d.A.; Investigation, A.R.V.N., P.T.d.S., S.B., C.D.d.R.B., and J.D. Methodology, A.R.V.N., C.D.d.R.B., L.H.d.A., and J.A.P.G.; Project administration, A.R.V.N., L.H.d.A., and J.A.P.G.; Resources, S.B., J.A.P.G., and L.H.d.A.; Supervision: L.H.d.A. and J.A.P.G.; Validation: A.R.V.N., C.D.d.R.B., J.D., J.A.P.G., and L.H.d.A.; Visualization, A.R.V.N., C.D.d.R.B., and G.G.C.; Writing—original draft, A.R.V.N. and C.D.d.R.B.; Writing—review and editing, J.D., J.A.P.G., and L.H.d.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by FAPERJ—Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) [Grants SEI-260003/019589/2022, E-26/200.947/2024, E-26/204.450/2021, E-26/204.160/2024].
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors gratefully acknowledge the financial support from the Brazilian research funding agencies and also extend their gratitude to Leonardo Sales Araujo and Laercio Rosignoli Guzela for their valuable contributions to this work.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kuroda, P.A.B.; da Silva, L.M.; Sousa, K.D.S.J.; Donato, T.A.G.; Grandini, C.R. Preparation; structural; microstructural; mechanical, and cytotoxic characterization of Ti-15Nb alloy for biomedical applications. Artif. Organs 2020, 44, 811–817. [Google Scholar] [CrossRef]
- Bocchetta, P.; Chen, L.Y.; Tardelli, J.D.C.; Reis, A.C.D.; Almeraya-Calderón, F.; Leo, P. Passive layers and corrosion resistance of biomedical ti-6al-4v and β-ti alloys. Coatings 2021, 11, 487. [Google Scholar] [CrossRef]
- Manivasagam, G.; Dhinasekaran, D.; Rajamanickam, A. Biomedical Implants: Corrosion and its Prevention—A Review. Recent Pat. Corros. Sci. 2010, 2, 40–54. [Google Scholar] [CrossRef]
- Atapour, M.; Pilchak, A.L.; Frankel, G.S.; Williams, J.C. Corrosion behavior of β titanium alloys for biomedical applications. Mater. Sci. Eng. C 2011, 31, 885–891. [Google Scholar] [CrossRef]
- Tardelli, J.D.C.; Bolfarini, C.; Reis, A.C.D. Comparative analysis of corrosion resistance between beta titanium and Ti-6Al-4V alloys: A systematic review. J. Trace Elem. Med. Biol. 2020, 62, 126618. [Google Scholar] [CrossRef]
- Ho, W.F.; Cheng, C.H.; Pan, C.H.; Wu, S.C.; Hsu, H.C. Structure, mechanical properties and grindability of dental Ti-10Zr-X alloys. Mater. Sci. Eng. C 2009, 29, 36–43. [Google Scholar] [CrossRef]
- Robin, A.; Carvalho, O.A.S. Influence of pH and fluoride species on the corrosion behavior of Ti-xNb-13Zr alloys in ringer’s solution. Adv. Mater. Sci. Eng. 2013, 2013, 434975. [Google Scholar] [CrossRef]
- Calderon-Moreno, J.M.; Vasilescu, C.; Drob, S.I.; Ivanescu, S.; Osiceanu, P.; Drob, P.; Popa, M.; Preda, S.; Vasilescu, E. Microstructural and mechanical properties, surface and electrochemical characterisation of a new Ti-Zr-Nb alloy for implant applications. J. Alloys Compd. 2014, 612, 398–410. [Google Scholar] [CrossRef]
- Shukla, A.K.; Balasubramaniam, R. Effect of surface treatment on electrochemical behavior of CP Ti, Ti–6Al–4V and Ti–13Nb–13Zr alloys in simulated human body fluid. Corros. Sci. 2006, 48, 1696–1720. [Google Scholar] [CrossRef]
- Cvijović-Alagić, I.; Cvijović, Z.; Mitrović, S.; Panić, V.; Rakin, M. Wear and corrosion behaviour of Ti–13Nb–13Zr and Ti–6Al–4V alloys in simulated physiological solution. Corros. Sci. 2011, 53, 796–808. [Google Scholar] [CrossRef]
- Chui, P.; Jing, R.; Zhang, F.; Li, J.; Feng, T. Mechanical properties and corrosion behavior of β-type Ti-Zr-Nb-Mo alloys for biomedical application. J. Alloys Compd. 2020, 842, 155693. [Google Scholar] [CrossRef]
- Nnamchi, P.S.; Obayi, C.S.; Todd, I.; Rainforth, M.W. Mechanical and electrochemical characterisation of new Ti-Mo-Nb-Zr alloys for biomedical applications. J. Mech. Behav. Biomed. Mater. 2016, 60, 68–77. [Google Scholar] [CrossRef]
- Xu, Y.; Zhao, S.; Cai, Y. Design, preparation and performance research of β-type Ti-Mo-Nb-Zr alloy. Appl. Phys. A 2022, 128, 426. [Google Scholar] [CrossRef]
- Lu, J.; Ge, P.; Li, Q.; Zhang, W.; Huo, W.; Hu, J.; Zhang, Y.; Zhao, Y. Effect of microstructure characteristic on mechanical properties and corrosion behavior of new high strength Ti-1300 beta titanium alloy. J. Alloys Compd. 2017, 727, 1126–1135. [Google Scholar] [CrossRef]
- Matsumoto, H.; Watanabe, S.; Hanada, S. Microstructures and mechanical properties of metastable β TiNbSn alloys cold rolled and heat treated. J. Alloys Compd. 2007, 439, 146–155. [Google Scholar] [CrossRef]
- Grosdidier, T.; Combres, Y.; Gautier, E.; Philippe, M.-J. Effect of microstructure variations on the formation of deformation-induced martensite and associated tensile properties in a β metastable Ti alloy. Metall. Mater. Trans. A 2000, 31, 1095–1106. [Google Scholar] [CrossRef]
- Lin, J.; Ozan, S.; Munir, K.; Wang, K.; Tong, X.; Li, Y.; Li, G.; Wen, C. Effects of solution treatment and aging on the microstructure, mechanical properties, and corrosion resistance of a β type Ti-Ta-Hf-Zr alloy. RSC Adv. 2017, 7, 12309–12317. [Google Scholar] [CrossRef]
- Zheng, Y.; Williams, R.E.A.; Wang, D.; Shi, R.; Nag, S.; Kami, P.; Sosa, J.M.; Banerjee, R.; Wang, Y.; Fraser, H.L. Role of ω phase in the formation of extremely refined intragranular α precipitates in metastable β-titanium alloys. Acta Mater. 2016, 103, 850–858. [Google Scholar] [CrossRef]
- Li, Q.; Li, J.; Ma, G.; Liu, X.; Pan, D. Influence of ω phase precipitation on mechanical performance and corrosion resistance of Ti–Nb–Zr alloy. Mater. Des. 2016, 111, 421–428. [Google Scholar] [CrossRef]
- Monteiro, E.D.S.; Elias, C.N.; de Souza Soares, F.M.; Ribeiro, K.S.; de Carvalho Santana, A.I.; Santos, C.D. Influence of heat treatment on the microstructure, wettability, and corrosion resistance of a Ti–30Nb–7Zr alloy used in biomedical applications. J. Mater. Res. Technol. 2024, 29, 1683–1690. [Google Scholar] [CrossRef]
- Su, B.; Luo, L.; Wang, B.; Su, Y.; Wang, L.; Ritchie, R.O.; Guo, E.; Li, T.; Yang, H.; Huang, H.; et al. Annealed microstructure dependent corrosion behavior of Ti-6Al-3Nb-2Zr-1Mo alloy. J. Mater. Sci. Technol. 2021, 62, 234–248. [Google Scholar] [CrossRef]
- Nunes, A.R.V.; Borborema, S.; Araújo, L.S.; Rodrigues, T.Z.L.; Malet, L.; Dille, J.; de Almeida, L.H. Influence of the Chemical Composition on the Phase Stability and Mechanical Properties of Biomedical Ti-Nb-Mo-Zr Alloys. Metals 2023, 13, 1889. [Google Scholar] [CrossRef]
- Nunes, A.R.V.; Borborema, S.; Cossú, C.M.F.A.; Araújo, L.S.; Dille, J.; Malet, L.; Kaufman, M.J.; de Almeida, L.H. Effects of Aging on the Microstructure and Mechanical Properties of Metastable β-type Ti-23.6Nb-5.1Mo-6.7Zr Alloy with Low Elastic Modulus. J. Mater. Res. Technol. 2024, 31, 4104–4112. [Google Scholar] [CrossRef]
- ASTM E1876-22; Standard Test Method for Dynamic Young’s Modulus, Shear Modulus, and Poisson’s Ratio by Impulse Excitation of Vibration. ASTM International: West Conshohocken, PA, USA, 2022.
- ASTM F136-13(2021); Standard Specification for Wrought Titanium-6Aluminum-4Vanadium ELI (Extra Low Interstitial) Alloy for Surgical Implant Applications. ASTM International: West Conshohocken, PA, USA, 2021.
- ISO 5832-4:2024; Implants for Surgery—Metallic Materials—Part 4: Cobalt-Chromium-Molybdenum Casting Alloy. International Organization for Standardization: Geneva, Switzerland, 2024.
- ASTM F2129-19a; Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices. ASTM International: West Conshohocken, PA, USA, 2019.
- Kolli, R.P. A Review of Metastable Beta Titanium Alloys. Metals 2018, 8, 506. [Google Scholar] [CrossRef]
- Gabriel, S.B.; De Almeida, L.H.; Nunes, C.A.; Dille, J.; Soares, G.A. Maximisation of the ratio of microhardness to the Young’s modulus of Ti-12Mo-13Nb alloy through microstructure changes. Mater. Sci. Eng. C 2013, 33, 3319–3324. [Google Scholar] [CrossRef]
- Barros, C.D.d.R.; Rocha, J.C.; Bastos, I.N.; Gomes, J.A.D.C.P. Tribocorrosion Resistance of Dental Implant Alloys—Assessment of cp-Ti, Ti6Al4V, and NiCr in Neutral and Acidified Saliva. J. Bio Tribo Corros. 2021, 7, 73. [Google Scholar]
- Dimah, M.K.; Albeza, F.D.; Borrás, V.A.; Muñoz, A.I. Study of the biotribocorrosion behaviour of titanium biomedical alloys in simulated body fluids by electrochemical techniques. Wear 2012, 294, 409–418. [Google Scholar] [CrossRef]
- Chelariu, R.; Bolat, G.; Izquierdo, J.; Mareci, D.; Gordin, D.M.; Gloriant, T.; Souto, R.M. Metastable beta Ti-Nb-Mo alloys with improved corrosion resistance in saline solution. Electrochim. Acta 2014, 137, 280–289. [Google Scholar] [CrossRef]
- Voorhees, T.; Villalpando, O.; Ravi, V.A. Characterization of the Corrosion Behavior of Advanced Titanium Alloys in Simulated Physiological Environments Using Electrochemical Impedance Spectroscopy. Corrosion 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Ji, P.F.; Li, B.; Chen, B.H.; Wang, F.; Ma, W.; Zhang, X.Y.; Ma, M.Z.; Liu, R.P. Effect of Nb addition on the stability and biological corrosion resistance of Ti-Zr alloy passivation films. Corros. Sci. 2020, 170, 108696. [Google Scholar] [CrossRef]
- Robles, K.; McCarthy, S.; Medina, J.; Nguyen, L.; Rodriguez, R.; Fly, J.; Ravi, V.A. Corrosion Behavior of Niobium-Containing Titanium Alloys in Biological Solutions. Corrosion 2017, 2017, NACE-2017-9549. [Google Scholar]
- Vasconcelos, D.M.; Santos, S.G.; Lamghari, M.; Barbosa, M.A. The two faces of metal ions: From implants rejection to tissue repair/regeneration. Biomaterials 2016, 84, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Hanawa, T. Metal ion release from metal implants. Mater. Sci. Eng. C 2004, 24, 745–752. [Google Scholar] [CrossRef]
- Kassab, E.J.; Barros, C.D.D.R.; Silva, P.G.; Silva, L.F.; Gomes, J.A.P. Corrosion of NiCr Alloys for Dental Applications: Effects of Environment, Chemical Composition and Casting Route. J. Mater. Eng. Perform. 2021, 30, 994–1000. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).