1. Introduction
High-strength cold-resistant and cryogenic steels are widely used for icebreakers, gas carriers, hydrogen storage, hydrocarbon production and transportation, wind turbines, offshore platforms, railroads, and the automotive industry. The history of traditional high-strength steel (HSS) metallurgy began with the creation of low-alloy Si-Mn ferritic–pearlitic steels with a carbon content of <0.2% [
1,
2,
3]. The micro-alloying of V, Nb, Ti, and Thermomechanical Controlled Processing (TMCP) allowed the achievement of a fine-grained microstructure, increasing strength and impact toughness with a decrease in carbon content < 0.15% [
1,
2,
3].
Subsequent development of TMCP and the application of accelerated cooling with tempering allowed for obtaining ferritic–bainitic and ferritic–martensitic microstructures of steels with reduced carbon content <0.1%. This microstructure has a fine-dispersed morphology with distributed and dispersed carbides and carbonitrides, and has a high impact toughness. Micro-alloying, optimization of TMCP, microstructure refinement, dispersion hardening, refining, and reduction of the anisotropy of rolled steel are used to improve plasticity and impact toughness of high-strength steels. Modern HSS have bainitic, bainitic–martensitic, martensitic–bainitic, and martensitic microstructures with low carbon content up to 0.05–0.02%. The production and development of high-strength steels are closely related to welding metallurgy and weldability, as arc and laser welding are used to make structures.
Research on the metallurgy and weldability of high-strength steels is actively developing all over the world. The main trends are increasing strength and impact toughness of steels, improving quality, and reducing the cost of alloying in steelmaking [
4,
5,
6,
7]. Among traditional and advanced high-strength steels for low-temperature and cryogenic service, about 15 groups of low- and high-alloy steels should be distinguished, as shown in
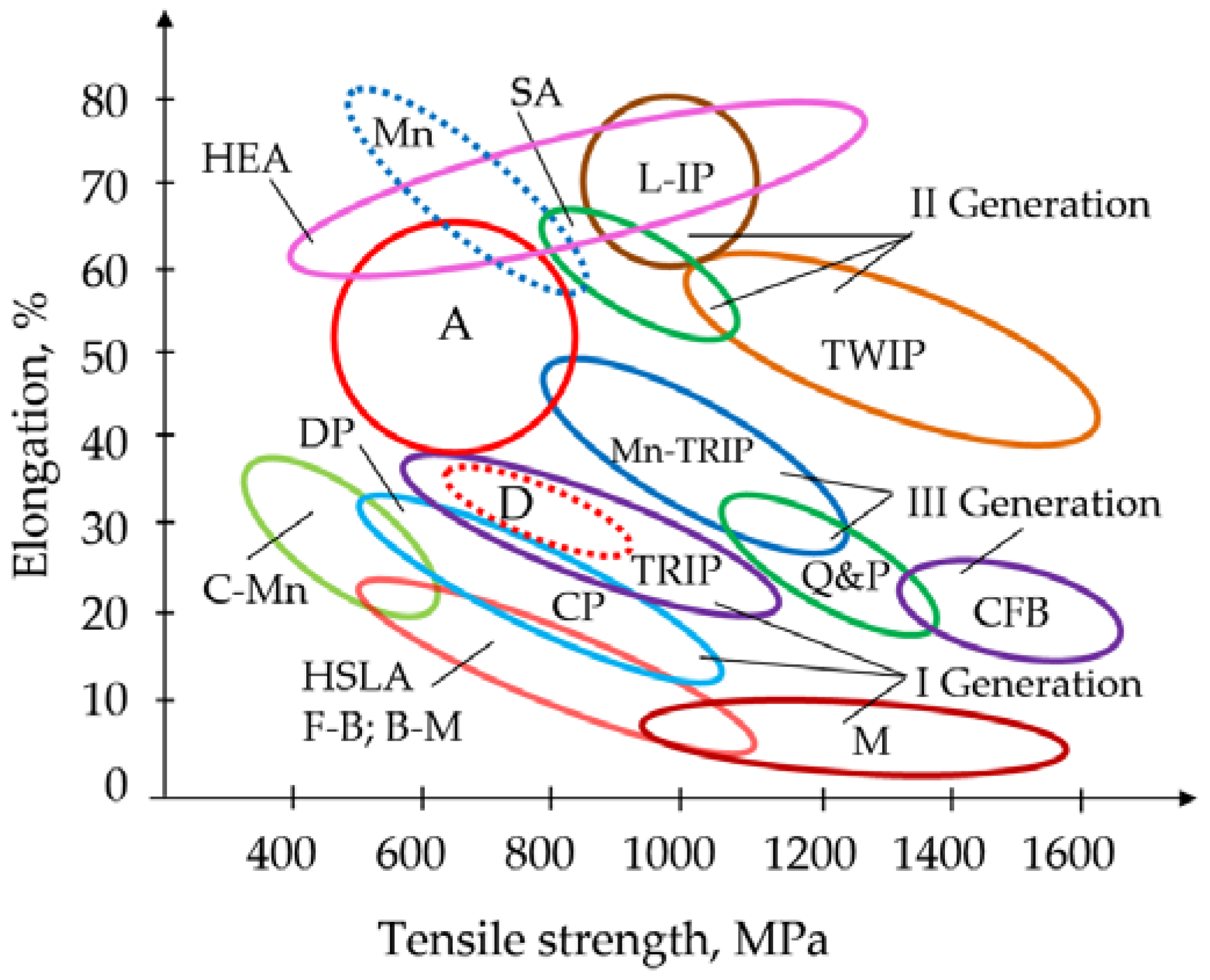
Figure 1.
Figure 1 contains the following designations: C-Mn—pearlitic steels; HSLA—high-strength low-alloy steels; F-B—ferritic–bainitic steels; B-M—bainitic–martensitic steels; DP (Dual-Phase)—two-phase steels; CP—complex phase steels; D—duplex steels; TRIP (Transformation-Induced-Plasticity)—metastable steels with induced plasticity; M—martensitic steels; TWIP (Twinning-Induced Plasticity)—austenitic high-manganese steels with induced plasticity; Mn-TRIP—medium-manganese TRIP steels; Q&P (Quenching and Partitioning)—martensitic–austenitic steels; CFB—carbide-free bainitic steels; A—austenitic steels; SA—superaustenitic steels; L-IP—lightweight steels with induced plasticity; Mn—high-manganese austenitic steels; HEA—high-entropy alloys.
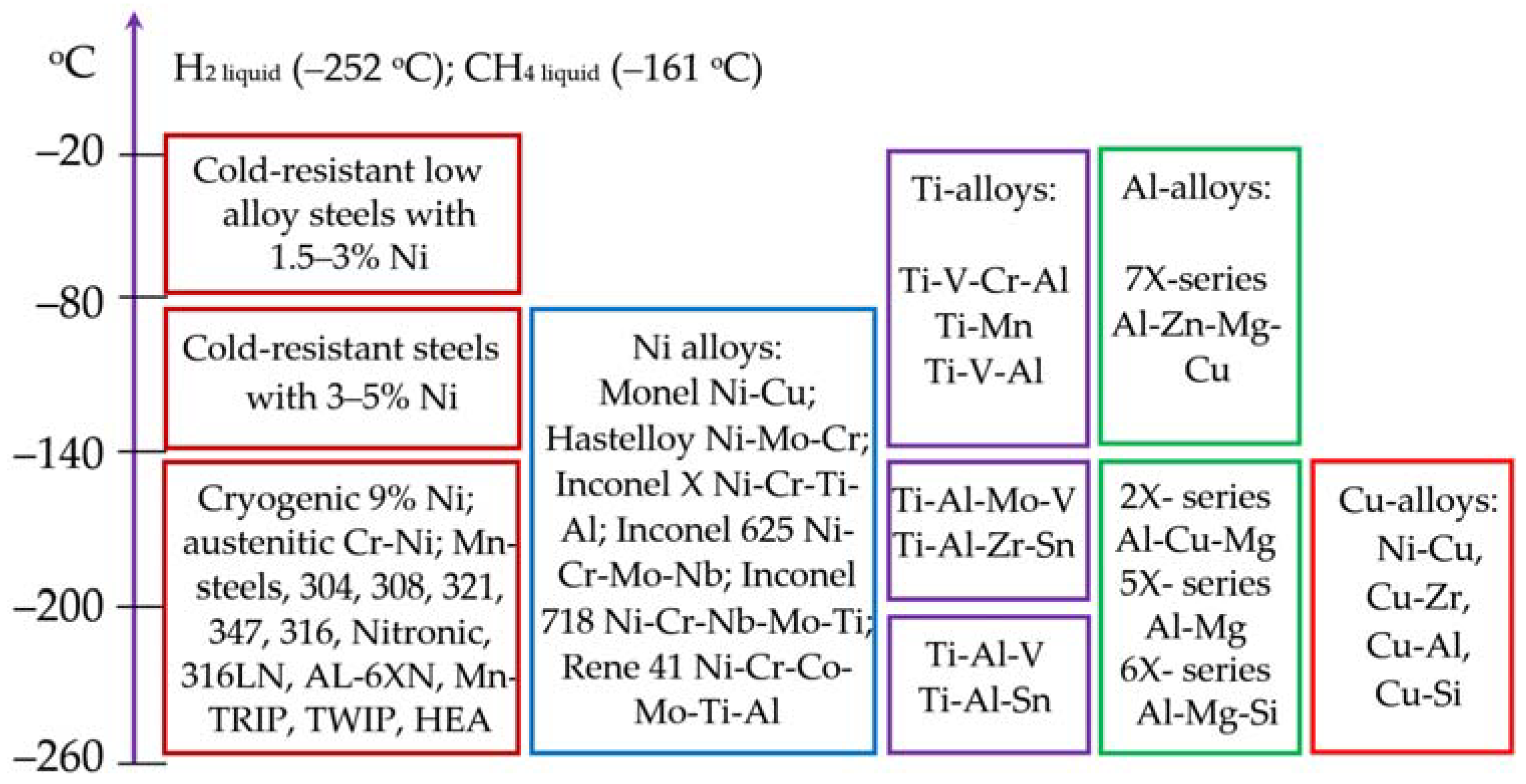
Advanced steels have high strength, corrosion resistance, and high viscoplasticity at low temperatures, and can be considered as alternatives to Ni-, Ti-, Al-, and Cu-alloys for low temperature and cryogenic service, including hydrogen storage systems, as shown in
Figure 2.
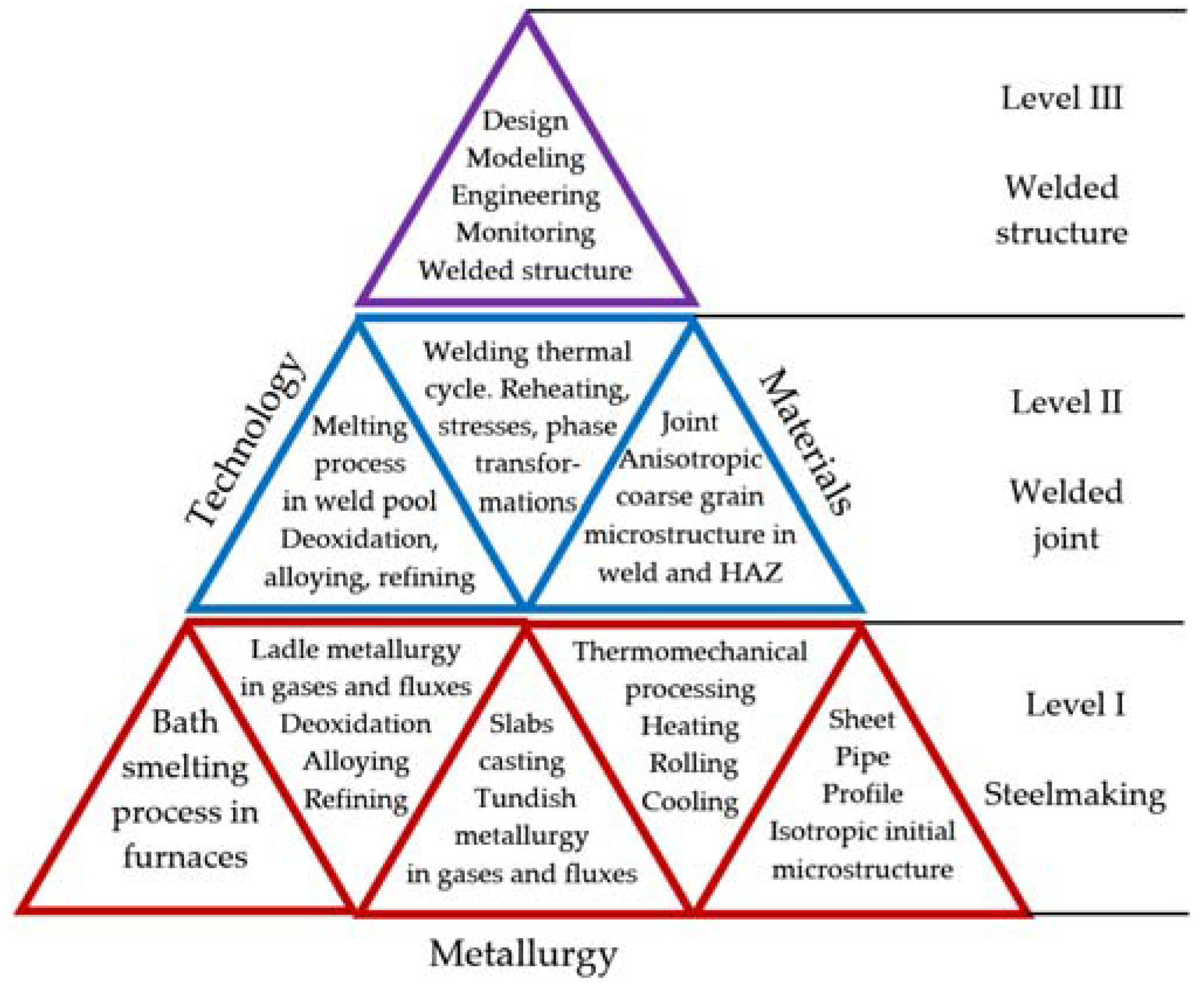
The scientific novelty of this paper consists in the analysis of problems of weldability and the systemic concept formulation of a relationship between the steelmaking, weldability and manufacturing of welded structures for low temperature and cryogenic service. The weldability of high-strength steels is linked to steelmaking, thermomechanical processing, and the microstructural design of steels, based on a three-level system of processes, as shown in
Figure 3.
The high quality of the manufacturing of welded structures is based on the quality in levels: steelmaking, welded joint, and welded structure. The most serious problem is the anisotropy and microstructure chemical-heterogeneity due to welding thermal cycle on level II. The anisotropy degree of the weld microstructure depends on the susceptibility of the initial microstructure of the rolled steels to the welding thermal cycle.
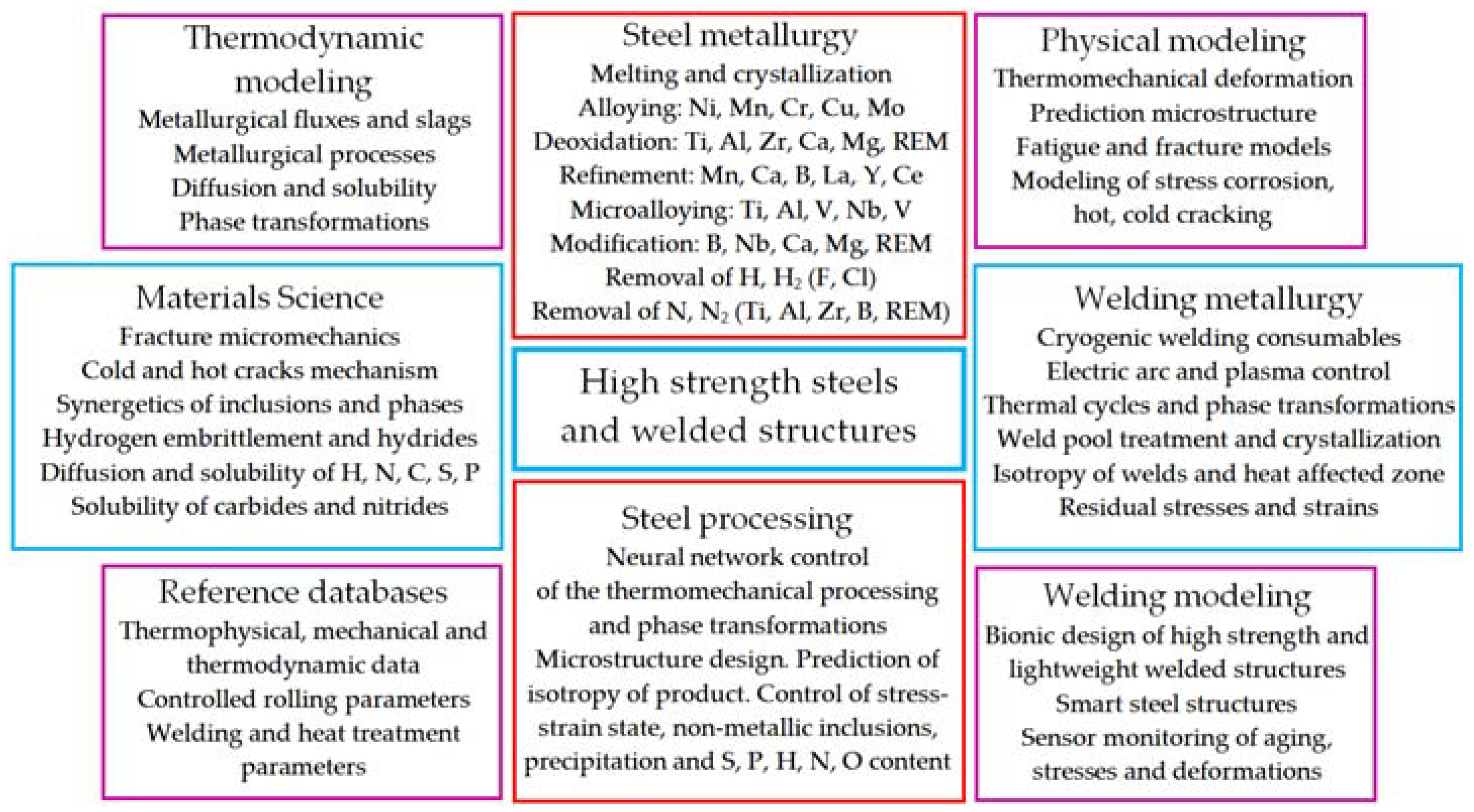
In our opinion, advanced current trends, and methods in the field of metallurgy and weldability for improving the mechanical properties, microstructure, and viscoplasticity of high-strength steels and welded joints can present as system, as shown in
Figure 4.
The system of advanced trends and methods is based on the metallurgical relationship between the steelmaking and the manufacturing of welded structures.
The purpose of this work is to analyze and systematize the advanced trends and methods in the field of metallurgy and weldability of high-strength cold-resistant and cryogenic steels to achieve the high quality of welded structures.
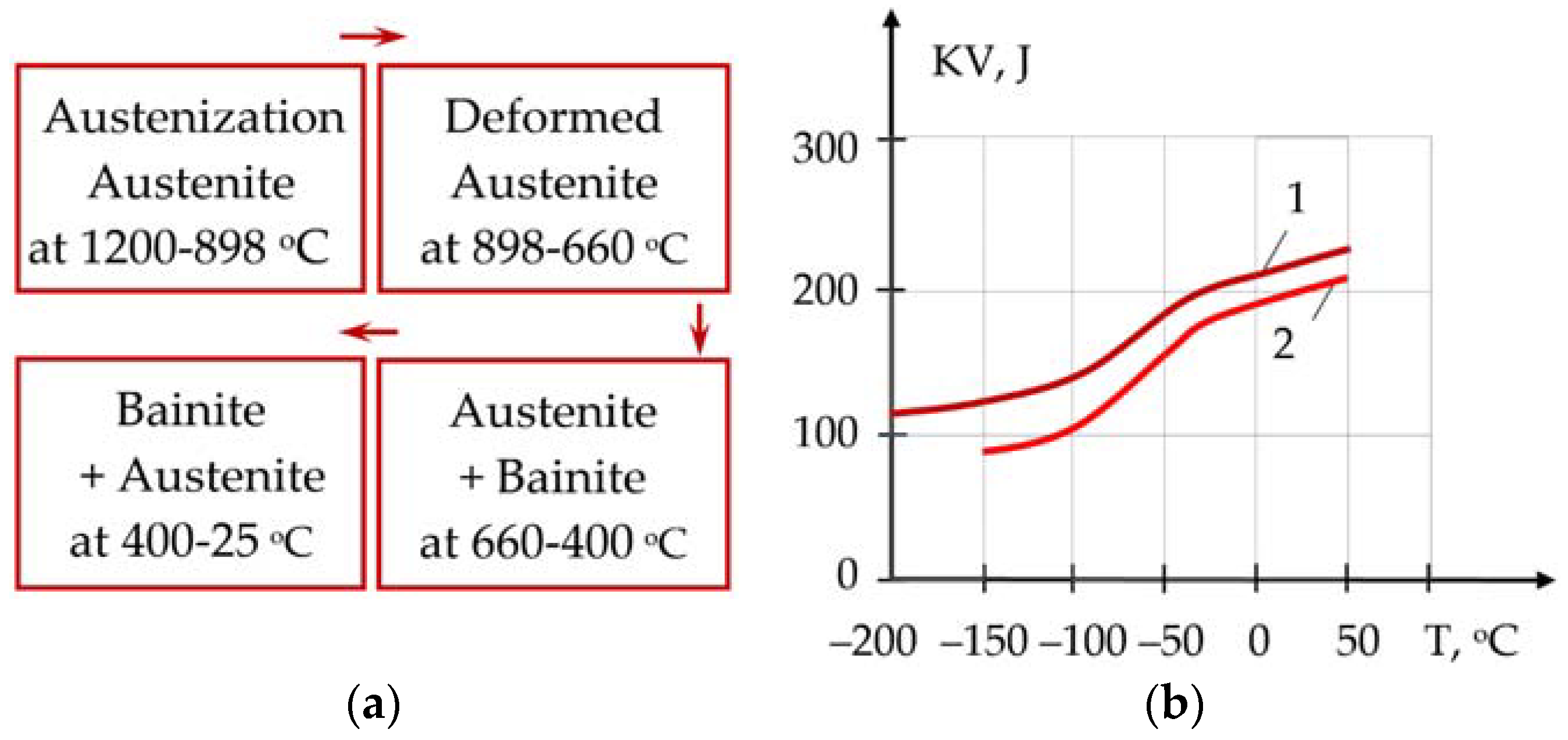
5. Improvement of Viscoplasticity of Steels
Theoretical foundations for improving the viscoplasticity of steels are based on the studies by R. Hadfield, J. Chipman, A. Samarin, A. Baikov, E. Bain, A. Cottrell, H. Bhadeshia, N. Liakishev, and I. Gorynin [
84,
85,
86,
87]. TMCP and the formation of nonmetallic inclusions leads to residual stresses at the interface between the inclusion and the steel matrix, due to different coefficients of thermal expansion and mechanical properties. The local stresses in the nonmetallic inclusions of Mg-Al-O, Al-Ca-O-S, and TiN, especially with sharp-edged shapes, can reach 750–2000 MPa, which leads to the fracture of brittle inclusions and the appearance of cracks [
88].
As shown by the Electron Backscatter Diffraction (EBSD) analysis, parameters of TMCP and the hardening of Cu-containing HSLA steels can lead to coagulation and irregular distribution of Cu precipitates. This reduces the isotropy of rolled steel and the weldability of high-strength steels, especially with errors in welding parameters [
89]. The composition, morphology, and micromechanical properties of nanoparticles smaller than 1 nm, including Cu precipitates, NiAl, Ni
2AlMn, Ni
2AlTi, Ni
3Ti, NbC, and Mo
2C intermetallics should be considered to improve impact toughness at low temperatures and weldability. For the isotropy of high-strength steels, the development of a synergistic combination of nanoparticles in the nanoscale hardening mechanism is required [
90].
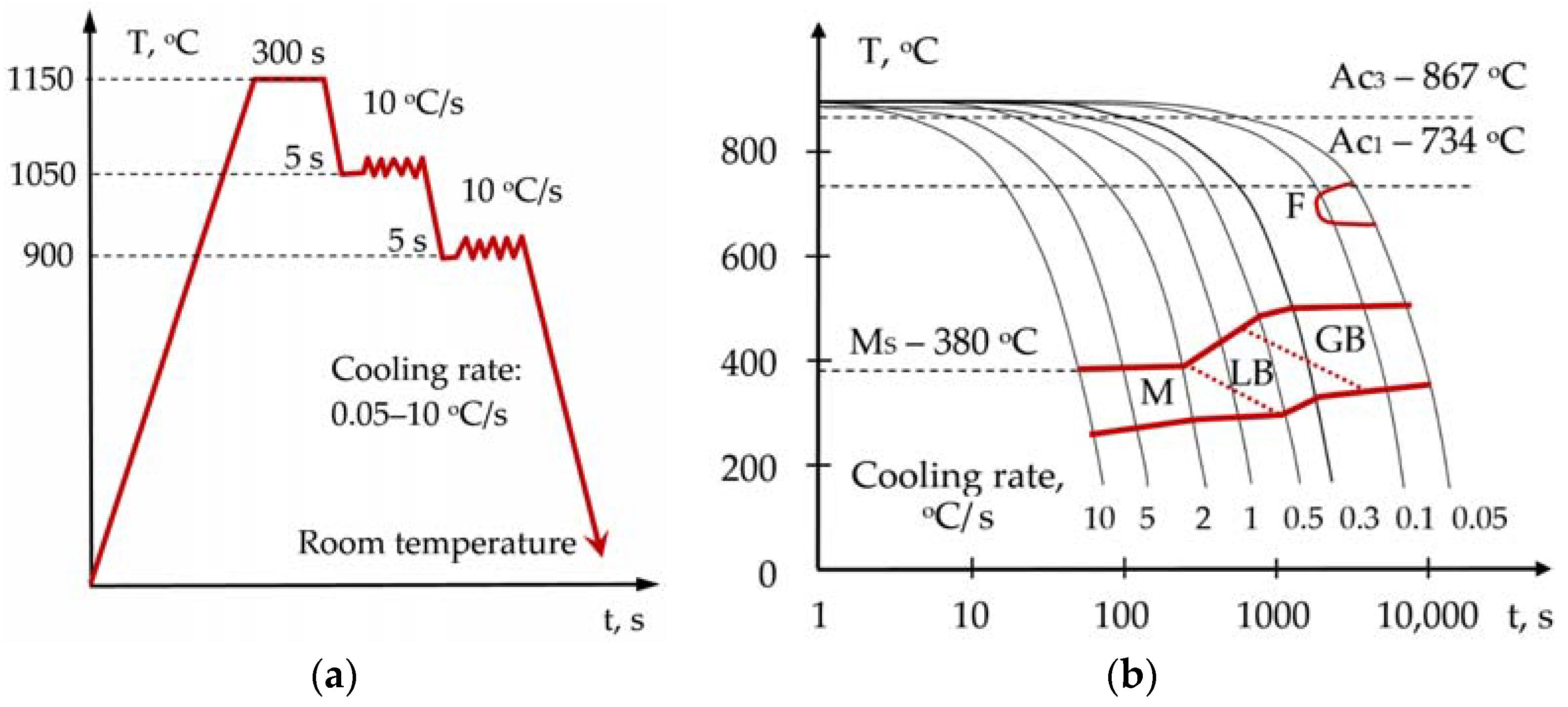
Reducing the ductile-brittle transition temperature to minus 120 °C and improving the weldability of high-strength cold-resistant steels can be achieved through developing methods of the microstructural design of rolled products and welded joints based on X-ray microstructure tomography [
91,
92]. An important field is the physical thermomechanical modeling to build CCT diagrams to determine the optimal concentrations of alloying elements, Cr, Nb, V, Ni, Ti, Mo, and N [
93]. An increase in the viscoplasticity of steels is also achieved through the refinement of the primary austenitic grain, through the control of solubility and distribution of dispersion carbides, nitrides, and complex carbonitrides, VC, VN, TiC, TiN, Nb (C, N), and NbTi (C, N) [
94,
95].
Optimization of the TMCP of austenitic cold-resistant stainless steels, application of cold rolling, and annealing for grain refinement have great potential for the improvement of viscoplasticity. The application of the TMCP reversion method for AISI 304L ultradisperse steel [
96,
97,
98] allowed achieving a tensile strength of 1010–1640 MPa with an elongation of 40–59% and excellent weldability during laser welding. High mechanical properties have products made of AISI 316L cryogenic steel from SLM (Selective Laser Melting) technology. The tensile strength of SLM samples at a temperature of minus 196 °C reached 1355–1510 MPa with an elongation of 35–54% [
99].
The key importance for the development of AHSS is the study of hardening mechanisms, the determination of dependence between dispersive precipitations and microstructure refinement, studies of thermodynamics, solubility, and crystallography of precipitations [
100]. The future development of second- and third-generation ANSS is connected to the development of the mechanisms of nanometer hardening, synergistic effect of dispersion precipitations, and volume isotropy of rolled steel [
101]. The solution to these problems is possible through the application of neural network technologies in material science and metallurgy [
102,
103].
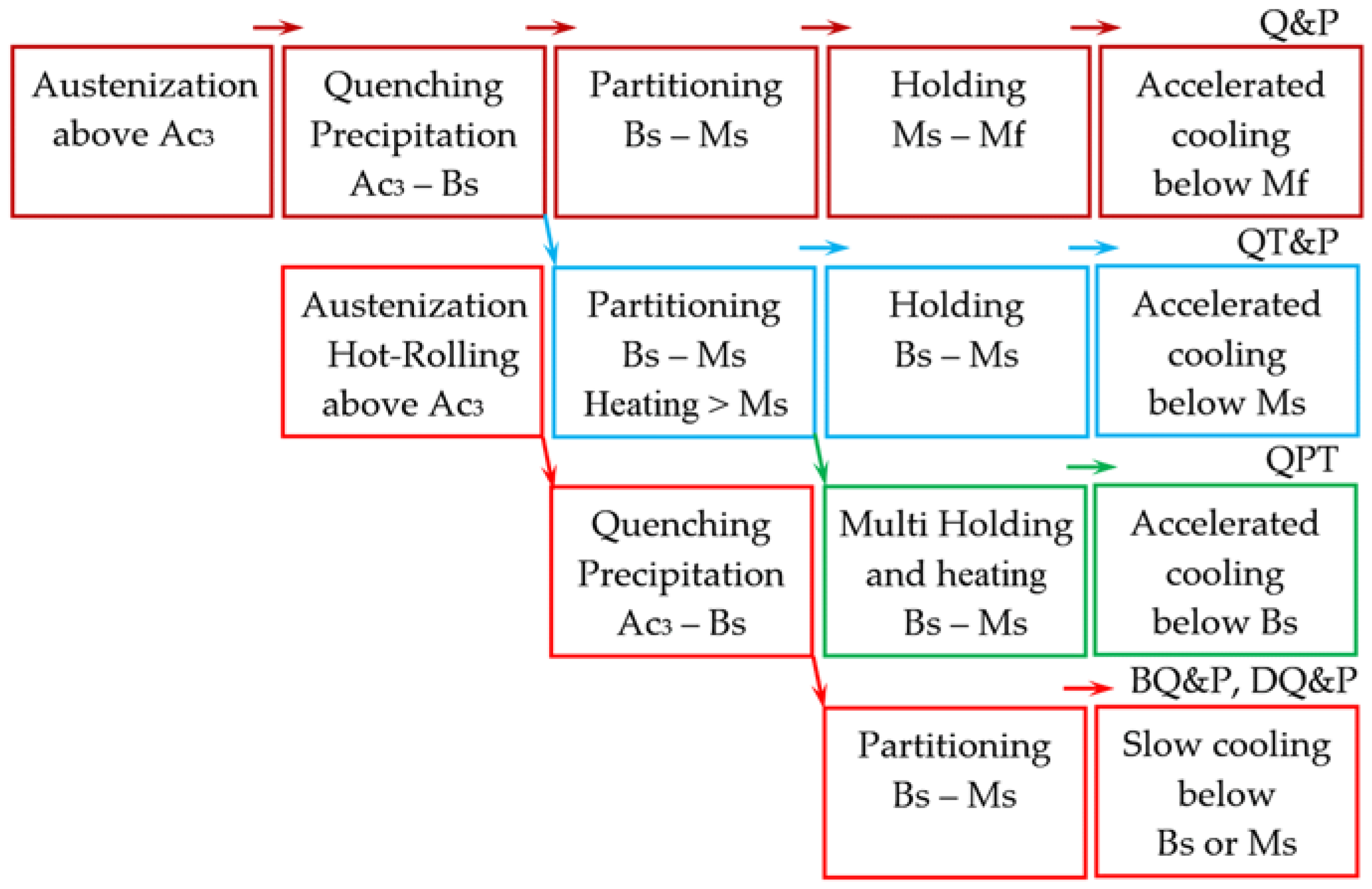
6. Weldability of High-Strength Steels
Laser welding of boron-containing ultra-high-strength steel Docol PHS CR 2000 leads to the formation of a softening layer in the heat-affected zone (HAZ) when heating in the temperature range Ac
1-Ac
3. The microstructure of this layer consists of tempered martensite, and tensile strength is 33% lower compared to that of the base metal [
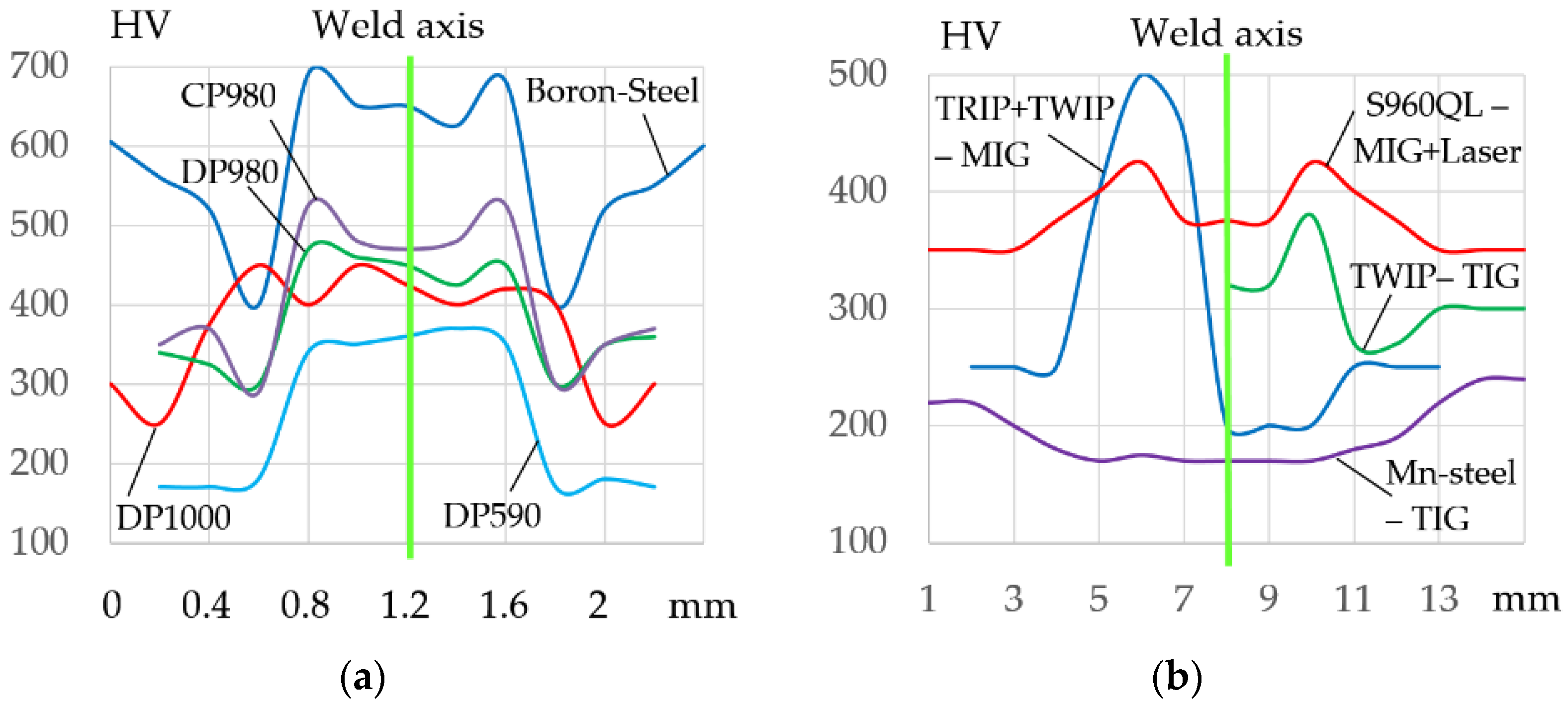
104]. In addition to the softening effect, a hardening effect may occur in the HAZ. During hybrid laser-arc welding of S960QL steel, the hardness in HAZ increases to 436 HV with 383 HV in the weld metal and 350 HV in the base metal [
105].
Kang et al. [
106] studied the MAG welding of DP590 GA, SAE 1008, HSLA HR 420, DP600 HR, Boron, Boron-HSLA, DQSK, TRIP780 GI, and DP780 GI steels and dissimilar joints. Typical weld fractures during cyclic tests occur along the weld metal, in HAZ, and in areas of high stress concentration. Steel thickness, stress concentrators in the structure, and the shape of the welds have a significant influence on the fracture of samples during cyclic tests. The main problem in the welding of DP steels is also the hardening and softening effect in the heat-affected zone. During laser welding of 0.15C-1.7Mn-steel, softening in the low-temperature HAZ (LT-HAZ) reached 20% due to martensite tempering. In the high-temperature HAZ (HT-HAZ), strength increased up to 28% due to martensite formation compared with base metal [
107]. Formation of a martensitic layer near the weld metal and softening results in fractures under static, impact, and cyclic dynamic loads. As the tensile strength of DP steel increases up to 1000 MPa, the softening increases up to 25–78% [
108,
109,
110]. During laser welding of DP 980, the softening layer strength decreases by 41%, which is the main cause of fractures under cyclic and vibration loads [
111].
To control the welding thermal cycle and microstructure of DP 590 steel, the change in laser pulse frequency can be used [
112]. Twin-spot laser welding is used to reduce the cooling rate and hardening when welding DP, CP, and TRIP steels [
113,
114]. Changing the ratio of power of two laser beams allows one to optimally distribute the heat input, reducing the width of HAZ, the volume of martensite, and the weld hardness by 10%. These effects are important for improving the isotropy of welded joints. Martin-Root [
115] investigated the laser welding of CP 980 steel. The hardness in the HT-HAZ increased from 460 to 520 HV.
Laser, arc, and resistance welding are also promising for welding TWIP steels [
116,
117,
118,
119]. The problem of Mn-TWIP steels is also high sensitivity to the welding thermal cycle. This results in weld metal softening of up to 20–40%, due to microstructural phase transformations [
120,
121], and a softening of up to 35% in HAZ [
122]. Ma et al. [
122] performed the TIG and laser welding of 25 Mn-3 Al-3 Si steel. The weld hardness during TIG welding increased to 360 HV and during laser welding to 420 HV with the base metal hardness of 320 HV. The HAZ hardness during TIG welding decreased to 230 HV and during laser welding to 290 HV. Elongation when testing samples for base metal, laser, and TIG welding was 95%, 67%, and 48%, respectively. The fracture of samples after laser welding during cyclic tests occurred along the base metal, and after TIG welding, fractures occur in red along the weld metal.
Kalacska et al. [
123] analyzed the properties of dissimilar joints made of TRIP 800 and TWIP 1000 steels after MIG welding with AWS 307 filler wire. On the TRIP 800 side, the hardness in HAZ increased from 250 HV to 500 HV. On the TWIP 1000 side, the hardness in HAZ decreased from 250 HV to 220 HV. Weldability analysis confirms the serious problem of hardening and softening in the weld and in HAZ when welding the second-generation AHSS, as shown in
Figure 13.
Excellent mechanical properties are achieved during hybrid laser-MAG welding of AHSS as well as when welding dissimilar joints of AHSS with AISI 316 steel [
124]. Similar problems with softening in HAZ occur when welding third-generation steels [
125]. Laser, resistance welding, and friction-stir welding are used to weld Mn-TRIP steels [
126,
127,
128]. To develop welding technologies for AHSS, the finite element method is used, which allows predicting the fusion zone, HAZ, the distribution of temperature fields, and mechanical stresses and strains during the welding thermal cycle.
7. Improvement of Viscoplasticity of Welded Joints
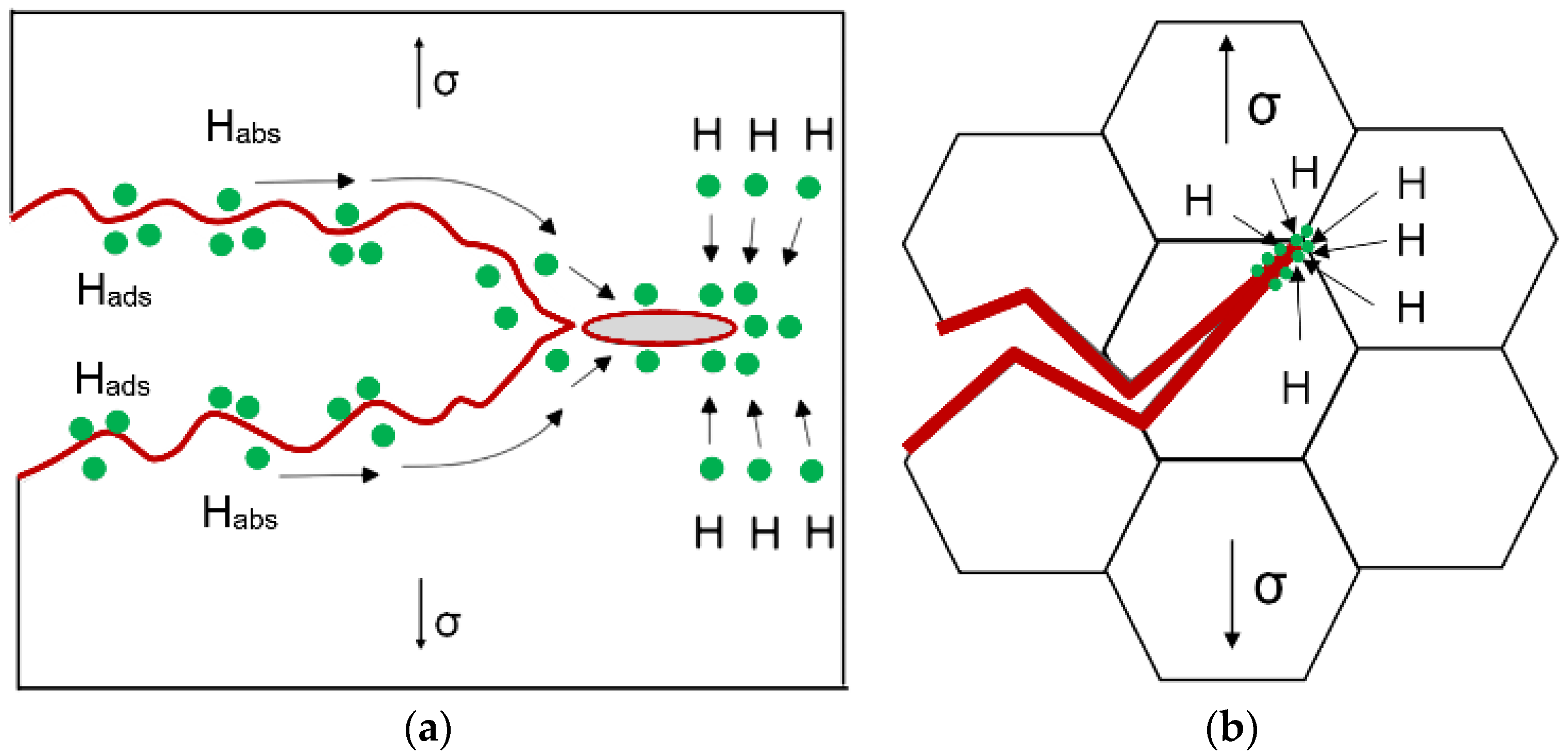
Welded structures are operated at low temperatures often in hydrogen-containing and corrosive environments. Weldability of steels is complicated by grain growth, hydrogen embrittlement, occurrence of HAC, and decreased impact toughness at low temperatures [
129]. The main methods of welding high-strength steels are MIG and MAG welding with solid wire. These methods ensure a minimum content of diffusible hydrogen in the weld metal [
130]. Application of hybrid laser-arc welding of HSLA steels with a thickness of over 40 mm also allows obtaining high impact toughness at a temperature of minus 50 °C [
131]. A hybrid laser-arc welding of HT780 steel with MGS-80 wire allowed the achievement of the impact energy of 100–150 J at minus 40 °C [
132]. Hybrid laser and plasma welding increases the productivity, weld quality, and corrosion resistance of duplex steel welds [
133]. A heat treatment after laser welding of 0.3 C-1 Cr-1 Si steel [
134] and controlling intermetallic formation during laser welding of NiTi/316L improves the viscoplasticity of welded joints [
135].
The microstructural state of the weld and HAZ is the most important factor in the mechanism of hydrogen embrittlement, HAC occurrence, and reduction of impact toughness [
136]. A fine-grain microstructure in welding is achieved through an optimal thermal cycle of heating and cooling [
137], through reduction of impurities and segregation at grain boundaries [
138].
Another effective metallurgical solution is microstructure refinement through the mechanism of heterogeneous nucleation of nonmetallic inclusions and acicular ferrite by forming refractory oxides, sulfides, and nitrides in a weld pool [
139]. An effective method of microstructure refinement and removal of impurities is the application of welding consumables with rare-earth elements [
140].
Refractory REM oxides and sulfides, such as REM
2O
3 and REM
2S
3, are nonmetallic inclusions of spherical shapes, similar to titanium oxides TiO, TiO
2, and Ti
2O
3 [
141]. REM oxides and sulfides have high melting temperatures, °C: La
2O
3 (2304); Y
2O
3 (2439); Ce
2O
3 (2250); ThO
2 (3350); La
2S
3 (2110); Y
2S
3 (1925); Ce
2S
3 (2450); Th
2S
3 (2300). Therefore, the formation of these compounds in the weld pool increases the number of crystallization nuclei. This process contributes to the refinement of microstructure as well as to the nucleation of acicular ferrite [
142]. Nonmetallic spherical inclusions of TiO, TiO
2, and Ti
2O
3 are also effective centers of acicular ferrite nucleation [
143]. However, REMs are more active in the reactions of desulfurization and deoxidation of the weld pool [
144].
REM borides play an important role in the mechanism of hardening and refinement of the microstructure; they contribute to the formation of refractory boron carbides and the nitrides B
4C and BN [
145]. The alloying of boron has a positive effect on the properties and microstructure of cold-resistant and cryogenic austenitic steel with a low residual nitrogen content of up to 0.04% and boron of up to 0.004%. This effect is associated with the solubility of boron in γ-Fe and α-Fe, reduction of impurities of sulfur S and phosphorus P, and the formation of borides Me
2B, Me
5B
3, Me
3(C, B), Me
23(C, B)
6, and FeMo
2B
4 [
146].
The formation of free fluorine when introducing fluorides of Ca, Mg, K, and Li into welding consumables is of great importance in welding metallurgy. Fluorine reduces the content of diffusible hydrogen in weld metal due to the formation of the HF compound [
147]. High effectiveness of fluorine and fluorides in hydrogen removal is due to the reactions of fluorine and fluorides with the H atom, H
2 molecule, and H
2O water vapor in the liquid slag and in the plasma of the welding arc [
148]. The addition of LaF
3, LaB
6, and SF
6 lead to the improvement of the thermophysical properties of plasma and the quality of arc additive manufacturing products, using G3Si1, 316L, AlMg5Mn1Ti, and CuCr
0.7 wires [
149]. The addition of activating fluxes of Li
3AlF
6-TiO
2-SiO
2 and CaF
2-BaF
2-SrF
2 in the laser welding of steel leads to an increase in the penetration depth up to 33–46% and in productivity up to 45% [
150].
In steels and welds, there are the harmful oxides, FeO, SiO
2, and MnO, the sulfides, FeS and MnS, as well as diffusible elements, S, H, and N, which reduce the impact toughness, plasticity of welds, and increase sensitivity to the formation of HAC. The alloying of REM and boron reduces the concentration of the detrimental impurities, S, O, and N. As a result of the interaction, nanodispersed refractory REM oxides, sulfides, and boron nitrides BN are formed, which are the nuclei of primary crystallization. The increase in the number of refractory crystallization nuclei leads to the refinement of large crystallites, dendrites, and microstructures of primary austenitic grains. Parshin et al. [
151] proposed the models of dendritic crystallization at the boundary of the “weld pool—base metal” transition without limitation of crystallite growth and with limitation of crystallite growth in the formation of additional crystallization nuclei based on REM oxides and sulfides.
A similar mechanism of dendritic crystallization with crystallization nuclei made of oxides and sulfides is considered in works [
152,
153,
154,
155]. The alloying of boron into the TWIP steel reduced the size of the austenitic grain from 770 to 344 μm in the columnar and equiaxial zones of the primary crystallites [
48]. The formation of nonmetallic spherical inclusions reduces the sensitivity of steels to crack initiation and propagation. After primary crystallization of HSLA steels, secondary recrystallization with pearlitic and bainitic transformation occurs. At that, inside the austenitic grains on the surface of refractory nonmetallic inclusions, intragranular acicular ferrite can nucleate, which inhibits the cracks propagation and increases its trajectory. Parshin et al. [
151] proposed the models of crack propagation trajectory in the prior-austenitic grain without acicular ferrite and with acicular ferrite nucleated on a spherical nonmetallic inclusion.
Thus, oxides and sulfides of rare-earth metals and boron nitride contribute to the refinement of primary austenitic microstructures and the nucleation of acicular ferrite in high-strength steels [
156,
157,
158]. Increasing the volume of acicular ferrite and refinement of weld microstructure is a requirement for increasing impact toughness, reducing the critical temperature of ductile-brittle transition, and reducing sensitivity to the occurrence of HAC. Similar solutions are relevant for underwater wet welding of high-strength steels, as offshore welded structures and pipelines are often located in the Arctic Ocean [
146,
159,
160].
8. Conclusions
1. The analysis of research in the field of metallurgy and weldability shows the significant potential of advanced steels for low temperature and cryogenic application. For cold-resistant welded structures HSLA, bainitic, duplex, 1.5–5% Ni steels, and all AHSS steels are recommended. For cryogenic welded structures austenitic Cr-Ni-N-, Mn-, 9% Ni-, Mn-TRIP, and TWIP steels are recommended. For the high quality of welded structures it is necessary to improve weldability and reduce the sensitivity of HSS and AHSS steels of the welding thermal cycle to low temperature and hydrogen embrittlement. The future of the welding of cold-resistant and cryogenic steels lies in microstructure control, reducing the anisotropy of the welded joint-based low heat input in the arc, laser, and hybrid welding with filler REM-containing wires.
2. As for the evolution of TMCP, metallurgy, and welding of high-strength steels, it is necessary for the application of physical and thermodynamic modeling to achieve synergistic effects in the microstructural design. For the manufacturing of isotropic rolled steel, it is necessary to achieve the synergistic effect of the first level considering the microstructure, TMCP parameters, and physical and mechanical properties. For the manufacturing of welded structures, it is necessary to achieve the synergistic effect of the second level considering the structural design, welding metallurgy, microstructure, and welding thermal cycle.
3. Metallurgical processing of high-strength steels for welded structures for cold-resistant and cryogenic service should be based on a special microstructural design. Metallurgy and microstructural design of steels should take into consideration the influence of welding thermal cycles and stresses and repetitive phase transformations in the weld metal and in the heat-affected zone. The distribution and morphology of microstructural phases and nonmetallic inclusions in the matrix are of key factors to achieve high impact toughness and perfect nanometer isotropy of rolled products. Neural network control of TMCP parameters, melting technologies, refining, O2, H2 removal, micro-alloying, development of metallurgical fluxes, and slags with rare-earth elements have great potential in the field of fundamental metallurgy.
4. In the field of material science and metallurgy of welding, the fracture micromechanics, models of diffusible hydrogen embrittlement, crystallization kinetics, phase transformations, mechanisms of hardening, and formation of nonmetallic inclusions have great potential. The development of welding consumables with REM, neural network control of the welding thermal cycle, reduction of the softening of welds, increase of the isotropy of welded joints, and monitoring of stresses are very important. Bionic design, weight reduction, application of self-heating, and self-diagnosing are necessary in the design of welded structures for low temperature and cryogenic service.