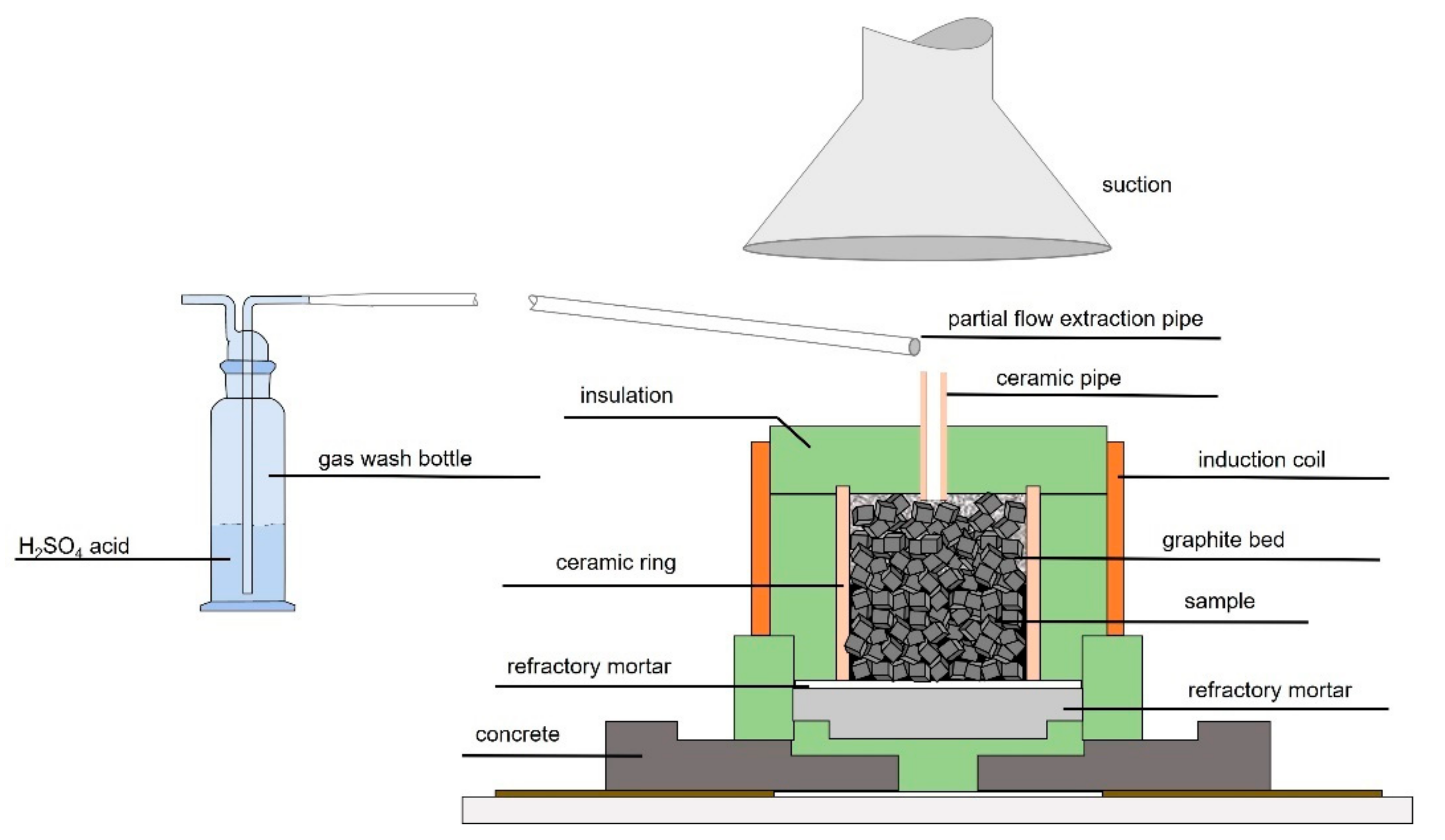
3.1. Simultaneous Thermal Analyses
So far, there are numerous studies that deal with the thermal stability of LIB cathode materials [
35,
36,
37,
38,
39,
40]. Since these are often only concerned with safety-related or performance-relevant issues, the analyses are usually carried out in temperature ranges below 300 °C. Pyrometallurgical recycling approaches on the other hand operate at temperatures of well above 1000 °C and cannot be adequately described by observed reactions and phenomena at lower temperatures [
41,
42,
43,
44,
45].
By looking at thermokinetic studies and publications that conducted investigations at sufficiently high temperatures, it can be concluded that lithium metal oxides basically tend to thermally collapse according to Equation (1) [
46,
47,
48]:
However, if a reductant, e.g., coke, is added, the reaction temperatures can be shifted towards lower temperatures and coupling reactions such as that in Equation (2) can occur. To provide an example for one certain lithium metal oxide, reference is made to Kwon and Sohn [
47], who present a detailed thermokinetic study on the behavior of LCO. The decomposition of lithium carbonate (Li
2CO
3), shown in Equation (3), is known to take place in two steps. First, from solid Li
2CO
3, which is expected to play a minor role, and the majority after Li
2CO
3 is melted [
28,
46,
49].
The further reaction of the metal oxide clearly depends on the type of cathode material that is examined. In general, we expect that the reduction of metal oxide can be direct or indirect, as shown in Equation (4) and Equation (5), respectively [
28]:
In case of NMC, i.e., LiNi
xMn
yCo
zO
2, we need to consider Ni, Co, and Mn, which have different oxygen potentials and call for different extents of reducing conditions. The reduction reaction of lithium oxide (Li
2O), formed during decomposition of Li
2CO
3, must also be considered. Thermodynamically, this reduction calls for higher temperatures and lower oxygen partial pressure values than for the other metals. If only thermodynamic considerations are made, we can expect nickel oxide and cobalt oxide to be reduced first. Manganese oxide, which can have various oxidation states and is reduced in several steps, is harder to predict. If Li
2O, which thermodynamically has the greatest requirements for its reduction, can be further reduced according to Equation (6), it should be possible to remove Li as a part of the gas phase, since the evaporation temperature of Li is only 1330 °C. This depends on whether sufficient reducing conditions can be reached or not.
Said reducing conditions, namely high temperatures, low oxygen partial pressures (pO
2), and a high carbon monoxide (CO) to carbon dioxide (CO
2) ratio, must be provided by the InduMelt reactor. In this regard, the high temperatures of approximately 1550 °C and the excessive supply of carbon, see the Boudouard reaction in Equation (7), should be sufficient to maintain low pO
2 and high CO/CO
2 values [
50].
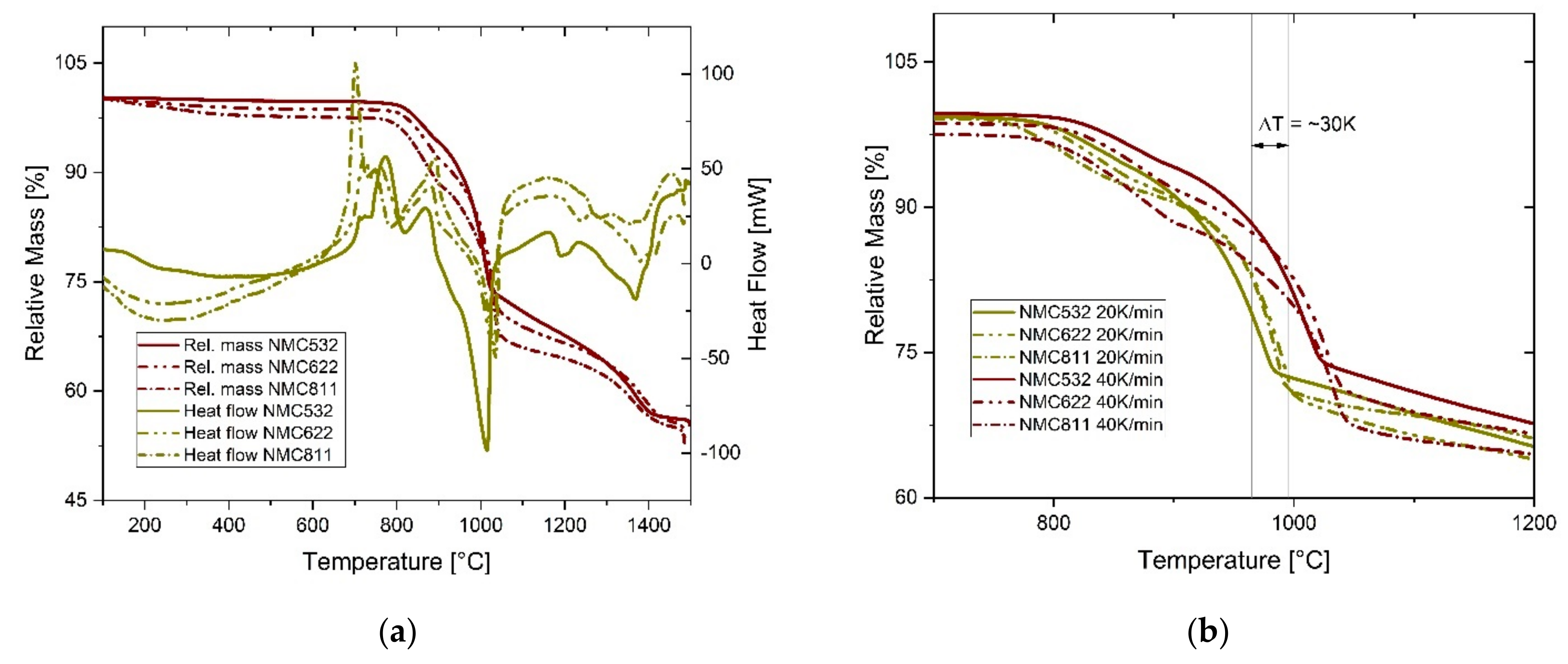
However, from the statements above, we can make some preliminary assumptions on how the NMC variations with coke addition may react. Considering these, as well as the results from the STA, which are summarized in
Figure 2, certain observations during the InduMelt experiments can be discussed and probably linked to certain reactions.
At first sight of
Figure 2a, it is worth mentioning that NMC532, NMC622, and NMC811 show a very similar behavior and share the same pattern. This indicates that the reaction scheme remains the same for all variants despite the significantly shifted ratios of Ni, Mn, and Co. As can be seen in
Figure 2a, the first significant observation can be made at approximately 700 °C, where an exothermic peak without a corresponding change of the sample mass can be seen. The first main weight loss occurs between 800 and 1000 °C and is accompanied by a significant endothermic peak of the heat flow. In that phase, the sample mass drops by about 30%, which is most likely caused by the reduction reactions of the metal oxides, see Equations (4) and (5). In this regard, attention should be paid to the fact that at this point, the relative mass decrease of NMC532 is noticeably lower than for NMC622 and NMC811. It seems that the higher the main mass decrease is, the higher the relative content of Ni is. After the steepest slope, a transition phase with relative constant mass decrease develops before another, more intense phase between 1300 and 1400 °C occurs. Thereafter, the curve of the relative mass seems to flatten out. At the end of the analysis, at 1500 °C, the overall mass decrease summed up to 45%, 46%, and 47% for NMC532, NMC622, and NMC811, respectively. The overall weight loss is 5–7% less than the sum of all potential volatile components (n
v), which can be found in
Table 1. This might be caused by insufficiently reducing conditions in the used laboratory analysis equipment that led to residual metal oxides, most likely containing Li or Mn.
From
Figure 2b, it can be seen that there is a slight shift of reaction temperatures with varying heating rates of 20 and 40 K·min
−1. For the interpretation of the InduMelt trials, where the heating rate is limited to less than 3.5 K·min
−1, this effect needs to be considered.
3.2. InduMelt Experiments
In the InduMelt experiments, the cathode materials with 20 wt.% coke addition were heated to approximately 1500 °C under highly reducing conditions. As mentioned in
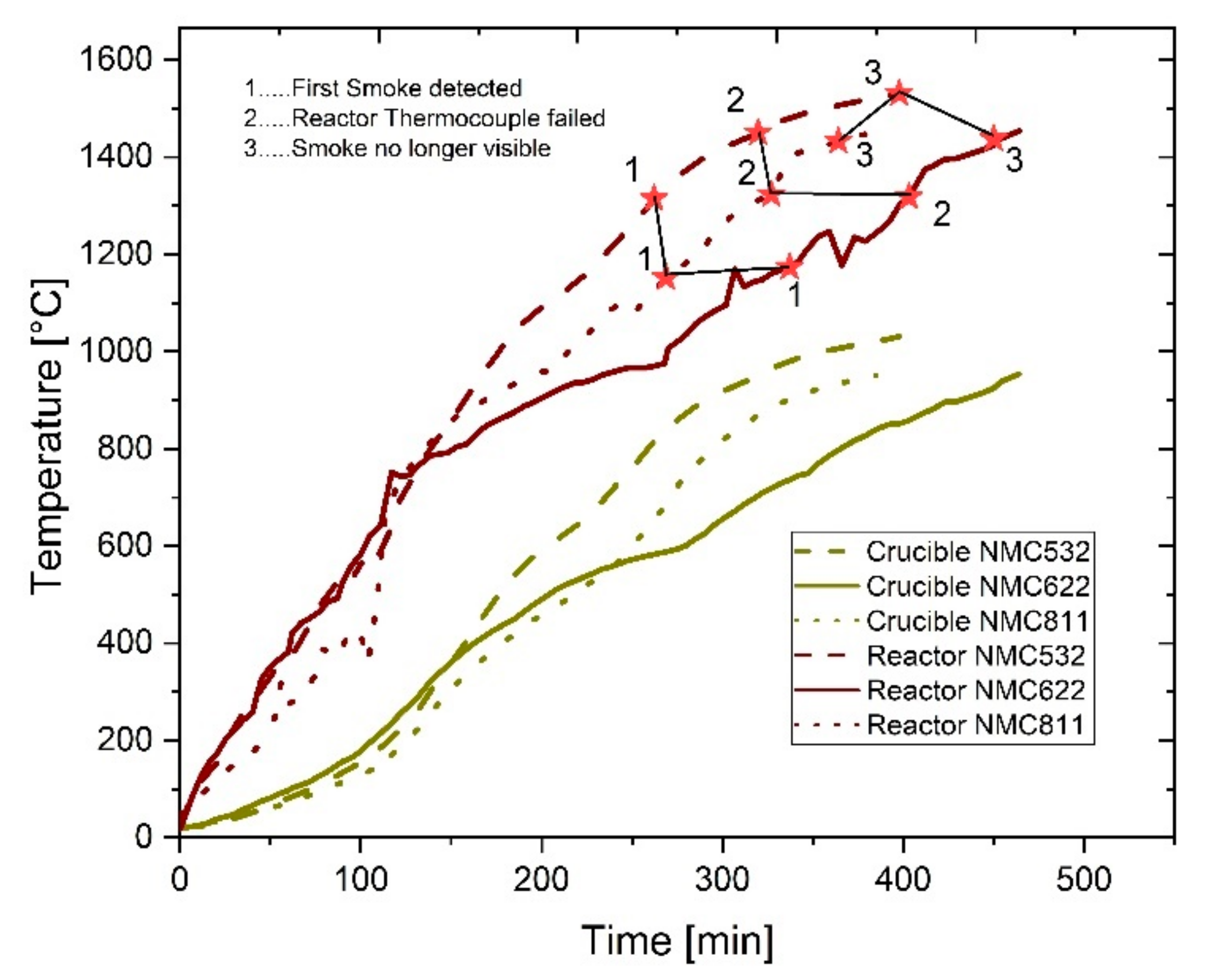
Section 2, striking observations that could have been made during heating were noted for later discussion. The most important of these observations as well as the trend of the temperatures inside and outside of the reactor can be seen in
Figure 3.
The first observation (1) that is highlighted in
Figure 3 was the detection of visible flue gas leaving the reactor. The appearance of the flue gas was white to greyish, which might be caused by small particles. From other reduction experiments in the InduMelt, it is known that generated CO never resulted in a visible flue gas. Therefore, this observation indicates the removal of other gaseous reaction products or fine particles. However, the discussion on that will be continued in
Section 3.3, where the gas phase is examined. For now, we wish to state that the phase of visible flue gas started at temperatures between 1100 and 1300 °C and ended between 1400 and 1500 °C, i.e., point (3) in
Figure 3. By comparing this temperature range with the results of the STA, it can be seen that the detected flue gas corresponds to either the constant or the second steeper mass decrease between 1100 and 1400 °C rather than to the main change of the sample mass starting at 800 °C. Combined with the knowledge of the reaction kinetics and the high-temperature behavior of NMC cathode materials, we can assume that the observed flue gas might be related to reactions including Mn and Li.
To clarify this and to examine the original research question, namely the investigation of possible recovery rates for Ni, Co, Mn, and Li with the desired reactor concept, hereinafter, a detailed look at the obtained products is provided. In general, it was possible to distinguish between the following product phases after each of the experiments.
Table 2 provides an overview on the masses of the charged input material and all obtained products that remained in the reactor.
Metal phase, which occurs as small metal spheres on the graphite cubes and bigger accumulations at the bottom of the reactor.
Slag phase, which can be found as thin, layered depositions on the graphite cubes.
Powder phase, which is left after sieving and magnetic separation.
Gas phase, which is not fully collected but is defined by the composition of the solution in the gas scrubber.
The metal phase is the largest contributor to the products in all conducted experiments and accounts for 77% to 80% of the found products. The amount of slag that was found differs noticeably more. In NMC811, only 3.3 g of slag was found, whereas in NMC532, the slag phase accumulated to 24 g. Thus, the slag phase contributes between 1.3% and 9.7% to the product sum. The mass of the obtained powder is again more uniform and accounts for 10% to 17% of the products. All in all, the mass loss that was observed in the experiments was 51.1%, 47.7%, and 49.9% for NMC532, NMC622, and NMC811 respectively, and therefore up to 6% higher than in the STA.
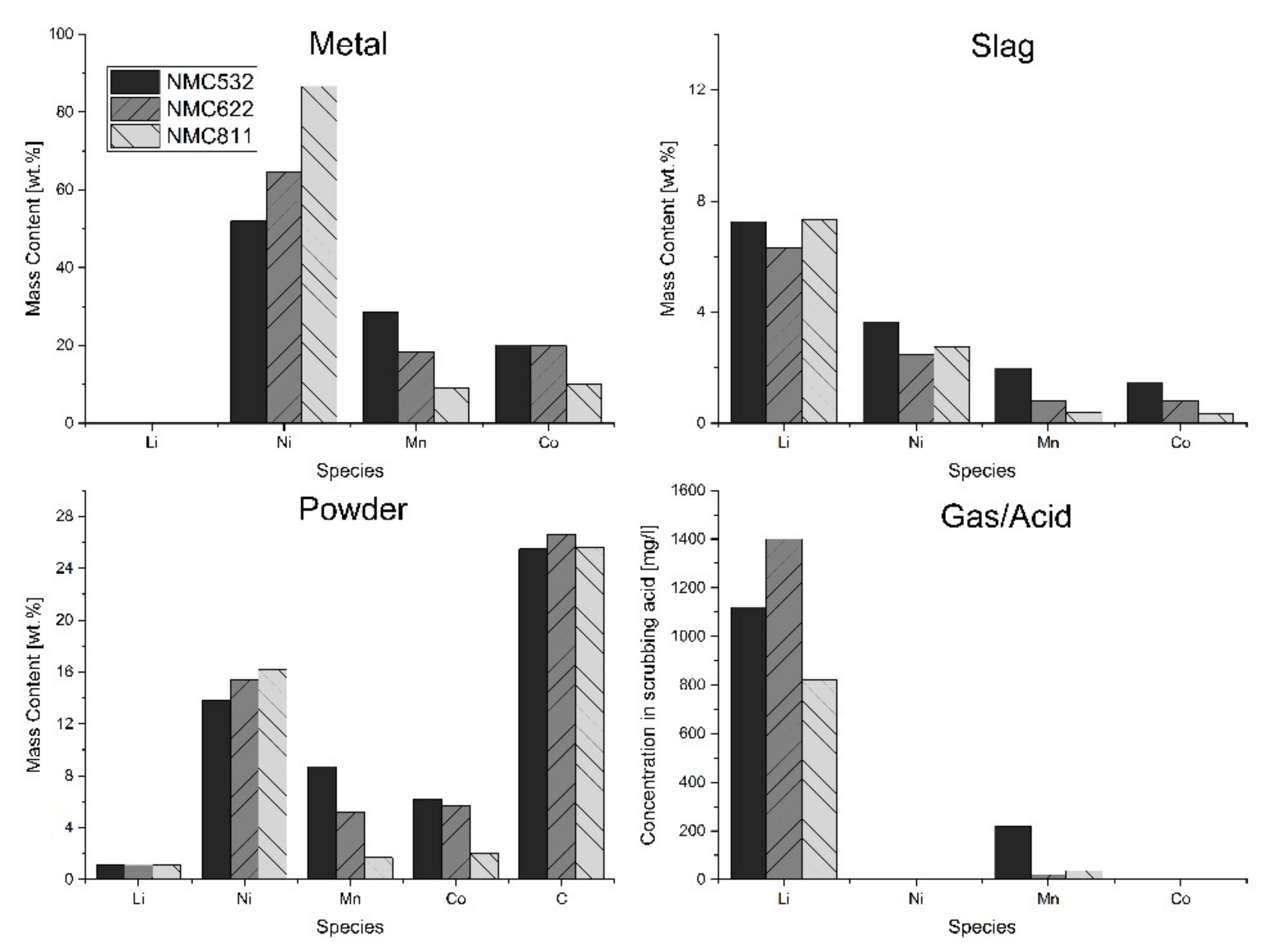
The detailed examination of the products starts with the metal phase, the chemical composition of which can be found in
Table 3.
In terms of that, it can be stated that the chemical composition of the metal phase follows the expectations and consists mainly of Ni, Co, and Mn. For all trials, the ratio Ni:Mn:Co also correlates very well with the stochiometric ratio of the respective input material. Accordingly, only the proportion of Mn is somewhat lower than it actually should be, which might be caused by its more complex reaction behavior. Regarding the fourth metal of interest, Li, there is almost no accumulation in the metal phase, which is in line with our assumption that Li would rather be in a gaseous state instead of being a part of the metal alloy. In
Figure 4a, an image of the small metal spheres is shown. The majority of the produced metal was found in this form, but there were also some bigger accumulations at the reactor bottom. In the SEM image in
Figure 4b, two areas are highlighted, the darker of which (indicated with “Spektrum 27”) consists almost exclusively of carbon. In the brighter spectrum (indicated with “Spektrum 27”), the conducted elemental analysis revealed a similar result as the ICP-MS analysis of the NMC622 metal phase. The metals in that area consists of 60.6 wt.% Ni, 20.0 wt.% Mn, and 19.4 wt.% Co. Therefore, the SEM analysis has shown that C mainly occurs sporadically as a particle enclosed in the metal phase. The statement regarding chemical composition and accumulation of the metallic products as well as the description of their optical appearance also applies for the experimental series with NMC532 and NMC811.
Metals that are not fully reduced during the experiment, and thus are still metal oxides, are expected to form a slag. Since we aimed to investigate possible recovery rates for said metals, the examination of the slag phase, illustrated in
Figure 5a,b, can provide information on which of the metals still have potential for improvement.
Most of the slag that was found after the InduMelt experiments accumulated as a thin slag layer on the graphite cubes, as is shown in
Figure 5a. Some small slag depositions on the inner wall of the ceramic crucible were also found and taken into account for the mass balance. The results of the ICP-MS analysis of the obtained slag phase in each of the trials can be seen in
Table 4. Relating to the four metals of interest in NMC cathode materials, Li is the largest contributor to the produced slag phase and has a mass content of between 6.3% and 7.3%. Most likely, Li is found as Li
2O, which was formed during the thermal decomposition of Li
2CO
3 but not further reduced. However, other compounds cannot be completely ruled out either. For Ni, the mass content of which is laying between 2.5% and 3.7%, it is harder to assume in which form it is bound in the slag. It could be very fine metallic particles, unreduced nickel oxide, or even residual NMC, which for some reason has not reacted. The same accounts for Co and Mn, which contribute between 0.3% and 2% to the composition of the slag phase. However, analogous to the metal phase composition, the ratio of Ni, Co, and Mn is again approximately correlated to that in the respective original cathode material.
The third product that was found was the so-called powder phase, which is shown in
Figure 6a,b. The powder phase is expected to consist of fine particles of both the metal and the slag phase, as well as carbon from the graphite cubes caused by abrasion during the product examination.
This assumption is also supported by a glance at the chemical composition of the powder phase, presented in
Table 5. The largest contributor to the powder phase with a mass content of approximately 26 wt.% is carbon, which mainly results from the abrasion of the graphite cubes and leftover reducing agent, i.e., coke. Besides carbon, significant amounts of Ni, Mn, Co, and Li can also be found in the powder phase. The fact that these are remnants of the original NMC material can be ruled out based on the temperatures reached. An XRD analysis performed in parallel with the SEM also revealed that there was no detection of the original lithium metal oxide pattern anymore.
On closer inspection, the components apart from the C are probably mainly very small metal spheres, some of which are contaminated with slag and have therefore not been separated by the magnet. In contrast to the metal phase, which was obviously an alloy without slag inclusions, part of the manganese may still be present in the powder as an oxide, thus making magnetic separation more difficult and causing the issue. A clear identification, for example by further separation of metal and slag, is not feasible due to the small particle size. In the final determination of the recovery rates, however, the powder fraction is nevertheless considered.
The last product that needs to be discussed is the gas phase; thus, all gaseous reaction products and particles that left the reactor via the flue gas pipe. To visualize said product phase, a series of images is presented in
Figure 7. Here,
Figure 7a illustrates the already described observation (visible gas leaving the reactor) between 1100 and 1500 °C. In
Figure 7b, the significant optical change of the scrubbing medium can be seen. Besides the visible gas, this was the second indicator for gaseous products, other than CO, leaving the reactor. The third indicator of this event can be seen in
Figure 7c,d, where deposits inside the gas pipe are shown.
In order to obtain certainty about the cause of the above observations, both the deposits inside the gas tube as well as the gas scrubbing medium were analyzed. The results of these ICP-MS analyses are shown in
Table 6 and
Table 7.
If we concentrate again on the four metals of interest in the NMC cathode materials, it can be seen that Li accumulated by far the most in the gas phase-related products. The amount of Li that was found in the scrubber medium is umpteen times higher than that of Ni or Co, which show almost no accumulation. For NMC622 for example, the value of Li measured was 1400 mg L
−1, which is more than 2000 times the value of Ni and Co. In comparison to that, the value for Mn lays between 20 and 220 mg L
−1, thus also showing a significant enrichment. If we now have a closer look at
Table 7, we can see that this supports the first measurement. Additionally, for the deposition in the flue gas tube (see
Figure 7c,d), Li is the most prominent of the desired metals, followed by Mn. For Ni and Co, the value was below the determination limit.
Finally, the results of the product examination are summarized in
Figure 8, which will be used to discuss possible recovery rates.