A General Vision for Reduction of Energy Consumption and CO2 Emissions from the Steel Industry
Abstract
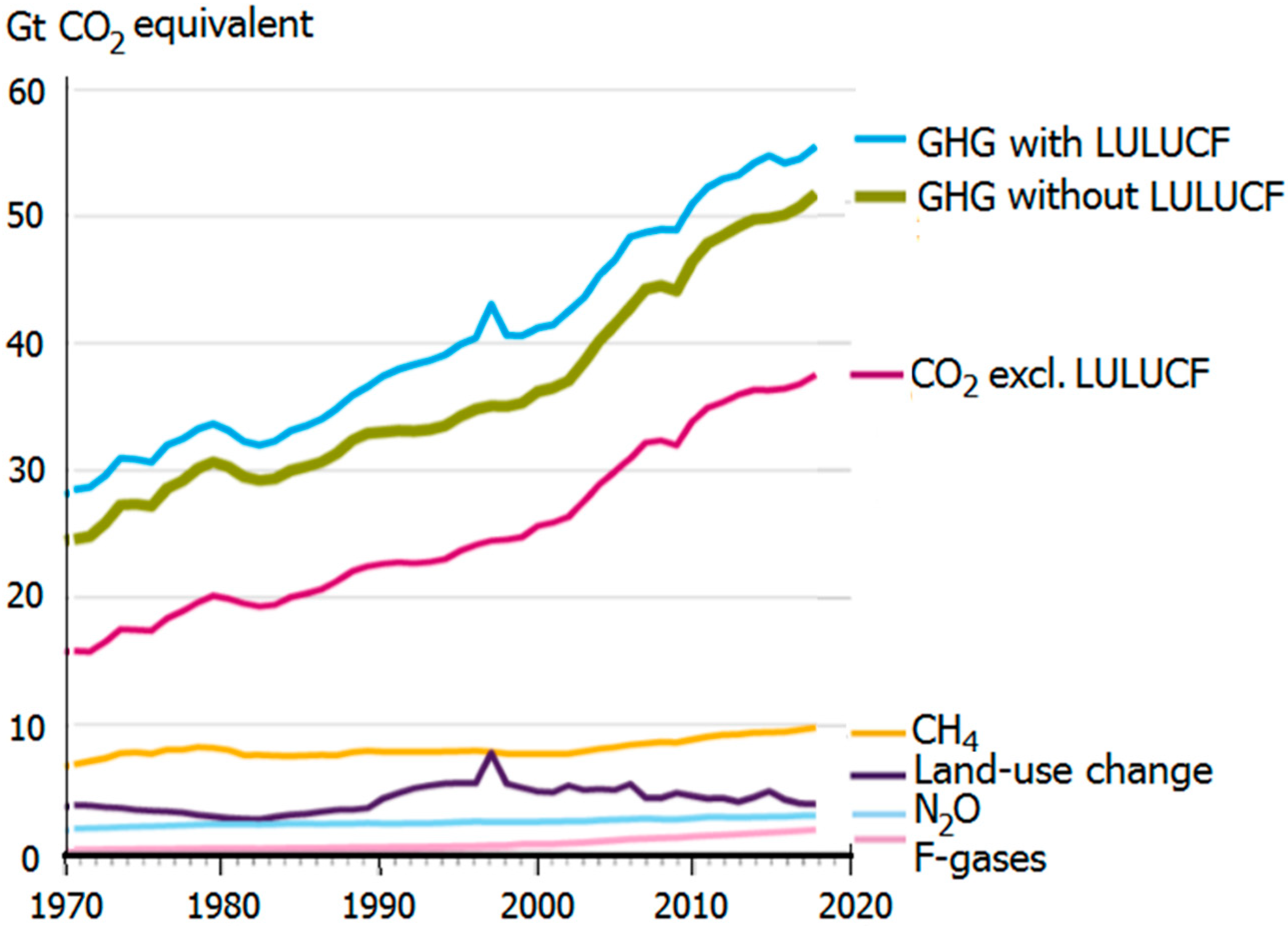
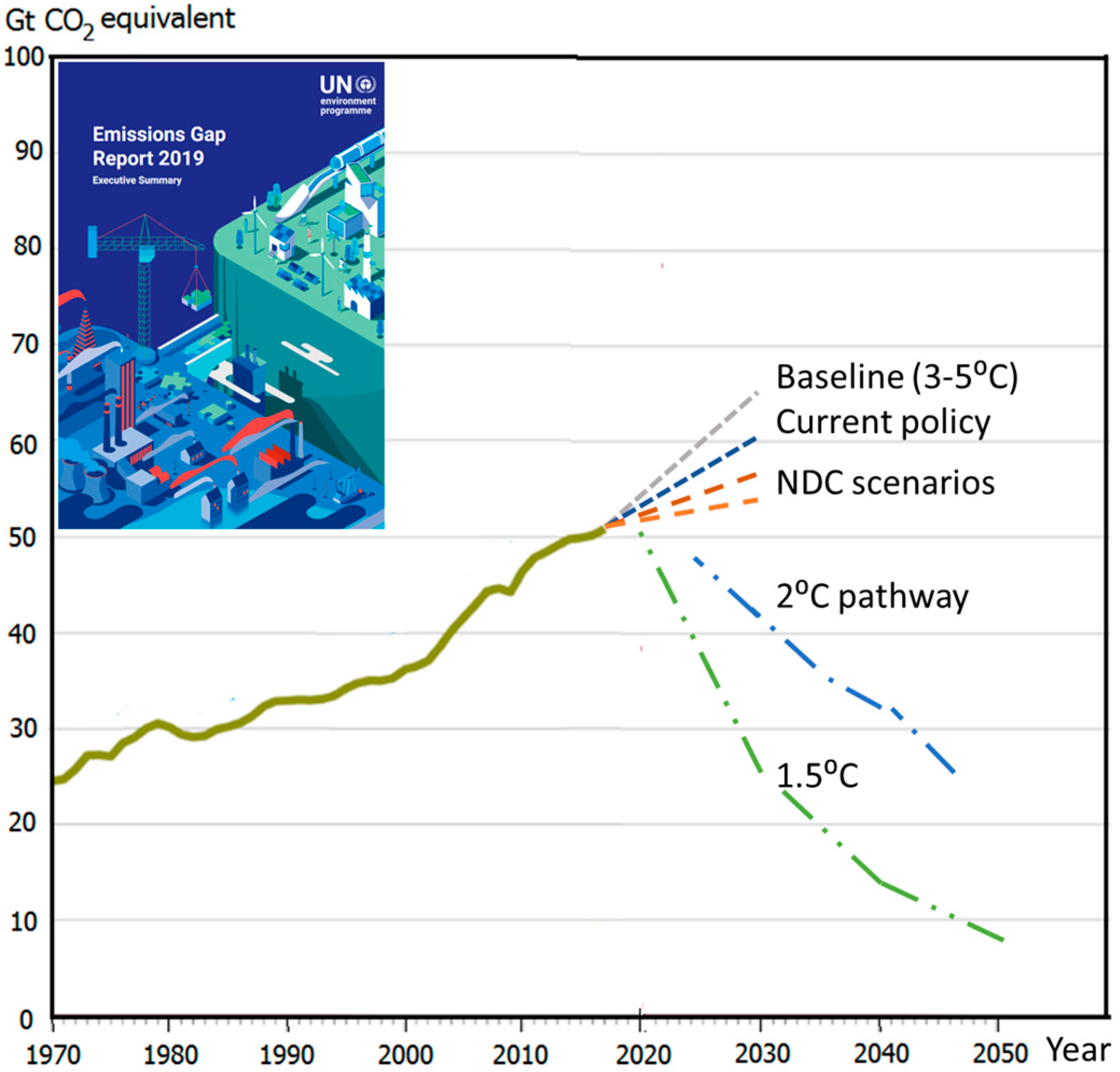
1. Global Challenge of Climate Warming and Its Rationale
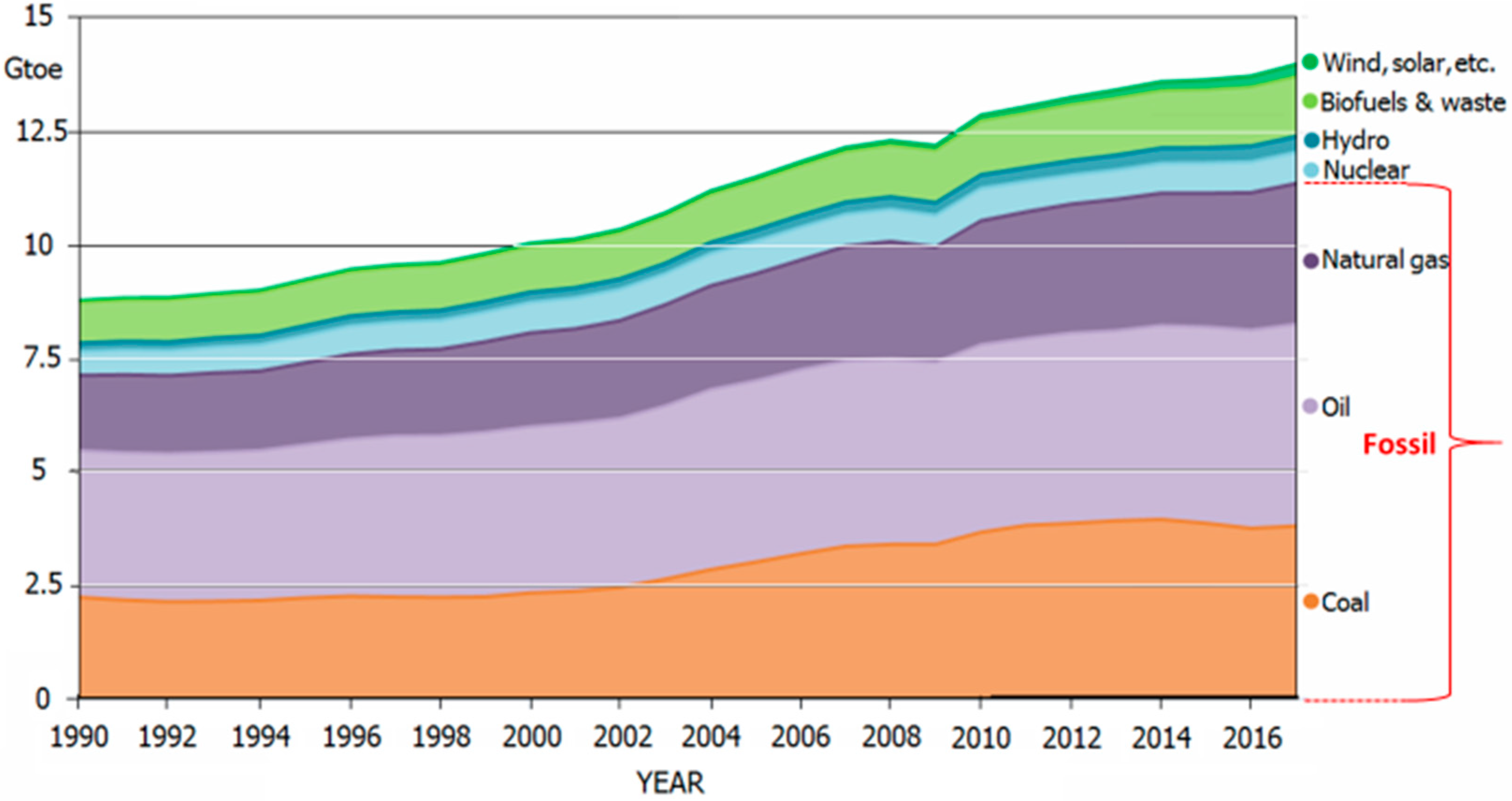
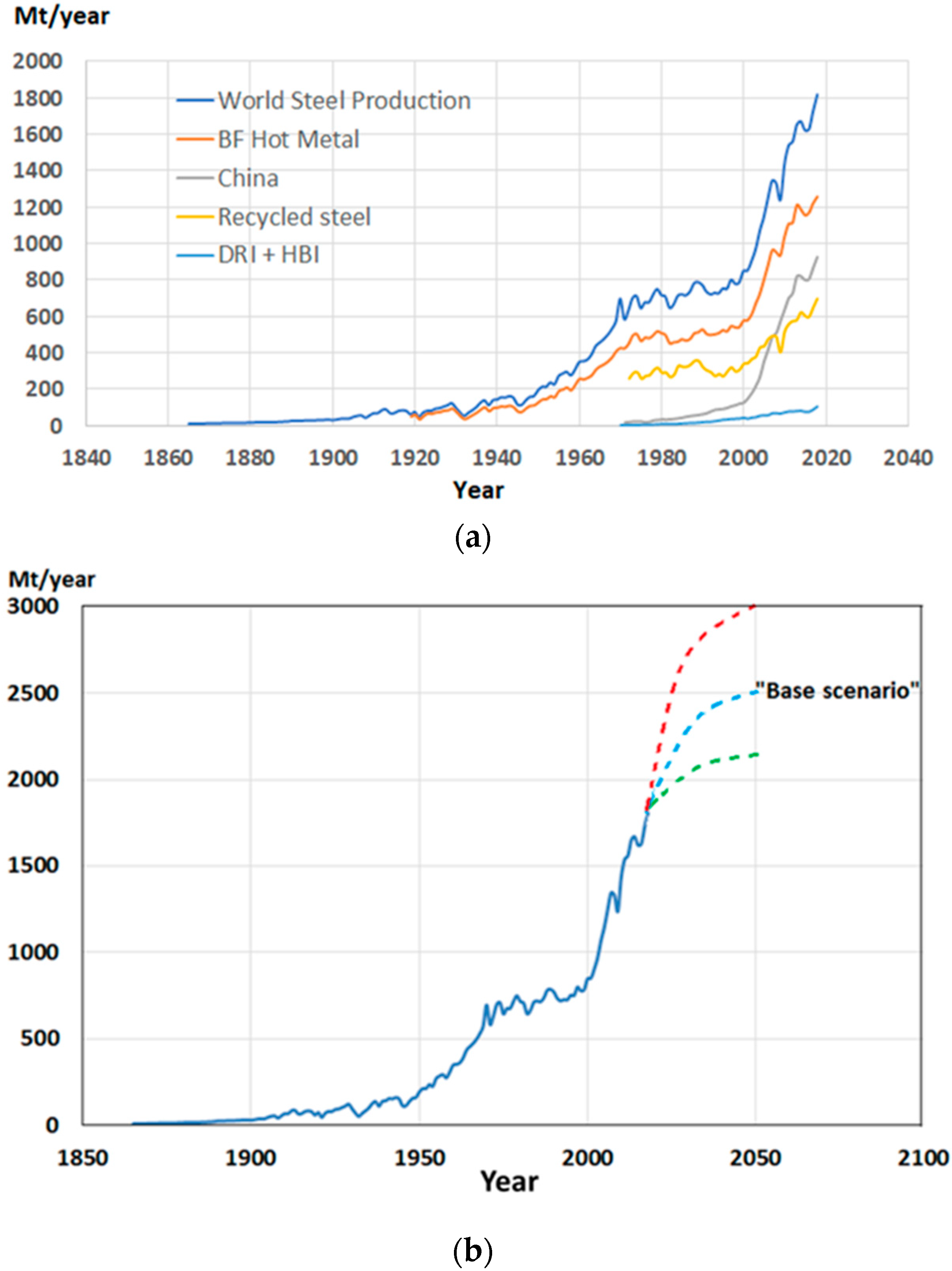
2. Progress of the Steel Industry and its Role in Energy Consumption and CO2 Emissions
3. Review of Means to Cut CO2 Emissions from the Steel Industry
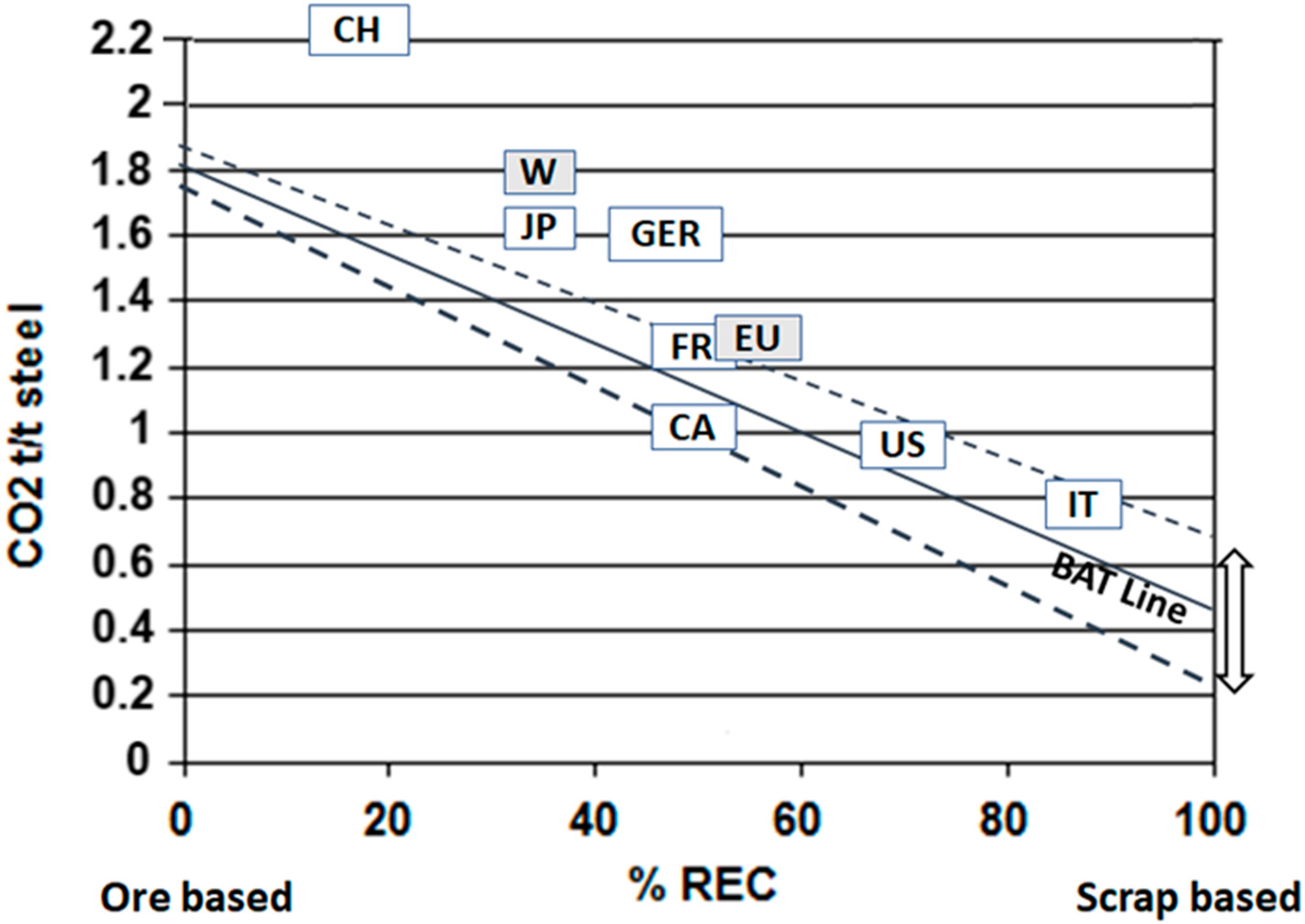
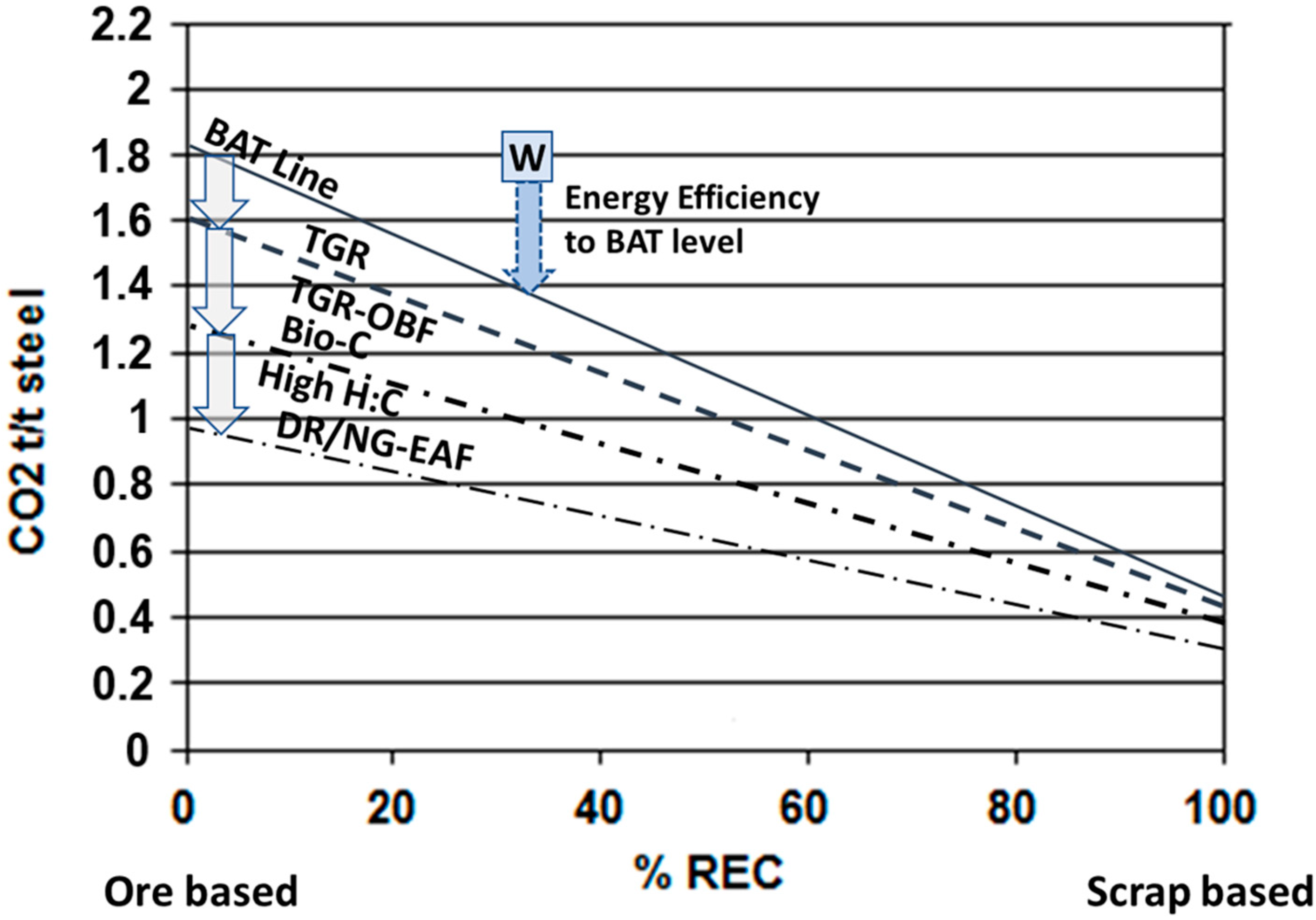
3.1. Improving Energy Efficiency
3.2. Potential Means to Mitigate CO2 Emissions in Ore-Based Production by Improving and Modifying Current Technology
- Transfer to coke dry quenching in coke making (CDQ)
- Production of high strength coke to better utilize hydrogen reduction in BF
- Top-pressure Recovery Turbine (TRT) technology in BFs and dry dedusting systems for BFG and BOFG (blast furnace and converter off-gases)
- Incorporation of oxygen enrichment technology in hot stoves
- Integrated optimized usage of off-gases in-plant, for electricity generation and district heating
- Top gas recycling in oxygen blast furnace (TGR-OBF)
- Enhanced utilization of unused steel plant waste heat (off-gases from sintering, coke plant, hot stoves, converters, reheating furnaces, etc.)
- Heat recovery from slags
3.3. Potential Means to Reduce Emissions by CO2 Capturing and Storage as well as Utilization
3.4. Hydrogen Economy—Definitive Solution Toward Carbon Neutral Society?
3.5. Clean Electricity—The Major Energy Form in the Future
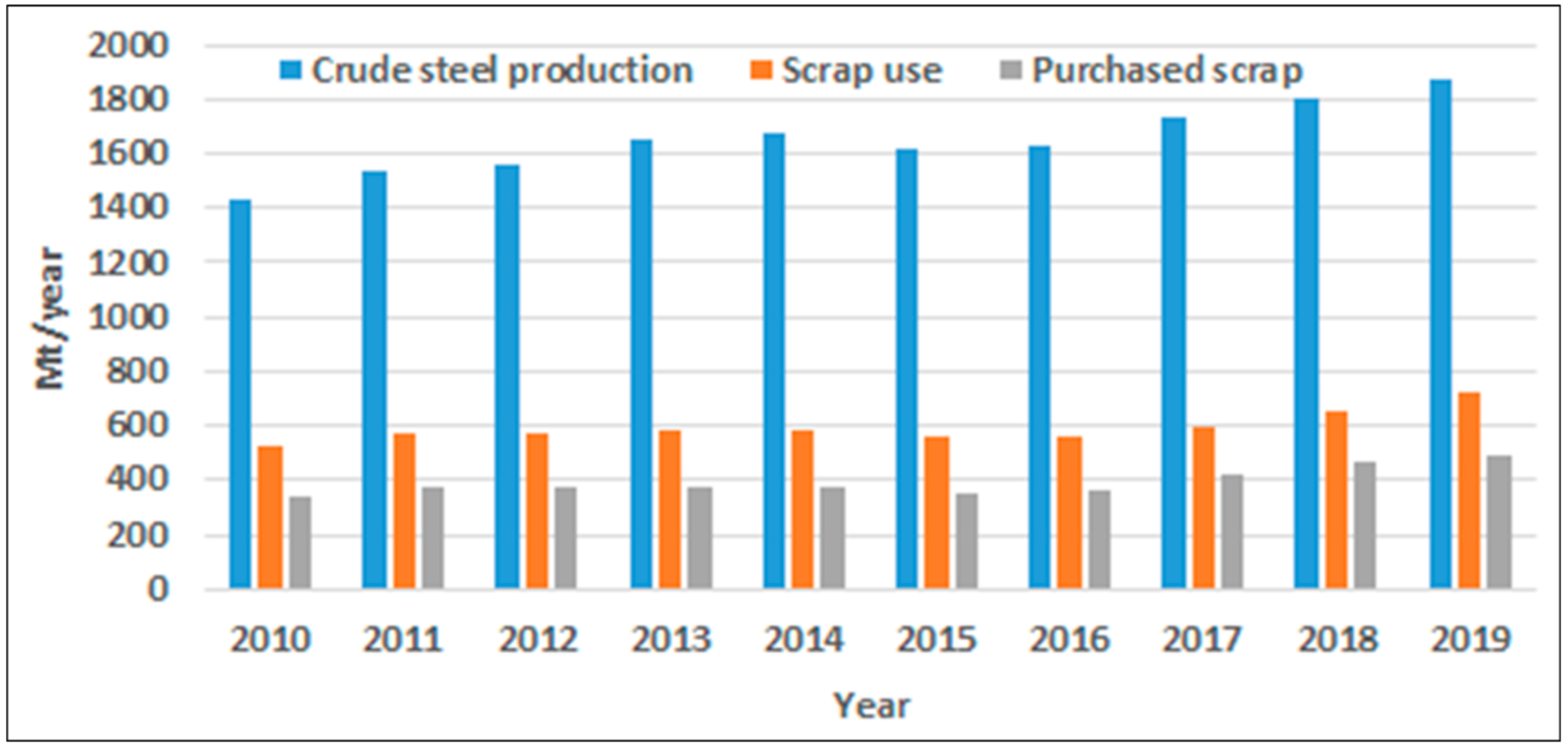
3.6. Increasing Recycling—A Key Factor in Overall Reduction of CO2 Emissions
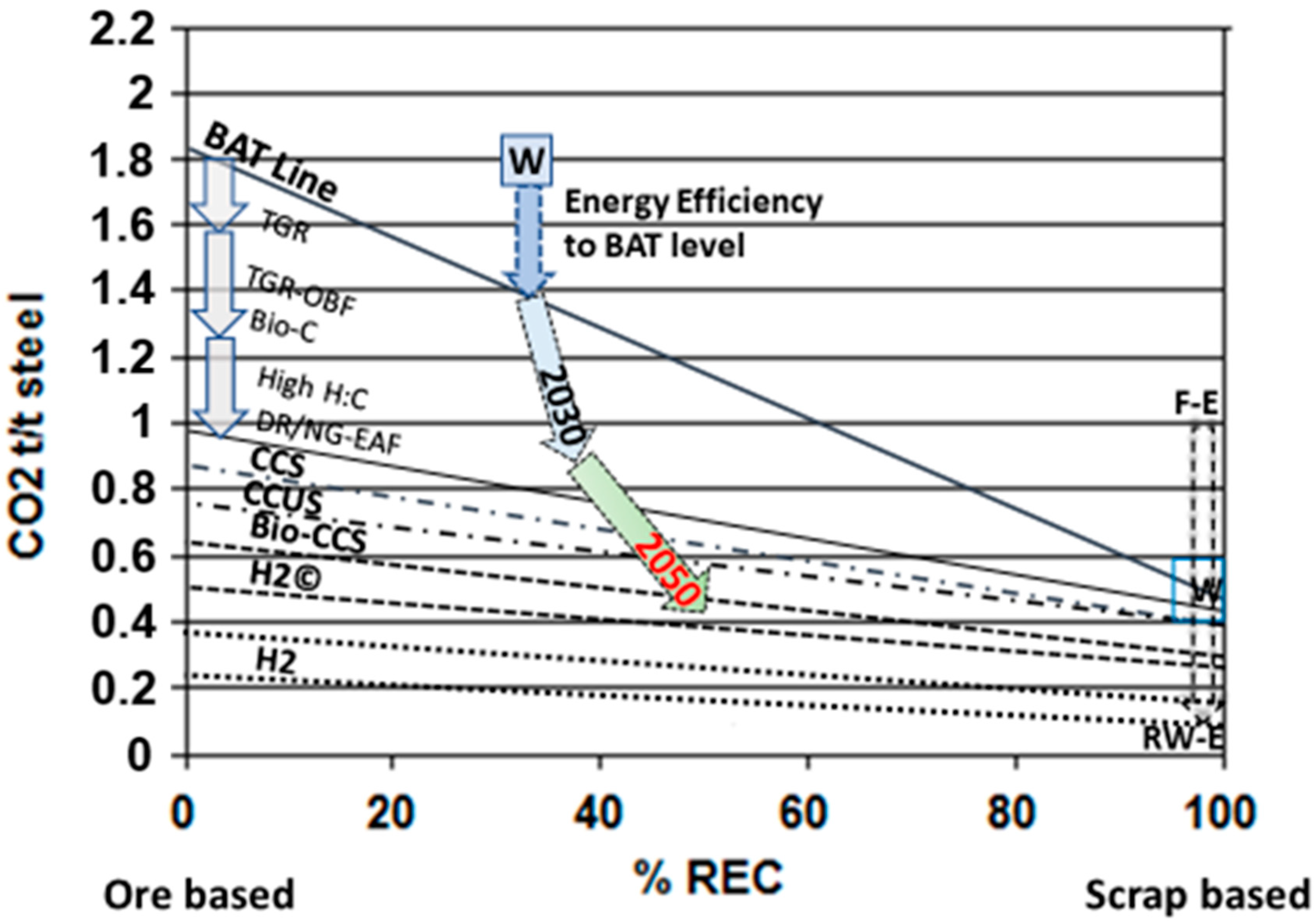
4. Summary of the Means to Cut CO2 Emissions from the Steel Industry—A General Vision
5. Conclusions
- (1)
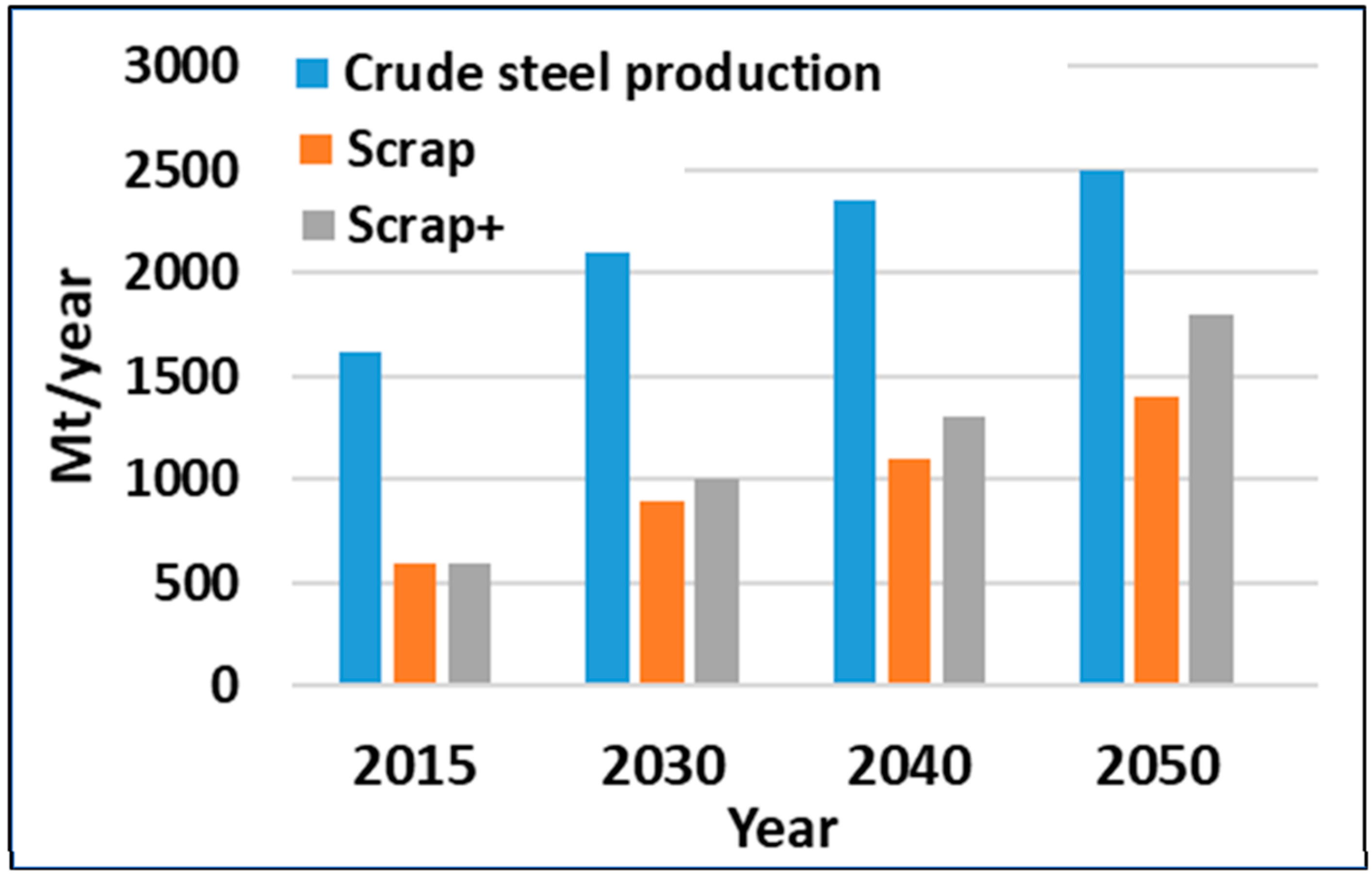
- The conservative forecast for steel demand/production in 2050 is 2.5 Gt/y. At the same time, the emissions should be cut down by 65–70%, which means that specific emissions, t CO2/t steel, should be reduced by approximately 75%. This will keep the steel industry in line with the IPCC´s target. This realization demands resolution and a strong commitment to wide-ranging and properly directed investments in reconstruction of the global steel industry. Practical implementation should be performed by steel enterprises and supervised by organizations like the World Steel Association, the United Nations, and the European Union.
- (2)
- In the short term, the most effective pathway is to improve energy efficiency and mitigate CO2 emissions by installing best available technologies in existing integrated steel plants (including sintering/pelletizing, coke making, BFs, BOFs, CC, rolling mills, etc.) as well as in EAF plants. Reducing C/H ratio in fuels and reductants, via efficient heat recovery and integrated management of energy flows as well as by adopting CCS from off gases, can decrease specific emissions by 40–50%.
- (3)
- More radical change is possible only via radical transmission from carbon-based metallurgy (coal, coke, oil, natural gas) to carbon-lean/carbon-free iron making. That can be realized via a combination of hydrogen reduction and melting with low-Carbon/C-free electricity. This provides major reconstruction of steel plants, replacement of blast furnaces with direct reduction furnaces, and production and storage for hydrogen. Pioneering attempts in this direction are under way, but for a wide quantum leap, a bigger wide-ranging transition to hydrogen economy throughout the society is necessary.
- (4)
- One positive trend that supports the steel industry in its endeavor to attain its targets is the growing share of recycled steel—from 30% to 50% level in steel production. This means a smaller need (share) for ore-based primary iron, higher share of EAF production, and strongly increasing demand for carbon-neutral electricity.
- (5)
- In order for the steel industry to make these implementations on a broad front and on schedule, certain policy tools are also necessary. Financial support to develop and deploy new technologies should be used as positive incentives. On the other hand, carbon pricing that favors low- and zero-carbon solutions and penalizes CO2 emissions is indispensable. The system should be transparent and based on both national conventions and wide international treaties.
Funding
Acknowledgments
Conflicts of Interest
References
- IPCC. AR5 SYR Summary for Policymakers. 2014. Available online: https://archive.ipcc.ch/pdf/assessment-report/ar5/syr/AR5_SYR_FINAL_SPM.pdf (accessed on 23 January 2020).
- Olivier, J.G.J.; Peters, J.A.H.W. Trends in Global CO2 and Total Greenhouse Gas Emissions: 2019 Report; PBL Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2019; p. 70. [Google Scholar]
- UNEP. Emissions Gap Report 2018. United Nations Environment Programme, Nairobi. Available online: https://www.unenvironment.org/resources/emissions-gap-report-2018 (accessed on 6 February 2020).
- Key World Energy Statistics. Energy and Climate Change. Available online: https://www.iea.org/statistics/kwes/ (accessed on 23 January 2020).
- Enerdata. Global Energy Statistical Yearbook 2019. Available online: https://yearbook.enerdata.net/total-energy/world-energy-production.html (accessed on 5 March 2020).
- World Steel Association. World Steel in Figures 2020. Available online: https://www.worldsteel.org/en/dam/jcr:f7982217-cfde-4fdc-8ba0-795ed807f513/World%2520Steel%2520in%2520Figures%25202020i.pdf (accessed on 5 July 2020).
- Morfeldt, J.; Nijs, W.; Silveira, S. The impact of climate targets on future steel production -an analysis based on a global energy system model. J. Clean. Prod. 2015, 103, 469–482. [Google Scholar] [CrossRef]
- World Steel Association. 2016. Available online: http://www.worldsteel.org/statistics/statistics-archive.html (accessed on 20 April 2017).
- World Steel Association. Steel’s Contribution to A Low Carbon Future and Climate Resilient Societies—World Steel Position Paper; World Steel Association: Brussels, Belgium, 2020; ISBN 978-2-930069-83-8. Available online: https://www.worldsteel.org/en/dam/jcr:7ec64bc1-c51c-439b-84b8-94496686b8c6/Position_paper_climate_2020_vfinal.pdf (accessed on 5 March 2020).
- Holappa, L. Toward Low Carbon Metallurgy in Iron and Steel Making. In Proceedings of the Guthrie Honorary Symposium, Montreal, QC, Canada, 6–9 June 2011; pp. 248–254. [Google Scholar]
- Holappa, L. Energy efficiency and sustainability in steel production. In An EPD Symposium in Honor of Professor Ramana, G. Reddy, Proceedings of the TMS Symposium on Applications of Process Engineering Principles in Materials Processing, Energy and Environmental Technologies, San Diego, CA, USA, 26 February–2 March 2017; Wang, S., Free, M.L., Alam, S., Zhang, M., Taylor, P.R., Eds.; Springer: Cham, Switzerland, 2017; pp. 401–410. [Google Scholar]
- IEA/OECD. Energy Technology Transitions for Industry, Strategies for the Next Industrial Revolution. Available online: http://www.oecd-ilibrary.org/energy/energy-technology-transitions-for-industry_9789264068612-en (accessed on 20 April 2017).
- Zhang, J.; Liu, Z.; Li, K.; Wang, G.; Jiao, K.; Yang, T. Current status and prospects of Chinese steel industry. In Proceedings of the Current Status and Prospects of Chinese Steel Industry. Scanmet V Conference, Luleå, Sweden, 12–15 June 2016; p. 15. [Google Scholar]
- Hasanbeigi, A.; Springer, C. How Clean is the U.S. Steel Industry? An International Benchmarking of Energy and CO2 Intensities; Global Efficiency Intelligence: San Francisco, CA, USA; Available online: https://www.globaleciencyintel.com (accessed on 5 March 2020).
- The European Commission. Document L:2012:070: TOC. Implementing Decision of 28 February 2012. Available online: http://data.europa.eu/eli/dec_impl/2012/135/oj (accessed on 5 March 2020).
- Worrell, E.; Price, L.; Neelis, M.; Galitsky, C.; Zhou, N. World Best Practice Energy Intensity Values for Selected Industrial Sectors; Ernest Orlando Lawrence Berkeley National Laboratory: Berkeley, CA, USA, 2007; p. 51, LBNL-62806. [Google Scholar]
- Zhang, Q.; Xu, J.; Wang, Y.; Hasanbeigi, A.; Zhang, W.; Lu, H.; Arens, M. Comprehensive assessment of energy conservation and CO2 emissions mitigation in China’s iron and steel industry based on dynamic material flows. Appl. Energy 2018, 209, 251–265. [Google Scholar] [CrossRef]
- Bureau of International Recycling. World Steel Recycling in Figures 2011–2015. Available online: https://www.bdsv.org/fileadmin/service/markt_und_branchendaten/weltstatistik_2011_2015.pdf (accessed on 16 March 2020).
- Eurofer. A Steel Roadmap for A Low Carbon Europe 2050. 2013. Available online: http://www.eurofer.org/News%26Events/PublicationsLinksList/2013-Roadmap.pdf (accessed on 5 March 2020).
- Wörtler, M.; Schuler, F.; Voigt, N.; Schmidt, T.; Dahlmann, P.; Lüngen, H.-B.; Ghenda, T. Steel’s Contribution to a Low Carbon Europe 2050. BCG & VDEh. Available online: https://www.stahl-online.de/wp-content/uploads/2013/09/Schlussbericht-Studie-Low-carbon-Europe-2050_-Mai-20131.pdf (accessed on 16 March 2020).
- Birat, J.-P. Update on the ULCOS program. In How will Employment and Labor Markets Develop in the Context of a Transition Towards a Low-Carbon Economy? ESTEP: Brussels, Belgium, 2014; Available online: http://erc-online.eu/wp-content/uploads/2014/04/2011-00612-E.pdf (accessed on 16 March 2020).
- van der Stel, J.; Hattink, M.; Zeilstra, C.; Louwerse, G.; Hirsch, A.; Janhsen, U.; Sert, D.; Grant, M.; Delebecque, A.; Diez-Brea, P.; et al. ULCOS top gas recycling blast furnace process (ULCOS TGRBF). In European Commission EUR 26414; Publications Office of the European Union: Luxembourg, 2014; p. 47. ISBN 978-92-79-35038-2. [Google Scholar] [CrossRef]
- van der Stel, J.; Meijer, K.; Santos, S.; Peeters, T.; Broersen, P. Opportunities for Reducing CO2 emissions from Steel Industry. In Proceedings of the EMECR 2017 1st International Conference on Energy and Material Efficiency and CO2 Reduction in the Steel Industry, Kobe, Japan, 11–13 October 2017; pp. 46–49. Available online: http://www.emecr2017.com (accessed on 16 March 2020).
- Nishioka, K.; Ujisawa, Y.; Tonomura, S.; Ishiwata, N.; Sikstrom, P. Sustainable Aspects of CO2 Ultimate Reduction in the Steelmaking Process (COURSE50 Project), Part 1: Hydrogen Reduction in the Blast Furnace. J. Sustain. Met. 2016, 2, 200–208. [Google Scholar] [CrossRef]
- Onoda, M.; Matsuzaki, Y.; Chowdhury, F.A.; Yamada, H.; Goto, K.; Tonomura, S. Sustainable Aspects of Ultimate Reduction of CO2 in the Steelmaking Process (COURSE50 Project), Part 2: CO2 Capture. J. Sustain. Met. 2016, 2, 209–215. [Google Scholar] [CrossRef]
- Tonomura, S.; Kikuchi, N.; Ishiwata, N.; Tomisaki, S.; Tomita, Y. Concept and Current State of CO2 Ultimate Reduction in the Steelmaking Process (COURSE50) Aimed at Sustainability in the Japanese Steel Industry. J. Sustain. Met. 2016, 2, 191–199. [Google Scholar] [CrossRef]
- Ariyama, T.; Takahashi, K.; Kawashiri, Y.; Nouchi, T. Diversification of the Ironmaking Process Toward the Long-Term Global Goal for Carbon Dioxide Mitigation. J. Sustain. Met. 2019, 5, 276–294. [Google Scholar] [CrossRef]
- Kim, J.; Ahn, Y.; Roh, T. Low-Carbon Management of POSCO in Circular Economy: Current Status and Limitations. In Towards a Circular Economy: Corporate Management and Policy Pathways. ERIA Research Project Report 2014-44; Anbumozhi, V., Kim, J., Eds.; ERIA: Jakarta, Indonesia, 2016; pp. 185–199. Available online: https://www.eria.org/RPR_FY2014_No.44_Chapter_11.pdf (accessed on 16 March 2020).
- Jahanshahi, S.; Mathieson, J.G.; Reimink, H. Low Emission Steelmaking. J. Sustain. Met. 2016, 2, 185–190. [Google Scholar] [CrossRef]
- Sohn, H.Y. Energy Consumption and CO2 Emissions in Ironmaking and Development of a Novel Flash Technology. Metals 2019, 10, 54. [Google Scholar] [CrossRef]
- Zhao, J.; Zuo, H.-B.; Wang, Y.; Wang, J.; Xue, Q. Review of green and low-carbon ironmaking technology. Ironmak. Steelmak. 2019, 47, 296–306. [Google Scholar] [CrossRef]
- He, K.; Wang, L.; Li, X. Review of the Energy Consumption and Production Structure of China’s Steel Industry: Current Situation and Future Development. Metals 2020, 10, 302. [Google Scholar] [CrossRef]
- Liao, W.-C.; Chen, J.-S.; Liu, C.-H.; Chen, J.-J.; Ko, F.-K. The Inspection of CO2 Emission Targets of Industry Sector in Taiwan. Available online: https://www.irena.org/assets/IEW/EventDocs/Session_3_Parallel_E_Liao_Wei_Chen_paper.pdf (accessed on 16 March 2020).
- Krishnan, S.S.; Vunnam, V.; Sunder, P.S.; Sunil, J.V.; Ramakrishnan, A.M. A Study of Energy Efficiency in the Indian Iron and Steel Industry; Center for Study of Science, Technology and Policy: Bangalore, India, 2013; p. 132. ISBN 978-81-903613-4-7. Available online: http://www.indiaenvironmentportal.org.in/files/file/A%20Study%20of%20Energy%20Efficiency%20in%20the%20Indian%20IS%20Industry.pdf (accessed on 24 June 2020).
- Wenzel, W.; Gudenau, H.W.; Fukushima, T. Blast Furnace Operating Methods. US Patent 3884677, 20 November 1973. [Google Scholar]
- Zhang, W.; Zhang, J.; Xue, Z.; Zou, Z.; Qi, Y. Unsteady Analyses of the Top Gas Recycling Oxygen Blast Furnace. ISIJ Int. 2016, 56, 1358–1367. [Google Scholar] [CrossRef]
- Arasto, A.; Tsupari, E.; Kärki, J.; Lilja, J.; Sihvonen, M. Oxygen blast furnace with CO2 capture and storage at an integrated steel mill—Part I: Technical concept analysis. Int. J. Greenh. Gas Control. 2014, 30, 140–147. [Google Scholar] [CrossRef]
- Tsupari, E.; Kärki, J.; Arasto, A.; Lilja, J.; Kinnunen, K.; Sihvonen, M. Oxygen blast furnace with CO2 capture and storage at an integrated steel mill—Part II: Economic feasibility in comparison with conventional blast furnace highlighting sensitivities. Int. J. Greenh. Gas Control. 2015, 32, 189–196. [Google Scholar] [CrossRef]
- Fu, J.-X.; Tang, G.-H.; Zhao, R.-J.; Hwang, W.-S. Carbon Reduction Programs and Key Technologies in global Steel Industry. J. Iron Steel Res. Int. 2014, 21, 275–281. [Google Scholar] [CrossRef]
- Wakuri, S.; Mochizuki, S.; Baba, M.; Misawa, J.; Anan, K. Operation of Pulverized Coal Injection into Large Blast Furnace with High Top Pressure. Trans. Iron Steel Inst. Jpn. 1984, 24, 622–630. [Google Scholar] [CrossRef]
- Janz, J.; Weiss, W. Injection of waste plastics into the blast furnace of Stahlwerke Bremen. Revue Métallurgie 1996, 93, 1219–1226. [Google Scholar] [CrossRef]
- Norgate, T.; Haque, N.; Somerville, M.; Jahanshahi, S. Biomass as a Source of Renewable Carbon for Iron and Steelmaking. ISIJ Int. 2012, 52, 1472–1481. [Google Scholar] [CrossRef]
- Mathieson, J.G.; Rogers, H.; Somerville, M.; Jahanshahi, S. Reducing Net CO2 Emissions Using Charcoal as a Blast Furnace Tuyere Injectant. ISIJ Int. 2012, 52, 1489–1496. [Google Scholar] [CrossRef]
- Mousa, E.A.; Wang, C.; Riesbeck, J.; Larsson, M. Biomass applications in iron and steel industry: An overview of challenges and opportunities. Renew. Sustain. Energy Rev. 2016, 65, 1247–1266. [Google Scholar] [CrossRef]
- Suopajärvi, H.; Kemppainen, A.; Haapakangas, J.; Fabritius, T. Extensive review of the opportunities to use biomass-based fuels in iron and steelmaking processes. J. Clean. Prod. 2017, 148, 709–734. [Google Scholar] [CrossRef]
- IEA IETS. Process Integration in the Iron and Steel Industry—IEA IETS Annex XIV Technical Report. Swerea MEFOS Report No: 1406; p. 41. Available online: https://iea-industry.org/app/uploads/mef14064-iets-annex-xiv-technical-report_2014.pdf (accessed on 5 March 2020).
- Lima Lopes, N.; de Pádua Nacif, A.; de Cássia Oliveira Carneiro, A.; de Assis, J.B.; Costa Oliveira, A. Brazilian Green Steel. Viçosa, Minas Gerais March. 2017. Available online: http://ciflorestas.com.br/arquivos/d_e_e_1099159498.pdf (accessed on 29 May 2020).
- De Castro, J.A. A Comprehensive Modeling as a tool for Developing New Mini Blast Furnace Technologies Based on Biomass and Hydrogen Operation. Invited Lecture at Nippon Steel, 11th December 2019 on “New trends on the development of green pig iron”. 50 Slides. 2019. Available online: https://www.researchgate.net/publication/340564870_Nippon_Steel 11th_December_2019_Castro_New_trends_on_the_development_of _green_pig_iron (accessed on 1 June 2020).
- Williams, M. Deforesting the Earth: From Prehistory to Global Crisis; The University of Chicago Press: Chicago, IL, USA, 2003; p. 689. ISBN 0-226-89926-8. [Google Scholar]
- Global CCS Institute. The Global Status of CCS: 2019. 2019. Available online: https://www.globalccsinstitute.com/wp-content/uploads/2019/12/GCC_GLOBAL_STATUS_REPORT_2019.pdf (accessed on 1 June 2020).
- Nuttall, W.J.; Bakenne, A.T. Carbon Capture, Utilisation and Storage. In Fossil Fuel Hydrogen, 1st ed.; Nuttall, W.J., Bakenne, A.T., Eds.; Springer: Cham, Switzerland, 2020; pp. 53–67. [Google Scholar]
- IEAGHG. Iron and Steel CCS Study (Techno-Economics Integrated Steel Mill). 2013. Available online: https://ieaghg.org/publications/technical-reports/reports-list/9-technical-reports/1001-2013-04-iron-and-steel-ccs-study-techno-economics-integrated-steel-mill (accessed on 5 March 2020).
- Gerdemann, S.J.; O’Connor, W.K.; Dahlin, D.C.; Penner, L.R.; Rush, H. Ex Situ Aqueous Mineral Carbonation. Environ. Sci. Technol. 2007, 41, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- Zevenhoven, R.; Fagerlund, J.; Songok, J.K. Review: CO2 mineral sequestration: Developments toward large-scale application. Greenh. Gas Sci Technol. 2011, 1, 48–57. [Google Scholar] [CrossRef]
- Zevenhoven, R. Metals Production, CO2 Mineralization and LCA. Metals 2020, 10, 342. [Google Scholar] [CrossRef]
- Said, A.; Laukkanen, T.; Järvinen, M. Pilot-scale experimental work on carbon dioxide sequestration using steelmaking slag. Appl. Energy 2016, 177, 602–611. [Google Scholar] [CrossRef]
- Singh, J.; Dhar, D.W. Overview of Carbon Capture Technology: Microalgal Biorefinery Concept and State-of-the-Art. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Otto, A.; Grube, T.; Schiebahn, S.; Stolten, D. Closing the loop: Captured CO2 as a feedstock in the chemical industry. Energy Environ. Sci. 2015, 8, 3283–3297. [Google Scholar] [CrossRef]
- Araki, K. CO2 Ultimate Reduction in Steelmaking Process (COURSE50 Project). In Proceedings of the EMECR 2017, 1st Int. Conf. on Energy and Material Efficiency and CO2 Reduction in the Steel Industry, Kobe, Japan, 11–13 October 2017; Paper 11A-KL1. pp. 32–35. [Google Scholar]
- Japan Iron and Steel Federation. JISF Long-Term Vision for Climate Change Mitigation—A Challenge Towards Zero-Carbon Steel. 2019. Available online: https://www.jisf.or.jp/en/activity/climate/documents/JISFLong-termvisionforclimatechangemitigation.pdf (accessed on 6 March 2020).
- JFE Group CSR REPORT. Protecting the Global Environment. Climate Change Mitigation. 2019. Available online: https://www.jfe-holdings.co.jp/en/csr/pdf/2019/2019_09_04.pdf (accessed on 1 June 2020).
- He, Q.; Shi, M.; Liang, F.; Xu, L.; Ji, L.; Yan, S. Renewable absorbents for CO2 capture: From biomass to nature. Greenh. Gases Sci. Technol. 2019, 9, 637–651. [Google Scholar] [CrossRef]
- IEA. May 2019 Report: Transforming Industry through CCUS. 2019. Available online: https://webstore.iea.org/download/direct/2778?fileName=Transforming_Industry_through_CCUS.pdf (accessed on 23 June 2020).
- Handler, R.M.; Shonnard, D.R.; Griffing, E.M.; Lai, A.; Palou-Rivera, I. Life Cycle Assessments of Ethanol Production via Gas Fermentation: Anticipated Greenhouse Gas Emissions for Cellulosic and Waste Gas Feedstocks. Ind. Eng. Chem. Res. 2015, 55, 3253–3261. [Google Scholar] [CrossRef]
- STEELANOL Project 2015-21. Production of Sustainable, Advanced Bio-ethANOL through an Innovative Gas-Fermentation Process Using Exhaust Gases Emitted in the STEEL Industry. On-Going Project under H2020-EU.3.3.3.1. Available online: http://www.steelanol.eu/en (accessed on 24 June 2020).
- Zhang, Y.; Ni, W.; Li, Z. Research on superclean polygeneration energy system of iron and steel industry. In Power and Energy Systems, Proceeding of the 7th IASTED International Conference on Power and Energy Systems; Acta Press: Anaheim, CA, USA, 2004; pp. 152–157. [Google Scholar]
- Ghanbari, H.; Saxén, H.; Grossmann, I.E. Optimal design and operation of a steel plant integrated with a polygeneration system. AIChE J. 2013, 59, 3659–3670. [Google Scholar] [CrossRef]
- IEA. The Future of Hydrogen—Report prepared by the IEA for the G20, Japan Seizing Today’s Opportunities. 2020. Available online: https://webstore.iea.org/download/direct/2803 (accessed on 24 June 2020).
- IRENA. Hydrogen: A Renewable Energy Perspective. 2019. Intern. Renewable Energy Agency, Abu Dhabi. Available online: https://www.irena.org/-/media/Files/IRENA/Agency/Publication/2019/Sep/IRENA_Hydrogen_2019.pdf (accessed on 24 June 2020).
- FCH. Hydrogen Roadmap Europe—A Sustainable Pathway for the European. Available online: https://www.fch.europa.eu/sites/default/files/Hydrogen%20Roadmap%20Europe_Report.pdf (accessed on 24 June 2020).
- Kapdan, I.K.; Kargi, F. Bio-hydrogen production from waste materials. Enzym. Microb. Technol. 2006, 38, 569–582. [Google Scholar] [CrossRef]
- Dou, B.; Zhang, H.; Song, Y.; Zhao, L.; Jiang, B.; He, M.; Ruan, C.; Chen, H.; Xu, Y. Hydrogen production from the thermochemical conversion of biomass: Issues and challenges. Sustain. Energy Fuels 2019, 3, 314–342. [Google Scholar] [CrossRef]
- Squires, A.M.; Johnson, C.A. The H-iron process. JOM 1957, 9, 586–590. [Google Scholar] [CrossRef]
- Guo, L.; Bao, Q.; Gao, J.; Zhu, Q.; Guo, Z. A Review on Prevention of Sticking during Fluidized Bed Reduction of Fine Iron Ore. ISIJ Int. 2020, 60, 1–17. [Google Scholar] [CrossRef]
- Nuber, D.; Eichberger, H.; Rollinger, B. Circored fine ore direct reduction—The future of modern electric steelmaking. Stahl Eisen 2006, 126, 47–51. [Google Scholar]
- Sohn, H.Y. From Nonferrous Flash Smelting to Flash Ironmaking: Development of an ironmaking technology with greatly reduced CO2 emissions and energy consumption. In Treatise on Process Metallurgy; Elsevier: Oxford, UK; Waltham, MA, USA, 2014; Volume 3 Industrial Processes Part B, Section 4.5.2.2; pp. 1596–1691. [Google Scholar]
- Cavaliere, P. Clean Ironmaking and Steelmaking Processes; Springer Nature: Cham, Switzerland, 2019; p. 624. Available online: https://link.springer.com/book/10.1007%2F978-3-030-21209-4 (accessed on 5 March 2020).
- Quader, M.A.; Ahmed, S.; Dawal, S.; Nukman, Y. Present needs, recent progress and future trends of energy-efficient Ultra-Low Carbon Dioxide (CO2) Steelmaking (ULCOS) program. Renew. Sustain. Energy Rev. 2016, 55, 537–549. Available online: https://www.sciencedirect.com/science/article/pii/S1364032115011806 (accessed on 1 June 2020). [CrossRef]
- Spreitzer, D.; Schenk, J.L. Reduction of Iron Oxides with Hydrogen—A Review. Steel Res. Int. 2019, 90, 17. [Google Scholar] [CrossRef]
- Patisson, F.; Mirgaux, O. Hydrogen Ironmaking: How it Works. Metals 2020, 10, 922. [Google Scholar] [CrossRef]
- H2FUTURE. Verbund Solutions GmbH. Available online: https://www.h2future-project.eu (accessed on 24 June 2020).
- GrInHy. Green Industrial Hydrogen. Salzgitter Mannesmann Forschung GmbH. Available online: https://www.green-industrial-hydrogen.com (accessed on 28 April 2020).
- Åhman, M.; Olsson, O.; Vogl, V.; Nyqvist, B.; Maltais, A.; Nilsson, L.J.; Hallding, K.; Skånberg, K.; Nilsson, M. Hydrogen Steelmaking for a Low-Carbon Economy. A Joint LU-SEI Working Paper for the HYBRIT Project. EESS Report No 109, SEI Working Paper WP 2018-07. 2018. Available online: https://www.sei.org/publications/hydrogen-steelmaking/ (accessed on 24 June 2020). Hydrogen Steelmaking for a Low-Carbon Economy.
- Vogl, V.; Åhman, M.; Nilsson, L.J. Assessment of hydrogen direct reduction for fossil-free steelmaking. J. Clean. Prod. 2018, 203, 736–745. [Google Scholar] [CrossRef]
- Pei, M.; Petäjäniemi, M.; Regnell, A.; Wijk, O. Toward a Fossil Free Future with HYBRIT: Development of Iron and Steelmaking Technology in Sweden and Finland. Metals 2020, 10, 972. [Google Scholar] [CrossRef]
- WNA. Comparison of Lifecycle Greenhouse Gas Emissions of Various Electricity Generation Sources. World Nuclear Association (WNA). 2011. Available online: http://www.world-nuclear.org/uploadedFiles/org/WNA/Publications/Working_Group_Reports/comparison_of_lifecycle.pdf (accessed on 1 July 2020).
- Schlömer, S.; Bruckner, T.; Fulton, L.; Hertwich, E.; McKinnon, A.; Perczyk, D.; Roy, J.; Schaeffer, R.; Sims, R.; Smith, P.; et al. Annex III: Technology-specific cost and performance parameters. In Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- EEA. CO2-Emission Intensity from Electricity Generation. 2018. Available online: https://www.eea.europa.eu/data-and-maps/daviz/sds/co2-emission-intensity-from-electricity-generation-2/@@view (accessed on 1 June 2020).
- IEA. Tracking the Decoupling of Electricity Demand and Associated CO2 Emissions, 1990–2017. 2019. Available online: https://www.iea.org/commentaries/tracking-the-decoupling-of-electricity-demand-and-associated-co2-emissions (accessed on 1 July 2020).
- Otto, A.; Robinius, M.; Grube, T.; Schiebahn, S.; Praktiknjo, A.; Stolten, D. Power-to-Steel: Reducing CO2 through the Integration of Renewable Energy and Hydrogen into the German Steel Industry. Energies 2017, 10, 451. [Google Scholar] [CrossRef]
- Bureau of International Recycling. World Steel Recycling in Figures 2011–2018. 2019. Available online: https://www.bdsv.org/fileadmin/user_upload/World-Steel-Recycling-in-Figures-2014-2018.pdf (accessed on 28 April 2020).
- Pauliuk, S.; Milford, R.L.; Müller, D.B.; Allwood, J.M. The Steel Scrap Age. Environ. Sci. Technol. 2013, 47, 3448–3454. [Google Scholar] [CrossRef] [PubMed]
- Geyer, R.; Davis, J.; Ley, J.; He, J.; Clift, R.; Kwan, A.; Sansom, M.; Jackson, T. Time-dependent material flow analysis of iron and steel in the UK. Resour. Conserv. Recycl. 2007, 51, 101–117. [Google Scholar] [CrossRef]
- Gauffin, A.; Andersson, N.Å.I.; Storm, P.; Tilliander, A.; Jönsson, P.G. Use of volume correlation model to calculate lifetime of end-of-life steel. Ironmak. Steelmak. 2014, 42, 88–96. [Google Scholar] [CrossRef]
- Haslehner, R.; Stelter, B.; Osio, N. Steel as a Model for a Sustainable Metal Industry in 2050. 2015. Available online: https://www.bcg.com/publications/2015/metals-mining-sustainability-steel-as-a-model-for-a-sustainable-metal-industry-in-2050.aspx (accessed on 5 March 2020).
- World Economic Forum. 2015 Mining & Metals in a Sustainable World 2050, REF 250815. Available online: http://www3.weforum.org/docs/WEF_MM_Sustainable_World_2050_report_2015.pdf (accessed on 1 July 2020).
- Rammer, B.; Millner, R.; Boehm, C. Comparing the CO2 Emissions of Different Steelmaking Routes. BHM Berg Hüttenmänn. Monatshefte 2017, 162, 7–13. [Google Scholar] [CrossRef]
- Bhaskar, A.; Assadi, M.; Somehsaraei, H.N. Decarbonization of the Iron and Steel Industry with Direct Reduction of Iron Ore with Green Hydrogen. Energies 2020, 13, 758. [Google Scholar] [CrossRef]
- World Steel Association. Global steel industry: Outlook, challenges and opportunities. 5th International Steel Industry & Sector Relations Conference; April 20th, 2017 Istanbul. Available online: https://www.worldsteel.org/en/dam/jcr:d9e6a3df-ff19-47ff-9e8ff8c136429fc4/International+Steel+Industry+and+Sector+Relations+Conference+Istanbul170420.pdf (accessed on 14 August 2020).
- Elkerbout, M.; Egenhofer, C. Tools to Boost Investment in Low-Carbon Technologies—Five Possible Ways to Create Low-Carbon Markets in the EU. CEPS Policy Insights No 2018/11. Available online: http://www.cepsech.eu/system/tdf/PI2018_11_ME_CE_Tools%20to%20boost%20investment%20in%20low%20carbon%20technologies_0.pdf?file=1&type=node&id=612&force= (accessed on 14 August 2020).
| Primary Energy—Fossil | Bio | New Technology with CCS | ||
| Coal | Natural gas | Biomass | Coal + CCS | NG + CCS |
| 820 | 490 | 740; 230 1 | 160–220 | 170 |
| Primary Energy | Renewable/Non-fossil | |||
| Geothermal | Hydro | Nuclear | Solar | Wind |
| 38 | 24 | 12 | 48 | 12 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holappa, L. A General Vision for Reduction of Energy Consumption and CO2 Emissions from the Steel Industry. Metals 2020, 10, 1117. https://doi.org/10.3390/met10091117
Holappa L. A General Vision for Reduction of Energy Consumption and CO2 Emissions from the Steel Industry. Metals. 2020; 10(9):1117. https://doi.org/10.3390/met10091117
Chicago/Turabian StyleHolappa, Lauri. 2020. "A General Vision for Reduction of Energy Consumption and CO2 Emissions from the Steel Industry" Metals 10, no. 9: 1117. https://doi.org/10.3390/met10091117
APA StyleHolappa, L. (2020). A General Vision for Reduction of Energy Consumption and CO2 Emissions from the Steel Industry. Metals, 10(9), 1117. https://doi.org/10.3390/met10091117





