High Speed Steel with Iron Addition Materials Sintered by Spark Plasma Sintering
Abstract
1. Introduction
2. Materials and Methods
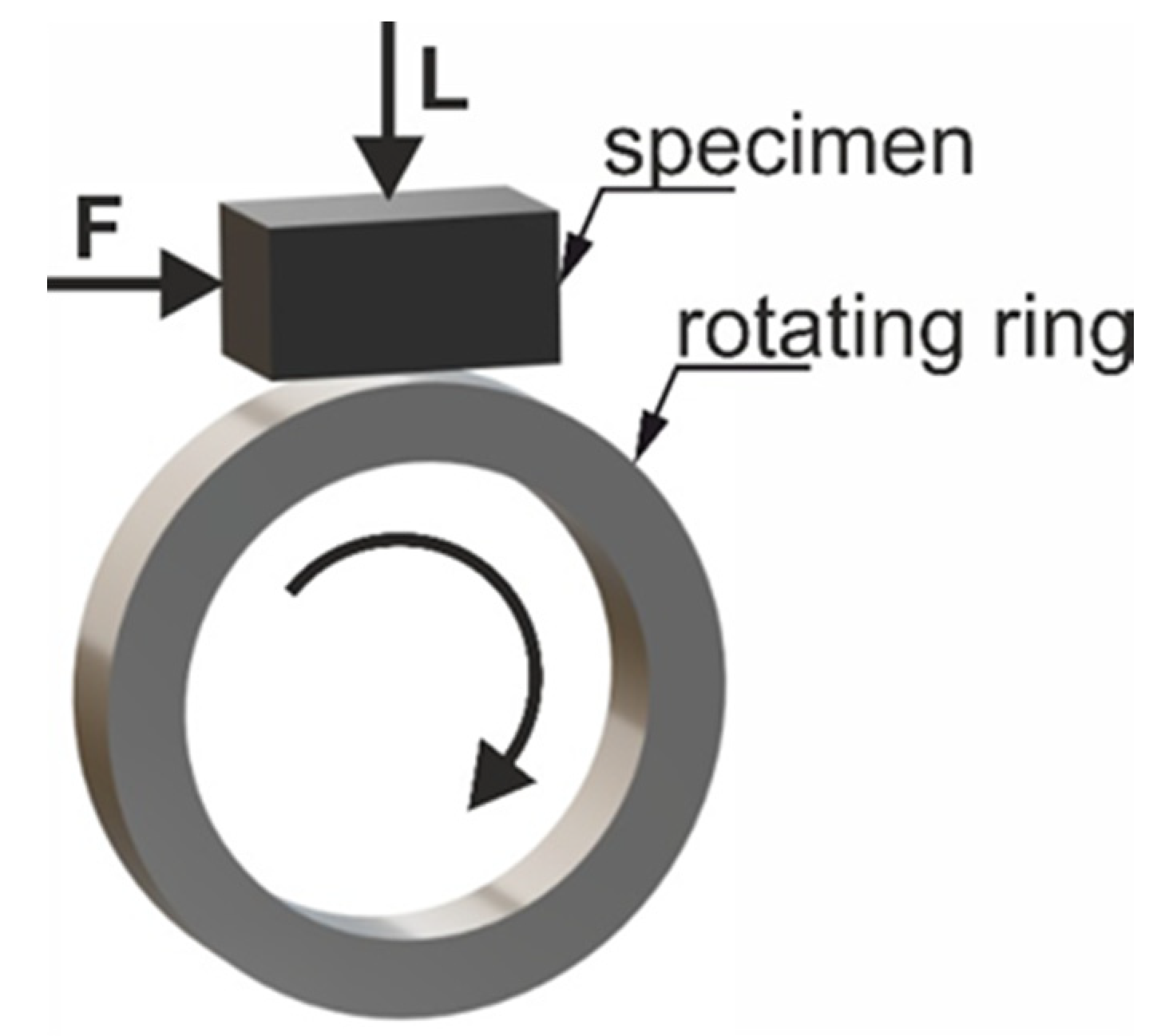
- dimensions of the test sample: 20 mm × 4 mm× 4 mm,
- rotating ring: heat-treated steel 100Cr6, 55 HRC, Ø49.5 mm × 8 mm,
- rotational speed: 136 rpm,
- velocity: 0.35 m/s,
- load: 200 N,
- sliding distance: 250 and 500 m.
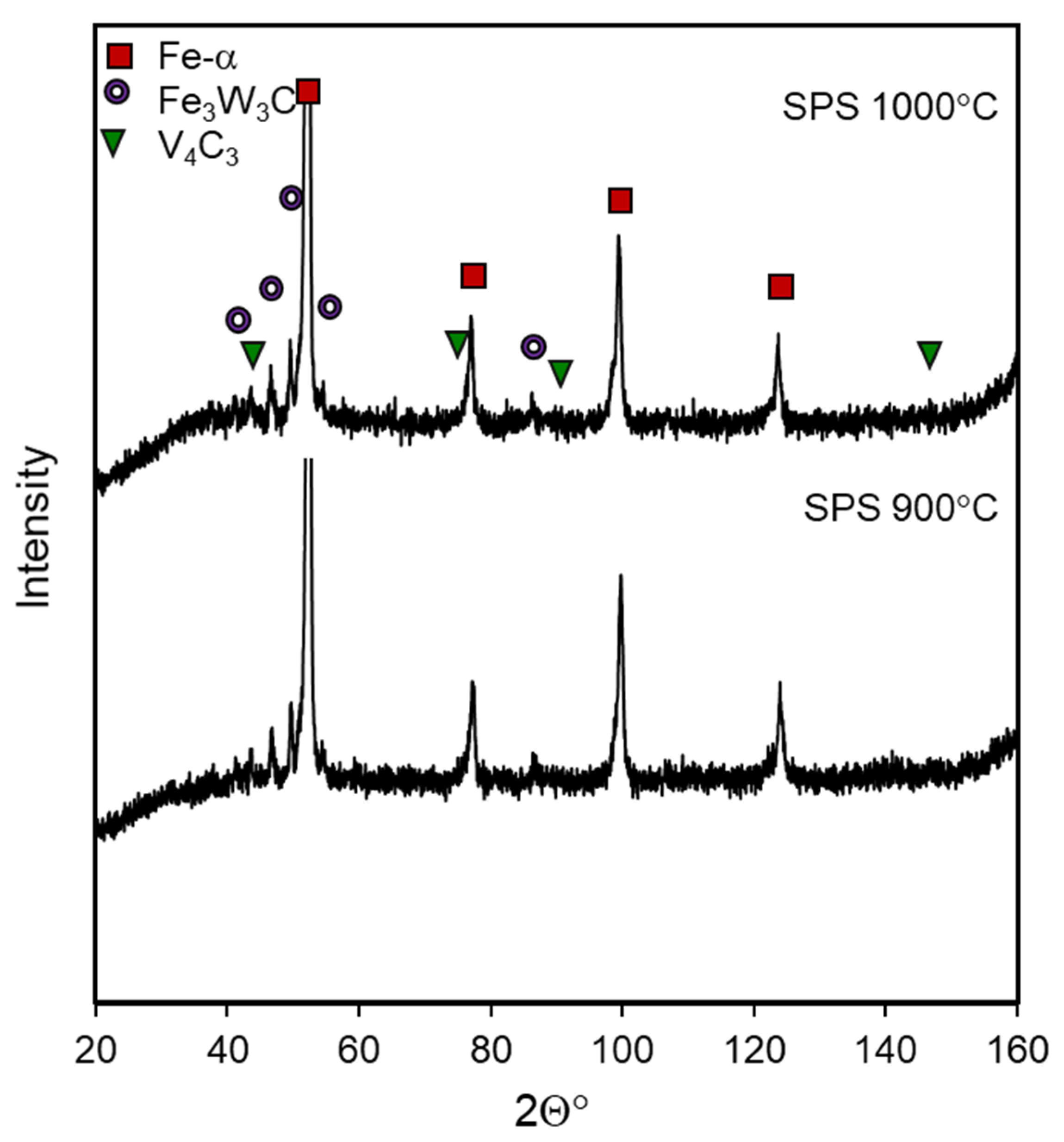
3. Results and Discussion
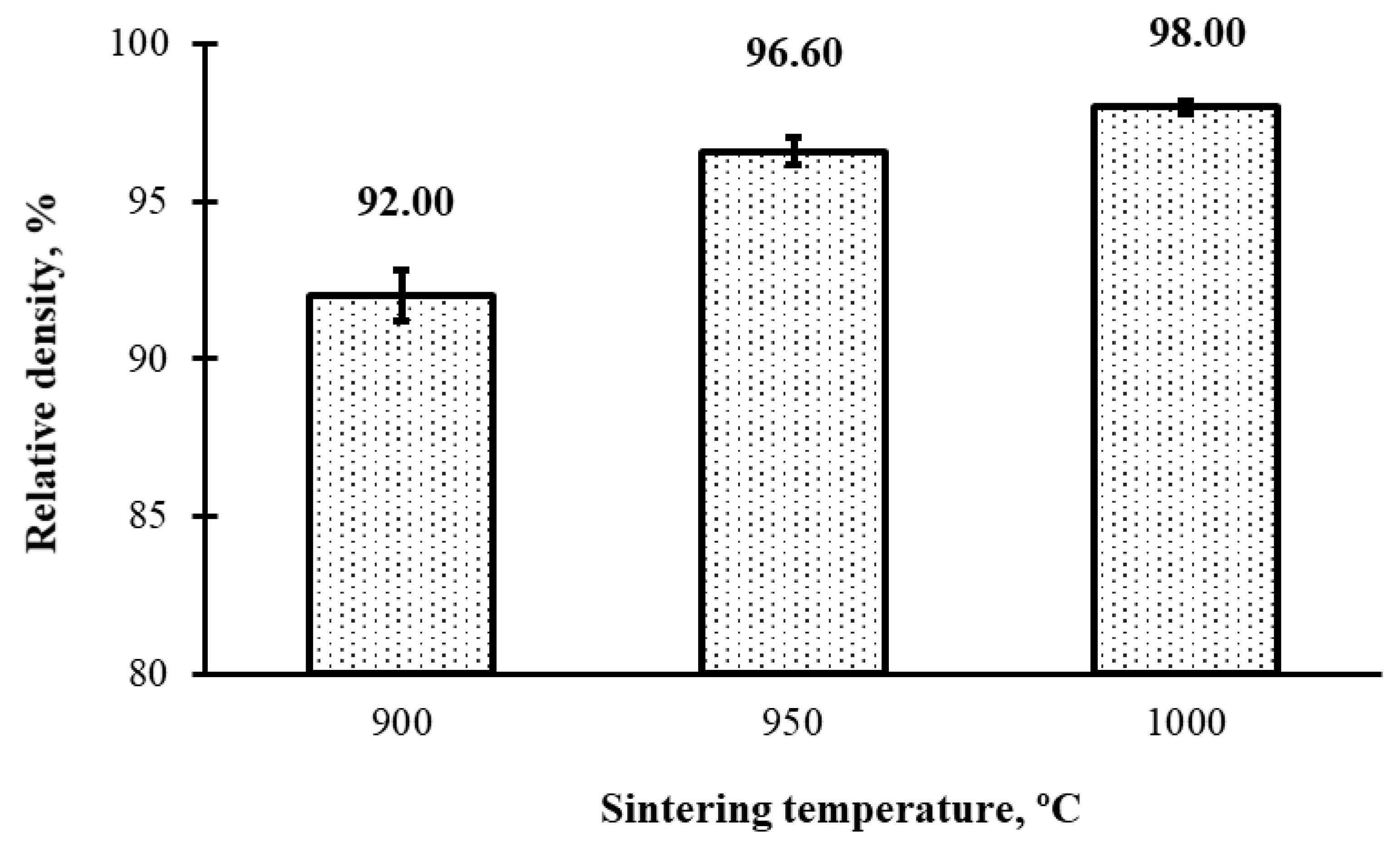
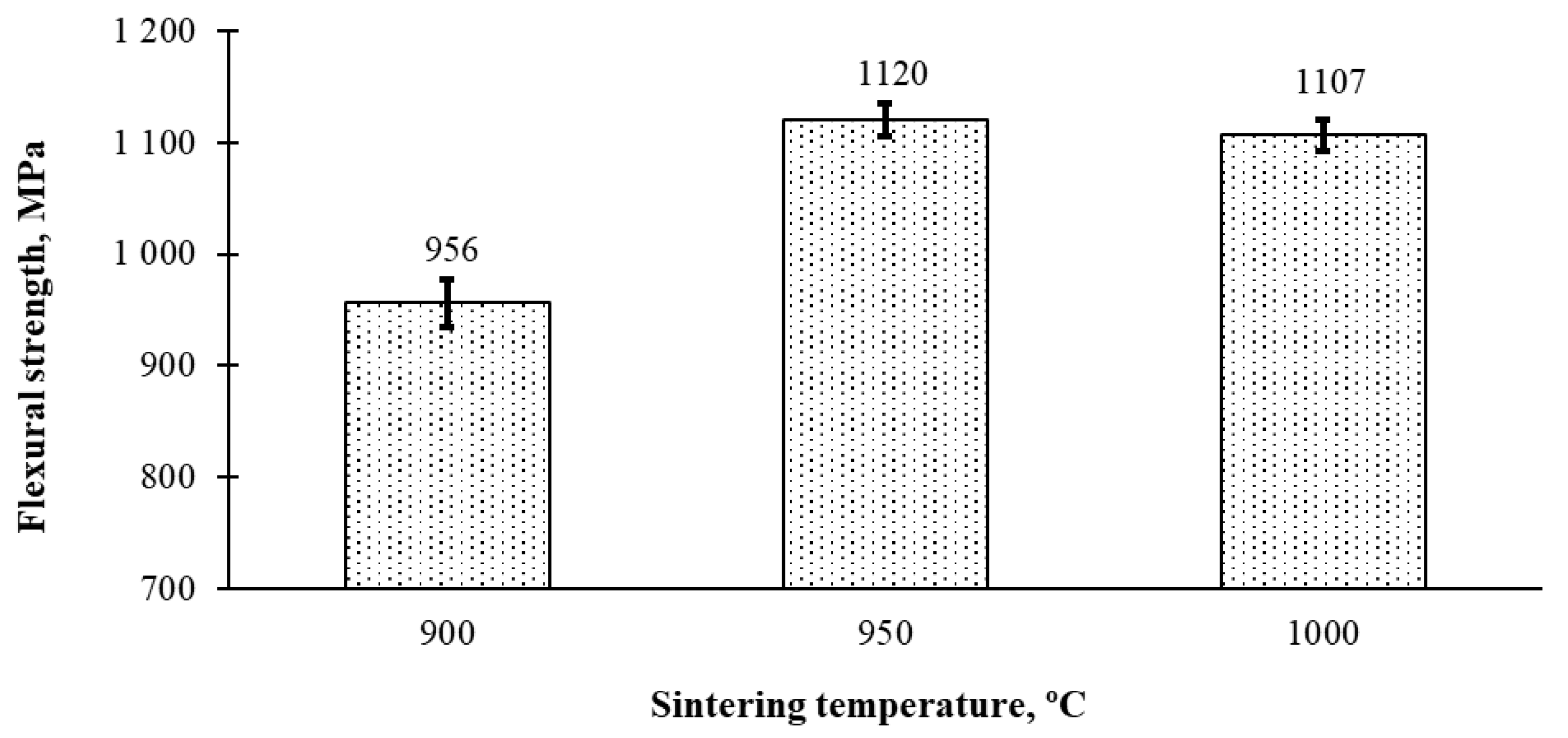
3.1. Density and Hardness
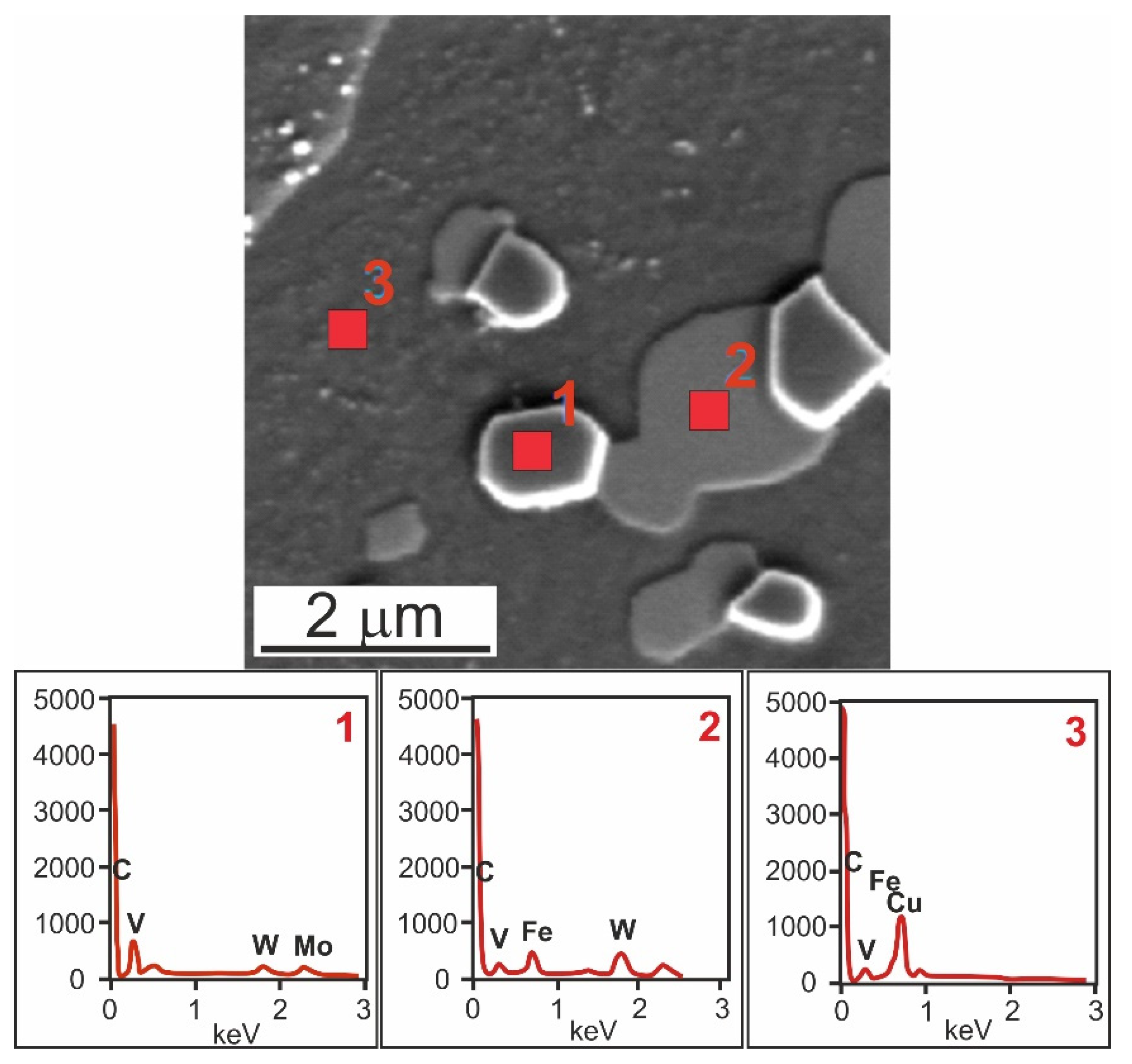
3.2. Microstructure
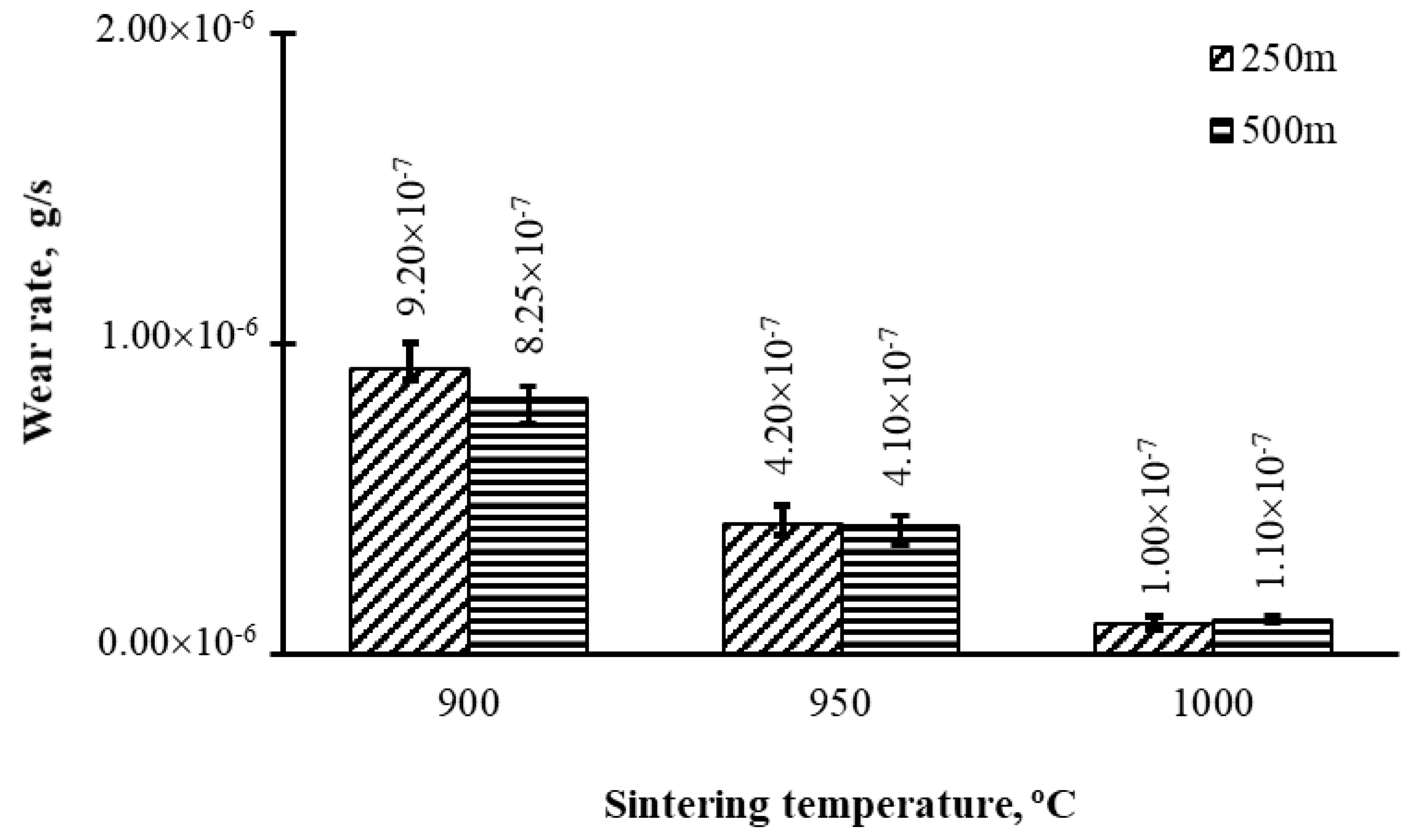
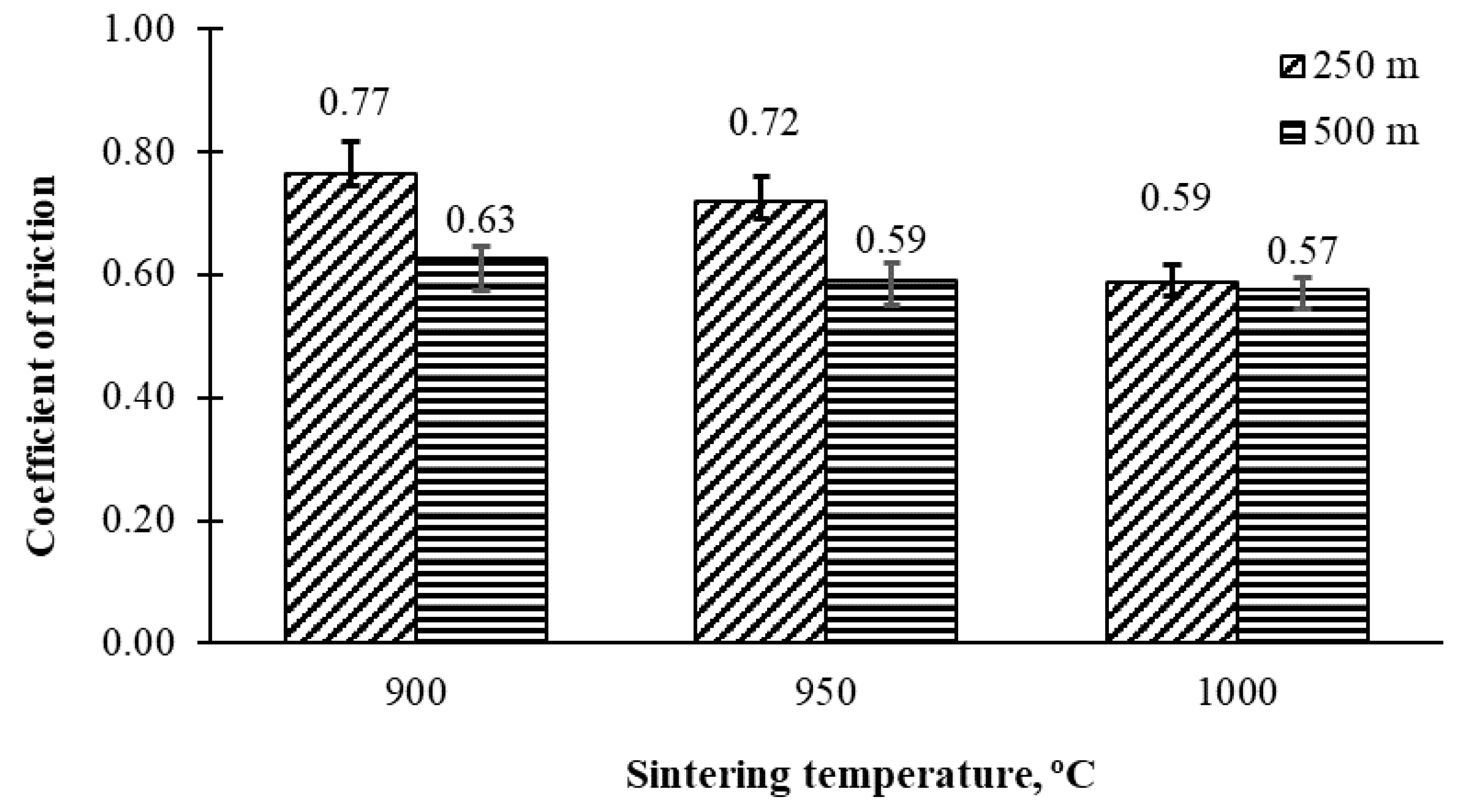
3.3. Tribological Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wright, C.S. The production and application of PM high-speed steels. Powder Metall. 1994, 3, 937–944. [Google Scholar]
- Dobrzański, L.A.; Matula, G.; Várez, A.; Levenfeld, B.; Torralba, J.M. Structure and mechanical properties of HSS HS6-5-2- and HS 12-1-5-5-type steel produced by modified powder injection moulding process. J. Mater. Process. Technol. 2004, 1, 157–158. [Google Scholar]
- Torralba, J.M.G.; Cambronero, J.M.; Ruiz-Pietro, M.M. Sinterability study of PM M2 and T15 HSS reinforced with tungsten and titanium carbides. Powder Metall. 1993, 36, 55–66. [Google Scholar] [CrossRef]
- Samal, P.K.; Newkirk, J.W. ASM Handbook: Powder Metallurgy; ASM International: Novelty, OH, USA, 2015; Volume 7, pp. 1–907. [Google Scholar]
- Farid, A. Microstructure evolution and wear properties of in situ synthesized TiB2 and TiC reinforced steel matrix composites. J. Alloy. Compd. 2008, 459, 491–497. [Google Scholar]
- Gordo, E.; Velasco, F.; Antón, N.; Torralba, J.M. Wear mechanisms in high speed steel reinforced with (NbC)p and (TaC)p MMCs. Wear 2000, 239, 251–259. [Google Scholar] [CrossRef]
- Shizhong, W.; Jinhua, Z.; Liujie, X. Effects of vanadium and carbon on microstructures and abrasive wear resistance of high speed steel. Tribol. Int. 2006, 39, 641–648. [Google Scholar]
- Madej, M.; Leżański, J. Copper infiltrated high speed steel based composites. Arch. Metall. Mater. 2005, 50, 871–877. [Google Scholar]
- Madej, M.; Leżański, J. The structure and properties of copper infiltrated HSS based. Arch. Metall. Mater. 2008, 53, 839–845. [Google Scholar]
- Madej, M. The tribological properties of high speed steel based composites. Arch. Metall. Mater. 2010, 55, 61–68. [Google Scholar]
- Dobrzanski, L.A.; Matula, G.; Varez, A.; Levenfeld, B.; Torralba, J.M. Fabrication methods and heat treatment conditions effect on tribological properties of high speed steels. J. Mater. Process. Technol. 2004, 157–158, 324–330. [Google Scholar] [CrossRef]
- Dobrzańśki, L.A. (Ed.) Fabrication, Structure, Properties and Application of Gradient Sintered Carbide-Steels with HS6-5-2 Matrix. In Powder Metallurgy—Fundamentals and Case Studies; IntechOpen: Rijeka, Croatia, 2017; pp. 199–222. [Google Scholar]
- Dobrzański, L.A.; Kloc-Ptaszna, A. Goals and contemporary position of powder metallurgy in products manufacturing. In Powder Metallurgy—Fundamentals and Case Studies; Dobrzańśki, L.A., Ed.; IntechOpen: Rijeka, Croatia, 2017; pp. 1–16. [Google Scholar]
- Hoyle, G. High. Speed Steels; Butterworth & Co: Cambridge, UK, 1998; pp. 121–130. [Google Scholar]
- Marnier, G.; Keller, C.; Noudem, J.; Hug, E. Functional properties of a spark plasma sintered ultrafine-grained 316L steel. Mater. Des. 2014, 63, 633–640. [Google Scholar] [CrossRef]
- Shashanka, R. Non-lubricated dry sliding wear behavior of spark plasma sintered nano-structured stainless steel. J. Mater. Environ. Sci. 2019, 10, 767–777. [Google Scholar]
- Hu, Z.; Ning, K.; Lu, K. Study of spark plasma sintered nanostructured ferritic steel alloy with silicon carbide addition. Mater. Sci. Eng. 2016, 670, 75–80. [Google Scholar] [CrossRef]
- Tanab, C.; Wangb, G.; Jib, L.; Tonga, Y.; Duanb, X. Investigation on 316L/W functionally graded materials fabricated by mechanical alloying and spark plasma sintering. J. Nucl. Mater. 2016, 469, 32–38. [Google Scholar]
- Gyu, L.; Khor, K.A.; Sundararajan, G. Boriding of mild steel using the spark plasma sintering (SPS) technique. Surf. Coat. Technol. 2002, 157, 226–230. [Google Scholar]
- Pellizzari, M.; Fedrizzi, A.; Zadra, A. Influence of processing parameters and particle size on the properties of hot work and high speed tool steels by Spark Plasma Sintering. Mater. Des. 2011, 32, 1796–1805. [Google Scholar] [CrossRef]
- Pellizzari, M.; Fedrizzi, A.; Zadra, A. Spark Plasma co-Sintering of hot work and high speed steel powders for fabrication of a novel tool steel with composite microstructure. Powder Technol. 2011, 214, 292–299. [Google Scholar] [CrossRef]
- Madej, M. Copper infiltrated high speed steel based composites with iron additions. Arch. Metall. Mater. 2009, 54, 1083–1091. [Google Scholar]









| C | Cr | Co | Mn | Mo | Ni | Si | V | W | O | Fe |
|---|---|---|---|---|---|---|---|---|---|---|
| 1.22 | 4.28 | 0.38 | 0.22 | 5.11 | 0.33 | 0.17 | 3.11 | 6.23 | 0.0626 | balance |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Madej, M.; Leszczyńska-Madej, B.; Garbiec, D. High Speed Steel with Iron Addition Materials Sintered by Spark Plasma Sintering. Metals 2020, 10, 1549. https://doi.org/10.3390/met10111549
Madej M, Leszczyńska-Madej B, Garbiec D. High Speed Steel with Iron Addition Materials Sintered by Spark Plasma Sintering. Metals. 2020; 10(11):1549. https://doi.org/10.3390/met10111549
Chicago/Turabian StyleMadej, Marcin, Beata Leszczyńska-Madej, and Dariusz Garbiec. 2020. "High Speed Steel with Iron Addition Materials Sintered by Spark Plasma Sintering" Metals 10, no. 11: 1549. https://doi.org/10.3390/met10111549
APA StyleMadej, M., Leszczyńska-Madej, B., & Garbiec, D. (2020). High Speed Steel with Iron Addition Materials Sintered by Spark Plasma Sintering. Metals, 10(11), 1549. https://doi.org/10.3390/met10111549







