Effects of Powder Atomisation on Microstructural and Mechanical Behaviour of L-PBF Processed Steels
Abstract
1. Introduction
2. Materials and Methods
2.1. Steel Powders and Laser Powder Bed Fusion
2.2. Material Characterisation
2.3. Thermal Analysis and Phase Modelling
2.4. Thermal Treatment
2.5. Mechanical Testing
3. Results and Discussion
3.1. Powder Properties
3.2. Microstructure Evolution
3.2.1. Relative Density
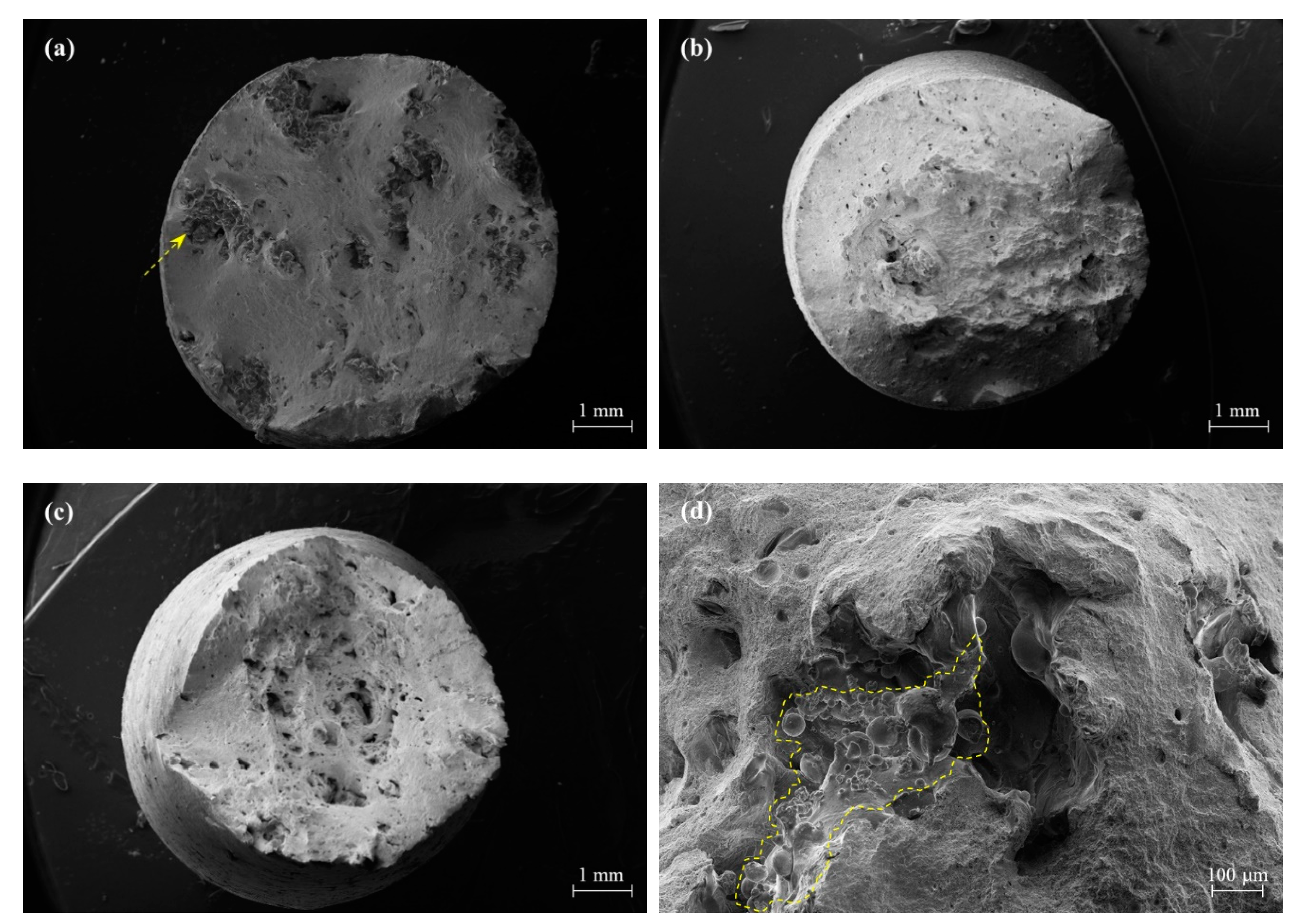
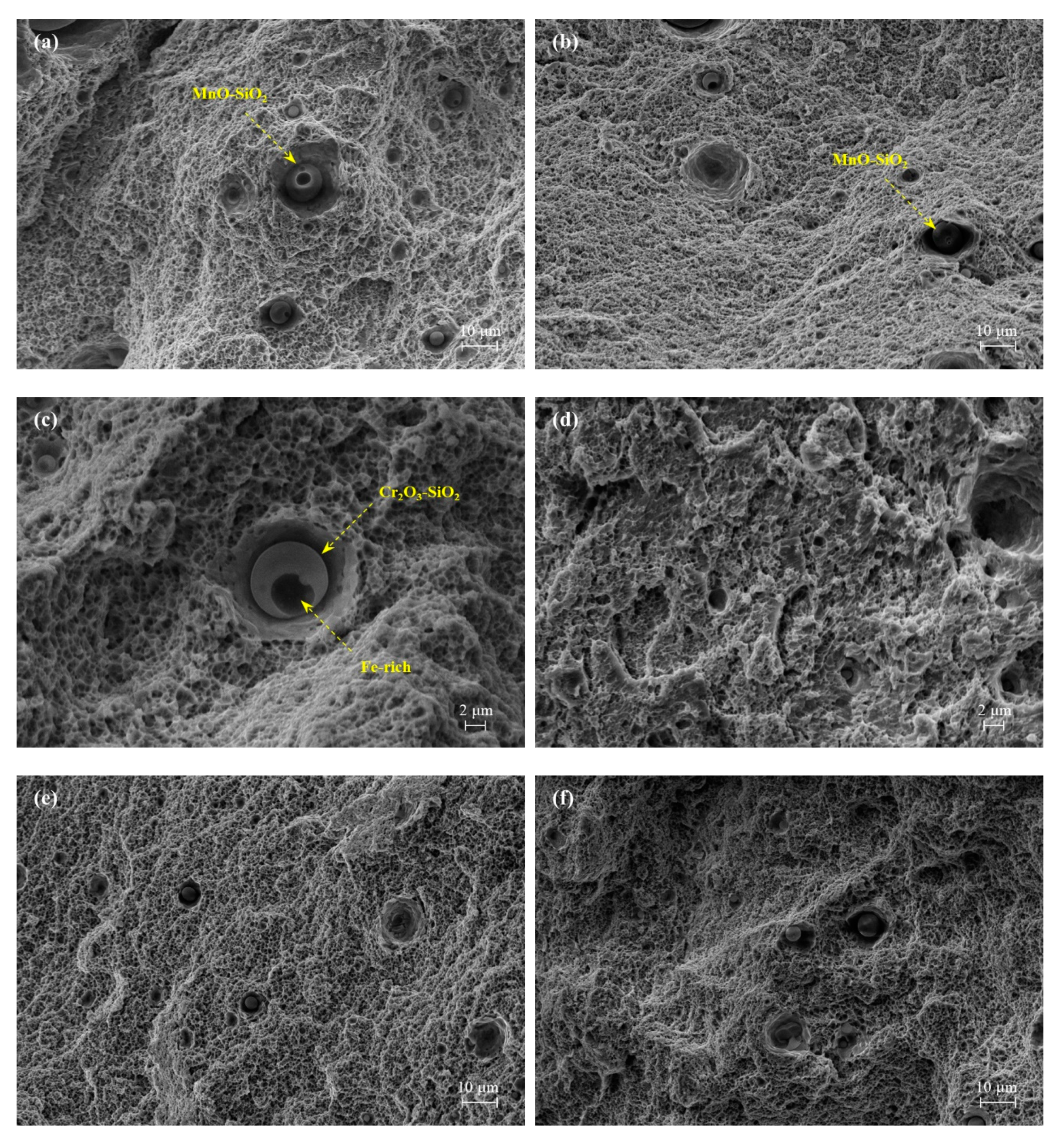
3.2.2. Nonmetallic Inclusions
3.2.3. As-Built Microstructure
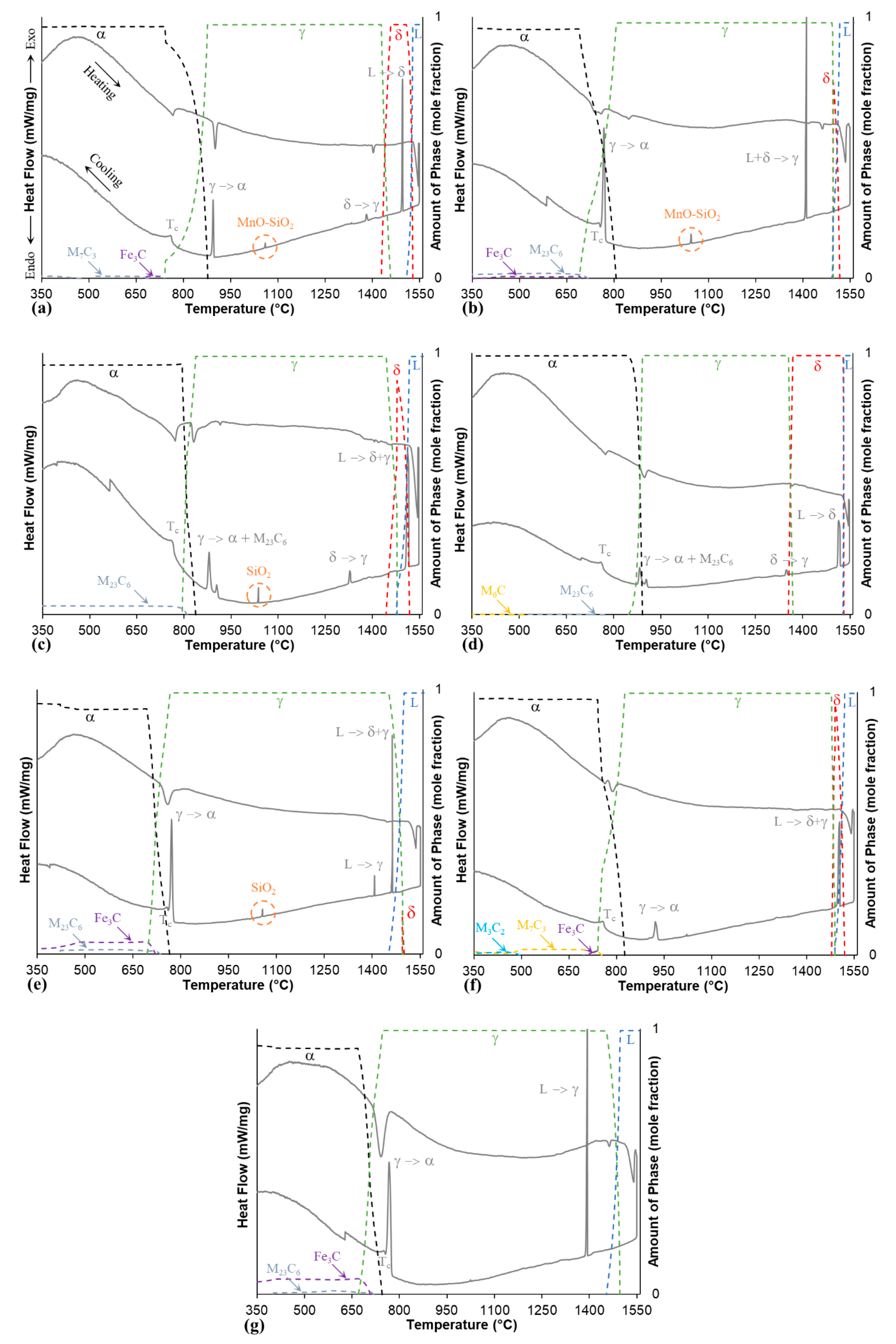
3.3. Thermal Analysis and Phase Modelling
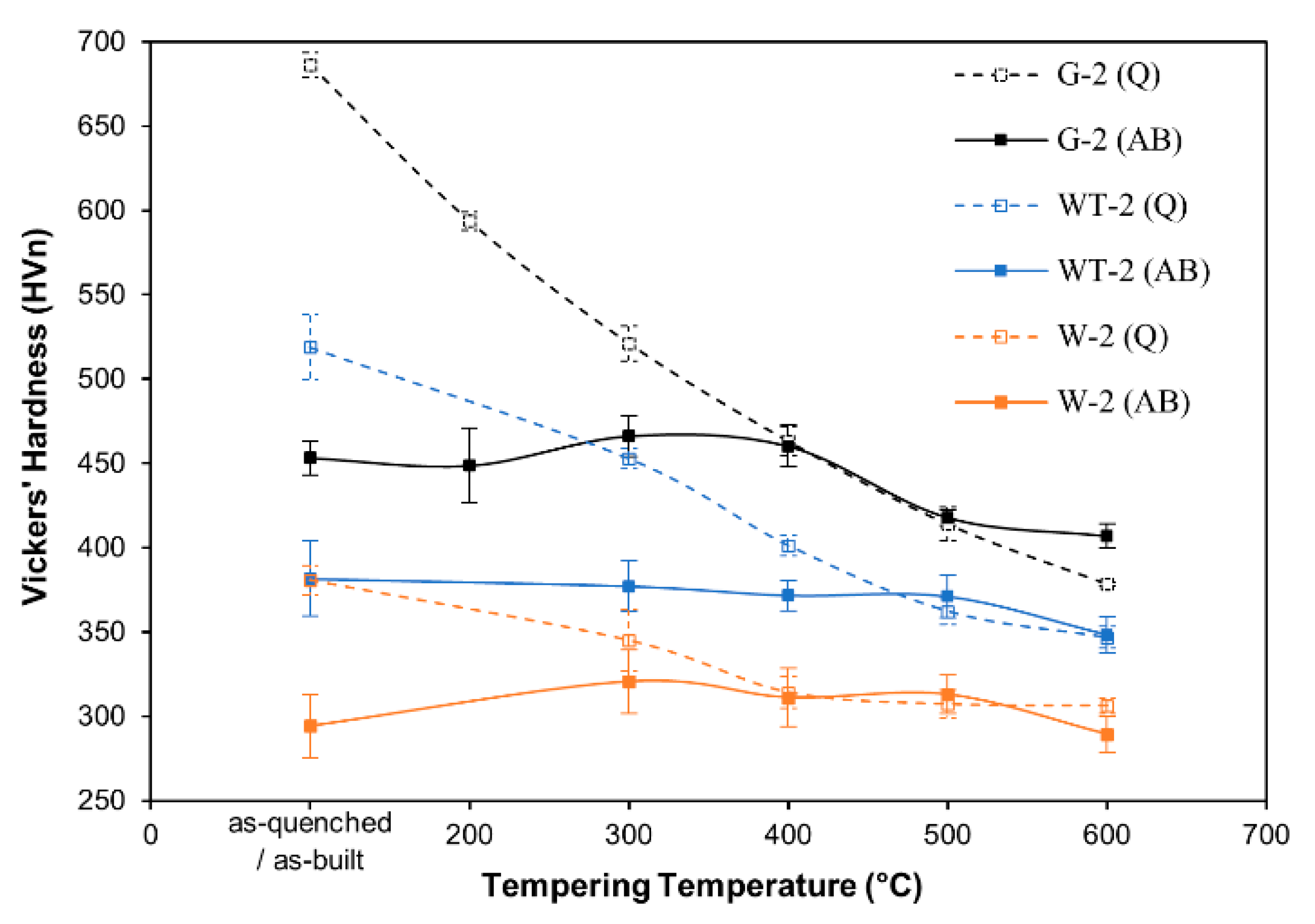
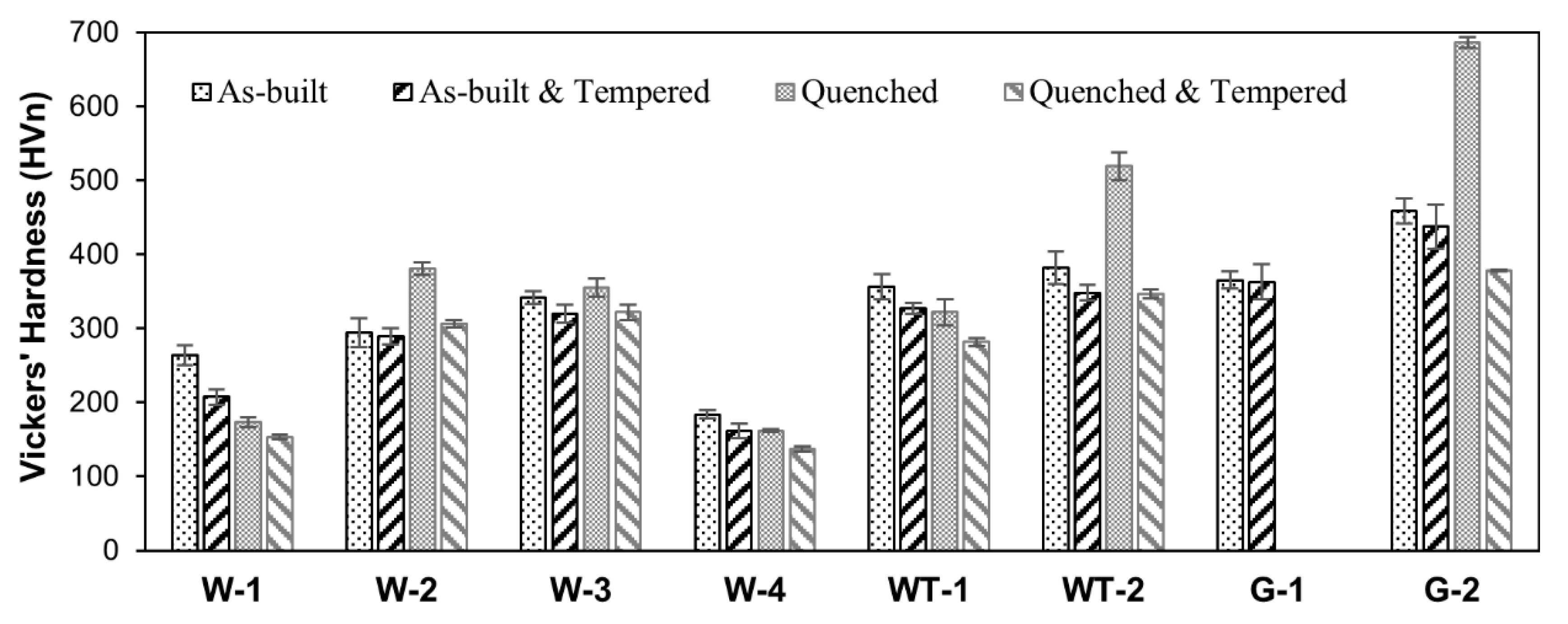
3.4. Thermal Treatment Strategies
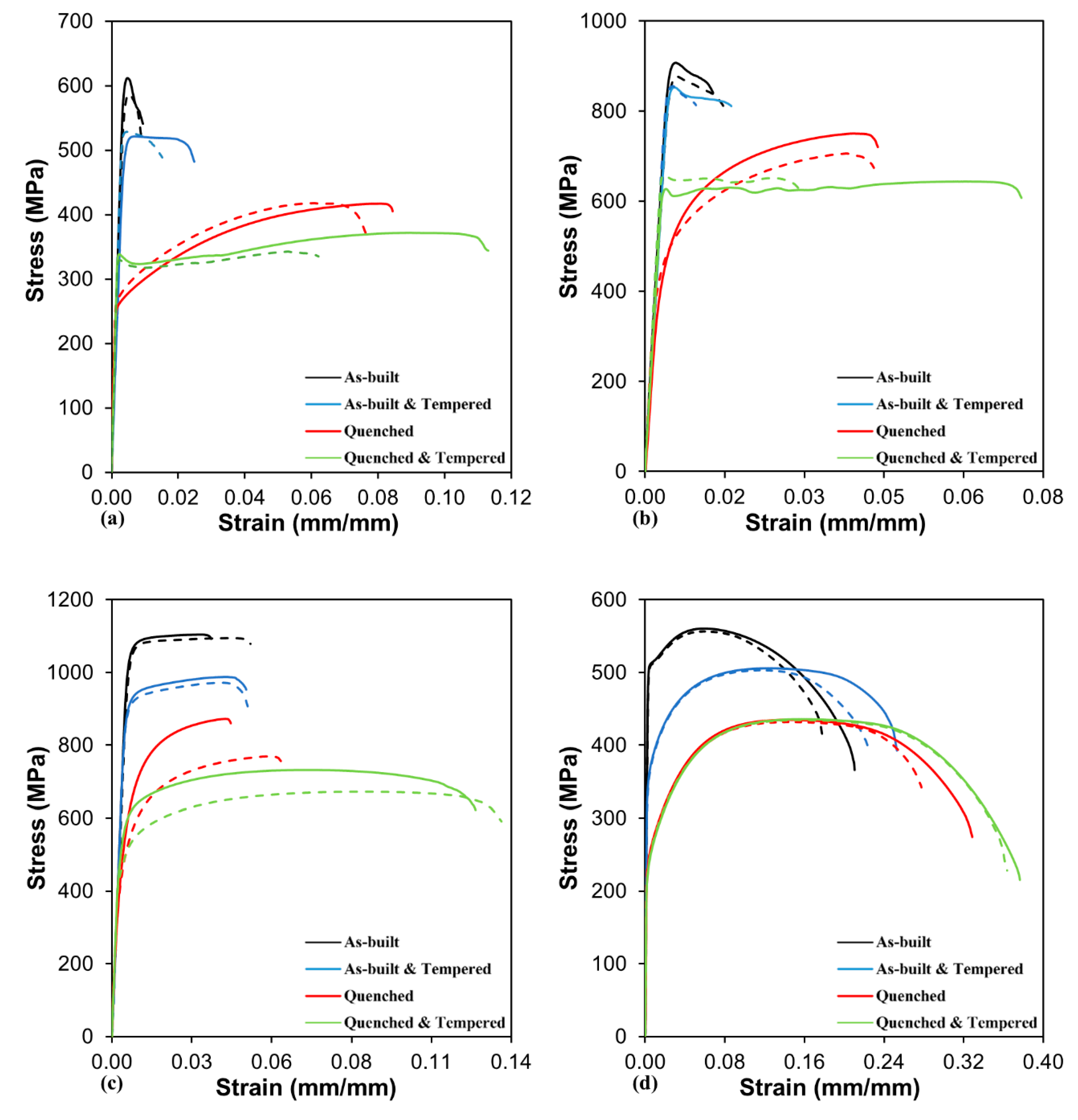
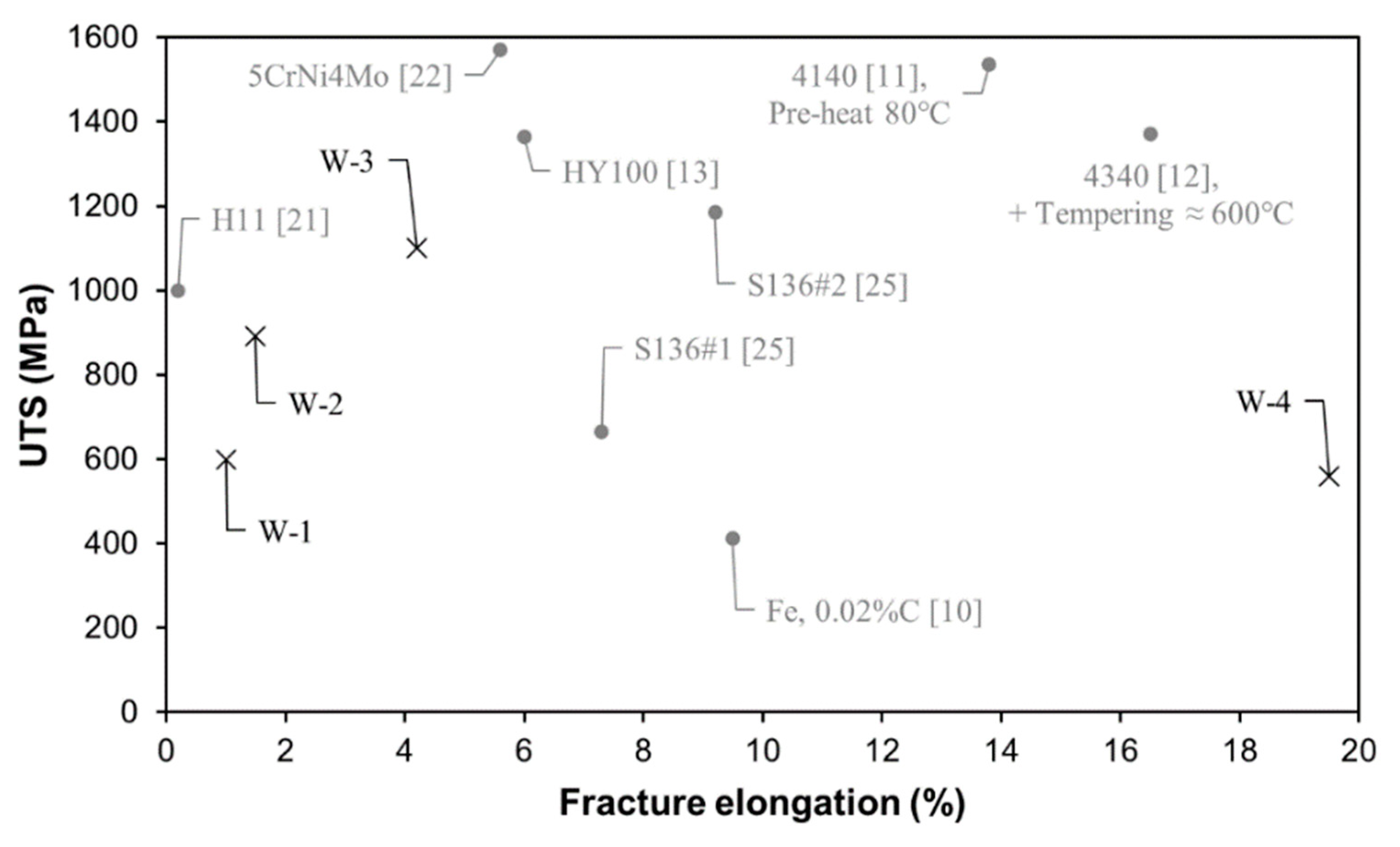
3.5. Tensile Properties
3.6. Fractographic Analysis
4. Concluding Remarks
- Production of steel powders by water atomisation, possibly followed by a post-treatment aimed at modifying powder morphology and its flowability, can lead to suitable feedstock materials for L-PBF. Indeed, despite a more irregular morphology of the water-atomised powders, after an accurate setting of the parameters for the powder bed deposition and for the laser melting, specimens having a density exceeding 99.5% could be produced.
- Depending on steel composition and microstructure achieved after the rapid solidification and cooling upon L-PBF processing, hardness values ranging from 182 up to 490 HV were achieved in the investigated steels in as-built condition.
- The hardenable steels, containing suitable amounts of C and of alloying elements (Cr, Mn, Mo, Ni), could benefit from post-processing quench and tempering heat treatments to achieve improved combination of properties. It was also demonstrated that the cooling and reheating cycles experienced by the steels during L-PBF processing resulted in a condition which is comparable to a tempering treatment performed around 400 °C.
- The low C W-1 and W-4 alloys showed a more homogeneous microstructure consisting of ferrite grains that provided their highest strength right in the as-built condition.
- The WA steel grades featured a higher oxygen content which resulted in a population of small micrometre- and submicrometric-size oxide inclusions. Their presence was detected on fracture surface of broken tensile specimens as nucleating sites for micro-dimples activating a ductile fracture mechanism.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DebRoy, T.; Wei, H.L.; Zuback, J.S.; Mukherjee, T.; Elmer, J.W.; Milewski, J.O.; Beese, A.M.; Wilson-Heid, A.; De, A.; Zhang, W. Additive manufacturing of metallic components—Process, structure and properties. Prog. Mater. Sci. 2018, 92, 112–224. [Google Scholar] [CrossRef]
- Sames, W.J.; List, F.A.; Pannala, S.; Dehoff, R.R.; Babu, S.S. The metallurgy and processing science of metal additive manufacturing. Int. Mater. Rev. 2016, 61, 315–360. [Google Scholar] [CrossRef]
- Heiden, M.J.; Deibler, L.A.; Rodelas, J.M.; Koepke, J.R.; Tung, D.J.; Saiz, D.J.; Jared, B.H. Evolution of 316L stainless steel feedstock due to laser powder bed fusion process. Addit. Manuf. 2019, 25, 84–103. [Google Scholar] [CrossRef]
- Casati, R.; Lemke, J.; Vedani, M. Microstructure and Fracture Behavior of 316L Austenitic Stainless Steel Produced by Selective Laser Melting. J. Mater. Sci. Technol. 2016, 32, 738–744. [Google Scholar] [CrossRef]
- Zhang, B.; Dembinski, L.; Coddet, C. The study of the laser parameters and environment variables effect on mechanical properties of high compact parts elaborated by selective laser melting 316L powder. Mater. Sci. Eng. A 2013, 584, 21–31. [Google Scholar] [CrossRef]
- LeBrun, T.; Nakamoto, T.; Horikawa, K.; Kobayashi, H. Effect of retained austenite on subsequent thermal processing and resultant mechanical properties of selective laser melted 17–4 PH stainless steel. Mater. Des. 2015, 81, 44–53. [Google Scholar] [CrossRef]
- Irrinki, H.; Jangam, J.S.D.; Pasebani, S.; Badwe, S.; Stitzel, J.; Kate, K.; Gulsoy, O.; Atre, S.V. Effects of particle characteristics on the microstructure and mechanical properties of 17-4 PH stainless steel fabricated by laser-powder bed fusion. Powder Technol. 2018, 331, 192–203. [Google Scholar] [CrossRef]
- Casalino, G.; Campanelli, S.L.; Contuzzi, N.; Ludovico, A.D. Experimental investigation and statistical optimisation of the selective laser melting process of a maraging steel. Opt. Laser Technol. 2015, 65, 151–158. [Google Scholar] [CrossRef]
- Casati, R.; Lemke, J.; Tuissi, A.; Vedani, M. Aging Behaviour and Mechanical Performance of 18-Ni 300 Steel Processed by Selective Laser Melting. Metals 2016, 6, 218. [Google Scholar] [CrossRef]
- Song, B.; Dong, S.; Deng, S.; Liao, H.; Coddet, C. Microstructure and tensile properties of iron parts fabricated by selective laser melting. Opt. Laser Technol. 2014, 56, 451–460. [Google Scholar] [CrossRef]
- Wang, W.; Kelly, S. A Metallurgical Evaluation of the Powder-Bed Laser Additive Manufactured 4140 Steel Material. JOM 2016, 68, 869–875. [Google Scholar] [CrossRef]
- Jelis, E.; Clemente, M.; Kerwien, S.; Ravindra, N.M.; Hespos, M.R. Metallurgical and Mechanical Evaluation of 4340 Steel Produced by Direct Metal Laser Sintering. JOM 2015, 67, 582–589. [Google Scholar] [CrossRef]
- Dilip, J.J.S.; Ram, G.D.J.; Starr, T.L.; Stucker, B. Selective laser melting of HY100 steel: Process parameters, microstructure and mechanical properties. Addit. Manuf. 2017, 13, 49–60. [Google Scholar] [CrossRef]
- Casati, R.; Coduri, M.; Lecis, N.; Andrianopoli, C.; Vedani, M. Microstructure and mechanical behavior of hot-work tool steels processed by Selective Laser Melting. Mater. Charact. 2018, 137, 50–57. [Google Scholar] [CrossRef]
- Krell, J.; Röttger, A.; Geenen, K.; Theisen, W. General investigations on processing tool steel X40CrMoV5-1 with selective laser melting. J. Mater. Process. Technol. 2018, 255, 679–688. [Google Scholar] [CrossRef]
- Schmitt, M.; Schlick, G.; Seidel, C.; Reinhart, G. Examination of the processability of 16MnCr5 by means of laser powder bed fusion. Procedia CIRP 2018, 74, 76–81. [Google Scholar] [CrossRef]
- Åsberg, M.; Fredriksson, G.; Hatami, S.; Fredriksson, W.; Krakhmalev, P. Influence of post treatment on microstructure, porosity and mechanical properties of additive manufactured H13 tool steel. Mater. Sci. Eng. A 2019, 742, 584–589. [Google Scholar] [CrossRef]
- Feuerhahn, F.; Schulz, A.; Seefeld, T.; Vollertsen, F. Microstructure and Properties of Selective Laser Melted High Hardness Tool Steel. Phys. Procedia 2013, 41, 843–848. [Google Scholar] [CrossRef]
- Sander, J.; Hufenbach, J.; Giebeler, L.; Wendrock, H.; Kühn, U.; Eckert, J. Microstructure and properties of FeCrMoVC tool steel produced by selective laser melting. Mater. Des. 2016, 89, 335–341. [Google Scholar] [CrossRef]
- Liu, Z.H.; Zhang, D.Q.; Chua, C.K.; Leong, K.F. Crystal structure analysis of M2 high speed steel parts produced by selective laser melting. Mater. Charact. 2013, 84, 72–80. [Google Scholar] [CrossRef]
- Huber, F.; Bischof, C.; Hentschel, O.; Heberle, J.; Zettl, J.; Nagulin, K.Y.; Schmidt, M. Laser beam melting and heat-treatment of 1.2343 (AISI H11) tool steel–microstructure and mechanical properties. Mater. Sci. Eng. A 2019, 742, 109–115. [Google Scholar] [CrossRef]
- Chen, H.; Gu, D.; Dai, D.; Ma, C.; Xia, M. Microstructure and composition homogeneity, tensile property, and underlying thermal physical mechanism of selective laser melting tool steel parts. Mater. Sci. Eng. A 2017, 682, 279–289. [Google Scholar] [CrossRef]
- Rännar, L.; Glad, A.; Gustafson, C. Efficient cooling with tool inserts manufactured by electron beam melting. Rapid Prototyp. J. 2007, 13, 128–135. [Google Scholar] [CrossRef]
- Brøtan, V.; Berg, O.Å.; Sørby, K. Additive Manufacturing for Enhanced Performance of Molds. Procedia CIRP 2016, 54, 186–190. [Google Scholar] [CrossRef]
- Zhou, Y.; Duan, L.; Ji, X.; Wen, S.; Wei, Q.; Ye, F.; Shi, Y. Comparisons on microstructure, mechanical and corrosion resistant property of S136 mold steel processed by selective laser melting from two pre-alloy powders with trace element differences. Opt. Laser Technol. 2018, 108, 81–89. [Google Scholar] [CrossRef]
- EU Science Hub-Raw Materials Information System (RMIS). Available online: https://rmis.jrc.ec.europa.eu/?page=crm-list-2017-09abb4 (accessed on 10 October 2020).
- Pinkerton, A.J.; Li, L. Direct additive laser manufacturing using gas- and water-atomised H13 tool steel powders. Int. J. Adv. Manuf. Technol. 2005, 25, 471–479. [Google Scholar] [CrossRef]
- Li, R.; Shi, Y.; Wang, Z.; Wang, L.; Liu, J.; Jiang, W. Densification behavior of gas and water atomized 316L stainless steel powder during selective laser melting. Appl. Surf. Sci. 2010, 256, 4350–4356. [Google Scholar] [CrossRef]
- Hoeges, S.; Zwiren, A.; Schade, C. Additive manufacturing using water atomized steel powders. Met. Powder Rep. 2017, 72, 111–117. [Google Scholar] [CrossRef]
- Shibata, H.; Kimura, K.; Tanaka, T.; Kitamura, S. Mechanism of Change in Chemical Composition of Oxide Inclusions in Fe–Cr Alloys Deoxidized with Mn and Si by Heat Treatment at 1473 K. ISIJ Int. 2011, 51, 1944–1950. [Google Scholar] [CrossRef]
- Shibata, H.; Tanaka, T.; Kimura, K.; Kitamura, S.-Y. Composition change in oxide inclusions of stainless steel by heat treatment. Ironmak. Steelmak. 2010, 37, 522–528. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kim, S.-J.; Shibata, H.; Kitamura, S. Reaction between MnO–SiO2–FeO Oxide and Fe–Mn–Si Solid Alloy during Heat Treatment. ISIJ Int. 2014, 54, 2144–2153. [Google Scholar] [CrossRef]
- Ren, Y.; Zhang, L.; Pistorius, P.C. Transformation of Oxide Inclusions in Type 304 Stainless Steels during Heat Treatment. Metall. Mater. Trans. B 2017, 48, 2281–2292. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, S.; Li, J.; Liu, C.; Hao, W. Evolution of Interfacial Features of MnO-SiO2 Type Inclusions/Steel Matrix during Isothermal Heating at Low Temperatures. High Temp. Mater. Process. 2019, 38, 347–353. [Google Scholar] [CrossRef]
- Raju, S.; Jeya Ganesh, B.; Rai, A.K.; Mythili, R.; Saroja, S.; Raj, B. A study on martensitic phase transformation in 9Cr–1W–0.23V–0.063Ta–0.56Mn–0.09C–0.02N (wt.%) reduced activation steel using differential scanning calorimetry. J. Nucl. Mater. 2010, 405, 59–69. [Google Scholar] [CrossRef]
- Van Genderen, M.J.; Isac, M.; Böttger, A.; Mittemeijer, E.J. Aging and tempering behavior of iron-nickel-carbon and iron-carbon martensite. Metall. Mater. Trans. A 1997, 28, 545–561. [Google Scholar] [CrossRef]
- Naraghi, R.; Selleby, M.; Ågren, J. Thermodynamics of stable and metastable structures in Fe–C system. Calphad 2014, 46, 148–158. [Google Scholar] [CrossRef]










| Alloy | Chemical Composition | Production Process | ||||||
|---|---|---|---|---|---|---|---|---|
| C | Cr | Mo | Si | Mn | Ni | O | ||
| W-1 | 0.05 | 0.70 | - | 0.50 | 0.89 | - | 0.509 | WA |
| W-2 | 0.12 | 0.44 | 0.27 | 0.36 | 0.58 | 1.77 | 0.395 | |
| W-3 | 0.15 | 3.10 | 0.50 | 0.33 | - | - | 0.254 | |
| W-4 | 0.005 | 3.10 | 0.51 | 0.03 | 0.06 | 0.048 | 0.159 | WA + OR |
| WT-1 | 0.15 | 3.10 | 0.50 | 0.33 | - | - | 0.250 | WA + PT |
| WT-2 | 0.32 | 0.82 | 0.24 | 0.44 | - | 1.89 | 0.262 | |
| G-1 | 0.15 | 1.27 | - | 0.08 | 0.54 | - | 0.061 | GA |
| G-2 | 0.37 | 0.52 | 0.26 | 0.07 | 0.64 | 1.83 | 0.082 | |
| Processing Parameters | Renishaw AM250 | EOS M270 |
|---|---|---|
| Laser power, W | 200 | 185 |
| Layer thickness, μm | 40 | 40 |
| Hatching distance, μm | 70–125 | 50–100 |
| Point distance, μm | 30–60 | - |
| Exposure time, μs | 80 | |
| Scanning velocity, mm/s | - | 450–750 |
| Alloy | Production Process | Particle Size Distribution (μm) | Flow (s/50 g) | ρapp. (g/cm3) | ρtap (g/cm3) | Relative Density (%) | ||
|---|---|---|---|---|---|---|---|---|
| D10 | D50 | D90 | ||||||
| W-1 | WA | 12 | 32 | 65 | 36.0 | 2.80 | 3.62 | 97.81 |
| W-2 | 15 | 35 | 65 | 30.7 | 2.70 | 3.39 | 98.54 | |
| W-3 | 29 | 50 | 80 | 31.0 | 2.80 | 3.47 | 99.80 | |
| W-4 | WA + OR | 35 | 59 | 88 | 28.0 | 2.80 | 3.53 | 99.88 |
| WT-1 | WA + PT | 26 | 46 | 72 | 18.0 | 3.60 | 4.37 | 99.84 |
| WT-2 | 21 | 37 | 59 | 21.0 | 3.44 | 4.37 | 99.55 | |
| G-1 | GA * | 22 | 38 | 57 | 12.0 | 4.36 | 5.05 | 99.93 |
| G-2 | 99.94 | |||||||
| Alloy | Method * | Solidification | Tγ −> α (°C) | TC (°C) | ||
|---|---|---|---|---|---|---|
| Temperature Range(°C) | ΔT(°C) | Mode | ||||
| W-1 | DSC | 1497.3–1488.2 | 9.1 | 898.1 | 760.0 | |
| TC | 1528.6–1508.0 | 20.6 | L → L+δ → δ | 877.1 | - | |
| W-2 | DSC | 1411.6–1403.5 | 8.1 | 775.1 | 753.8 | |
| TC | 1517.4–1492.3 | 25.1 | L → L+δ+γ → γ | 807.2 | - | |
| W-3 | DSC | 1518.1–1499.1 | 19 | 912.5 | 758.8 | |
| TC | 1518.5–1477.9 | 40.6 | L → L+δ+γ → γ | 838.1 | - | |
| W-4 | DSC | 1520.6–1506.7 | 13.9 | 909.9 | 757.9 | |
| TC | 1530.6–1528.1 | 2.5 | L → L+δ → δ | 890.8 | - | |
| WT-2 | DSC | 1464.9–1457.4 | 7.5 | 783.2 | 756.4 | |
| TC | 1499.9–1451.3 | 48.6 | L → L+δ+γ → γ | 765.5 | - | |
| G-1 | DSC | 1504.8–1490.7 | 14.1 | 934.1 | 755.9 | |
| TC | 1520.7–1489.6 | 31.1 | L → L+δ+γ → γ | 825.7 | - | |
| G-2 | DSC | 1393.6–1385.5 | 8.1 | 776.9 | 750.8 | |
| TC | 1496.4–1453.8 | 42.6 | L → L+γ → γ | 745.8 | - | |
| Alloy | As-Built | As-Built & Tempered | Quenched | Quenched & Tempered |
|---|---|---|---|---|
| W-1 | 263.65 ± 12.99 | 207.16 ± 10.14 | 173.02 ± 6.250 | 153.42 ± 3.310 |
| W-2 | 294.15 ± 18.93 | 289.29 ± 10.98 | 380.66 ± 8.680 | 306.16 ± 4.610 |
| W-3 | 342.12 ± 8.570 | 319.87 ± 11.93 | 355.36 ± 12.51 | 321.68 ± 10.65 |
| W-4 | 183.77 ± 5.470 | 161.17 ± 9.560 | 161.56 ± 1.850 | 136.98 ± 3.780 |
| WT-1 | 356.29 ± 16.83 | 326.95 ± 7.750 | 321.82 ± 17.93 | 281.62 ± 5.680 |
| WT-2 | 381.60 ± 22.24 | 348.30 ± 10.57 | 518.96 ± 18.91 | 346.55 ± 6.450 |
| G-1 | 365.22 ± 11.20 | 362.86 ± 24.02 | - | - |
| G-2 | 458.97 ± 16.83 | 437.59 ± 29.77 | 686.29 ± 7.330 | 378.43 ± 1.020 |
| Alloy | Property | As-Built | As-Built & Tempered | Quenched | Quenched & Tempered |
|---|---|---|---|---|---|
| W-1 | YS (MPa) | 599 ± 12 | 524 ± 4 | 274 ± 6 | 329 ± 4 |
| UTS (MPa) | 599 ± 12 | 525 ± 4 | 418 ± 1 | 357 ± 15 | |
| Fracture elongation (%) | 0.92 ± 0.03 | 2.00 ± 0.48 | 8.06 ± 0.37 | 8.75 ± 2.55 | |
| W-2 | YS (MPa) | 891 ± 16 | 852 ± 1 | 493 ± 22 | 632 ± 17 |
| UTS (MPa) | 892 ± 15 | 854 ± 1 | 729 ± 23 | 652 ± 7 | |
| Fracture elongation (%) | 1.48 ± 0.12 | 1.37 ± 0.36 | 4.63 ± 0.04 | 5.36 ± 2.21 | |
| W-3 | YS (MPa) | 1062 ± 3 | 909 ± 7 | 507 ± 40 | 537 ± 47 |
| UTS (MPa) | 1100 ± 5 | 980 ± 8 | 822 ± 52 | 703 ± 30 | |
| Fracture elongation (%) | 4.16 ± 0.69 | 4.73 ± 0.02 | 5.10 ± 0.94 | 13.20 ± 0.46 | |
| W-4 | YS (MPa) | 510 ± 2 | 356 ± 1 | 242 ± 1 | 233 ± 1 |
| UTS (MPa) | 559 ± 2 | 505 ± 2 | 434 ± 2 | 436 ± 1 | |
| Fracture elongation (%) | 19.42 ± 1.59 | 23.90 ± 1.39 | 30.29 ± 2.55 | 37.00 ± 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelwahed, M.; Casati, R.; Bengtsson, S.; Larsson, A.; Riccio, M.; Vedani, M. Effects of Powder Atomisation on Microstructural and Mechanical Behaviour of L-PBF Processed Steels. Metals 2020, 10, 1474. https://doi.org/10.3390/met10111474
Abdelwahed M, Casati R, Bengtsson S, Larsson A, Riccio M, Vedani M. Effects of Powder Atomisation on Microstructural and Mechanical Behaviour of L-PBF Processed Steels. Metals. 2020; 10(11):1474. https://doi.org/10.3390/met10111474
Chicago/Turabian StyleAbdelwahed, Marawan, Riccardo Casati, Sven Bengtsson, Anna Larsson, Martina Riccio, and Maurizio Vedani. 2020. "Effects of Powder Atomisation on Microstructural and Mechanical Behaviour of L-PBF Processed Steels" Metals 10, no. 11: 1474. https://doi.org/10.3390/met10111474
APA StyleAbdelwahed, M., Casati, R., Bengtsson, S., Larsson, A., Riccio, M., & Vedani, M. (2020). Effects of Powder Atomisation on Microstructural and Mechanical Behaviour of L-PBF Processed Steels. Metals, 10(11), 1474. https://doi.org/10.3390/met10111474








