Effects of Endurance Exercise Training and Crataegus Extract WS® 1442 in Patients with Heart Failure with Preserved Ejection Fraction – A Randomized Controlled Trial
Abstract
:1. Introduction
2. Method, Results, Discussion
2.1. Method
2.1.1. Design
2.1.2. Patients
2.1.3. Treatments
2.1.4. Outcomes
2.1.5. Analysis
2.2. Results
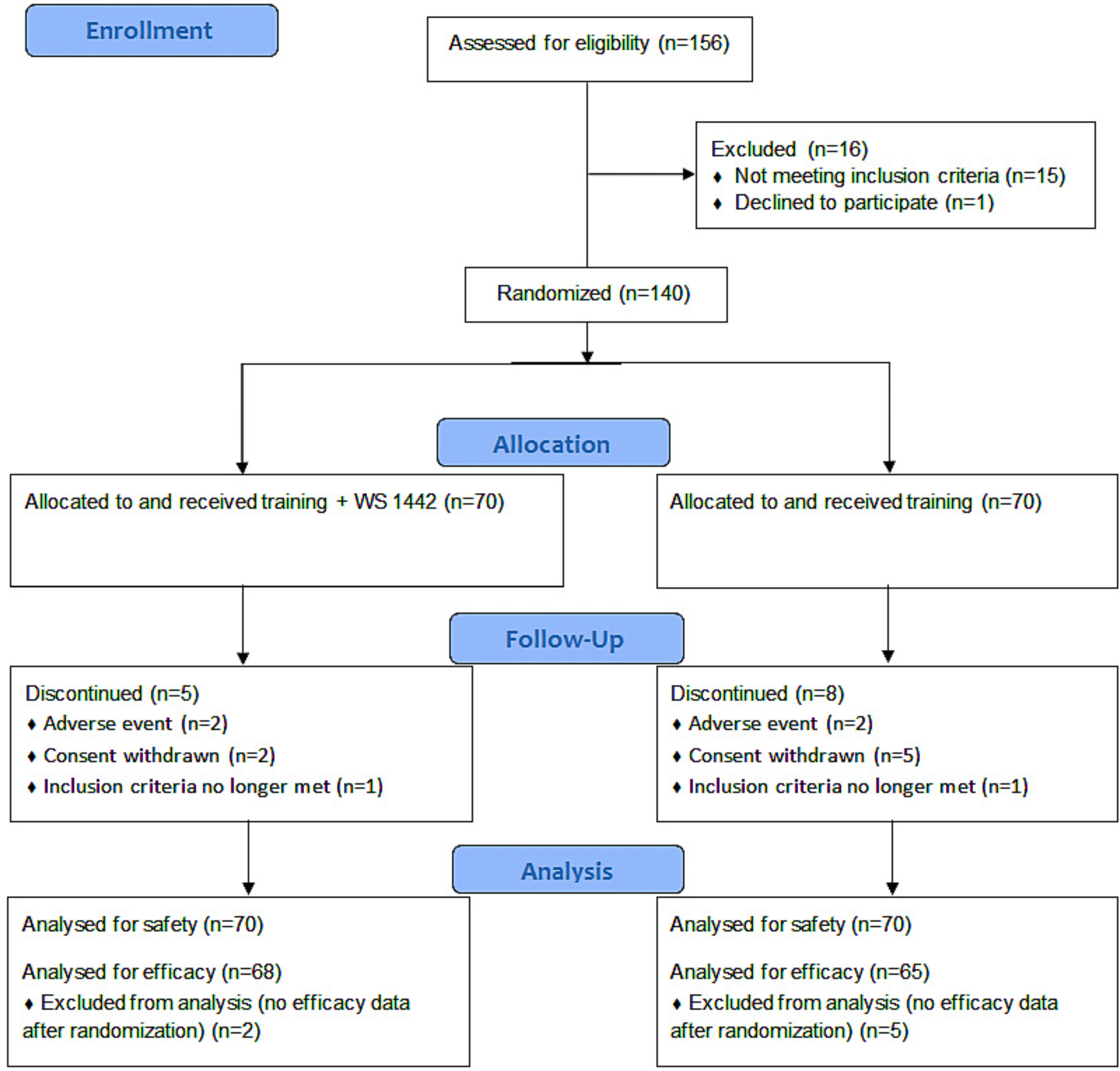
2.2.1. Patients and baseline data
2.2.2. Study treatment
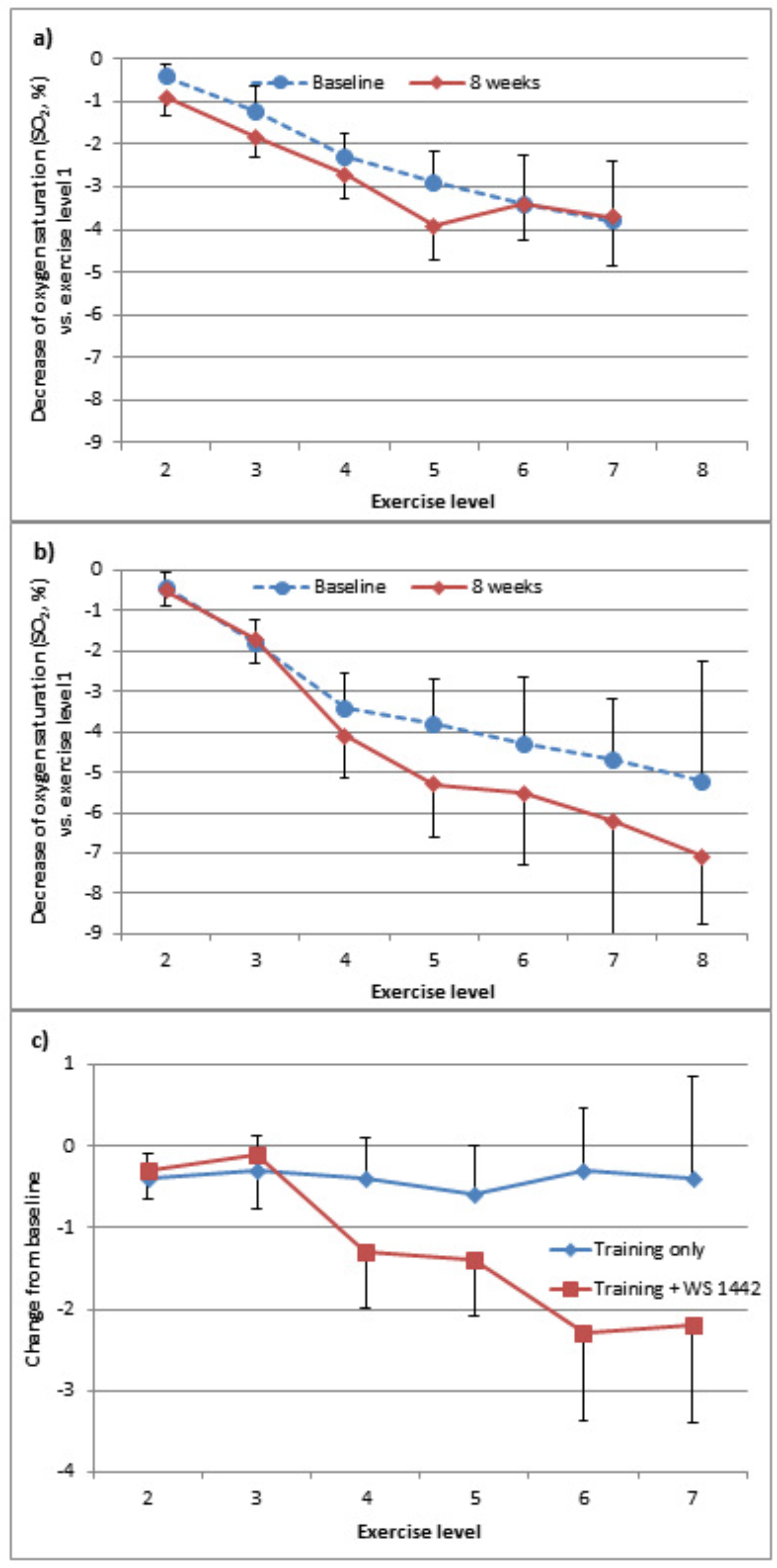
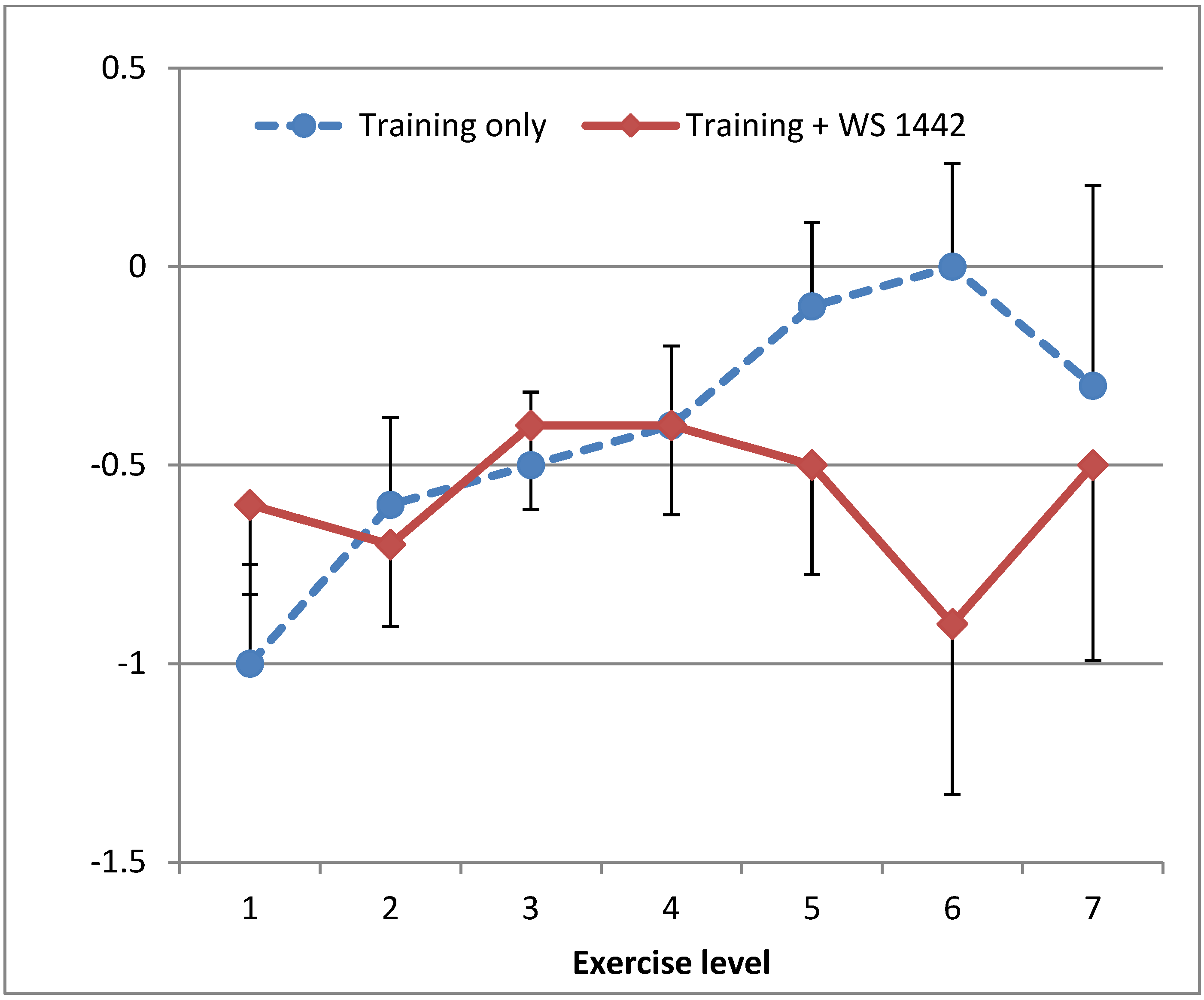
2.2.3. Treatment effects in training only group
| percent of patients or mean value (± SD) | Treatment Group | |
|---|---|---|
| Training + WS 1442 (n=70) | Training (n=70) | |
| Age (years) | 62 ± 9.0 | 62 ± 7.8 |
| Cardiovascular disease/ risk factor | ||
| Hypertension | 91 % | 87 % |
| Ischemic heart disease | 43 % | 36 % |
| Mitral insufficiency | 11 % | 10 % |
| Hyperlipidaemia | 33 % | 33 % |
| Diabetes mellitus | 16 % | 9 % |
| Thyroid disorder | 17 % | 13 % |
| Smoking | 9 % | 9 % |
| Body mass index (kg/m2) | 30.2 ± 5.5 | 29.7 ± 4.7 |
| Concomitant medication | 99 % | 96 % |
| ACE-inhibitors, AT2-antagonists | 69 % | 64 % |
| Beta blockers | 64 % | 57 % |
| Platelet aggregation inhibitors, coumarines | 64 % | 49 % |
| Lipid lowering drugs | 61 % | 56 % |
| Calcium-antagonists | 27 % | 21 % |
| Anti-diabetics | 20 % | 11 % |
| Nitrates, vasodilating drugs, other cardiacs | 19 % | 11 % |
| Diuretics | 17 % | 10 % |
| Thyroid therapeutics | 23 % | 14 % |
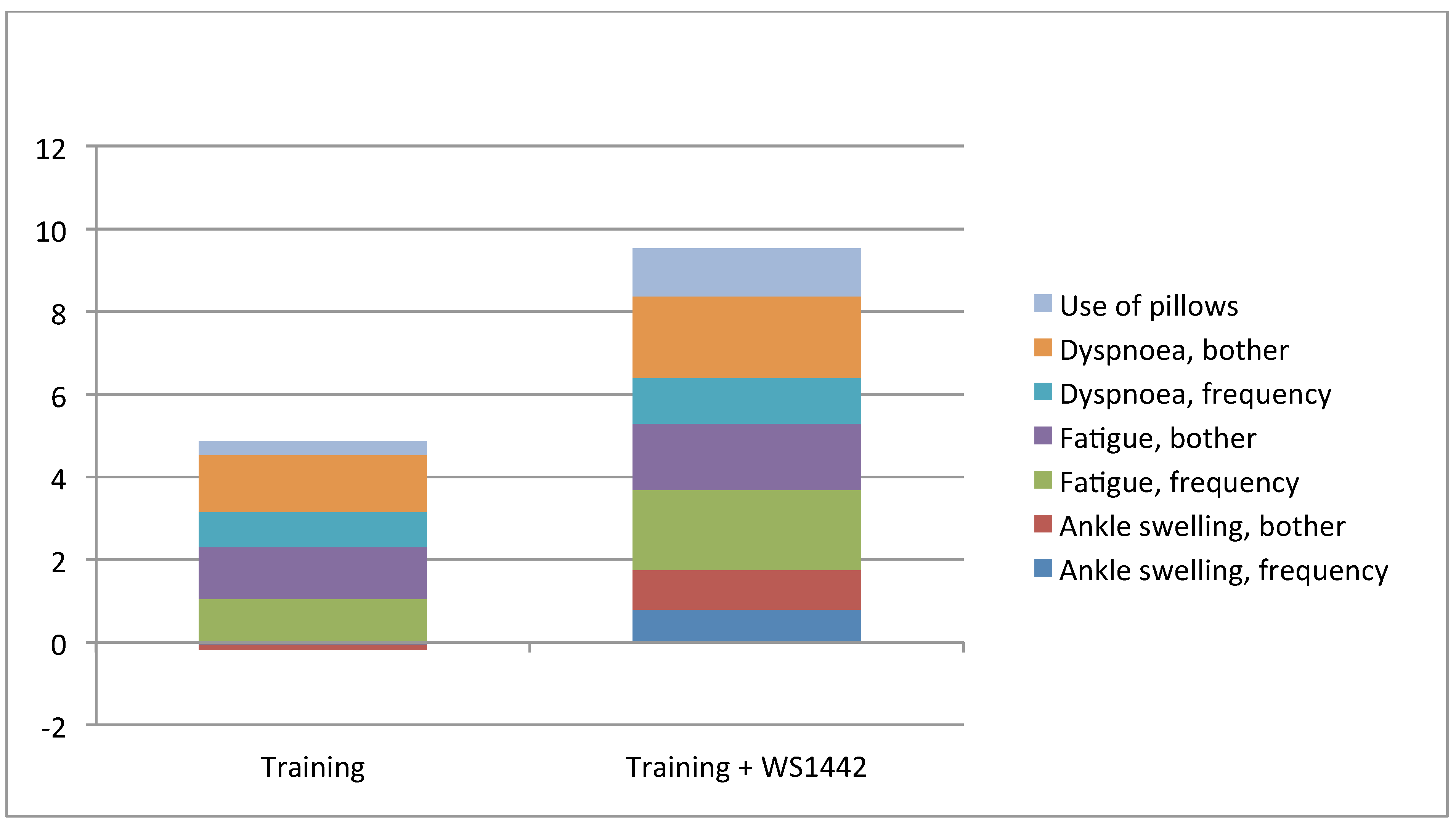
2.2.4. Effects of WS 1442
| Treatment | |||||
|---|---|---|---|---|---|
| Training only | Training + WS 1442 | ||||
| Baseline | Change | Baseline | Change | ||
| KCCQ | |||||
| Physical limitation | 85 ± 17 | 4 ± 15* | 82 ± 17 | 6 ± 15 | |
| Symptom stability | 57 ± 29 | 9 ± 35 | 60 ± 29 | 3 ± 29 | |
| Symptoms | 76 ± 18 | 5 ± 14* | 70 ± 22 | 10 ± 17 | |
| Self-efficacy | 70 ± 21 | 2 ± 17 | 70 ± 20 | 3 ± 15 | |
| Social limitation | 79 ± 23 | 5 ± 16* | 76 ± 25 | 9 ± 20 | |
| Quality of life | 71 ± 24 | 9 ± 17* | 69 ± 23 | 11 ± 18 | |
| Exercise capacity | |||||
| Freiburg Questionnaire of Physical Activity | 27 ± 23 | 14 ± 27* | 27 ± 26 | 14 ± 29 | |
| Time for 2 km walking test (min) | 21.4 ± 2.4 | -1.9 ± 2.0* | 23.0 ± 3.1 | -2.9 ± 2.4† | |
| Treadmill test, time to termination (min) | 11.1 ± 3.0 | 1.3 ± 2.0* | 9.6 ± 3.7 | 1.3 ± 2.7 | |
| Treadmill test, maximum exercise | |||||
| relative O2 uptake (ml/min/kg) | 22.2 ± 4.2 | 0.0 ± 2.5 | 21.9 ± 4.6 | -0.2 ± 2.8 | |
| cardiac output (l/min) | 14.5 ± 3.6 | 0.8 ± 2.4* | 14.4 ± 3.8 | 0.5 ± 2.7 | |
| muscular efficiency (mlO2/W) | 16.9 ± 4.3 | -1.4 ± 4.1* | 18.6 ± 5.4 | -0.8 ± 4.7 | |
| Borg subjective exertion scale | 15.7 ± 2.0 | -0.1 ± 1.7 | 15.5 ± 2.1 | -0.6 ± 2.0 | |
| Time to lactate threshold (min) | 6.8 ± 2.4 | 0.9 ± 2.2* | 5.4 ± 2.8 | 1.1 ± 2.2 | |
2.2.5. Tolerability and Safety
| percent of patients | Treatment Group | |
|---|---|---|
| Training + WS 1442 (n=70) | Training (n=70) | |
| Adverse events | ||
| total | 13 % | 17 % |
| musculoskeletal and injuries | 7 % | 7 % |
| infections | 6 % | 7 % |
| cardiac | 0 % | 1 % |
| gastrointestinal | 0 % | 1 % |
| psychiatric | 0 % | 1 % |
| serious | 1 % | 0 % |
| fatal | 0 % | 0 % |
| causal relationship to drug likely or possible | 0 % | |
2.3. Discussion
2.3.1. Training effects
2.3.2. Effects of WS 1442
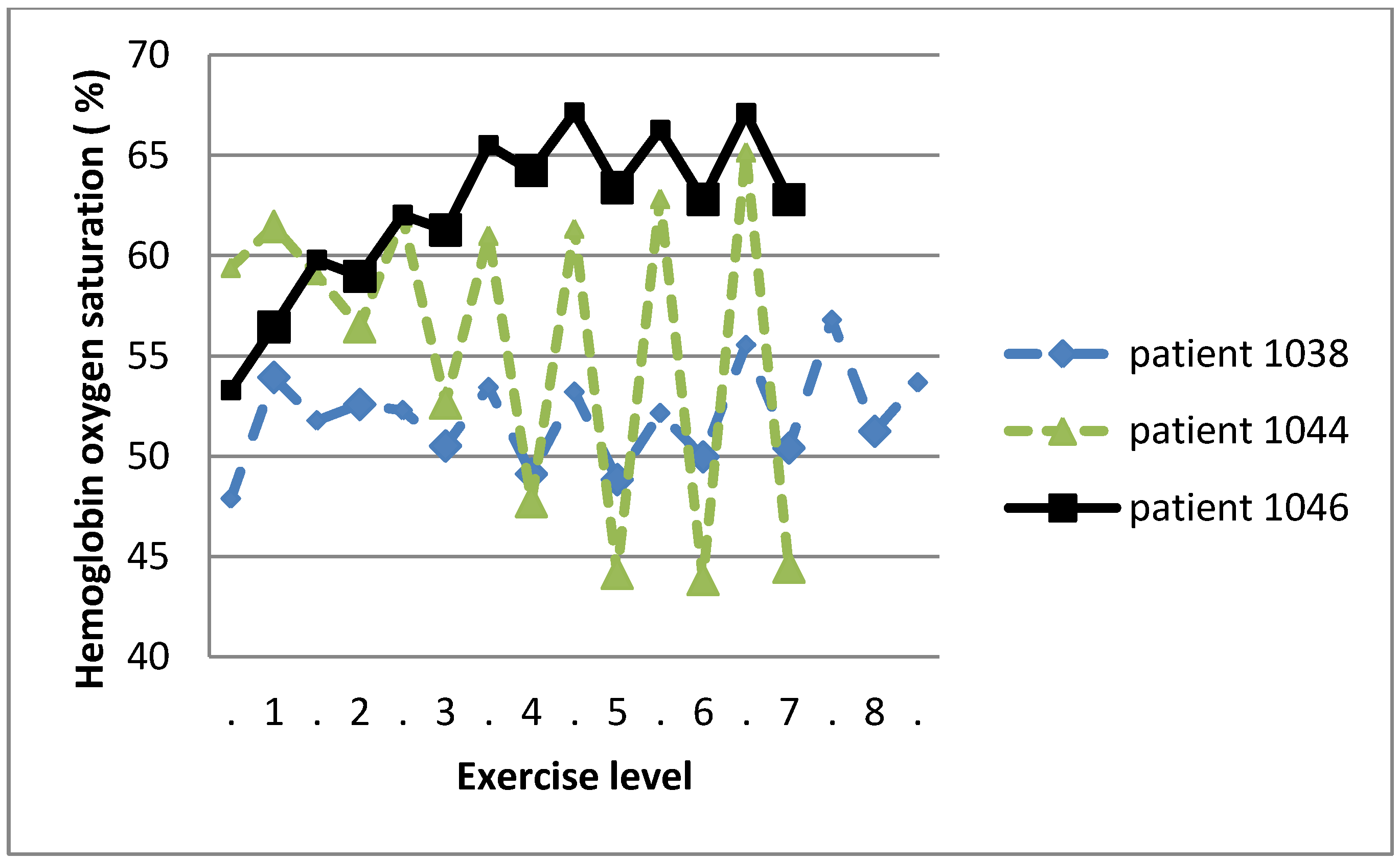
2.3.3. Skeletal muscle oxygenation
2.3.4. Limitations
4. Conclusion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Haq, M.A.; Wong, C.; Mutha, V.; Anavekar, N.; Lim, K.; Barlis, P.; Hare, D.L. Therapeutic interventions for heart failure with preserved ejection fraction: A summary of current evidence. World J. Cardiol. 2014, 6, 67–76. [Google Scholar] [PubMed]
- Paulus, W.J.; Tschope, C. A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J. Am. Coll. Cardiol. 2013, 62, 263–271. [Google Scholar] [PubMed]
- Shibata, K.; Shimokawa, H.; Yanagihara, N.; Otsuji, Y.; Tsutsui, M. Nitric oxide synthases and heart failure - lessons from genetically manipulated mice. J. UOEH. 2013, 35, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Zouein, F.A.; de Castro Bras, L.E.; da Costa, D.V.; Lindsey, M.L.; Kurdi, M.; Booz, G.W. Heart failure with preserved ejection fraction: emerging drug strategies. J. Cardiovasc. Pharmacol. 2013, 62, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Marti, C.N.; Gheorghiade, M.; Kalogeropoulos, A.P.; Georgiopoulou, V.V.; Quyyumi, A.A.; Butler, J. Endothelial dysfunction, arterial stiffness, and heart failure. J. Am. Coll. Cardiol. 2012, 60, 1455–1469. [Google Scholar] [CrossRef] [PubMed]
- Borlaug, B.A. Mechanisms of exercise intolerance in heart failure with preserved ejection fraction. Circ. J. 2013, 78, 20–32. [Google Scholar] [PubMed]
- Bundesärztekammer (BÄK); Kassenärztliche Bundesvereinigung (KBV); Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF). Nationale VersorgungsLeitlinie Chronische Herzinsuffizienz – Langfassung, 1. Auflage Version 7 Dezember 2009, zuletzt geändert: August 2013. 2013. [Google Scholar]
- McMurray, J.J.; Adamopoulos, S.; Anker, S.D.; Auricchio, A.; Bohm, M.; Dickstein, K.; Falk, V.; Filippatos, G.; Fonseca, C.; Gomez-Sanchez, M.A.; Jaarsma, T.; Kober, L.; Lip, G.Y.; Maggioni, A.P.; Parkhomenko, A.; Pieske, B.M.; Popescu, B.A.; Ronnevik, P.K.; Rutten, F.H.; Schwitter, J.; Seferovic, P.; Stepinska, J.; Trindade, P.T.; Voors, A.A.; Zannad, F.; Zeiher, A. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2012, 33, 1787–1847. [Google Scholar]
- Pittler, M.H.; Guo, R.; Ernst, E. Hawthorn extract for treating chronic heart failure. Cochrane. Database. Syst. Rev. 2008, CD005312. [Google Scholar]
- Eggeling, T.; Regitz-Zagrosek, V.; Zimmermann, A.; Burkart, M. Baseline severity but not gender modulates quantified Crataegus extract effects in early heart failure--a pooled analysis of clinical trials. Phytomedicine. 2011, 18, 1214–1219. [Google Scholar] [CrossRef] [PubMed]
- Peters, W.; Drueppel, V.; Kusche-Vihrog, K.; Schubert, C.; Oberleithner, H. Nanomechanics and sodium permeability of endothelial surface layer modulated by hawthorn extract WS 1442. PLoS. One. 2012, 7, e29972. [Google Scholar] [CrossRef] [PubMed]
- Anselm, E.; Socorro, V.F.; Dal-Ros, S.; Schott, C.; Bronner, C.; Schini-Kerth, V.B. Crataegus special extract WS 1442 causes endothelium-dependent relaxation via a redox-sensitive Src- and Akt-dependent activation of endothelial NO synthase but not via activation of estrogen receptors. J. Cardiovasc. Pharmacol. 2009, 53, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Brixius, K.; Willms, S.; Napp, A.; Tossios, P.; Ladage, D.; Bloch, W.; Mehlhorn, U.; Schwinger, R.H. Crataegus special extract WS 1442 induces an endothelium-dependent, NO-mediated vasorelaxation via eNOS-phosphorylation at serine 1177. Cardiovasc. Drugs Ther. 2006, 20, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Rieckeheer, E.; Schwinger, R.H.; Bloch, W.; Brixius, K. Hawthorn special extract WS(R) 1442 increases red blood cell NO-formation without altering red blood cell deformability. Phytomedicine. 2011, 19, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Idris-Khodja, N.; Auger, C.; Koch, E.; Schini-Kerth, V.B. Crataegus special extract WS((R))1442 prevents aging-related endothelial dysfunction. Phytomedicine. 2012, 19, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Schini-Kerth, V.B. The standardized Crataegus extract WS® 1442 prevents systolic hypertension in the DOCA-salt hypertensive rat: Beneficial effects on target organs (blood vessels, heart and kidney). Personal communication, 2013. [Google Scholar]
- Schwinger, R.H.; Pietsch, M.; Frank, K.; Brixius, K. Crataegus special extract WS 1442 increases force of contraction in human myocardium cAMP-independently. J. Cardiovasc. Pharmacol. 2000, 35, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.S.; Bleske, B.E.; Ghannam, M.M.; Converso, K.; Russell, M.W.; Hunter, J.C.; Boluyt, M.O. Effects of hawthorn on cardiac remodeling and left ventricular dysfunction after 1 month of pressure overload-induced cardiac hypertrophy in rats. Cardiovasc. Drugs Ther. 2008, 22, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.S.; Boluyt, M.O.; Converso, K.; Russell, M.W.; Bleske, B.E. Effects of hawthorn on the progression of heart failure in a rat model of aortic constriction. Pharmacotherapy 2009, 29, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Koch, E.; Spörl-Aich, G. Oral treatment with the Crataegus special extract WS(R) 1442 inhibits cardiac hypertrophy in rats with DOCA-salt or aortic banding induced hypertension. Planta Medica 2006, 72, 12–15. [Google Scholar]
- Chen, J.; Brixius, K.; Burkart, M.; Regitz-Zagrosek, V.; Schubert, C. Influence of Crataegus Extract WS1442® on Age-related Alterations in Skeletal Muscle. In Presented at the 78th Annual Meeting of the German Cardiac Society, Mannheim, Germany, 12-4-2012; p. P696.
- Faller, H.; Steinbuchel, T.; Schowalter, M.; Spertus, J.A.; Stork, S.; Angermann, C.E. The Kansas City Cardiomyopathy Questionnaire (KCCQ) -- a new disease-specific quality of life measure for patients with chronic heart failure. Psychother. Psychosom. Med. Psychol. 2005, 55, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Green, C.P.; Porter, C.B.; Bresnahan, D.R.; Spertus, J.A. Development and evaluation of the Kansas City Cardiomyopathy Questionnaire: a new health status measure for heart failure. J. Am. Coll. Cardiol. 2000, 35, 1245–1255. [Google Scholar] [PubMed]
- Frey, I.; Berg, A.; Grathwohl, D.; Keul, J. Freiburg Questionnaire of physical activity--development, evaluation and application. Soz. Praventivmed. 1999, 44, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Bös, K. Der 2-km-Walking-Test. Alters- und geschlechtsspezifische Normwerte. Gesundheitssport und Sporttherapie 2003, 19, 201–207. [Google Scholar]
- Röcker, K.; Schotte, O.; Niess, A.M.; Horstmann, T.; Huth, H.H. Predicting competition performance in long-distance running by means of a treadmill test. Medicine and Science in Sports and Exercise 1998, 30, 1552–1557. [Google Scholar] [CrossRef] [PubMed]
- Borg, G. Anstrengungsempfinden und körperliche Aktivität. Dtsch. Ärztblatt 2004, 1001, A1016–A1021. [Google Scholar]
- Schardt, F. W. Kardiopulmonale Leistungsdiagnostik, 1st ed.; MKM Marketinginstitut GmbH: Lenggries, Germany, 2005. [Google Scholar]
- Kroidl, F.R.; Schwarz, S.; Lehnigk, B. Kursbuch Spiroergometrie: Technik und Befundung verständlich gemacht; Stuttgart, Germany, 2007. [Google Scholar]
- Westphal, E.; Bös, K. Lebensqualität von Herzinsuffizienz-Patienten beim moderaten Ausdauertraining unter Crataegus-Spezialextrakt WS® 1442. Der Hausarzt 2008, Supp1/08. [Google Scholar]
- Kline, R.B. Principles and practice of structural equation modeling; Guildford Press: New York, 1998. [Google Scholar]
- Gasiorowski, A.; Dutkiewicz, J. Comprehensive rehabilitation in chronic heart failure. Ann. Agric. Environ. Med. 2013, 20, 606–612. [Google Scholar] [PubMed]
- Kitzman, D.W.; Brubaker, P.H.; Herrington, D.M.; Morgan, T.M.; Stewart, K.P.; Hundley, W.G.; Abdelhamed, A.; Haykowsky, M.J. Effect of endurance exercise training on endothelial function and arterial stiffness in older patients with heart failure and preserved ejection fraction: a randomized, controlled, single-blind trial. J. Am. Coll. Cardiol. 2013, 62, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Nolte, K.; Herrmann-Lingen, C.; Wachter, R.; Gelbrich, G.; Dungen, H.D.; Duvinage, A.; Hoischen, N.; von Oehsen, K.; Schwarz, S.; Hasenfuss, G.; Halle, M.; Pieske, B.; Edelmann, F. Effects of exercise training on different quality of life dimensions in heart failure with preserved ejection fraction: the Ex-DHF-P trial. Eur. J. Prev. Cardiol. 2014. Epub ahead of print. [Google Scholar]
- Kitzman, D.W.; Upadhya, B. Heart failure with preserved ejection fraction: a heterogenous disorder with multifactorial pathophysiology. J. Am. Coll. Cardiol. 2014, 63, 457–459. [Google Scholar] [CrossRef] [PubMed]
- Edelmann, F.; Gelbrich, G.; Duvinage, A.; Stahrenberg, R.; Behrens, A.; Prettin, C.; Kraigher-Krainer, E.; Schmidt, A.G.; Dungen, H.D.; Kamke, W.; Tschope, C.; Herrmann-Lingen, C.; Halle, M.; Hasenfuss, G.; Wachter, R.; Pieske, B. Differential interaction of clinical characteristics with key functional parameters in heart failure with preserved ejection fraction - results of the Aldo-DHF trial. Int. J. Cardiol. 2013, 169, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Holubarsch, C.J.; Colucci, W.S.; Meinertz, T.; Gaus, W.; Tendera, M. The efficacy and safety of Crataegus extract WS 1442 in patients with heart failure: the SPICE trial. Eur. J. Heart Fail. 2008, 10, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Holubarsch, C.J.F.; Koehler, S.; Malek, F.A.; Gaus, W. Sichere Kombinationstherapie mit Weissdorn-Extrakt WS(R) 1442 bei Herzinsuffizienz NYHA II-III Ergebnisse der SPICE-Studie, der ersten Mortalitaets- und Morbiditaetsstudie mit einem pflanzlichen Arzneimittel. Perfusion 2010, 23, 104–109. [Google Scholar]
- Gerz, E.; Geraskin, D.; Neary, P.; Franke, J.; Platen, P.; Kohl-Bareis, M. Tissue oxygenation during exercise measured with NIRS: a quality control study. In Presented at the Clinical and Biomedical Spectroskopy Meeting, Munich, Germany, 14-6-2009; p. 736816.
- Härtel, S.; Kutzner, C.; Schneider, D.; Grieger, S.; Neumaier, M.; Kohl-Bareis, M. Oxygenation of the calf muscle during an incremental, intermittent walking exercise assessed by NIRS. In Presented at the Diffuse Optical Imaging Meeting III, Munich, Germany, 22-5-2011; Hielscher, A.A., Taroni, P., Eds.; p. 80881G-6.
- Haykowsky, M.J.; Brubaker, P.H.; Morgan, T.M.; Kritchevsky, S.; Eggebeen, J.; Kitzman, D.W. Impaired aerobic capacity and physical functional performance in older heart failure patients with preserved ejection fraction: role of lean body mass. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Haykowsky, M.J.; Kouba, E.J.; Brubaker, P.H.; Nicklas, B.J.; Eggebeen, J.; Kitzman, D.W. Skeletal muscle composition and its relation to exercise intolerance in older patients with heart failure and preserved ejection fraction. Am. J. Cardiol. 2014, 113, 1211–1216. [Google Scholar] [CrossRef]
- Kitzman, D.W.; Nicklas, B.J.; Kraus, W.E.; Lyles, M.F.; Eggebeen, J.; Morgan, T.M.; Haykowsky, M.J. Skeletal Muscle Abnormalities and Exercise Intolerance in Older Patients with Heart Failure and Preserved Ejection Fraction. Am. J. Physiol Heart Circ. Physiol 2014, 13, 64–70. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Härtel, S.; Kutzner, C.; Westphal, E.; Limberger, M.; Burkart, M.; Ebner-Priemer, U.; Kohl-Bareis, M.; Bös, K. Effects of Endurance Exercise Training and Crataegus Extract WS® 1442 in Patients with Heart Failure with Preserved Ejection Fraction – A Randomized Controlled Trial. Sports 2014, 2, 59-75. https://doi.org/10.3390/sports2030059
Härtel S, Kutzner C, Westphal E, Limberger M, Burkart M, Ebner-Priemer U, Kohl-Bareis M, Bös K. Effects of Endurance Exercise Training and Crataegus Extract WS® 1442 in Patients with Heart Failure with Preserved Ejection Fraction – A Randomized Controlled Trial. Sports. 2014; 2(3):59-75. https://doi.org/10.3390/sports2030059
Chicago/Turabian StyleHärtel, Sascha, Claire Kutzner, Eva Westphal, Matthias Limberger, Martin Burkart, Ulrich Ebner-Priemer, Matthias Kohl-Bareis, and Klaus Bös. 2014. "Effects of Endurance Exercise Training and Crataegus Extract WS® 1442 in Patients with Heart Failure with Preserved Ejection Fraction – A Randomized Controlled Trial" Sports 2, no. 3: 59-75. https://doi.org/10.3390/sports2030059
APA StyleHärtel, S., Kutzner, C., Westphal, E., Limberger, M., Burkart, M., Ebner-Priemer, U., Kohl-Bareis, M., & Bös, K. (2014). Effects of Endurance Exercise Training and Crataegus Extract WS® 1442 in Patients with Heart Failure with Preserved Ejection Fraction – A Randomized Controlled Trial. Sports, 2(3), 59-75. https://doi.org/10.3390/sports2030059




