Effect of Mat Pilates Training on Blood Pressure, Inflammatory, and Oxidative Profiles in Hypertensive Elderly
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Mat Pilates Training Program
2.3. Physiological Variables and Measurements
2.4. Blood Collection and Biochemical Analysis
2.5. Statistical Analysis
3. Results
3.1. General Characteristics
3.2. Cardiovascular Function
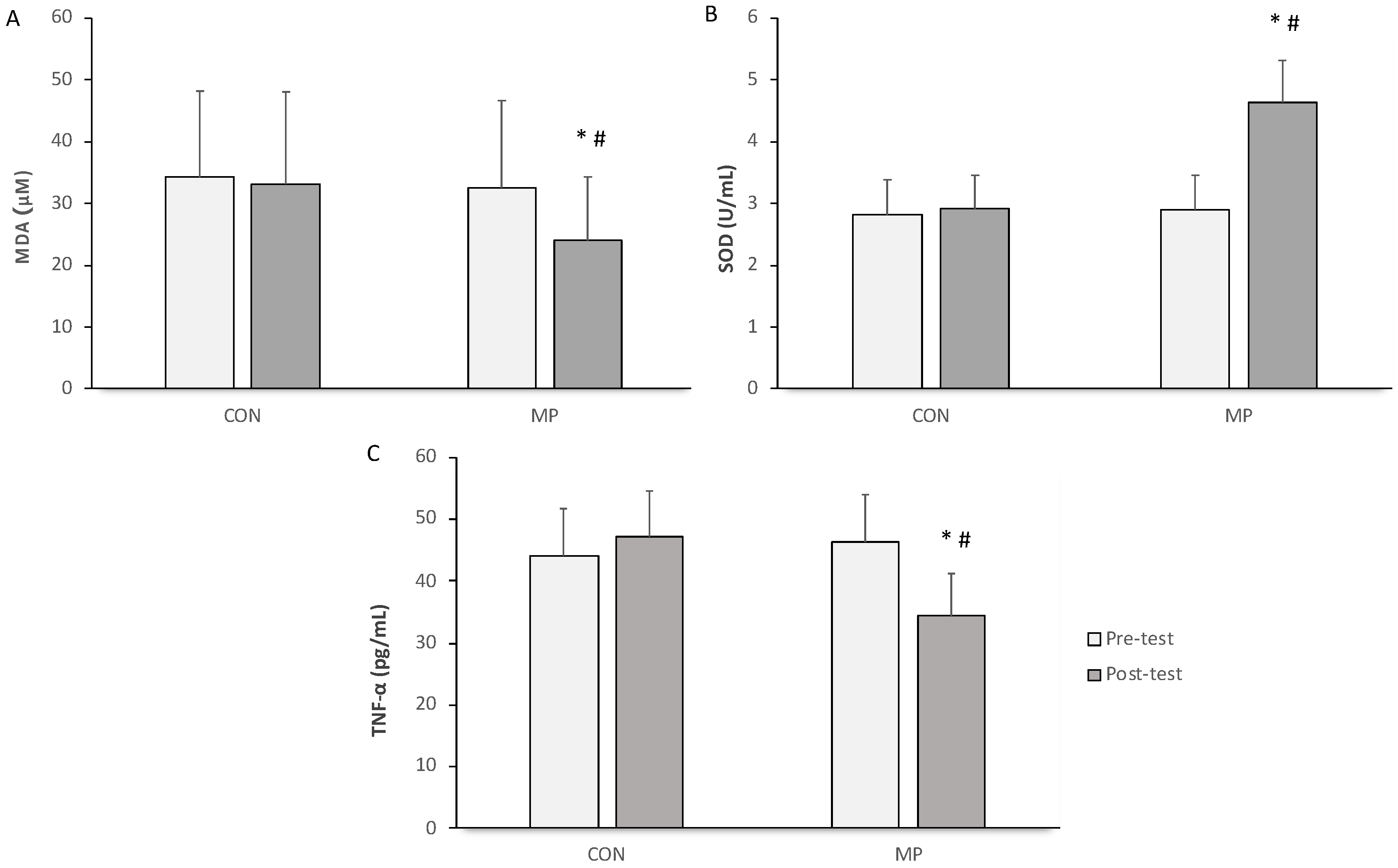
3.3. Anti-Inflammatory and Antioxidative Markers
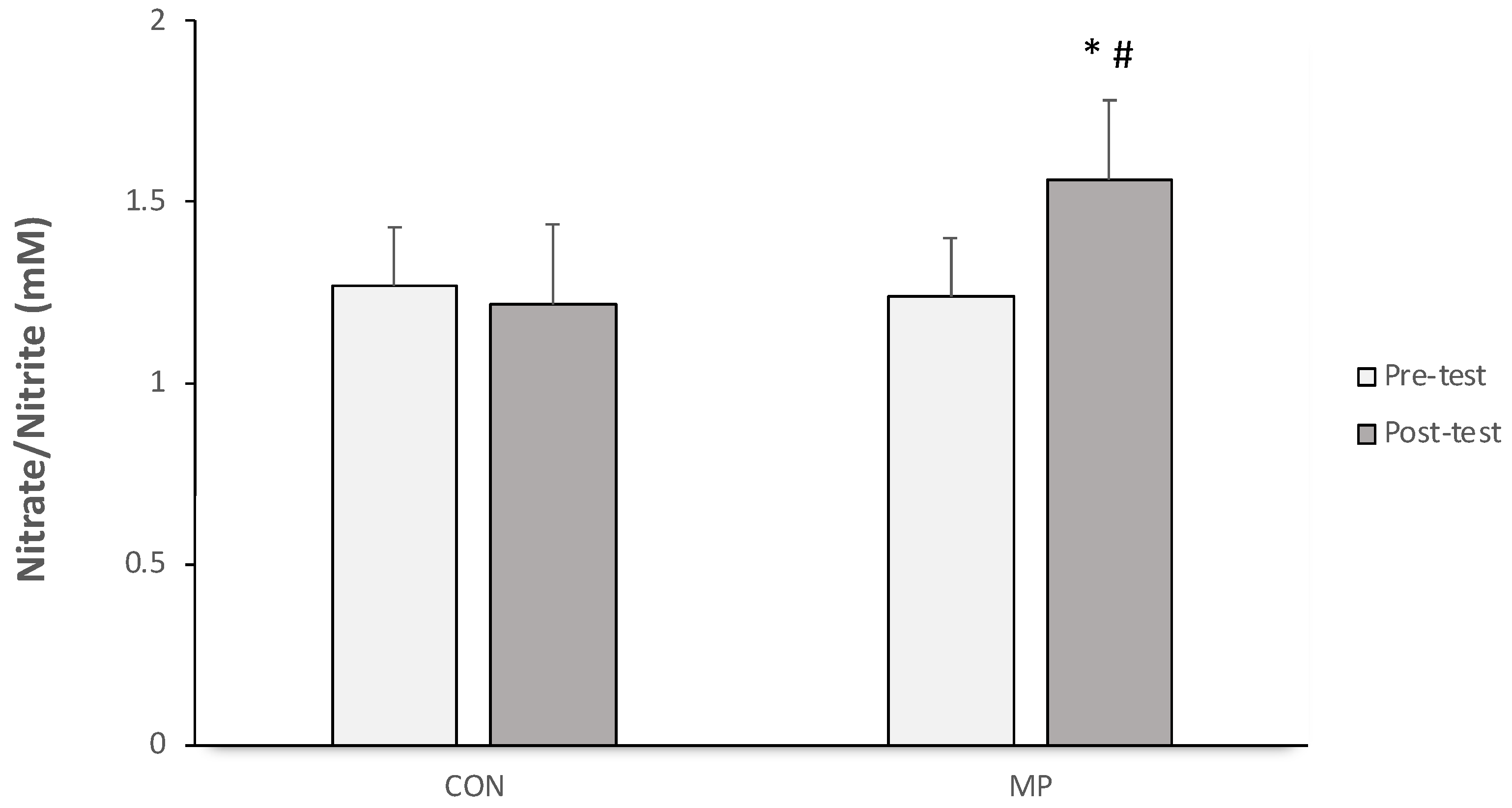
3.4. Serum Nitric Oxide Level
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kumma, W.P.; Lindtjørn, B.; Loha, E. Prevalence of hypertension, and related factors among adults in Wolaita, southern Ethiopia: A community-based cross-sectional study. PLoS ONE 2021, 16, e0260403. [Google Scholar] [CrossRef]
- Saheera, S.; Krishnamurthy, P. Cardiovascular Changes Associated with Hypertensive Heart Disease and Aging. Cell Transplant 2020, 29, 963689720920830. [Google Scholar] [CrossRef]
- Cuspidi, C.; Tadic, M.; Grassi, G.; Mancia, G. Treatment of hypertension: The ESH/ESC guidelines recommendations. Pharmacol. Res. 2018, 128, 315–321. [Google Scholar] [CrossRef]
- Lionakis, N. Hypertension in the elderly. World J. Cardiol. 2012, 4, 135–147. [Google Scholar] [CrossRef]
- Masodsai, K.; Lin, Y.-Y.; Lin, S.-Y.; Su, C.-T.; Lee, S.-D.; Yang, A.-L. Aging Additively Influences Insulin- and Insulin-Like Growth Factor-1-Mediated Endothelial Dysfunction and Antioxidant Deficiency in Spontaneously Hypertensive Rats. Biomedicines 2021, 9, 676. [Google Scholar] [CrossRef]
- Dharmashankar, K.; Widlansky, M.E. Vascular Endothelial Function and Hypertension: Insights and Directions. Curr. Hypertens. Rep. 2010, 12, 448–455. [Google Scholar] [CrossRef]
- Hansen, L.; Taylor, W.R. Is increased arterial stiffness a cause or consequence of atherosclerosis? Atherosclerosis 2016, 249, 226–227. [Google Scholar] [CrossRef]
- Ambrosino, P.; Bachetti, T.; D’anna, S.E.; Galloway, B.; Bianco, A.; D’agnano, V.; Papa, A.; Motta, A.; Perrotta, F.; Maniscalco, M. Mechanisms and Clinical Implications of Endothelial Dysfunction in Arterial Hypertension. J. Cardiovasc. Dev. Dis. 2022, 9, 136. [Google Scholar] [CrossRef]
- Benincasa, G.; Coscioni, E.; Napoli, C. Cardiovascular risk factors and molecular routes underlying endothelial dysfunction: Novel opportunities for primary prevention. Biochem. Pharmacol. 2022, 202, 115108. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Yang, A.-L.; Wong, J.K.; Masodsai, K.; Lee, S.-D.; Lin, Y.-Y. Exercise intervention prevents early aged hypertension-caused cardiac dysfunction through inhibition of cardiac fibrosis. Aging 2022, 14, 4390–4401. [Google Scholar] [CrossRef] [PubMed]
- Myers, J.; Kaykha, A.; George, S.; Abella, J.; Zaheer, N.; Lear, S.; Yamazaki, T.; Froelicher, V. Fitness versus physical activity patterns in predicting mortality in men. Am. J. Med. 2004, 117, 912–918. [Google Scholar] [CrossRef]
- Cornelissen, V.A.; Smart, N.A. Exercise training for blood pressure: A systematic review and meta-analysis. J. Am. Heart Assoc. 2013, 2, e004473. [Google Scholar] [CrossRef]
- 13 DiMeo, F.; Pagonas, N.; Seibert, F.; Arndt, R.; Zidek, W.; Westhoff, T.H. Aerobic Exercise Reduces Blood Pressure in Resistant Hypertension. Hypertens. 2012, 60, 653–658. [Google Scholar] [CrossRef]
- Esmailiyan, M.; Amerizadeh, A.; Vahdat, S.; Ghodsi, M.; Doewes, R.I.; Sundram, Y. Effect of Different Types of Aerobic Exercise on Individuals With and Without Hypertension: An Updated Systematic Review. Curr. Probl. Cardiol. 2021, 48, 101034. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, E.C.; Abrahin, O.; Ferreira, A.L.L.; Rodrigues, R.P.; Alves, E.A.C.; Vieira, R.P. Resistance training alone reduces systolic and diastolic blood pressure in prehypertensive and hypertensive individuals: Meta-analysis. Hypertens. Res. 2017, 40, 927–931. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Wei, W.R.; Can, Z. Effects of 12-week brisk walking training on exercise blood pressure in elderly patients with essential hypertension: A pilot study. Clin. Exp. Hypertens. 2018, 40, 673–679. [Google Scholar] [CrossRef]
- Guimaraes, G.V.; Cruz, L.G.d.B.; Fernandes-Silva, M.M.; Dorea, E.L.; Bocchi, E.A. Heated water-based exercise training reduces 24-hour ambulatory blood pressure levels in resistant hypertensive patients: A randomized controlled trial (HEx trial). Int. J. Cardiol. 2014, 172, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.Z.; Yan, S.; Yuan, W.X. Effect of isometric handgrip training on resting blood pressure in adults: A meta-analysis of randomized controlled trials. J. Sports Med. Phys. Fit. 2017, 57, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Inder, J.D.; Carlson, D.J.; Dieberg, G.; McFarlane, J.R.; Hess, N.C.; A Smart, N. Isometric exercise training for blood pressure management: A systematic review and meta-analysis to optimize benefit. Hypertens. Res. 2015, 39, 88–94. [Google Scholar] [CrossRef]
- Edwards, J.J.; Deenmamode, A.H.P.; Griffiths, M.; Arnold, O.; Cooper, N.J.; Wiles, J.D.; O’Driscoll, J.M. Exercise training and resting blood pressure: A large-scale pairwise and network meta-analysis of randomised controlled trials. Br. J. Sports Med. 2023, 57, 1317–1326. [Google Scholar] [CrossRef]
- Gambardella, J.; Morelli, M.B.; Wang, X.; Santulli, G. Pathophysiological mechanisms underlying the beneficial effects of physical activity in hypertension. J. Clin. Hypertens. 2020, 22, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Roque, F.R.; Briones, A.M.; García-Redondo, A.B.; Galán, M.; Martínez-Revelles, S.; Avendaño, M.S.; Cachofeiro, V.; Fernandes, T.; Vassallo, D.V.; Oliveira, E.M.; et al. Aerobic exercise reduces oxidative stress and improves vascular changes of small mesenteric and coronary arteries in hypertension. Br. J. Pharmacol. 2013, 168, 686–703. [Google Scholar] [CrossRef] [PubMed]
- Boeno, F.P.; Ramis, T.R.; Munhoz, S.V.; Farinha, J.B.; Moritz, C.E.; Leal-Menezes, R.; Ribeiro, J.L.; Christou, D.D.; Reischak-Oliveira, A. Effect of aerobic and resistance exercise training on inflammation, endothelial function and ambulatory blood pressure in middle-aged hypertensive patients. J. Hypertens. 2020, 38, 2501–2509. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, D. Using Physical Activity to Enhance Health Outcomes Across the Life Span. J. Funct. Morphol. Kinesiol. 2020, 5, 2. [Google Scholar] [CrossRef] [PubMed]
- Lopes, S.; Félix, G.; Mesquita-Bastos, J.; Figueiredo, D.; Oliveira, J.; Ribeiro, F. Determinants of exercise adherence and maintenance among patients with hypertension: A narrative review. Rev. Cardiovasc. Med. 2021, 22, 1271–1278. [Google Scholar] [CrossRef] [PubMed]
- Casonatto, J.; Yamacita, C.M. Pilates exercise and postural balance in older adults: A systematic review and meta-analysis of randomized controlled trials. Complement. Ther. Med. 2019, 48, 102232. [Google Scholar] [CrossRef] [PubMed]
- Pinto, J.R.; Santos, C.S.; Soares, W.J.S.; Ramos, A.P.S.; Scoz, R.D.; de Júdice, A.F.T.; Ferreira, L.M.A.; Mendes, J.J.B.; Amorim, C.F. Is pilates better than other exercises at increasing muscle strength? A systematic review. Heliyon 2022, 8, e11564. [Google Scholar] [CrossRef] [PubMed]
- Hyun, A.-H.; Cho, J.-Y.; Koo, J.-H. Effect of Home-Based Tele-Pilates Intervention on Pregnant Women: A Pilot Study. Healthcare 2022, 10, 125. [Google Scholar] [CrossRef]
- Lim, E.-J.; Hyun, E.-J. The Impacts of Pilates and Yoga on Health-Promoting Behaviors and Subjective Health Status. Int. J. Environ. Res. Public Health 2021, 18, 3802. [Google Scholar] [CrossRef]
- Zaras, N.; Kavvoura, A.; Gerolemou, S.; Hadjicharalambous, M. Pilates-mat training and detraining: Effects on body composition and physical fitness in pilates-trained women. J. Bodyw. Mov. Ther. 2023, 36, 38–44. [Google Scholar] [CrossRef]
- Carrasco-Poyatos, M.; Ramos-Campo, D.J.; Rubio-Arias, J.A. Pilates versus resistance training on trunk strength and balance adaptations in older women: A randomized controlled trial. PeerJ 2019, 7, e7948. [Google Scholar] [CrossRef] [PubMed]
- da Luz, M.A.; Costa, L.O.P.; Fuhro, F.F.; Manzoni, A.C.T.; Oliveira, N.T.B.; Cabral, C.M.N. Effectiveness of Mat Pilates or Equipment-Based Pilates Exercises in Patients With Chronic Nonspecific Low Back Pain: A Randomized Controlled Trial. Phys. Ther. 2014, 94, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Almeida, I.d.S.; Andrade, L.d.S.; de Sousa, A.M.M.; Junior, G.C.; Catai, A.M.; Mota, Y.L.; Durigan, J.L.Q. Is the Combination of Aerobic Exercise with Mat Pilates Better than Mat Pilates Training Alone on Autonomic Modulation Related to Functional Outcomes in Hypertensive Women? Secondary Analysis of a Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2022, 19, 10577. [Google Scholar] [CrossRef]
- Fourie, M.; Gildenhuys, G.; Shaw, I.; Shaw, B.; Toriola, A.; Goon, D. Effects of a Mat Pilates Programme on Body Composition in Elderly Women. West Indian Med. J. 2013, 62, 524–528. [Google Scholar] [CrossRef] [PubMed]
- González-Devesa, D.; Varela, S.; Diz-Gómez, J.C.; Ayán-Pérez, C. The efficacy of Pilates method in patients with hypertension: Systematic review and meta-analysis. J. Hum. Hypertens. 2024, 38, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Martins-Meneses, D.T.; Antunes, H.K.M.; de Oliveira, N.R.C.; Medeiros, A. Mat Pilates training reduced clinical and ambulatory blood pressure in hypertensive women using antihypertensive medications. Int. J. Cardiol. 2014, 179, 262–268. [Google Scholar] [CrossRef]
- Rocha, J.; Cunha, F.A.; Cordeiro, R.; Monteiro, W.; Pescatello, L.S.; Farinatti, P. Acute Effect of a Single Session of Pilates on Blood Pressure and Cardiac Autonomic Control in Middle-Aged Adults With Hypertension. J. Strength Cond. Res. 2020, 34, 114–123. [Google Scholar] [CrossRef]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. Circ. 2000, 37, 153–156. [Google Scholar] [CrossRef]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Gomez, J.M.; Heitmann, B.L.; Kent-Smith, L.; Melchior, J.-C.; Pirlich, M.; et al. Bioelectrical impedance analysis? Part I: Review of principles and methods. Clin. Nutr. 2004, 23, 1226–1243. [Google Scholar] [CrossRef]
- Masodsai, K.; Chaunchaiyakul, R. Determination of Cardiac Function Using Impedance Cardiography During Jogging With and Without Breast Support. In Enhancing Health and Sports Performance by Design. MoHE 2019. Lecture Notes in Bioengineering; Springer: Singapore. [CrossRef]
- Gambassi, B.B.; Neves, V.R.; Brito, E.Z.A.; Fernandes, D.S.D.S.; Sá, C.A.; Nogueira, R.M.D.R.; Almeida, F.D.J.F.; Cavalcanti, P.A.D.A.; E Silva, D.C.G.G.; Neto, D.S.; et al. A validation study of a smartphone application for heart rate variability assessment in asymptomatic adults. Am. J. Cardiovasc. Dis. 2020, 10, 219–229. [Google Scholar]
- Tedla, Y.G.; Bautista, L.E. Drug Side Effect Symptoms and Adherence to Antihypertensive Medication. Am. J. Hypertens. 2015, 29, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Hellsten, Y.; Nyberg, M. Cardiovascular Adaptations to Exercise Training. Compr Physiol. 2015, 15, 1–32. [Google Scholar]
- Gomez-Cabrera, M.-C.; Domenech, E.; Viña, J. Moderate exercise is an antioxidant: Upregulation of antioxidant genes by training. Free. Radic. Biol. Med. 2008, 44, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Mann, N.; Rosenzweig, A. Can Exercise Teach Us How to Treat Heart Disease? Circulation 2012, 126, 2625–2635. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Radavelli-Bagatini, S.; Ho, S. Potential benefits of exercise on blood pressure and vascular function. J. Am. Soc. Hypertens. 2013, 7, 494–506. [Google Scholar] [CrossRef] [PubMed]
- Makki, K.; Froguel, P.; Wolowczuk, I. Adipose tissue in obesity-related inflammation and insulin resistance: Cells, cytokines, and chemokines. ISRN Inflamm. 2013, 2013, 139239. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in Inflammation, Immunity, and Disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef] [PubMed]
- Tsiotra, P.; Tsigos, C.; Raptis, S. TNFα and leptin inhibit basal and glucose-stimulated insulin secretion and gene transcription in the HIT-T15 pancreatic cells. Int. J. Obes. 2001, 25, 1018–1026. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Maldonado, A.; Montero, S.; Lemus, M.; Cerna-Cortés, J.; Rodríguez-Hernández, A.; Mendoza, M.A.; Melnikov, V.; Gamboa-Domínguez, A.; Muñiz, J.; Virgen-Ortiz, A.; et al. Moderate and high intensity chronic exercise reduces plasma tumor necrosis factor alpha and increases the Langerhans islet area in healthy rats. J. Musculoskelet. Neuronal Interact. 2019, 19, 354–361. [Google Scholar]
- Talebi-Garakani, E.; Safarzade, A. Resistance training decreases serum inflammatory markers in diabetic rats. Endocrine 2012, 43, 564–570. [Google Scholar] [CrossRef]
- Ito, Y.; Nomura, S.; Ueda, H.; Sakurai, T.; Kizaki, T.; Ohno, H.; Izawa, T. Exercise training increases membrane bound form of tumor necrosis factor-α receptors with decreases in the secretion of soluble forms of receptors in rat adipocytes. Life Sci. 2002, 71, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Seals, D.R.; Nagy, E.E.; Moreau, K.L. Aerobic exercise training and vascular function with ageing in healthy men and women. J. Physiol. 2019, 597, 4901–4914. [Google Scholar] [CrossRef] [PubMed]
- Pedralli, M.L.; Waclawovsky, G.; Camacho, A.; Markoski, M.M.; Castro, I.; Lehnen, A.M. Study of endothelial function response to exercise training in hypertensive individuals (SEFRET): Study protocol for a randomized controlled trial. Trials 2016, 17, 84. [Google Scholar] [CrossRef]
- Touyz, R.M. Reactive Oxygen Species, Vascular Oxidative Stress, and Redox Signaling in Hypertension. Hypertension 2004, 44, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Fukai, T.; Folz, R.J.; Landmesser, U.; Harrison, D.G. Extracellular superoxide dismutase and cardiovascular disease. Cardiovasc. Res. 2002, 55, 239–249. [Google Scholar] [CrossRef]
- Graham, D.A.; Rush, J.W.E. Exercise training improves aortic endothelium-dependent vasorelaxation and determinants of nitric oxide bioavailability in spontaneously hypertensive rats. J. Appl. Physiol. 2004, 96, 2088–2096. [Google Scholar] [CrossRef]
- Higashi, Y.; Sasaki, S.; Kurisu, S.; Yoshimizu, A.; Sasaki, N.; Matsuura, H.; Kajiyama, G.; Oshima, T. Regular aerobic exercise augments endothelium-dependent vascular relaxation in normotensive as well as hypertensive subjects: Role of endothelium-derived nitric oxide. Circulation 1999, 100, 1194–1202. [Google Scholar] [CrossRef]
- Goto, C.; Higashi, Y.; Kimura, M.; Noma, K.; Hara, K.; Nakagawa, K.; Kawamura, M.; Chayama, K.; Yoshizumi, M.; Nara, I. Effect of Different Intensities of Exercise on Endothelium-Dependent Vasodilation in Humans: Role of Endothelium-Dependent Nitric Oxide and Oxidative Stress. Circulation 2003, 108, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Z.; Tang, D. Aerobic Exercise Alleviates Inflammation, Oxidative Stress, and Apoptosis in Mice with Chronic Obstructive Pulmonary Disease. Int. J. Chronic Obstr. Pulm. Dis. 2021, 16, 1369–1379. [Google Scholar] [CrossRef]
- Takabe, W.; Jen, N.; Ai, L.; Hamilton, R.; Wang, S.; Holmes, K.; Dharbandi, F.; Khalsa, B.; Bressler, S.; Barr, M.L.; et al. Oscillatory Shear Stress Induces Mitochondrial Superoxide Production: Implication of NADPH Oxidase and c-Jun NH2-Terminal Kinase Signaling. Antioxidants Redox Signal. 2011, 15, 1379–1388. [Google Scholar] [CrossRef]
- Fujii, J.; Homma, T.; Osaki, T. Superoxide Radicals in the Execution of Cell Death. Antioxidants 2022, 11, 501. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Lin, H.; Wan, M.; Qiu, P.; Xia, R.; He, J.; Tao, J.; Chen, L.; Zheng, G. The Effects of Aerobic Exercise on Oxidative Stress in Older Adults: A Systematic Review and Meta-Analysis. Front. Physiol. 2021, 12, 701151. [Google Scholar] [CrossRef] [PubMed]
| Mat Pilates | Principles |
|---|---|
| Half rollback | Head, cervical, and ribcage placement Shoulder blade stability |
| Oblique rollback | Head, cervical, and ribcage placement Shoulder blade stability |
| Hundred prep | Breathing control Ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Dead bug | Breathing control Ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Tabletop toe tapping | Ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Single leg stretch | Breathing control Ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Scissors | Breathing control Ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Side leg: lower and lift | Breathing control Pelvic and ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Side leg: kick | Breathing control Pelvic placement |
| Side leg: circle | Pelvic placement |
| Side leg: bicycle | Pelvic and ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Shoulder bridge | Breathing control Pelvic and ribcage placement |
| Knee push-up | Breathing control Pelvic and ribcage placement Shoulder blade movement and stability Head and cervical placement |
| Swimming prep | Pelvic and ribcage placement |
| Superman | Shoulder blade movement and stability |
| Swan | Head and cervical placement |
| Variables | CON (n = 17) (Males/Females: 5/12) | MP (n = 17) (Males/Females: 5/12) | Time | Group | Time × Group | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre-Test | Post-Test | Pre-Test | Post-Test | ES | p-Value | ES | p-Value | ES | p-Value | |
| Body mass (kg) | 58.06 ± 7.87 | 57.91 ± 8.35 | 55.27 ± 8.80 | 55.04 ± 8.95 # | 0.098 | 0.411 | 0.599 | 0.014 | 0.634 | 0.010 |
| BMI (kg/m2) | 22.82 ± 1.84 | 23.12 ± 2.38 | 22.07 ± 2.07 | 22.24 ± 2.72 | 0.185 | 0.248 | 0.418 | 0.060 | 0.346 | 0.095 |
| Waist/hip ratio | 0.85 ± 0.05 | 0.86 ± 0.06 | 0.86 ± 0.05 | 0.84 ± 0.05 # | 0.124 | 0.352 | 0.526 | 0.027 | 0.196 | 0.226 |
| Body fat (%) | 29.98 ± 3.88 | 29.73 ± 4.13 | 27.42 ± 6.05 | 25.84 ± 4.57 *# | 0.322 | 0.111 | 0.578 | 0.017 | 0.282 | 0.141 |
| Body fat (kg) | 16.07 ± 2.10 | 17.08 ± 2.47 | 15.03 ± 3.59 | 14.35 ± 3.83 *# | 0.349 | 0.094 | 0.317 | 0.114 | 0.014 | 0.765 |
| LBM (kg) | 38.03 ± 6.89 | 37.58 ± 7.05 | 35.09 ± 6.37 | 42.06 ± 5.88 * | 0.617 | 0.012 | 0.030 | 0.656 | 0.398 | 0.068 |
| Variables | CON (n = 17) | MP (n = 17) | Time | Group | Time × Group | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre-Test | Post-Test | Pre-Test | Post-Test | ES | p-Value | ES | p-Value | ES | p-Value | |
| HR (bpm) | 77.47 ± 9.26 | 74.65 ± 9.22 | 72.76 ± 13.03 | 71.82 ± 9.97 | 0.303 | 0.125 | 0.127 | 0.346 | 0.103 | 0.400 |
| SBP (mmHg) | 145.12 ± 17.23 | 143.12 ± 17.99 | 141.12 ± 7.83 | 127.65 ± 11.85 *# | 0.757 | 0.002 | 0.578 | 0.017 | 0.568 | 0.019 |
| DBP (mmHg) | 82.82 ± 10.87 | 79.71 ± 7.91 | 78.53 ± 8.12 | 70.82 ± 9.86 | 0.035 | 0.628 | 0.138 | 0.326 | 0.037 | 0.621 |
| MABP (mmHg) | 103.59 ± 12.12 | 99.12 ± 9.97 | 98.02 ± 7.80 | 91.41 ± 9.05 *# | 0.411 | 0.063 | 0.346 | 0.096 | 0.148 | 0.307 |
| PP (mmHg) | 62.29 ± 11.79 | 63.41 ± 14.80 | 64.65 ± 8.94 | 56.82 ± 9.06 *# | 0.761 | 0.002 | 0.555 | 0.021 | 0.645 | 0.009 |
| CO (L/min) | 5.97 ± 1.81 | 5.82 ± 1.28 | 6.08 ± 0.98 | 6.58 ± 1.02 | 0.195 | 0.234 | 0.103 | 0.399 | 0.076 | 0.472 |
| SV (mL/min) | 81.71 ± 24.81 | 74.88 ± 21.37 | 80.68 ± 15.37 | 91.14 ± 15.89 *# | 0.001 | 0.950 | 0.418 | 0.060 | 0.023 | 0.696 |
| SVR (Dynes·s/cm⁵) | 1587.47 ± 812.26 | 1500.71 ± 372.61 | 1296.59 ± 306.82 | 1172.47 ± 198.48 *# | 0.139 | 0.324 | 0.320 | 0.112 | 0.172 | 0.267 |
| Right baPWV (cm/s) | 1677 ± 215.14 | 1650.41 ± 196.57 | 1661.88 ± 318.99 | 1542.76 ± 138.27 | 0.102 | 0.401 | 0.007 | 0.828 | 0.004 | 0.869 |
| Left baPWV (cm/s) | 1660 ± 207.88 | 1645.00 ± 174.36 | 1666.94 ± 314.87 | 1542.18 ± 154.71 | 0.058 | 0.534 | 0.003 | 0.896 | 0.002 | 0.910 |
| IMT (mm) | 0.63 ± 0.76 | 0.65 ± 0.83 | 0.61 ± 0.11 | 0.57 ± 0.81 # | 0.013 | 0.769 | 0.541 | 0.024 | 0.231 | 0.190 |
| LF/HF ratio | 2.31 ± 2.34 | 1.61 ± 1.50 | 1.29 ± 1.12 | 2.52 ± 1.32 * | 0.638 | 0.01 | 0.266 | 0.155 | 0.249 | 0.172 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woramontri, C.; Chaunchaiyakul, R.; Yang, A.-L.; Lin, Y.-Y.; Masodsai, K. Effect of Mat Pilates Training on Blood Pressure, Inflammatory, and Oxidative Profiles in Hypertensive Elderly. Sports 2024, 12, 120. https://doi.org/10.3390/sports12050120
Woramontri C, Chaunchaiyakul R, Yang A-L, Lin Y-Y, Masodsai K. Effect of Mat Pilates Training on Blood Pressure, Inflammatory, and Oxidative Profiles in Hypertensive Elderly. Sports. 2024; 12(5):120. https://doi.org/10.3390/sports12050120
Chicago/Turabian StyleWoramontri, Chutima, Rungchai Chaunchaiyakul, Ai-Lun Yang, Yi-Yuan Lin, and Kunanya Masodsai. 2024. "Effect of Mat Pilates Training on Blood Pressure, Inflammatory, and Oxidative Profiles in Hypertensive Elderly" Sports 12, no. 5: 120. https://doi.org/10.3390/sports12050120
APA StyleWoramontri, C., Chaunchaiyakul, R., Yang, A.-L., Lin, Y.-Y., & Masodsai, K. (2024). Effect of Mat Pilates Training on Blood Pressure, Inflammatory, and Oxidative Profiles in Hypertensive Elderly. Sports, 12(5), 120. https://doi.org/10.3390/sports12050120







