Plant Selection for the Establishment of Push–Pull Strategies for Zea mays–Spodoptera frugiperda Pathosystem in Morelos, Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Selection and Propagation of Plants in Greenhouse Conditions
2.3. Oviposition Preference of Spodoptera frugiperda in Different Grasses
2.4. Influence of Trichome Density on Oviposition Preference
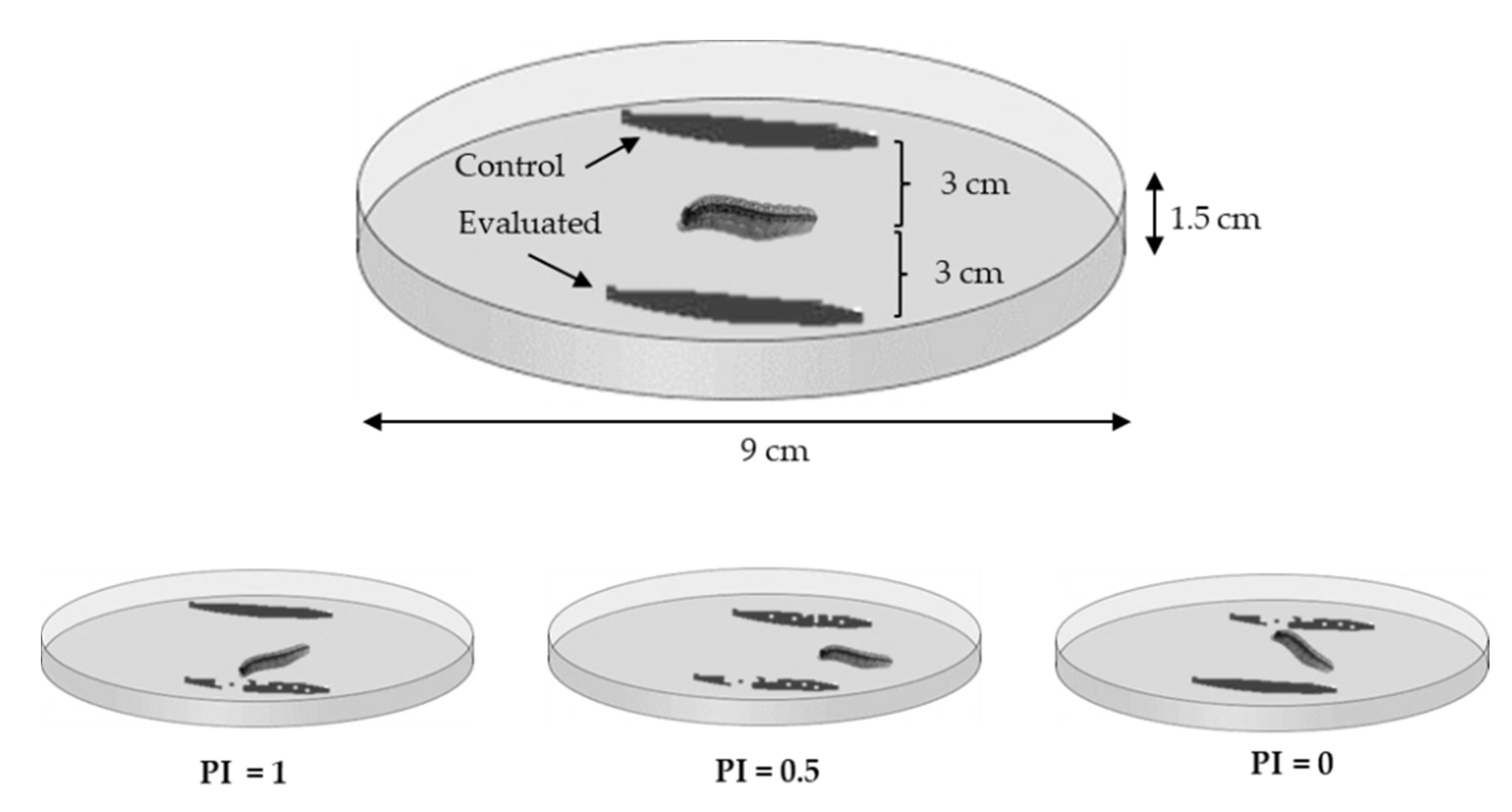
2.5. Evaluation of Attraction and Repulsion of Spodoptera frugipeda Larvae to Plants
2.6. Dispersion and Feeding Preference of Spodoptera frugiperda Larvae among the Different Grasses
2.7. Performance of Spodoptera frugiperda on Potential Trap Plants
2.8. Construction of Trap Plant Selection Index (TRAPS)
2.9. Statistical Data Analysis
3. Results
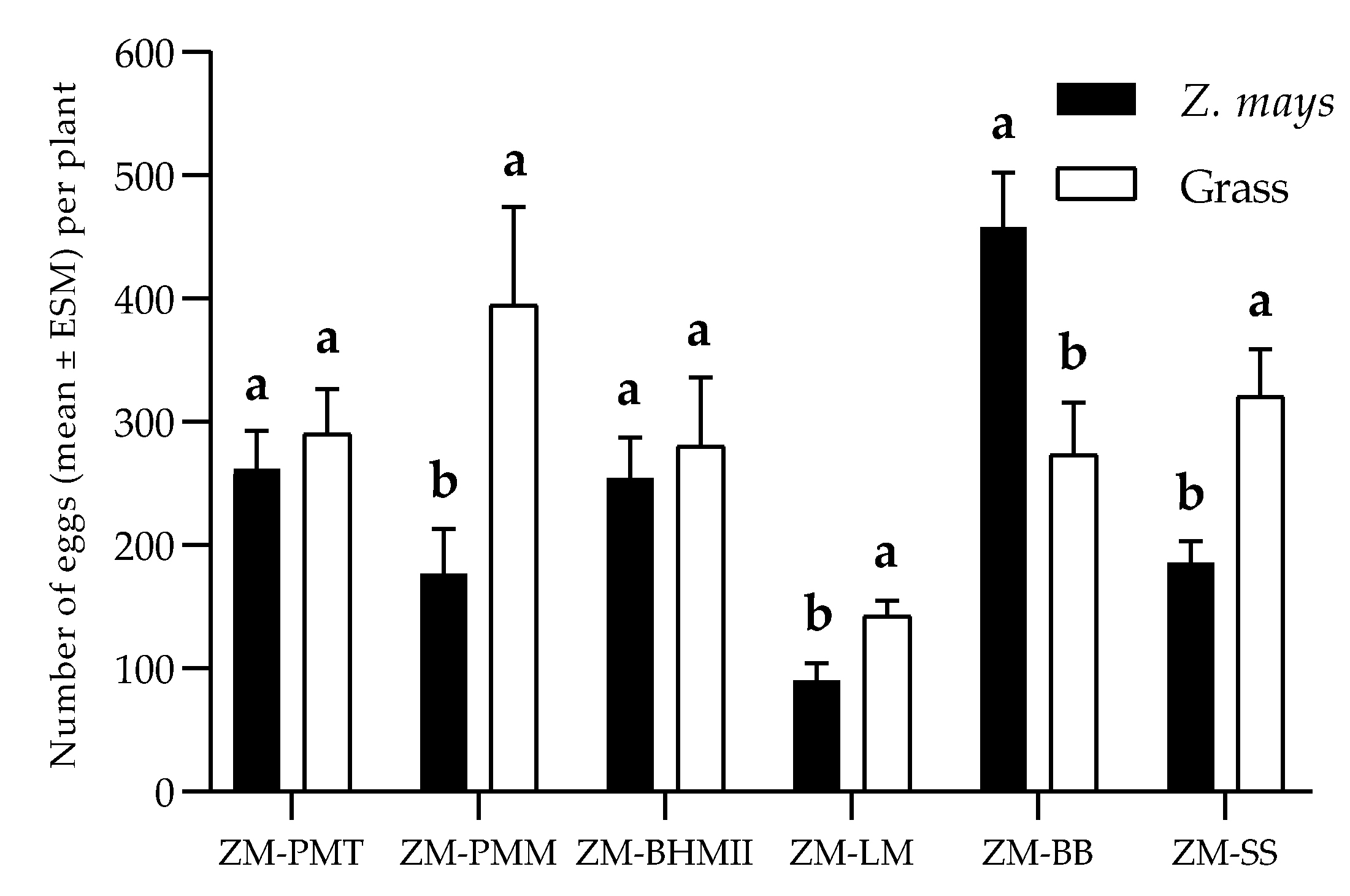
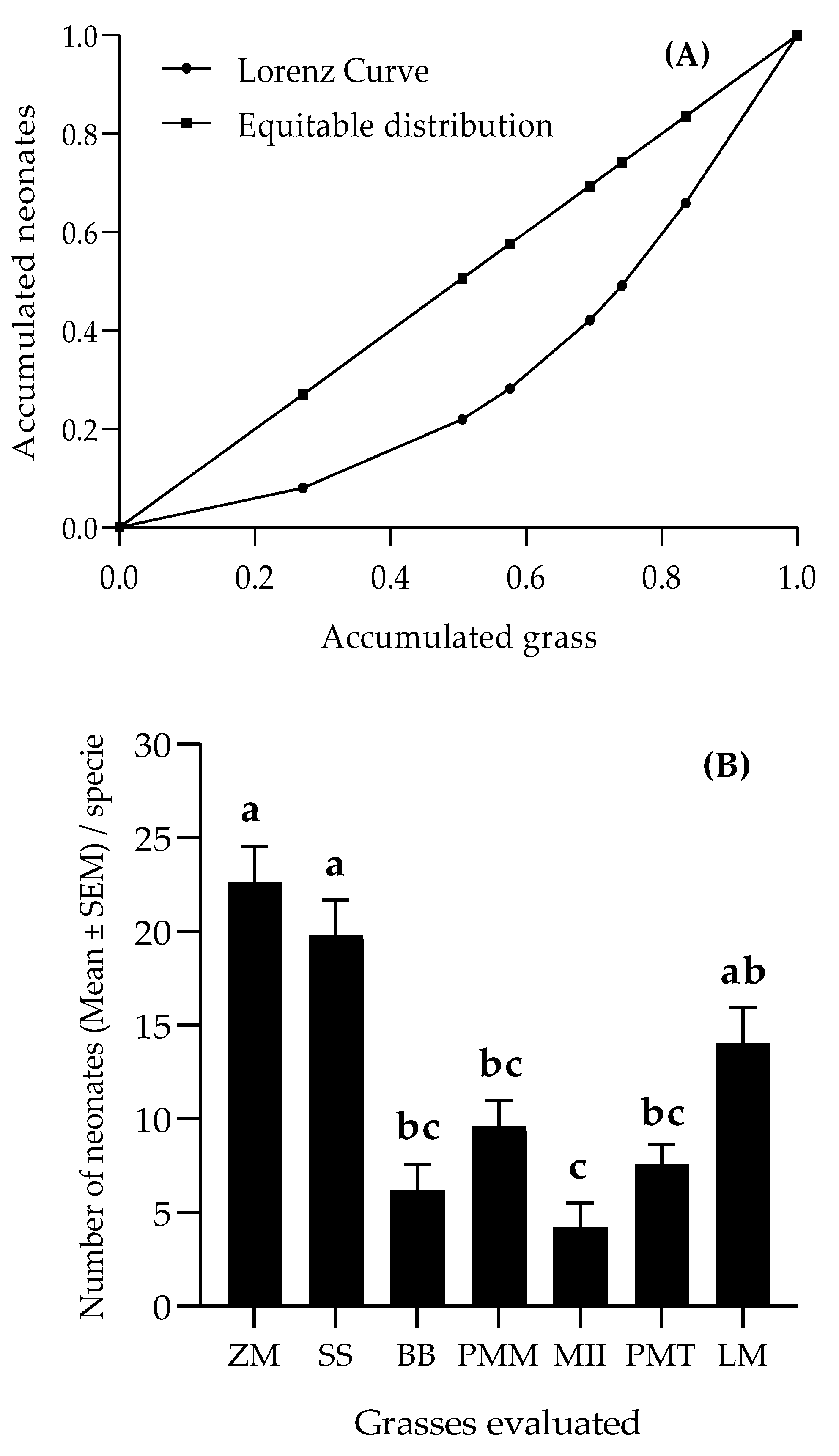
3.1. Oviposition Preference of Spodoptera frugiperda to Different Grasses
3.2. Influence of Trichome Density on Oviposition Preference
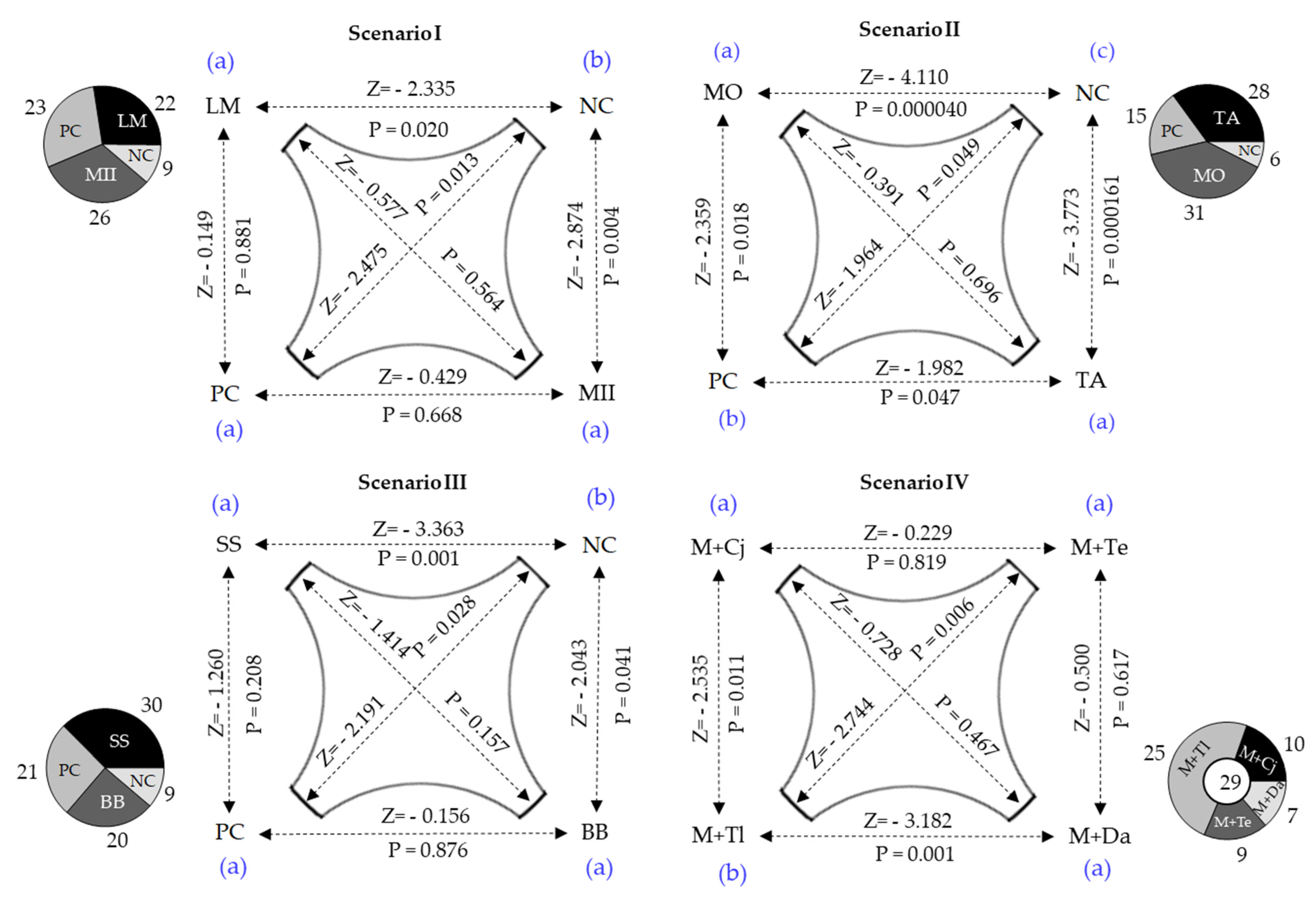
3.3. Response of Attraction or Repulsion of Spodoptera frugiperda Larvae
3.4. Dispersion of Spodoptera frugiperda Larvae on Grasses
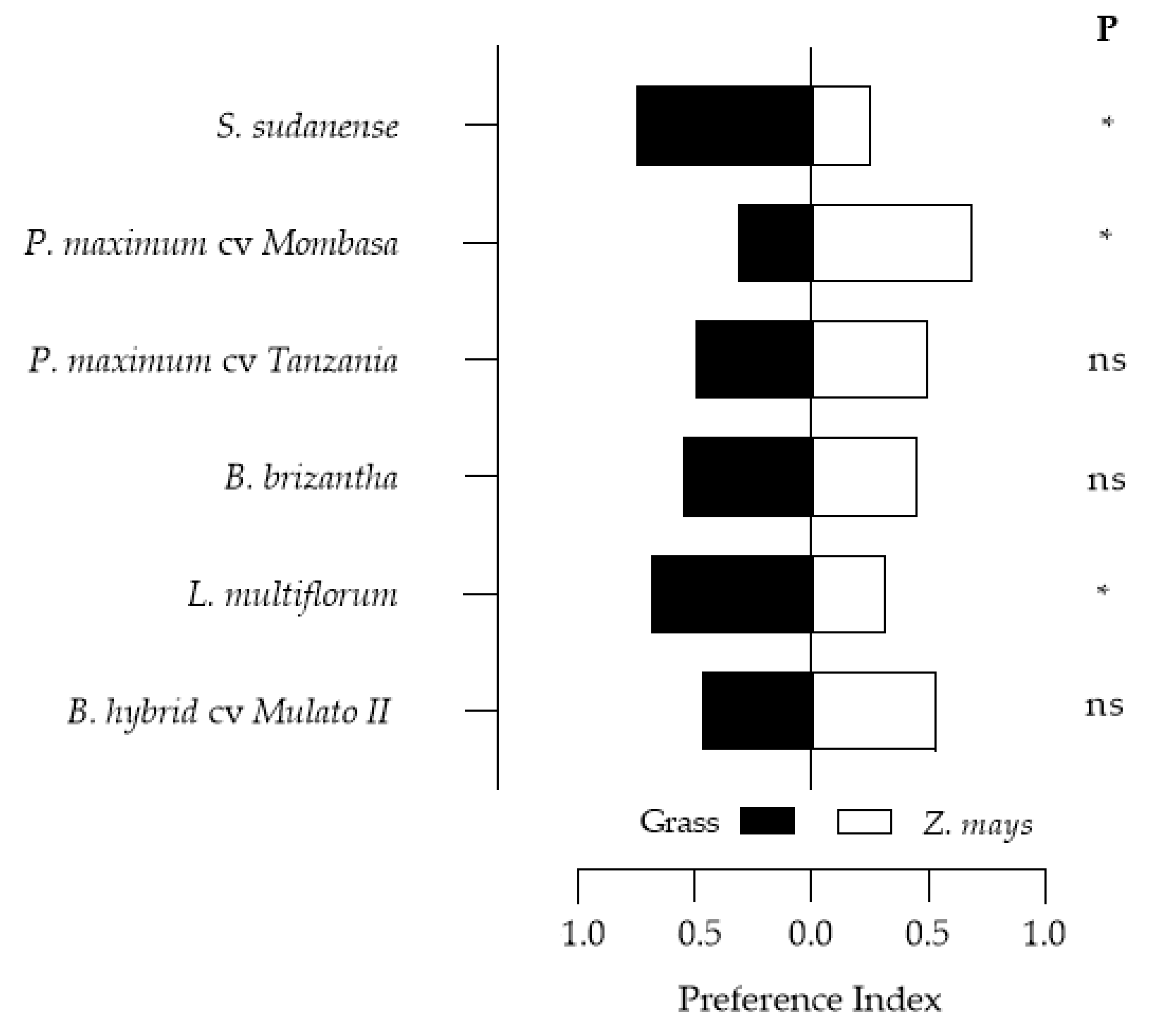
3.5. Feeding Preference of Spodoptera frugiperda Larvae on the Grasses
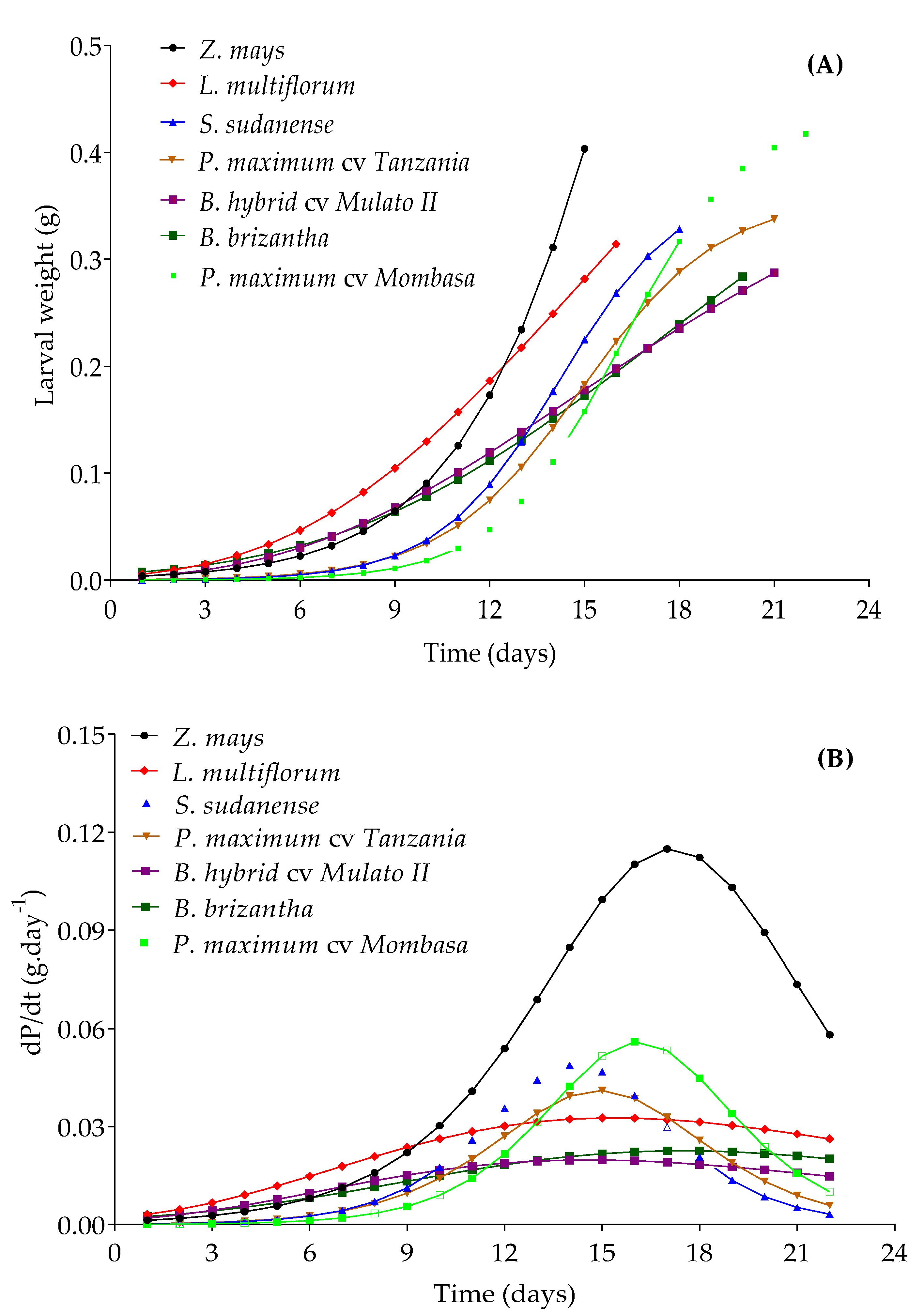
3.6. Performance of Spodoptera frugiperda in Potential Trap Plants
3.7. Trap Plant Selection Index (TRAPS)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bernays, E.A.; Funk, D.J. Specialists make faster decisions than generalists: Experiments with aphids. Proc. R. Soc. B 1999, 266, 151–156. [Google Scholar] [CrossRef]
- Vogel, H.; Musser, R.O.; Mancera, M.C. Transcriptome responses in herbivorous insects towards host plant and toxin feeding. Annu. Plant Rev. 2014, 47, 197–233. [Google Scholar] [CrossRef]
- Meijden, E.V.D. Herbivorous Insects-A Threat for Crop Production. In Principles of Plant-Microbe Interactions: Microbes for Sustainable Agriculture; Springer International Publishing: Cham, Switzerland, 2014; pp. 103–114. [Google Scholar] [CrossRef]
- Turcotte, M.M.; Turley, N.E.; Johnson, M.T. The impact of domestication on resistance to two generalist herbivores across 29 independent domestication events. New Phytol. 2014, 204, 671–681. [Google Scholar] [CrossRef]
- Rodriguez-Saona, C.; Cloonan, K.R.; Sanchez-Pedraza, F.; Zhou, Y.; Giusti, M.M.; Benrey, B. Differential Susceptibility of Wild and Cultivated Blueberries to an Invasive Frugivorous Pest. J. Chem. Ecol. 2019, 45, 286–297. [Google Scholar] [CrossRef]
- Vigoroux, Y.; Glaubitz, J.C.; Matsuoka, Y.; Goodman, M.M.; Sanchez, G.J.; Doebley, J. Population structure and genetic diversity of New World maize landraces assessed by DNA microsatellites. Am. J. Bot. 2008, 95, 1240–1253. [Google Scholar] [CrossRef] [PubMed]
- Bellota, E.; Medina, R.F.; Bernal, J.S. Physical leaf defenses – altered by Zea life-history evolution, domestication, and breeding – mediate oviposition preference of a specialist leafhopper. Entomol. Exp. Appl. 2013, 149, 185–195. [Google Scholar] [CrossRef]
- Iltis, H.H.; Doebley, J.F. Taxonomic of Zea (Gramineae). II. Subspecific categories in the Zea mays complex and a generic synopsis. Am. J. Bot. 1980, 67, 994–1004. [Google Scholar] [CrossRef]
- EPPO. European and Mediterranean Plant Protection Organization. Global Database. EPPO Global Database. Available online: https://gd.eppo.int/ EPPO,Paris,France (accessed on 19 December 2019).
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamò, M. First report of outbreaks of the fall armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in west and central Africa. PLoS ONE 2016, 11, e0165632. [Google Scholar] [CrossRef] [PubMed]
- Day, R.; Abrahams, P.; Bateman, M.; Beale, T.; Clottey, V.; Cock, M.; Colmenarez, Y.; Corniani, N.; Early, R.; Godwin, J.; et al. Fall armyworm: Impacts and implications for Africa. Outlooks Pest Manag. 2017, 28, 196–201. [Google Scholar] [CrossRef]
- Chormule, A.; Shejawal, N.; Sharanabasappa, D.; Kalleshwaraswamy, C.M.; Asokan, R.; Mahadeva Swamy, H.M. First report of the fall Armyworm, Spodoptera frugiperda (J. E. Smith) (Lepidoptera, Noctuidae) on sugarcane and other crops from Maharashtra, India. J. Entomol. Zool. Stud. 2019, 7, 114–117. [Google Scholar]
- Casmuz, A.; Juárez, M.L.; Socías, M.G.; Murúa, M.G.; Prieto, S.; Medina, S.; Willink, E.; Gastaminza, G. Revisión de los hospederos del gusano cogollero del maíz, Spodoptera frugiperda (Lepidoptera: Noctuidae). Rev. Soc. Entomol. Arge 2010, 69, 209–231. [Google Scholar]
- Montezano, D.G.; Specht, A.; Sosa-Gómez, D.R.; Roque-Specht, V.F.; Sousa-Silva, J.C.; Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 2018, 26, 286–300. [Google Scholar] [CrossRef]
- Rodriguez-Saona, C.; Stelinski, L.L. Behavior-Modifying Strategies in IPM: Theory and Practice. In Integrated Pest Management: Innovation – Development Process. Theory and Practice; Peshin, R., Dhawan, A.K., Eds.; Springer: New York, NY, USA, 2009; pp. 261–312. [Google Scholar] [CrossRef]
- Foster, S.P.; Harris, M.O. Behavioral manipulation methods for insect pest management. Annu. Rev. Entomol. 1997, 42, 123–146. [Google Scholar] [CrossRef] [PubMed]
- Midega, C.A.O.; Pittchar, J.O.; Pickett, J.A.; Hailu, G.W.; Khan, Z.R.A. Climate-adapted push-pull system effectively controls fall armyworm, Spodoptera frugiperda (J. E. SMITH), in maize in east Africa. Crop Prot. 2018, 105, 10–15. [Google Scholar] [CrossRef]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The use of push-pull strategies in integrated pest management. Annu. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef]
- Alkema, J.T.; Dicke, M.; Wertheim, B. Context-Dependence and the Development of Push-Pull Approaches for Integrated Management of Drosophila suzukii. Insects 2019, 10, 454. [Google Scholar] [CrossRef]
- Bernays, E.A.; Chapman, R.F. Host-Plant Selection by Phytophagous Insects; Chapman and Hall: New York, NY, USA, 1994; p. 312. [Google Scholar] [CrossRef]
- Whittaker, R.H.; Feeny, P.P. Allelochemics: Chemical interactions between species. Science 1971, 171, 757–770. [Google Scholar] [CrossRef]
- Ali, J.G.; Agrawal, A.A. Specialist versus generalist insect herbivores and plant defense. Trends Plant Sci. 2012, 17, 293–302. [Google Scholar] [CrossRef]
- Bernays, E.A.; Minkenberg, O.P.J.M. Insect herbivores: Different reasons for being a generalist. Ecology 1997, 78, 1157–1169. [Google Scholar] [CrossRef]
- Dall, S.R.X.; Cuthill, I.C. The information costs of generalism. Oikos 1997, 80, 197–202. [Google Scholar] [CrossRef]
- Jermy, T. On the nature of the oligophagy in Leptinotarsa decemlineata Say (Coleoptera: Chrysomelidae). Acta Zool. Acad. Sci. H 1961, 7, 119–132. [Google Scholar]
- Senna, L.D.; Maia, A.G.; Medeiros, J.D.F. The use of principal component analysis for the construction of the Water Poverty Index. Rev. Bras. Recur. Hídr. 2019, 24, 1–14. [Google Scholar] [CrossRef]
- Vyas, S.; Kumaranayake, L. Constructing socio-economic status indices: How to use principal components analysis. Health Policy Plan. 2006, 21, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.R.; Midega, C.A.; Wadhams, L.J.; Pickett, J.A.; Mumuni, A. Evaluation of napier grass (Pennisetum purpureum) varieties for use as trap plants for the management of African stemborer (Busseola fusca) in a push-pull strategy. Entomol. Exp. Appl. 2007, 124, 201–211. [Google Scholar] [CrossRef]
- Khan, Z.R.; Midega, C.A.O.; Bruce, T.J.A.; Hooper, A.M.; Pickett, J.A. Exploiting phytochemicals for developing a ‘push-pull’ crop protection strategy for cereal farmers in Africa. J. Exp. Bot. 2010, 61, 4185–4196. [Google Scholar] [CrossRef]
- Khan, Z.R.; Midega, C.A.O.; Pittchar, J.O.; Murage, A.W.; Birkett, M.A.; Bruce, T.J.A.; Pickett, J.A. Achieving food security for one million sub-Saharan African poor through push-pull innovation by 2020. Philos. Trans. R. Soc. B 2014, 369, 1–12. [Google Scholar] [CrossRef]
- Khan, Z.; Midega, C.A.O.; Hooper, A.; Pickett, J. Push-pull: Chemical ecology-based integrated pest management (IPM) technology. J. Chem. Ecol. 2016, 42, 689–697. [Google Scholar] [CrossRef]
- Khan, Z.R.; Pittchar, J.O.; Midega, C.A.O.; Pickett, J.A. Push-pull farming system controls Fall Armyworm: Lessons from Africa. Outlooks Pest Manag. 2018, 29, 220–224. [Google Scholar] [CrossRef]
- Pickett, J.A.; Woodcock, C.M.; Midega, C.A.O.; Khan, Z.R. Push-pull farming systems. Curr. Opin. Biotech. 2014, 26, 125–132. [Google Scholar] [CrossRef]
- Hailu, G.; Niassy, S.; Khan, R.Z.; Ochatum, N.; Subramanian, S. Maize–Legume Intercropping and Push–Pull for Management of Fall Armyworm, Stemborers, and Striga in Uganda. Agron. J. 2018, 110, 1–10. [Google Scholar] [CrossRef]
- Burton, L.R.; Perkins, D. Rearing the corn earworm and fall armyworm for maize resistance studies. In Proceedings of the International Symposium on Methodologies for Developings Host Plant Resistance to Maize Insects, Cimmyt, México, 9–14 March 1987. [Google Scholar]
- Cancino, S.J.; Joaquín-Torres, B.M.; Rojas-García, A.R.; Estrada-Drouaillet, B.; Hernández Meléndez, J.; Limas Martínez, A.G. Evaluación de la fecha de cosecha en el rendimiento y calidad de semilla de Brachiaria brizantha. Interciencia 2016, 41, 616–621. [Google Scholar]
- Carvajal, A.J.; Lara, R.M. Producción y calidad de semillas de los pastos Insurgente, Guinea y Llanero. Livest. Res. Rural. Dev. 2003, 15, 1–8. [Google Scholar]
- Rendón-Carmona, N.; Ishiki-Ishihara, M.; Terrazas, T.; Nieto-López, M.G. Indumento y tricomas en la caracterización de un grupo de nueve especies del género Mortoniodendron (Tiliaceae). Rev. Mex Biodivers 2006, 77, 169–176. [Google Scholar]
- Turlings, T.C.J.; Davison, A.C.; Tamo, C. A six-arm olfactometer permitting simultaneous observation of insect attraction and odour trapping. Physiol. Entomol. 2004, 29, 45–55. [Google Scholar] [CrossRef]
- Martínez, G.; Finozzi, M.V.; Cantero, G.; Soler, R.; Dicke, M.; González, A. Oviposition preference but not adult feeding preference matches with offspring performance in the bronze bug Thaumastocoris peregrinus. Entomol. Exp. Appl. 2017, 163, 101–111. [Google Scholar] [CrossRef]
- Friel, C. Notes on Factor Analysis. Criminal Justice Centre; Sam Houston State University: Huntsville, TX, USA, 2009; 45p. [Google Scholar]
- Kaiser, H.F. The Application of Electronic Computers to Factor Analysis. Educ. Psychol. Meas. 1960, 20, 141–151. [Google Scholar] [CrossRef]
- Fenemore, P.G. Oviposition of potato tuber moth, Phthorimaea operculella Zell. (Lepidoptera: Gelechiidae); identification of host-plant factors influencing oviposition response. N. Z. J. Zool 1980, 7, 435–439. [Google Scholar] [CrossRef]
- Glass, G.V.; Hopkins, K.D. Statistical Methods in Education and Psychology, 3rd ed.; Pearson: London, UK, 1995; p. 608. [Google Scholar]
- Lorenz, M.O. Methods of Measuring the Concentration of Wealth. Pub. Am. Statist. Assoc. 1905, 9, 209–219. [Google Scholar] [CrossRef]
- Gini, C. Measurement of Inequality of Incomes. Econ. J. 1921, 31, 124–126. [Google Scholar] [CrossRef]
- Noce, R.; Silva, M.L.; Carvalho, R.M.M.A.; Soares, T.S. Concentração das exportações no mercado internacional de madeira serrada. Rev. Árvore 2005, 29, 431–437. [Google Scholar] [CrossRef]
- Gompertz, B. On the nature of the law of human mortality, and on a new mode of determining the value of life contingencies. Philos. Trans. R. Soc. 1825, 115, 513–585. [Google Scholar]
- Richards, F.J. A flexible growth function for empirical use. J. Exp. Bot. 1959, 10, 290–300. [Google Scholar] [CrossRef]
- Verhulst, P.F. Notice sur la loi que la population suit dans son accroissement. Corr. Math. Phys. 1838, 10, 113–121. [Google Scholar]
- Kolmogorov, A.N. Sulla Determinazione Empirica Di Una Legge Di Distribuzione. Giornale dell’Istituto Italiano Degli Attuari 1933, 4, 83–91. [Google Scholar]
- Smirnov, N. Table for Estimating the Goodness of Fit of Empirical Distributions. Ann. Math. Statist. 1948, 19, 279–281. [Google Scholar] [CrossRef]
- White, H.A. Heterocedasticity-Consistent Covariance Matrix Estimator and a Direct Test for Heterocedasticity. Econometrica 1980, 48, 817–838. [Google Scholar] [CrossRef]
- Durbin, J.; Watson, G.S. Testing for Serial Correlation in Least Squares Regression, II. Biometrika 1951, 38, 159–179. [Google Scholar] [CrossRef]
- Kaplan, E.L.; Meier, P. Nonparametric estimation from incomplete observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Levene, H. Robust tests of equality of variances. In Contributions to Probability and Statistics: Essays in Honor of Harold Hotelling; Olkin, I., Ed.; Stanford University Press: Palo Alto, CA, USA, 1960; pp. 278–292. [Google Scholar]
- SPSS. International Business Machines Corporation (IBM). IBM SPSS Base 20; IBM: New York, NY, USA, 2011. [Google Scholar]
- Pitre, H.N.; Mulrooney, J.E.; Hogg, D.B. Fall armyworm (Lepidoptera: Noctuidae) oviposition: Crop preferences and egg distribution on plants. J. Econ. Entomol. 1983, 76, 463–466. [Google Scholar] [CrossRef]
- Ali, A.; Luttrell, R.G.; Pitre, H.N.; Davis, F.M. Distribution of fall armyworm (Lepidoptera: Noctuidae) egg masses on cotton. Environ. Entomol. 1989, 18, 881–885. [Google Scholar] [CrossRef]
- Beserra, E.B.; Dias, C.T.S.; Parra, J.R.P. Distribution and natural parasitism of Spodoptera frugiperda (Lepidoptera: Noctuidae) eggs at different phenological stages of corn. Fla Entomol. 2002, 85, 588–593. [Google Scholar] [CrossRef]
- Cheruiyot, D.; Midega, C.A.O.; Van den Berg, J.; Pickett, J.A.; Khan, Z.R. Suitability of brachiaria grass as a trap crop for management of Chilo partellus. Entomol. Exp. Appl. 2018, 166, 139–148. [Google Scholar] [CrossRef]
- Kumar, H. Resistance in maize to Chile partellus (Swinhoe) (Lepidoptera: Pyralidae): An overview. Crop Prot. 1997, 16, 243–250. [Google Scholar] [CrossRef]
- Rojas, J.C.; Virgen, A.; Cruz-Lopez, L. Chemical and tactile cues influencing oviposition of a generalist moth, Spodoptera frugiperda (Lepidoptera: Noctuidae). Environ. Entomol. 2003, 32, 1386–1392. [Google Scholar] [CrossRef]
- Carroll, M.J.; Schmelz, E.A.; Meagher, R.L.; Teal, P.E.A. Attraction of Spodoptera frugiperda larvae to volatiles from herbivore-damaged maize seedlings. J. Chem. Ecol. 2006, 32, 1911–1924. [Google Scholar] [CrossRef]
- Zalucki, M.P.; Clarke, A.R.; Malcolm, S.B. Ecology and behavior of first instar larval lepidoptera. Annu. Rev. Entomol. 2002, 47, 361–393. [Google Scholar] [CrossRef]
- Rojas, J.C.; Kolomiets, M.V.; Bernal, J.S. Nonsensical choices? Fall armyworm moths choose seemingly best or worst hosts for their larvae, but neonate larvae make their own choices. PLoS ONE 2018, 3, e0197628. [Google Scholar] [CrossRef]
- Bruce, T.J.A.; Midega, C.A.O.; Birkett, M.A.; Pickett, J.A.; Khan, Z.R. Is quality more important than quantity? Insect behavioral responses to changes in a volatile blend after oviposition on an African grass. Biol. Lett. 2010, 6, 314–317. [Google Scholar] [CrossRef]
- Malo, E.A.; Castrejón-Gómez, V.R.; Cruz-López, L.; Rojas, J.C. Antennal Sensilla and Electrophysiological Response of Male and Female Spodoptera frugiperda (Lepidoptera: Noctuidae) to Conspecific Sex Pheromone and Plant Odors. Ann. Entomol. Soc. Am. 2004, 97, 1273–1284. [Google Scholar] [CrossRef]
- Díaz-Cedillo, F.; Serrato-Cruz, M. Composición esencial de Tagetes parryi A. Gray. Rev. Fitotec. Mex. 2011, 34, 145–148. [Google Scholar]
- Calumpang, S.M.F.; Ohsawa, K. Repellency of marigold, Tagetes erecta L. (Asteraceae) volatile organic chemicals to eggplant fruit and shoot borer, Leucinodes orbonalis Guenee (Lepidoptera: Crambidae). J. ISSAAS 2015, 21, 119–128. [Google Scholar]
- Aldana-Llanos, L.; Salinas-Sánchez, D.O.; Valdés Estrada, M.E.; Gutiérrez-Ochoa, M.; Rodríguez-Flores, E.Y.; Navarro-García, V.M. Biological Activity of Dose Extracts of Tagetes erecta L. on Spodoptera frugiperda (J. E. Smith). Southwest Entomol. 2012, 37, 31–38. [Google Scholar] [CrossRef]
- Al-Snafi, A.E. The contents and pharmacology of Crotalaria juncea- A review. IOSR J. Pharm. 2016, 6, 77–86. [Google Scholar]
- Sagrero-Nieves, L.; Bartley, J.P. Volatile constituents from the leaves of Chenopodium ambrosioides L. J. Essent. Oil Res. 1995, 7, 221–223. [Google Scholar] [CrossRef]
- Moya-Raygoza, G. Early development of leaf trichomes is associated with decreased damage in teosinte, compared with maize, by Spodoptera frugiperda (Lepidoptera: Noctuidae). Ann. Entomol. Soc. Am. 2016, 109, 737–743. [Google Scholar] [CrossRef]
- Barfield, C.S.; Smith, J.W., Jr.; Carlysle, C.; Mitchell, E.R. Impact of peanut phenology on select population parameters of Fall Armyworm. Environ. Entomol. 1980, 9, 381–384. [Google Scholar] [CrossRef]
- Silva, D.M.; Bueno, A.F.; Andrade, K.; Stecca, C.S.; Neves, P.M.O.J.; Oliveira, M.C.N. Biology and nutrition of Spodoptera frugiperda (Lepidoptera: Noctuidae) fed on different food sources. Sci. Agric. 2017, 74, 18–31. [Google Scholar] [CrossRef]
- Loader, C.; Damman, H. Nitrogen Content of Food Plants and Vulnerability of Pieris Rapae to Natural Enemies. Ecology 1991, 72, 1586–1590. [Google Scholar] [CrossRef]
- Slansky, F.; Feeny, P. Stabilization of the rate of nitrogen accumulation by larvae of the cabbage butterfly on wild and cultivated food plants. Ecol. Monogr. 1977, 47, 209–228. [Google Scholar] [CrossRef]
- Frey, M.; Chomet, P.; Glawischnig, E.; Stettner, C.; Grün, S.; Winklmair, A.; Eisenreich, W.; Bacher, A.; Meeley, R.B.; Briggs, S.P.; et al. Analysis of a chemical plant defense mechanism in grasses. Science 1997, 277, 696–699. [Google Scholar] [CrossRef]
- Niemeyer, H.M. Hydroxamic acids derived from 2-hydroxy-2H-1,4-benzoxazin-3(4H)-one: Key defense chemicals of cereals. J. Agric. Food Chem. 2009, 57, 1677–1696. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Richter, A.; Jander, G. Beyond Defense: Multiple Functions of Benzoxazinoids in Maize Metabolism. Plant Cell Physiol. 2018, 59, 1528–1537. [Google Scholar] [CrossRef] [PubMed]
- Sicker, D.; Frey, M.; Schulz, M.; Gierl, A. Role of natural benzoxazinones in the survival strategy of plants. Int. Rev. Cytol. 2000, 198, 319–346. [Google Scholar] [CrossRef] [PubMed]
- Handrick, V.; Robert, C.A.; Ahern, K.; Zhou, S.; Machado, R.A.; Maag, D.; Glauser, G.; Fernandez-Penny, F.E.; Chandran, J.N.; Rodgers-Melnik, E.; et al. Biosynthesis of 8-O-methylated benzoxazinoid defense compounds in maize. Plant Cell 2016, 28, 1682–16700. [Google Scholar] [CrossRef]
- Maag, D.; Dalvit, C.; Thevenet, D.; Kohler, A.; Wouters, F.C.; Vassao, D.G.; Gershenzon, J.; Wolfender, J.L.; Turlings, T.C.; Erb, M.; et al. 3-b-D-glucopyranosyl-6-methoxy-2-benzoxazolinone (MBOA-N-Glc) is an insect detoxification product of maize 1,4-benzoxazin-3-ones. Phytochemistry 2014, 102, 97–105. [Google Scholar] [CrossRef]
- Wouters, F.C.; Gershenzon, J.; Vassão, D.G. Benzoxazinoids: Reactivity and modes of action of a versatile class of plant chemical defenses. J. Brazil. Chem. Soc. 2016, 27, 1379–1397. [Google Scholar] [CrossRef]
- Copaja, S.V.; Barria, B.N.; Niemeyer, H.M. Hydroxamic acid content of perennial triticeae. Phytockemistry 1991, 30, 1531–1534. [Google Scholar] [CrossRef]
- Moran, N.; Hamilton, W.D. Low nutritive quality as defense against herbivores. J. Theor. Biol. 1980, 86, 247–254. [Google Scholar] [CrossRef]
- Levins, R.; MacArthur, R. An hypothesis to explain the incidence of monophagy. Ecology 1969, 50, 910–911. [Google Scholar] [CrossRef]
- Jaenike, J. On optimal oviposition behavior in phytophagous insects. Theor. Popul. Biol. 1978, 14, 350–356. [Google Scholar] [CrossRef]
- Foster, S.P.; Howard, A.J. Adult female and neonate larval plant preferences of the generalist herbivore, Epiphyas postvittana. Entomol. Exp. Appl. 1999, 92, 53–62. [Google Scholar] [CrossRef]
- Mayhew, P.J. Adaptive patterns of host-plant selection by phytophagous insects. Oikos 1997, 79, 417–428. [Google Scholar] [CrossRef]
- Mayhew, P.J. Herbivore host choice and optimal bad motherhood. Trends Ecol. Evol. 2001, 16, 165–167. [Google Scholar] [CrossRef]
- García-Robledo, C.; Horvitz, C.C. Parent-offspring conflicts, ‘optimal bad motherhood’ and the ´mother knows best’ principles in insect herbivores colonizing novel host plants. Ecol. Evol. 2012, 2, 1446–1457. [Google Scholar] [CrossRef]
- Gripenberg, S.; Mayhew, P.J.; Parnell, M.; Roslin, T. A meta-analysis of preference–performance relationships in phytophagous insects. Ecol. Lett. 2010, 13, 383–393. [Google Scholar] [CrossRef]
- Jones, R.E. Movement patterns and egg distribution in cabbage butterflies. J. Anim. Ecol. 1977, 46, 195–212. [Google Scholar] [CrossRef]
- Gómez-Jiménez, M.I.; Sarmiento, C.E.; Díaz, M.F.; Chautá, A.; Peraza, A.; Ramírez, A.; Poveda, K. Oviposition, larval preference, and larval performance in two polyphagous species: Does the larva know best? Entomol. Exp. Appl. 2014, 153, 24–33. [Google Scholar] [CrossRef]
- Hopkins, A.D. Economic Investigations of the Scolytidae Bark and Timber Beetles of North America; US Department of Agriculture Program of Work for 1917: Washington, DC, USA, 1916; p. 353. [Google Scholar]
- Fox, L.R.; Eisenbach, J. Contrary choices: Posible exploitation of enemy-free space by herbivorous insects in cultivated vs. wild crucifers. Oecologia 1992, 89, 574–579. [Google Scholar] [CrossRef]



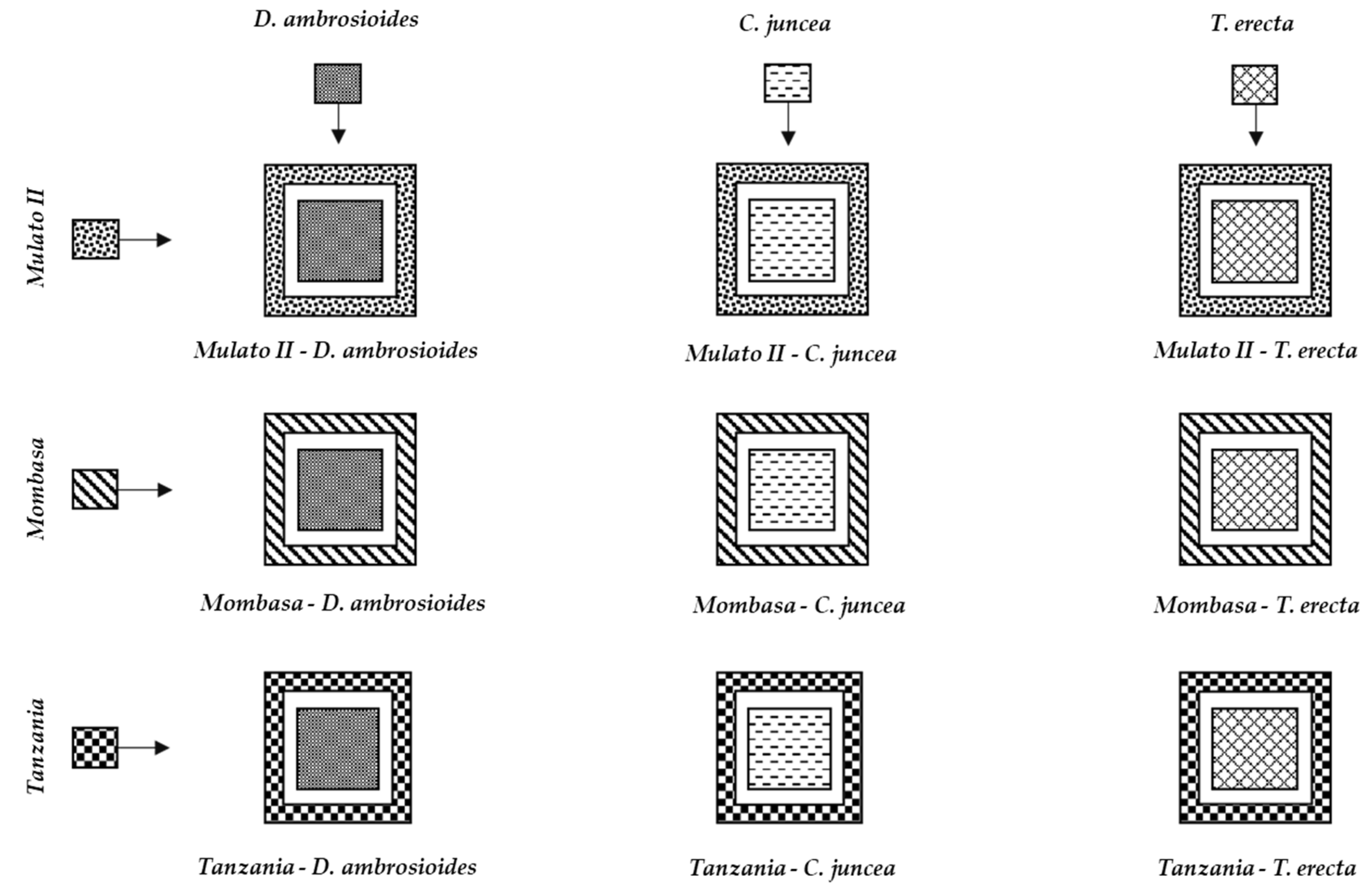
| Plants | Origin | Plant Height (m) | Altitude (m) | Main Uses | |
|---|---|---|---|---|---|
| Attractants | Brachiaria brizantha‡,§ | Uganda, Botswana | 2.50 | 0–1800 | Forage, Live Barrier |
| Panicum maximum cv. Tanzania ‡,* | Tropical and subtropical Africa | 2.00 | 0–1800 | Forage, grazing | |
| Panicum maximum cv. Mombasa ‡,* | 2.50 | 0–2000 | |||
| Brachiaria hybrid cv. Mulato II ‡, § | 3 generations of brachiaria pasture crossing | 1.00 | 0–1800 | Forage | |
| Lolium multiflorum‡,†,* | Africa, Europe and Asia | 1.30 | 0–2400 | Forage, Lawns | |
| Sorghum sudanense‡,§ | Africa | 3.00 | 0–1800 | Forage, Cut | |
| Repellents | Dysphania ambrosioides * | America | 1.00 | 0–3000 | Condiments, Medicinal |
| Crotalaria juncea * | India and Pakistan | 2.40 | 0–1900 | Forage, green manure | |
| Tagetes erecta * | Mesoamerica | 1.00 | 800–2300 | Ornamental, Religious, Medicinal | |
| Tagetes lucida * | Mexico and Guatemala | 0.80 | 800–2700 |
| Species Evaluated/Reference | Preference | t | p | |
|---|---|---|---|---|
| P. maximum cv. Tanzania/Z. mays | No | - | - | - |
| P. maximum cv. Mombasa */Z. mays | Yes | −0.156 | −1.712 | 0.089 |
| B. hybrid cv. Mulato II/Z. mays | No | - | - | - |
| L. multiflorum */Z. mays | Yes | 0.441 | 5.338 | <0.0001 |
| B. brizantha/Z. mays * | Yes | −0.765 | −12.884 | <0.0001 |
| S. sudanense */Z. mays | Yes | −0.094 | −1.021 | 0.310 |
| Species | Selected Model | Parameter Estimates | Inflection Point | (%) | RMSE (%) | p | |||
|---|---|---|---|---|---|---|---|---|---|
| Z. mays | Logistic | 1.277 * | 480.94 * | 0.360 * | - | t * = 17.15 days | 95.00 | 2.28 | <0.05 |
| Pt = 0.64 g | |||||||||
| P. maximum cv. Mombasa | Logistic | 0.438 * | 3839 * | 0.512 * | - | T* = 16.12 days | 98.48 | 1.71 | <0.05 |
| Pt = 0.22 g | |||||||||
| S. sudanense | Logistic | 0.373 * | 1705 * | 0.524 * | - | t * = 14.20 days | 98.85 | 1.23 | <0.05 |
| Pt = 0.18 g | |||||||||
| P. maximum cv. Tanzania | Logistic | 0.358 * | 932.6 * | 0.459 * | - | t * = 14.91 days | 98.84 | 1.35 | <0.05 |
| Pt = 0.18 g | |||||||||
| L. multiflorum | Gompertz | 0.796 * | 1.712 * | 0.112 * | - | t * = 15.34 days | 94.82 | 2.42 | <0.05 |
| Pt = 0.29 g | |||||||||
| B. hybrid cv. Mulato II | Gompertz | 0.459 * | 1.705 * | 0.117 * | - | t * = 14.55 days | 96.47 | 1.27 | <0.05 |
| Pt = 0.17 g | |||||||||
| B. brizantha | Richards | 0.553 * | 0.823 * | 0.125 * | 0.257 * | t * = 17.45 days | 90.10 | 2.54 | <0.05 |
| Pt = 0.23 g | |||||||||
| Grass | Duration of Cycle Components (Days) | Complete Cycle (Days) | Pupae Weight (g) | ||||
|---|---|---|---|---|---|---|---|
| Egg | Larva | Prepupae | Pupae | Adult | |||
| Z. mays | 3.25 ± 0.09 B | 14.00 ± 0.00 C | 1.62 ± 0.18 | 6.86 ± 0.40 | 10.43 ± 0.20 | 36.08 ± 0.60 C | 0.125 ± 0.012 A |
| P. maximum cv. Mombasa | 3.55 ± 0.11 AB | 21.25 ± 0.37 A | 2.62 ± 0.18 | 7.50 ± 0.38 | 10.25 ± 0.17 | 45.17 ± 0.53 A | 0.119 ± 0.006 AB |
| S. sudanense | 3.30 ± 0.10 B | 17.22 ± 0.15 B | 2.16 ± 0.16 | 6.83 ± 0.65 | 11.33 ± 0.33 | 40.80 ± 0.72 B | 0.104 ± 0.007 AB |
| P. maximum cv. Tanzania | 3.60 ± 0.11 AB | 20.28 ± 0.68 A | 2.50 ± 0.34 | 7.40 ± 0.40 | 10.80 ± 0.20 | 45.10 ± 0.86 A | 0.115 ± 0.005 AB |
| L. multiflorum | 3.30 ± 0.10 B | 15.22 ± 0.22 C | 1.55 ± 0.24 | 6.28 ± 0.28 | 10.86 ± 0.34 | 36.73 ± 0.29 C | 0.099 ± 0.007 AB |
| B. brizantha | 3.80 ± 0.09 A | 19.57 ± 0.67 A | 2.14 ± 0.55 | 7.43 ± 0.29 | 10.57 ± 0.29 | 45.00 ± 0.49 A | 0.088 ± 0.007 B |
| B. hybrid cv. Mulato II | 3.65 ± 0.11 AB | 20.33 ± 0.50 A | 2.33 ± 0.29 | 7.67 ± 0.17 | 10.78 ± 0.28 | 45.27 ± 0.46 A | 0.095 ± 0.003 AB |
| F | 3.994 | 45.118 | 2.007 | 1.864 | 1.608 | 55.559 | 3.434 |
| p | 0.001 | <0.05 | 0.084 | 0.110 | 0.169 | <0.05 | 0.007 |
| Variables | Principal Components | Communalities | |
|---|---|---|---|
| CP1 | CP2 | ||
| Larval growth performance | −0.932 | −0.090 | 0.877 |
| Cycle duration | 0.919 | −0.098 | 0.854 |
| Oviposition | 0.829 | 0.379 | 0.831 |
| Attraction | 0.723 | 0.537 | 0.810 |
| Feeding | −0.545 | 0.523 | 0.570 |
| Survival | −0.578 | 0.712 | 0.841 |
| Eigenvalues | 3.553 | 1.230 | - |
| Variance (%) | 59.21 | 20.50 | - |
| Cumulative variance (%) | 59.21 | 79.71 | - |
| Species | TRAPS | Ranking |
|---|---|---|
| Panicum maximum cv. Mombasa | 388.699 | 1 |
| Panicum maximum cv. Tanzania | 299.441 | 2 |
| Brachiaria hybrid cv. Mulato II | 290.397 | 3 |
| Sorghum sudanense | 289.642 | 4 |
| Brachiaria brizantha | 278.859 | 5 |
| Zea mays | 239.517 | 6 |
| Lolium multiflorum | 164.394 | 7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guera, O.G.M.; Castrejón-Ayala, F.; Robledo, N.; Jiménez-Pérez, A.; Sánchez-Rivera, G. Plant Selection for the Establishment of Push–Pull Strategies for Zea mays–Spodoptera frugiperda Pathosystem in Morelos, Mexico. Insects 2020, 11, 349. https://doi.org/10.3390/insects11060349
Guera OGM, Castrejón-Ayala F, Robledo N, Jiménez-Pérez A, Sánchez-Rivera G. Plant Selection for the Establishment of Push–Pull Strategies for Zea mays–Spodoptera frugiperda Pathosystem in Morelos, Mexico. Insects. 2020; 11(6):349. https://doi.org/10.3390/insects11060349
Chicago/Turabian StyleGuera, Ouorou Ganni Mariel, Federico Castrejón-Ayala, Norma Robledo, Alfredo Jiménez-Pérez, and Georgina Sánchez-Rivera. 2020. "Plant Selection for the Establishment of Push–Pull Strategies for Zea mays–Spodoptera frugiperda Pathosystem in Morelos, Mexico" Insects 11, no. 6: 349. https://doi.org/10.3390/insects11060349
APA StyleGuera, O. G. M., Castrejón-Ayala, F., Robledo, N., Jiménez-Pérez, A., & Sánchez-Rivera, G. (2020). Plant Selection for the Establishment of Push–Pull Strategies for Zea mays–Spodoptera frugiperda Pathosystem in Morelos, Mexico. Insects, 11(6), 349. https://doi.org/10.3390/insects11060349






