Large- and Small-Scale Environmental Factors Drive Distributions of Ant Mound Size Across a Latitudinal Gradient
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Sampling Design
2.3. Statistical Analyses
3. Results
4. Discussions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Research Involving Human and Animal Participants
References
- Folgarait, P.J. Ant biodiversity and its relationship to ecosystem functioning: A review. Biodivers. Conserv. 1998, 7, 1221–1244. [Google Scholar] [CrossRef]
- Johnson, F.S.; Mo, T.; Green, A.E.S. Average latitudinal variation in ultraviolet radiation at the Earth’s surface. Photochem. Photobiol. 1976, 23, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Tabony, R.C. The variation of surface temperature with altitude. Met. Mag. 1985, 114, 37–48. [Google Scholar]
- Bergmann, K.G.L.C. Ueber die Verhaltnisse der Warmeokonomie der thiere zu ihrer grosse. Gottinger Studien 1847, 3, 595–708. [Google Scholar]
- Geist, V. Bergmann’s rule is invalid. Can. J. Zool. 1987, 65, 1035–1038. [Google Scholar] [CrossRef]
- Hölldobler, B.; Wilson, E.O. The Ants; Harvard Univ. Press: Cambridge, MA, USA, 1990; pp. 23–33, 378–435. [Google Scholar]
- Samson, D.A.; Rickart, E.A.; Gonzales, P.C. Ant diversity and abundance along an elevational gradient in the Philippines. Biotropica 1997, 29, 349–363. [Google Scholar] [CrossRef]
- Blackburn, T.M.; Gaston, K.J.; Loder, N. Geographic gradients in body size: A clarification of Bergmann’s rule. Divers. Distrib. 1999, 5, 165–174. [Google Scholar] [CrossRef]
- Machac, A.; Janda, M.; Dunn, R.R.; Sanders, N.J. Elevational gradients in phylogenetic structure of ant communities reveal the interplay of biotic and abiotic constraints on diversity. Ecography 2011, 34, 364–371. [Google Scholar] [CrossRef]
- Vandegehuchte, M.L.; Wermelinger, B.; Fraefel, M.; Baltensweiler, A.; Düggelind, C.; Brändli, U.-B.; Freitag, A.; Bernasconi, C.; Cherix, D.; Risch, A.C. Distribution and habitat requirements of red wood ants in Switzerland: Implications for conservation. Biol. Conserv. 2017, 212, 366–375. [Google Scholar] [CrossRef]
- Economo, E.P.; Narula, N.; Friedman, N.R.; Weiser, M.D.; Guénard, B. Macroecology and macroevolution of the latitudinal diversity gradient in ants. Nat. Commun. 2018, 9, 1778. [Google Scholar] [CrossRef]
- Alhajeri, B.H.; Steppan, S.J. Association between climate and body size in rodents: A phylogenetic test of Bergmann’s rule. Mamm. Biol. 2016, 81, 219–225. [Google Scholar] [CrossRef]
- Gohli, J.; Voje, K.L. An interspecific assessment of Bergmann’s rule in 22 mammalian families. BMC Evol. Biol. 2016, 16, 222. [Google Scholar] [CrossRef] [PubMed]
- Torres-Romero, E.J.; Morales-Castilla, I.; Olalla-Tárraga, M.Á. Bergmann’s rule in the oceans? Temperature strongly correlates with global interspecific patterns of body size in marine mammals. Glob. Ecol. Biogeogr. 2016, 25, 1206–1215. [Google Scholar] [CrossRef]
- Mousseau, T.A. Ectotherms follow the converse to Bergmann’s rule. Evolution 1997, 51, 630–632. [Google Scholar] [CrossRef]
- Blanckenhorn, W.U.; Demont, M. Bergmann and converse Bergmann latitudinal clines in arthropods: Two ends of a continuum? Integr. Comp. Biol. 2004, 44, 413–424. [Google Scholar] [CrossRef]
- Feldman, A.; Meiri, S. Australian Snakes Do Not Follow Bergmann’s Rule. Evol. Biol. 2014, 41, 327–335. [Google Scholar] [CrossRef]
- Beerli, N.; Bärtschi, F.; Ballesteros-Mejia, L.; Kitching, I.J.; Beck, J. How has the environment shaped geographical patterns of insect body sizes? A test of hypotheses using sphingid moths. J. Biogeogr. 2019, 46, 1687–1698. [Google Scholar] [CrossRef]
- Zamora-Camacho, F.J.; Reguera, S.; Moreno-Rueda, G. Bergmann’s Rule rules body size in an ectotherm: Heat conservation in a lizard along a 2200-metre elevational gradient. J. Evol. Biol. 2014, 27, 2820–2828. [Google Scholar] [CrossRef]
- Gérard, M.; Vanderplanck, M.; Franzen, M.; Kuhlmann, M.; Potts, S.G.; Rasmont, P.; Schweiger, O.; Michez, D. Patterns of size variation in bees at a continental scale: Does Bergmann’s rule apply? Oikos 2018, 127, 1095–1103. [Google Scholar] [CrossRef]
- Yu, T.L.; Wang, D.L.; Busam, M.; Deng, Y.H. Altitudinal variation in body size in Bufo minshanicus supports Bergmann’s rule. Evol. Ecol. 2019, 33, 449–460. [Google Scholar] [CrossRef]
- Wheeler, W.M. The ant-colony as an organism. J. Morphol. 1911, 22, 307–325. [Google Scholar] [CrossRef]
- Wilson, E.O.; Hölldobler, B. The Superorganism: The Beauty, Elegance, and Strangeness of Insect Societies; W. W. Norton & Company: New York, NY, USA, 2008; pp. 3–10. [Google Scholar]
- Kadochová, Š.; Frouz, J. Red wood ants Formica polyctena switch off active thermoregulation of the nest in autumn. Insect. Soc. 2014, 61, 297–306. [Google Scholar] [CrossRef]
- Kadochová, Š.; Frouz, J. Thermoregulation strategies in ants in comparison to other social insects, with a focus on red wood ants (Formica rufa group). F1000Research 2014, 2, 280. [Google Scholar] [CrossRef] [PubMed]
- Czechowski, W.; Radchenko, A.; Czechowska, W. The Ants (Hymenoptera, Formicidae) of Poland; Museum and Institute of Zoology PAS: Warsaw, Poland, 2002; pp. 71–80. [Google Scholar]
- Stabentheiner, A.; Pressl, J.; Papst, T.; Hrassnigg, N.; Crailsheim, K. Endothermic heat production in honeybee winter clusters. J. Exp. Biol. 2003, 206, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Hozumi, S.; Mateus, S.; Kudô, K.; Kuwahara, T.; Yamane, S.; Zucchi, R. Nest thermoregulation in Polybia scutellaris (White) (Hymenoptera: Vespidae). Neotrop. Entomol. 2010, 39, 826–828. [Google Scholar] [CrossRef]
- Punttila, P.; Kilpeläinen, J. Distribution of mound-building ant species (Formica spp., Hymenoptera) in Finland: Preliminary results of a national survey. Ann. Zool. Fenn. 2009, 46, 1–15. [Google Scholar] [CrossRef][Green Version]
- Kaspari, M.; Vargo, E.L. Colony size as a buffer against seasonality: Bergmann’s rule in social insects. Am. Nat. 1995, 145, 610–632. [Google Scholar] [CrossRef]
- Cushman, J.H.; Lawton, J.H.; Manly, B.F.J. Latitudinal patterns in European ant assemblages: Variation in species richness and body size. Oecologia 1993, 95, 30–37. [Google Scholar] [CrossRef]
- Heinze, J.; Foitzik, S.; Fischer, B.; Wanke, T.; Kipyatkov, V.E. The significance of latitudinal variation in body size in a holarctic ant, Leptothorax acervorum. Ecography 2003, 26, 349–355. [Google Scholar] [CrossRef]
- Geraghty, M.J.; Dunn, R.R.; Sanders, N.J. Body size, colony size, and range size in ants (Hymenoptera: Formicidae): Are patterns along elevational and latitudinal gradients consistent with Bergmann’s Rule? Myrmecol. News 2007, 10, 51–58. [Google Scholar]
- Bernadou, A.; Römermann, C.; Gratiashvili, N.; Heinze, J. Body size but not colony size increases with altitude in the holarctic ant, Leptothorax acervorum. Ecol. Entomol. 2016, 41, 733–736. [Google Scholar] [CrossRef]
- Purcell, J.; Pirogan, D.; Avril, A.; Bouyarden, F.; Chapuisat, M. Environmental influence on the phenotype of ant workers revealed by common garden experiment. Behav. Ecol. Sociobiol. 2016, 70, 357–367. [Google Scholar] [CrossRef]
- Seifert, B. The Ants of Central and North Europe; Lutra Verlags-und Vertiebsgesellschaft: Tauer, Germany, 2018; pp. 299–332. [Google Scholar]
- Stockan, J.A.; Robinson, E.J. Wood Ant Ecology and Conservation; Cambridge Univ. Press: Cambridge, UK, 2016. [Google Scholar]
- Sorvari, J.; Hakkarainen, H. Deforestation reduces nest mound size and decreases the production of sexual offspring in the wood ant Formica aquilonia. Ann. Zool. Fenn. 2005, 42, 259–267. [Google Scholar]
- Sorvari, J.; Hakkarainen, H. Forest clear-cutting causes small workers in the polydomous wood ant Formica aquilonia. Ann. Zool. Fenn. 2009, 46, 431–438. [Google Scholar] [CrossRef]
- Chen, J.-H.; Robinson, E.J.H. The relationship between canopy cover and colony size of the wood ant Formica lugubris-implications for the thermal effects on a keystone ant species. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Chen, J.; Franklin, J.F.; Spies, T.A. Growing-season microclimatic gradients from clearcut edges into old-growth Douglas-fir forests. Ecol. Appl. 1995, 5, 74–86. [Google Scholar] [CrossRef]
- Sorvari, J.; Haatanen, M.-K.; Vesterlund, S.-R. Combined effects of overwintering temperature and habitat degradation on the survival of boreal wood ant. J. Insect Conserv. 2011, 15, 727–731. [Google Scholar] [CrossRef]
- Frouz, J.; Finer, L. Diurnal and seasonal fluctuations in wood ant (Formica polyctena) nest temperature in two geographically distant populations along a south–north gradient. Insect. Soc. 2007, 54, 251–259. [Google Scholar] [CrossRef]
- Jílková, V.; Cajthaml, T.; Frouz, J. Respiration in wood ant (Formica aquilonia) nests as affected by altitudinal and seasonal changes in temperature. Soil Biol. Biochem. 2015, 86, 50–57. [Google Scholar] [CrossRef]
- Punttila, P.; Haila, Y.; Pajunen, T.; Tukia, H. Colonization of clear-cut forests by ants in the Southern Finnish taiga: A quantitative survey. Oikos 1991, 61, 250–262. [Google Scholar] [CrossRef]
- Maeto, K.; Sato, S. Impacts of forestry on ant species richness and composition in warm-temperate forests of Japan. For. Ecol. Manag. 2004, 187, 213–223. [Google Scholar] [CrossRef]
- Procter, D.S.; Cottrell, J.; Watts, K.; Robinson, E.J.H. Do non-native conifer plantations provide benefits for a native forest specialist, the wood ant Formica lugubris? For. Ecol. Manag. 2015, 357, 22–32. [Google Scholar] [CrossRef]
- Arnan, X.; Cerdá, X.; Retana, J. Relationships among taxonomic, functional, and phylogenetic ant diversity across the biogeographic regions of Europe. Ecography 2017, 40, 448–457. [Google Scholar] [CrossRef]
- Jones, J.C.; Oldroyd, B.P. Nest thermoregulation in social insects. Adv. Insect Physiol. 2007, 33, 153–191. [Google Scholar]
- Kilpeläinen, J.; Punttila, P.; Finér, L.; Niemelä, P.; Domisch, T.; Jurgensen, M.F.; Neuvonen, S.; Ohashi, M.; Risch, A.C.; Sundström, L. Distribution of ant species and mounds (Formica) in different-aged managed spruce stands in Eastern Finland. J. Appl. Entomol. 2008, 132, 315–325. [Google Scholar] [CrossRef]
- Sondej, I.; Domisch, T.; Finér, L.; Czechowski, W. Wood ants in the Bialowieza Forest and factors affecting their distribution. Ann. Zool. Fenn. 2018, 55, 103–114. [Google Scholar] [CrossRef]
- Hijmans, E.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Risch, A.C.; Jurgensen, M.F.; Schütz, M.; Page-Dumroese, D.S. The Contribution of Red Wood Ants to Soil C and N Pools and CO2 Emissions in Subalpine Forests. Ecology 2005, 86, 419–430. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. lme4: Linear Mixed-Effects Models Using Eigen and S4. 2013 R Package Version 1.0-5. Available online: http://CRAN.R-project.org/package=lme4 (accessed on 22 October 2019).
- Bartoń, K. MuMIn: Multi-Model Inference. 2013 R Package Version 1.9.13. Available online: http://CRAN.R-project.org/package=MuMIn (accessed on 22 October 2019).
- Russell, L. Emmeans: Estimated Marginal Means, Aka LEAST-Squares Means. 2019 R Package Version 1.4.2. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 22 October 2019).
- Ellis, S.; Franks, D.W.; Robinson, E.J.H. Ecological consequences of colony structure in dynamic ant nest networks. Ecol. Evol. 2017, 7, 1170–1180. [Google Scholar] [CrossRef]
- Savolainen, R.; Vepsäläinen, K. A competition hierarchy among boreal ants: Impact on resource partitioning and community structure. Oikos 1988, 51, 135–155. [Google Scholar] [CrossRef]
- Punttila, P.; Haila, Y.; Tukia, H. Ant communities in taiga clearcuts: Habitat effects and species interactions. Ecography 1996, 19, 16–28. [Google Scholar] [CrossRef]
- Klimetzek, D. Population studies on hill building wood-ants of the Formica rufa-group. Oecologia 1981, 48, 418–421. [Google Scholar] [CrossRef] [PubMed]
- Härkönen, S.K.; Sorvari, J. Species richness of associates of ants in the nests of red wood ant Formica polyctena (Hymenoptera, Formicidae). Insect. Conserv. Divers. 2014, 7, 485–495. [Google Scholar] [CrossRef]
- Seidl, R.; Baier, P.; Rammer, W.; Schopf, A.; Lexer, M.J. Modelling tree mortality by bark beetle infestation in Norway spruce forests. Ecol. Model. 2007, 206, 383–399. [Google Scholar] [CrossRef]
- Linder, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Sorvari, J.; Hakkarainen, H. Forest clearing and sex ratio in forest-dwelling wood ant Formica aquilonia. Naturwissenschaften 2007, 94, 392–395. [Google Scholar] [CrossRef]
- Pisarski, B.; Czechowski, W. The course of artificial colonization of red wood ants in the Gorce National Park. Mem. Zool. 1990, 44, 37–46. [Google Scholar]
- Sorvari, J.; Huhta, E.; Hakkarainen, H. Survival of transplanted nests of the red wood ant Formica aquilonia (Hymenoptera: Formicidae): The effects of intraspecific competition and forest clear-cutting. Insect. Sci. 2014, 21, 486–492. [Google Scholar] [CrossRef]
- Way, J.M.; Khoo, K.C. Role of ants in pest management. Annu. Rev. Entomol. 1992, 37, 479–503. [Google Scholar] [CrossRef]
- Cilbircioglu, C.; Ünal, S. Bark beetles and their natural enemies on oriental spruce from the Black Sea region of Turkey. J. Agric. Urban Entomol. 2012, 28, 42–56. [Google Scholar] [CrossRef]
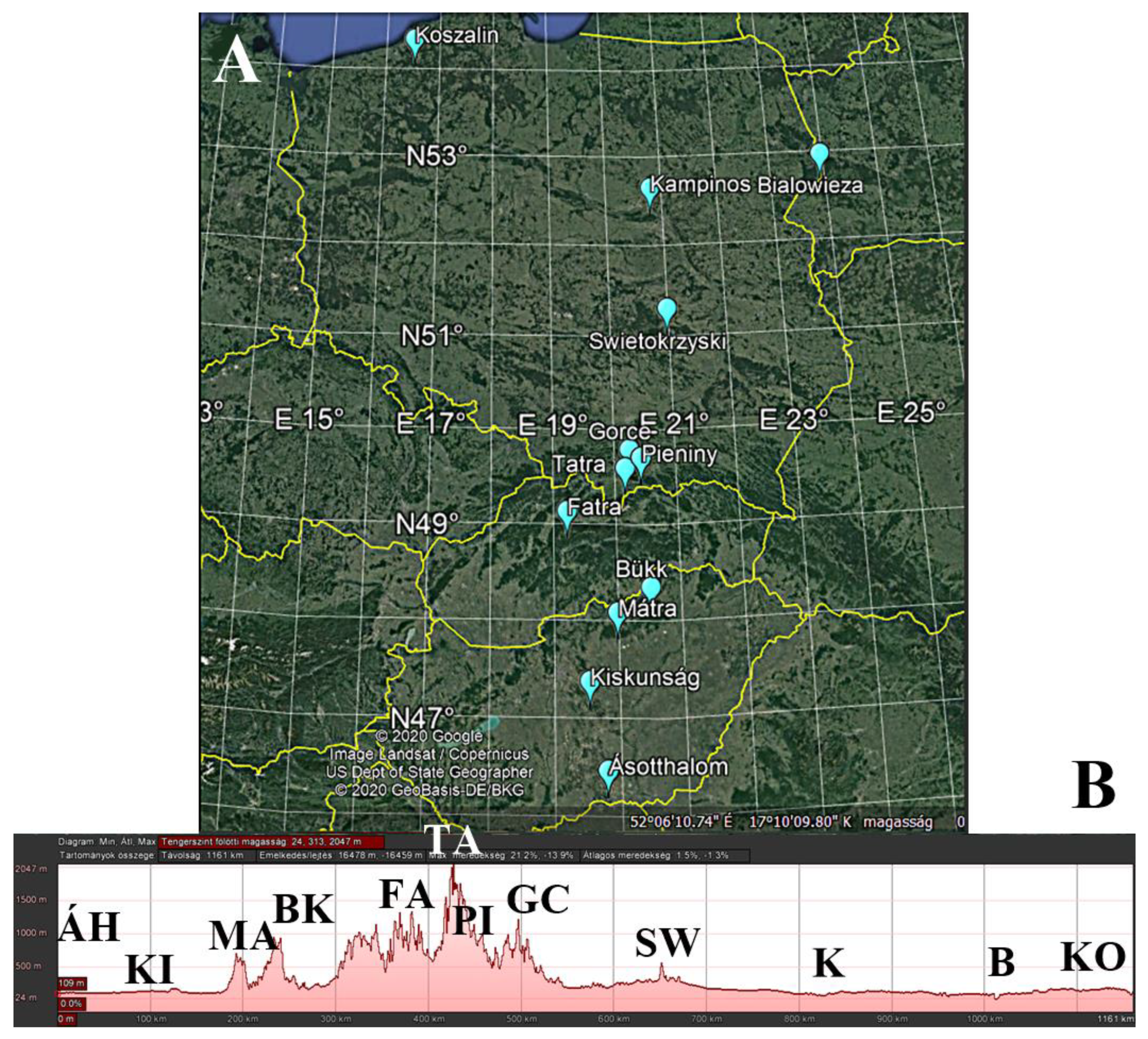
| Sampling Site | Distance Value | Latitude (N) | Longitude (E) | Average Altitude (m) |
|---|---|---|---|---|
| Ásotthalom | 1 | 46.215283° | 19.782783° | 118 |
| Kiskunság | 6 | 47.121150° | 19.504833° | 122 |
| Mátra | 10 | 47.827283° | 19.974950° | 429 |
| Bükk | 13 | 48.081000° | 20.502667° | 789 |
| Fatra | 20 | 48.882450° | 19.211333° | 871 |
| Tatra | 24 | 49.325650° | 20.153967° | 787 |
| Pieniny | 25 | 49.435850° | 20.423150° | 602 |
| Gorce | 26 | 49.520267° | 20.232833° | 724 |
| Świętokrzyska | 35 | 50.886933° | 21.094567° | 302 |
| Kampinos | 43 | 52.361400° | 20.792717° | 88 |
| Białowieza | 45 | 52.698183° | 23.891283° | 178 |
| Koszalin | 61 | 54.069650° | 16.535000° | 90 |
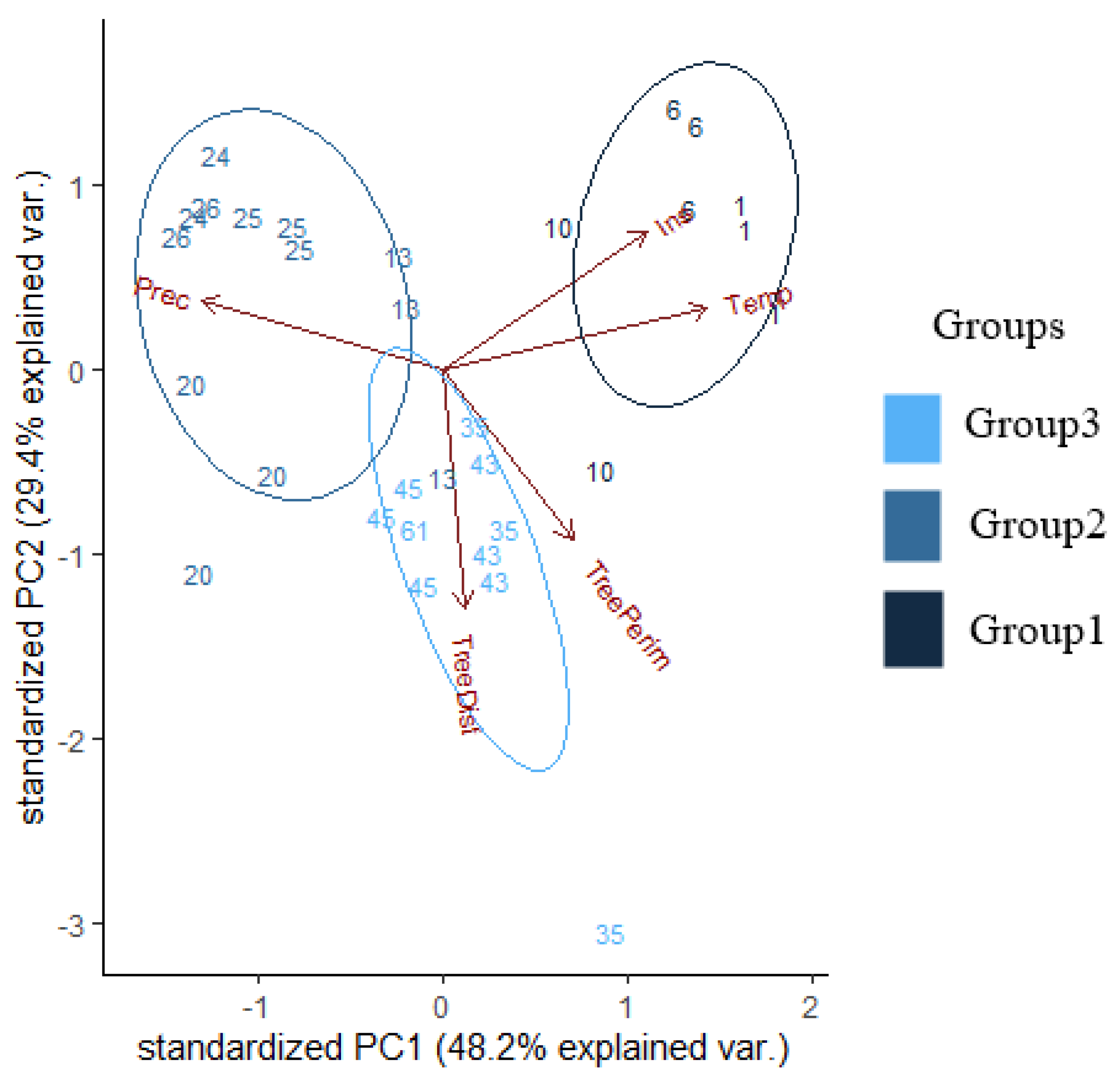
| Background Variables | PC1 | PC2 | PC3 |
|---|---|---|---|
| Distance of trees | 5.2% | −71% | 54.87% |
| Perimeter of trees | 30.11% | −50.69% | −76.3% |
| Temperature | 61.24% | 19.9% | −6.58% |
| Precipitation | −55.56% | 20.2% | −28.44% |
| Irradiation | 47.19% | 40.72% | 17.69% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juhász, O.; Bátori, Z.; Trigos-Peral, G.; Lőrinczi, G.; Módra, G.; Bóni, I.; Kiss, P.J.; Aguilon, D.J.; Tenyér, A.; Maák, I. Large- and Small-Scale Environmental Factors Drive Distributions of Ant Mound Size Across a Latitudinal Gradient. Insects 2020, 11, 350. https://doi.org/10.3390/insects11060350
Juhász O, Bátori Z, Trigos-Peral G, Lőrinczi G, Módra G, Bóni I, Kiss PJ, Aguilon DJ, Tenyér A, Maák I. Large- and Small-Scale Environmental Factors Drive Distributions of Ant Mound Size Across a Latitudinal Gradient. Insects. 2020; 11(6):350. https://doi.org/10.3390/insects11060350
Chicago/Turabian StyleJuhász, Orsolya, Zoltán Bátori, Gema Trigos-Peral, Gábor Lőrinczi, Gábor Módra, Imola Bóni, Péter János Kiss, Dianne Joy Aguilon, Anna Tenyér, and István Maák. 2020. "Large- and Small-Scale Environmental Factors Drive Distributions of Ant Mound Size Across a Latitudinal Gradient" Insects 11, no. 6: 350. https://doi.org/10.3390/insects11060350
APA StyleJuhász, O., Bátori, Z., Trigos-Peral, G., Lőrinczi, G., Módra, G., Bóni, I., Kiss, P. J., Aguilon, D. J., Tenyér, A., & Maák, I. (2020). Large- and Small-Scale Environmental Factors Drive Distributions of Ant Mound Size Across a Latitudinal Gradient. Insects, 11(6), 350. https://doi.org/10.3390/insects11060350





