The Effect of Temperature on the Development of Spodoptera frugiperda (Lepidoptera: Noctuidae)
Abstract
1. Introduction
2. Materials and Methods
2.1. Spodoptera Frugiperda Stock Colony
2.2. Temperature-Dependent Egg Development
2.3. Temperature-Dependent Larval and Pupal Development
2.4. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamo, M. First report of outbreaks of the fall armyworm Spodoptera frugiperda (J E Smith) (Lepidoptera: Noctuidae), a new alien invasive pest in west and Central Africa. PLoS ONE 2016, 11, e0165632. [Google Scholar] [CrossRef] [PubMed]
- Du Plessis, H.; Van den Berg, J.; Ota, N.; Kriticos, D.J. Spodoptera frugiperda. Available online: http://natural-sciences.nwu.ac.za/uesm/news (accessed on 23 January 2020).
- Early, R.; Gonzalez-Moreno, P.; Murphy, S.T.; Day, R. Forecasting the global extent of invasion of the cereal pest Spodoptera frugiperda, the fall armyworm. bioRxiv 2018. [Google Scholar] [CrossRef]
- Kalleshwaraswamy, C.M.; Asokan, R.; Swamy, H.M.; Maruthi, M.S.; Pavithra, H.B.; Hegde, K.; Navi, S.; Prabhu, S.T.; Goergen, G. First report of the Fall armyworm, Spodoptera frugiperda (J E Smith) (Lepidoptera: Noctuidae), an alien invasive pest on maize in India. Pest Manag. Hort. Ecosyst. 2018, 24, 23–29. [Google Scholar]
- FAO. First Detection of Fall Army Worm on the Border of Thailand. IPPC Official Pest Report, No. THA-03/1. 2019. Available online: https://www.ippc.int/en/countries/thailand/pestreports/2018/12/first-detection-of-fall-army-worm-on-the-border-of-thailand/ (accessed on 23 January 2020).
- FAO. First detection report of the Fall Armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae) on maize in Myanmar. IPPC Official Pest Report, No. MMR-19/6. 2019. Available online: https://www.ippc.int/ en/countries/Myanmar/pestreports/2019/01/first-detection-report-of-the-fall-armyworm-spodoptera-frugiperda-lepidoptra-noctuidae-on-maize-in-myanma/ (accessed on 23 January 2020).
- FAO. First Detection of Fall Armyworm in China. 2019. Available online: https://www.ippc.int/fr/news/first-detection-of-fall-armyworm-in-china/ (accessed on 23 January 2020).
- FAO. RepReport of first detection of Fall Armyworm (FAW) in Republic of Korea IPPC Official Pest Report, No. KOR-08/2. 2019. Available online: https://www.ippc.int/en/countries/republic-of-korea/pestreports/2019/06/report-of-first-detection-of-fall-armywormfaw-in-republic-of-korea/ (accessed on 23 January 2020).
- FAO. Report of first detection of Spodoptera frugiperda—Fall Armyworm (FAW) in Japan. IPPC Official Pest Report, No. JPN-08/6. 2019. Available online: https://www.ippc.int/ en/countries/japan/pestreports/2019/07/report-of-first-detection-of-spodoptera-frugipedera-fall-armyworm-faw-in-japan/ (accessed on 23 January 2020).
- FAO. Report of first detection of Fall Army Worm (FAW) in the Republic of the Philippines. In: IPPC Official Pest Report, (No. PHL-02/1). 2019. Available online: https://www.ippc.int/ en/countries/philippines/pestreports/2019/10/report-of-first-detection-of-fall-army-worm-faw-in-the-republic-of-the-philippines/ (accessed on 23 January 2020).
- FAO. Report of first detection of Spodoptera frugiperda—Fall Armyworm (FAW) in Indonesia. In: IPPC Official Pest Report, No. IDN-04/1. 2019. Available online: https://www.ippc.int/ countries/indonesia/pestreports/2019/07/the-occurence-of-fall-armyworm-spodoptera-frugiperda-in-indonesia/ (accessed on 23 January 2020).
- ABC News. Fall armyworm found in Torres Strait, Prompting Biosecurity alert amid Fears for Australian Crops. Available online: https://www.abc.net.au/news/2020-02-12/worm-moth-fall-armyworm-detected-found-australia-torres-strait/11957838 (accessed on 12 February 2020).
- Tobin, P.C.; Nagarkatti, S.; Saunders, M.C. Phenology of Grape berry moth (Lepidoptera: Tortricidae) in cultivated grape at selected geographic locations. Environ. Entomol. 2003, 32, 340–346. [Google Scholar] [CrossRef]
- Howe, R.W. Temperature effects on embryonic development in insects. Annu. Rev. Entomol. 1967, 12, 15–42. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, T.; Hilszczański, J. The effect of temperature and humidity changes on insect development and their impact on forest ecosystems in the context of expected climate change. For. Res. Pap. 2013, 74, 345–355. [Google Scholar]
- Begon, M.; Townsend, C.R.; Harper, J.L. Ecology: From Individuals to Ecosystems; Blackwell Publishing Ltd.: Oxford, UK, 2006; Volume 3, pp. 30–57. [Google Scholar]
- Aguilon, D.J.D.; Velasco, L.R.I. Effects of larval rearing temperature and host plant condition on the development, survival, and coloration of African armyworm, Spodoptera exempta Walker (Lepidoptera: Noctuidae). J. Environ. Sci. Manag. 2015, 18, 54–60. [Google Scholar]
- Porter, J.H.; Parry, M.L.; Carter, T.R. The potential effects of climatic change on agricultural insect pests. Agric. For. Meteorol. 1991, 57, 221–240. [Google Scholar] [CrossRef]
- Calvo, D.; Molina, J.M. Developmental rates of the lappet moth Streblote panda Hübner (1820) (Lepidoptera: Lasiocampidae) at constant temperatures. Span. J. Agric. Res. 2005, 3, 1–8. [Google Scholar] [CrossRef]
- Hagstrum, D.W.; Hagstrum, W.R. A simple device for producing fluctuating temperatures, with an evaluation of the ecological significance of fluctuating temperatures. Ann. Entomol. Soc. Am. 1970, 63, 1385–1389. [Google Scholar] [CrossRef]
- Mironidis, G.K. Development, survivorship and reproduction of Helicoverpa armigera (Lepidoptera: Noctuidae) under fluctuating temperatures. Bull. Entomol. Res. 2014, 104, 751–764. [Google Scholar] [CrossRef] [PubMed]
- Shanower, T.G.; Schulthess, F.; Bosque-Pérez, N.A. The effect of larval diet on the growth and development of Sesamia calamistis Hampson (Lepidoptera: Noctuidae) and Eldana saccharina Walker (Lepidoptera: Pyralidae). Int. J. Trop. Insect Sci. 1993, 14, 681–685. [Google Scholar] [CrossRef]
- Kruger, M.; Van Rensburg, J.B.J.; Van den Berg, J. Transgenic Bt maize: Farmers’ perceptions refuge compliance and reports of stem borer resistance in South Africa. J. Appl. Entomol. 2012, 136, 38–50. [Google Scholar] [CrossRef]
- Solomon, M.E. Control of humidity with potassium hydroxide, sulphuric acid or other solutions. Bull. Entomol. Res. 1951, 42, 543–554. [Google Scholar] [CrossRef]
- Campbell, A.; Frazer, B.D.; Gilbert, N.; Gutierrez, A.P.; Mackauer, M. Temperature requirements of some aphids and their parasites. J. Appl. Ecol. 1974, 11, 431–438. [Google Scholar] [CrossRef]
- Jackson, J.J.; Elliot, N.C. Temperature-dependent development of immature stages of the western corn rootworm, Diabrotica virgifera (Coleoptera: Chrysomelidae). Environ. Entomol. 1988, 17, 166–171. [Google Scholar] [CrossRef]
- TIBCO Software, Inc. Statistica (Data Analysis Software System), Version 13.3. Available online: https://www.tibco.com/ (accessed on 23 January 2020).
- Wagner, T.L.; Wu, H.; Sharpe, P.J.H.; Schoolfield, R.M.; Coulson, R.M. Modelling distributions of insect development time: A literature review and application of the Weibull function. Ann. Èntomol. Soc. Am. 1984, 77, 475–487. [Google Scholar] [CrossRef]
- Ali, A.; Luttrell, R.G.; Schneider, J.C. Effects of temperature and larval diet on development of the fall army worm (Lepidoptera: Noctuidae). Ann. Entomol. Soc. Am. 1990, 83, 725–733. [Google Scholar] [CrossRef]
- Luginbill, P. The Fall Army Worm; USDA: Washington, DC, USA, 1928.
- Busato, G.R.; Grützmacher, A.D.; Garcia, M.S.; Giolo, F.P.; Zotti, M.J.; Bandeira, J.D.M. Exigências térmicas e estimative do número de gerações dos biótipos “milho” e “arroz”de Spodoptera frugiperda. Pesq. Agropec. Bras. 2005, 40, 329–335. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Sosa-Gómez, D.R.; Roque-Specht, V.F.; de Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Developmental parameters of Spodoptera frugiperda (Lepidoptera: Noctuidae) immature stages under controlled and standardized conditions. J. Agric. Sci. 2019, 11, 76–89. [Google Scholar] [CrossRef]
- Jarošík, V.; Honek, A.; Dixon, A.F.G. Developmental rate isomorphy in insects and mites. Am. Nat. 2002, 160, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Sparks, A.N. A review of the biology of the fall armyworm. Fla. Entomol. 1979, 62, 82–87. [Google Scholar] [CrossRef]
- Capinera, J.L. Handbook of Vegetable Pests; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Simmons, A.M. Fall armyworm symposium: Effects of constant and fluctuating temperatures and humidities on the survival of Spodoptera frugiperda pupae (Lepidoptera: Noctuidae). Fla. Entomol. 1993, 76, 333–340. [Google Scholar] [CrossRef]
- Hogg, D.B.; Pitre, H.N.; Anderson, R.E. Assessment of early-season phenology of the fall armyworm (Lepidoptera: Noctuidae) in Mississippi. Environ. Entomol. 1982, 11, 705–710. [Google Scholar] [CrossRef]
- Barfield, C.S.; Mitchell, E.R.; Poe, S.L. A temperature-dependent model for fall armyworm development. Ann. Entomol. Soc. Am. 1978, 71, 70–74. [Google Scholar] [CrossRef]
- Pitre, H.N.; Hogg, D.B. Development of the fall armyworm on cotton, soybean and corn. J. Ga. Entomol. Soc. 1983, 18, 187–194. [Google Scholar]
- Nagoshi, R.N.; Goergen, G.; Du Plessis, H.; Van den Berg, J.; Meagher, R., Jr. Genetic comparisons of fall armyworm populations from 11 countries spanning sub-Saharan Africa provide insights into strain composition and migratory behaviors. Sci. Rep. 2019, 9, 8311. [Google Scholar] [CrossRef]
- Wood, J.R.; Poe, S.L.; Leppla, N.C. Winter survival of fall armyworm pupae in Florida. Environ. Entomol. 1979, 8, 249–252. [Google Scholar] [CrossRef]
- Vickery, R.A. Studies on the fall armyworm in the Gulf coast district of Texas. United Sates Department of Agriculture, Washington, D.C. Tech. Bull. 1929, 138, 64. [Google Scholar] [CrossRef]
- Gullan, P.J.; Cranston, P.S. The Insects. An Outline of Entomology, 4th ed.; Wiley-Blackwell: West Sussex, UK, 2010; pp. 151–187. [Google Scholar]
- Perkins, W.D. Laboratory rearing of the fall armyworm. Fla. Entomol. 1979, 62, 87–91. [Google Scholar] [CrossRef]
- Morrill, W.L. Ecology, Economics and Behavior of the Fall Armyworm in Field Corn. Ph.D. Thesis, University of Florida, Gainesville, FL, USA, 1971. [Google Scholar]
- Kelty, J.D.; Lee, R.E. Rapid cold-hardening of Drosophila melanogaster (Diptera: Drosophilidae) during ecologically based thermoperiodic cycles. J. Exp. Biol. 2001, 204, 1659–1666. [Google Scholar] [PubMed]
- Richard, E.; Lee, R.E., Jr.; Denlinger, D.L. Rapid cold-hardening: Ecological significance and underpinning mechanisms. In Low Temperature Biology of Insects; Cambridge University Press: Cambridge, UK, 2010; pp. 35–58. [Google Scholar]
- Cammell, M.E.; Knight, J.D. Effects of climatic-change on the population dynamics of crop pests. Adv. Ecol. Res. 1992, 22, 117–162. [Google Scholar]
- Marco, V.; Taberner, A.; Castañera, P. Development and survival of immature Aubeonymus mariaefranciscae (Coleoptera: Curculionidae) at constant temperatures. Ann. Entomol. Soc. Am. 1997, 90, 169–176. [Google Scholar] [CrossRef]
| Development Stage | Temperature (±1 °C) | ||||
|---|---|---|---|---|---|
| 18 | 22 | 26 | 30 | 32 | |
| Egg | 6.38 ± 0.05 a | 4.00 ± 0.00 b | 3.00 ± 0.00 b | 2.0 ± 0.0 c | 2.0 ± 0.0 c |
| (6–7) | −4 | −3 | −2 | −2 | |
| Instar 1 | 4.94 ± 0.15 a | 3.70 ± 0.10 b | 2.90 ± 0.21 c | 2.70 ± 0.08 c | 2.70 ± 0.08 c |
| (3–7) | (3–5) | (1–4) | (2–3) | (1–3) | |
| Instar 2 | 4.52 ± 0.11 a | 3.00 ± 0.12 b | 2.14 ± 0.16 bc | 1.90 ± 0.12 c | 1.33 ± 0.08 c |
| (3–7) | (2–5) | (1–3) | (1–3) | (1–2) | |
| Instar 3 | 5.00 ± 0.12 a | 2.48 ± 0.09 b | 2.00 ± 0.10 bc | 1.43 ± 0.11 cd | 1.06 ± 0.04 d |
| (4–6) | (2–3) | (1–3) | (1–2) | (1–2) | |
| Instar 4 | 5.16 ± 0.09 a | 2.85 ± 0.11 b | 2.10 ± 0.10 bc | 1.62 ± 0.11 c | 1.52 ± 0.09 c |
| (4–6) | (2–5) | (1–3) | (1–2) | (1–2) | |
| Instar 5 | 6.16 ± 0.16 a | 3.42 ± 0.15 b | 2.33 ± 0.11 c | 2.19 ± 0.13 c | 1.79 ± 0.07 c |
| (4–8) | (1–5) | (2–3) | (1–4) | (1–2) | |
| Instar 6 | 8.61 ± 0.24 a | 5.12 ± 0.16 b | 3.38 ± 0.16 b | 2.00 ± 0.00 c | 2.06 ± 0.04 c |
| (6–12) | (4–9) | (3–6) | −2 | (2–3) | |
| Larvae | 34.39 ± 0.41 a | 20.58 ± 015 b | 14.86 ± 0.31 bc | 11.38 ± 0.25 cd | 10.45 ± 0.10 d |
| (28–37) | (19–22) | (13–19) | (10–14) | (10–12) | |
| Pupae | 30.68 ± 0.28 a | 17.06 ± 0.24 b | 11.43 ± 0.22 bc | 9.00 ± 0.12 cd | 7.82 ± 0.10 d |
| (28–34) | (14–20) | (10–13) | (8–10) | (7–9) | |
| Egg to adult | 71.44 ± 0.40 a | 41.64 ± 0.32 b | 29.29 ± 0.29 bc | 22.38 ± 0.27 cd | 20.27 ± 0.15 d |
| (67–77) | (38–46) | (27–32) | (20–25) | (19–22) | |
| Larval mortality (%) | 71 | 37 | 15 | 4 | 28 |
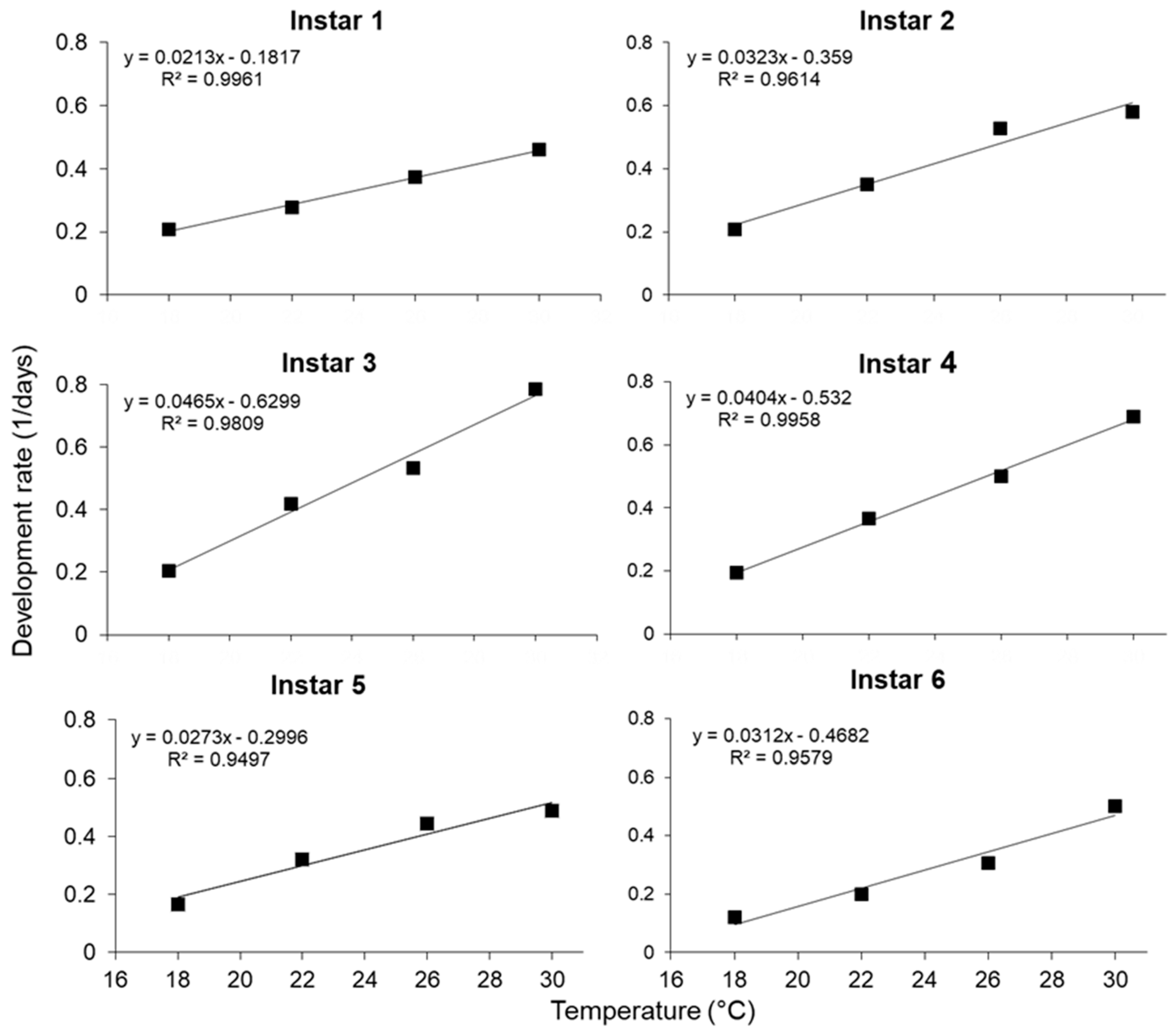
| Development Stage | Regression model | k ± S.E. | t ± S.E. | R2-Value |
|---|---|---|---|---|
| Eggs | y = 0.0280x − 0.3641 | 35.73 ± 0.31 | 13.01 ± 0.10 | 0.96 |
| First instar | y = 0.0212x − 0.1801 | 47.14 ± 3.13 | 8.49 ± 0.97 | 0.69 |
| Second instar | y = 0.0313x − 0.3315 | 31.98 ± 3.26 | 10.60 ± 1.28 | 0.48 |
| Third instar | y = 0.0463x − 0.6242 | 21.58 ± 1.49 | 13.47 ± 0.67 | 0.67 |
| Fourth instar | y = 0.0404x − 0.5293 | 24.78 ± 1.80 | 13.11 ± 0.73 | 0.65 |
| Fifth instar | y = 0.0278x − 0.3113 | 36.03 ± 3.13 | 11.21 ± 1.04 | 0.56 |
| Sixth instar | y = 0.0307x − 0.4560 | 32.57 ± 0.94 | 14.85 ± 0.24 | 0.92 |
| Pupal stages | y = 0.0067x − 0.0869 | 150.29 ± 2.79 | 13.06 ± 0.19 | 0.97 |
| Larval stage | y = 0.0049x − 0.0598 | 202.66 ± 4.45 | 12.12 ± 0.24 | 0.95 |
| Egg- to-adult | y = 0.0026x − 0.0322 | 390.41 ± 4.83 | 12.57 ± 0.13 | 0.98 |
| Developmental Stage | Temperature (°C) | n | Development time (days ± S.E.) | Range | Mean number of °D ± S.E. |
|---|---|---|---|---|---|
| Egg | 18 | 93 | 6.38 ± 0.05 | 6–7 | 31.84 ± 0.00 |
| 22 | 130 | 4.00 ± 0.00 | 4 | 35.96 ± 0.00 | |
| 26 | 151 | 3.00 ± 0.00 | 3 | 38.97 ± 0.00 | |
| 30 | 128 | 2.00 ± 0.00 | 2 | 33.98 ± 0.00 | |
| 32 | 162 | 2.00 ± 0.00 | 2 | 37.98 ± 0.00 | |
| Mean | 35.68 ± 0.22 | ||||
| Larvae | 18 | 31 | 34.39 ± 0.41 | 28–37 | 202.20 ± 2.43 |
| 22 | 33 | 20.58 ± 0.15 | 19–22 | 203.29 ± 1.49 | |
| 26 | 21 | 14.86 ± 0.31 | 13–19 | 206.22 ± 4.31 | |
| 30 | 21 | 11.38 ± 0.25 | 10–14 | 203.49 ± 4.53 | |
| 32 | 33 | 10.45 ± 0.10 | 10–12 | 207.84 ± 1.95 | |
| Mean | 204.60 ± 1.23 | ||||
| Pupae | 18 | 31 | 30.68 ± 0.28 | 28–34 | 151.55 ± 1.36 |
| 22 | 33 | 17.06 ± 0.24 | 14–20 | 152.52 ± 2.16 | |
| 26 | 21 | 11.43 ± 0.22 | 10–13 | 147.89 ± 2.90 | |
| 30 | 21 | 9.00 ± 0.12 | 8–10 | 152.46 ± 2.03 | |
| 32 | 33 | 7.82 ± 0.10 | 7–9 | 148.08 ± 1.93 | |
| Mean | 150.54 ± 0.93 | ||||
| Egg to adult | 18 | 31 | 71.44 ± 0.40 | 67–77 | 387.94 ± 2.17 |
| 22 | 33 | 41.64 ± 0.32 | 38–46 | 392.63 ± 2.98 | |
| 26 | 21 | 29.29 ± 0.29 | 27–32 | 393.31 ± 3.84 | |
| 30 | 21 | 22.38 ± 0.27 | 20–25 | 390.10 ± 4.73 | |
| 32 | 33 | 20.27 ± 0.15 | 19–22 | 393.90 ± 2.84 | |
| Mean | 391.61 ± 1.42 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du Plessis, H.; Schlemmer, M.-L.; Van den Berg, J. The Effect of Temperature on the Development of Spodoptera frugiperda (Lepidoptera: Noctuidae). Insects 2020, 11, 228. https://doi.org/10.3390/insects11040228
Du Plessis H, Schlemmer M-L, Van den Berg J. The Effect of Temperature on the Development of Spodoptera frugiperda (Lepidoptera: Noctuidae). Insects. 2020; 11(4):228. https://doi.org/10.3390/insects11040228
Chicago/Turabian StyleDu Plessis, Hannalene, Marie-Louise Schlemmer, and Johnnie Van den Berg. 2020. "The Effect of Temperature on the Development of Spodoptera frugiperda (Lepidoptera: Noctuidae)" Insects 11, no. 4: 228. https://doi.org/10.3390/insects11040228
APA StyleDu Plessis, H., Schlemmer, M.-L., & Van den Berg, J. (2020). The Effect of Temperature on the Development of Spodoptera frugiperda (Lepidoptera: Noctuidae). Insects, 11(4), 228. https://doi.org/10.3390/insects11040228





