Phylogenetic Community Structure and Niche Differentiation in Termites of the Tropical Dry Forests of Colombia
Abstract
1. Introduction
2. Materials and Methods
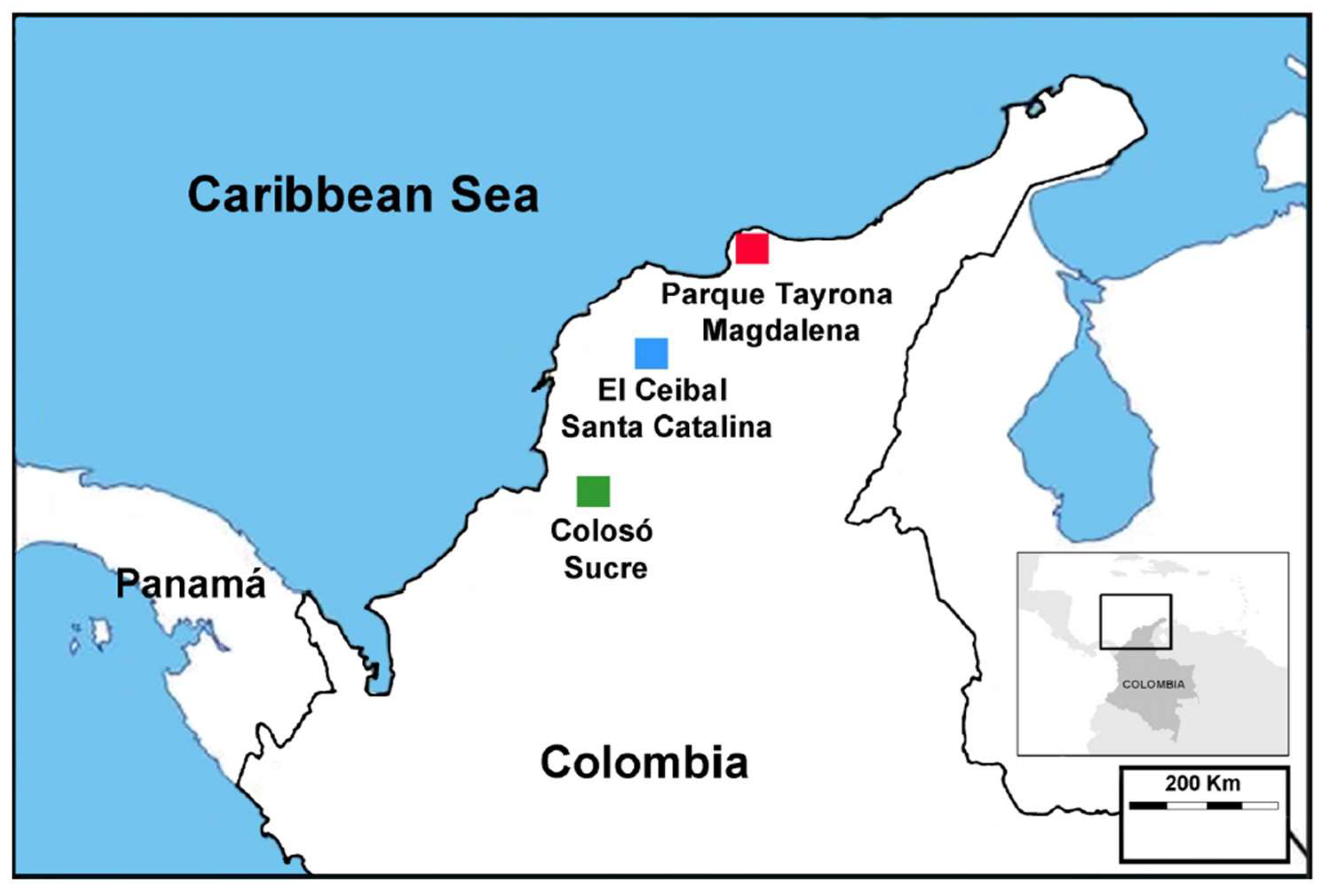
2.1. Study Sites and Termite Sampling
2.2. Determination of Food Niche
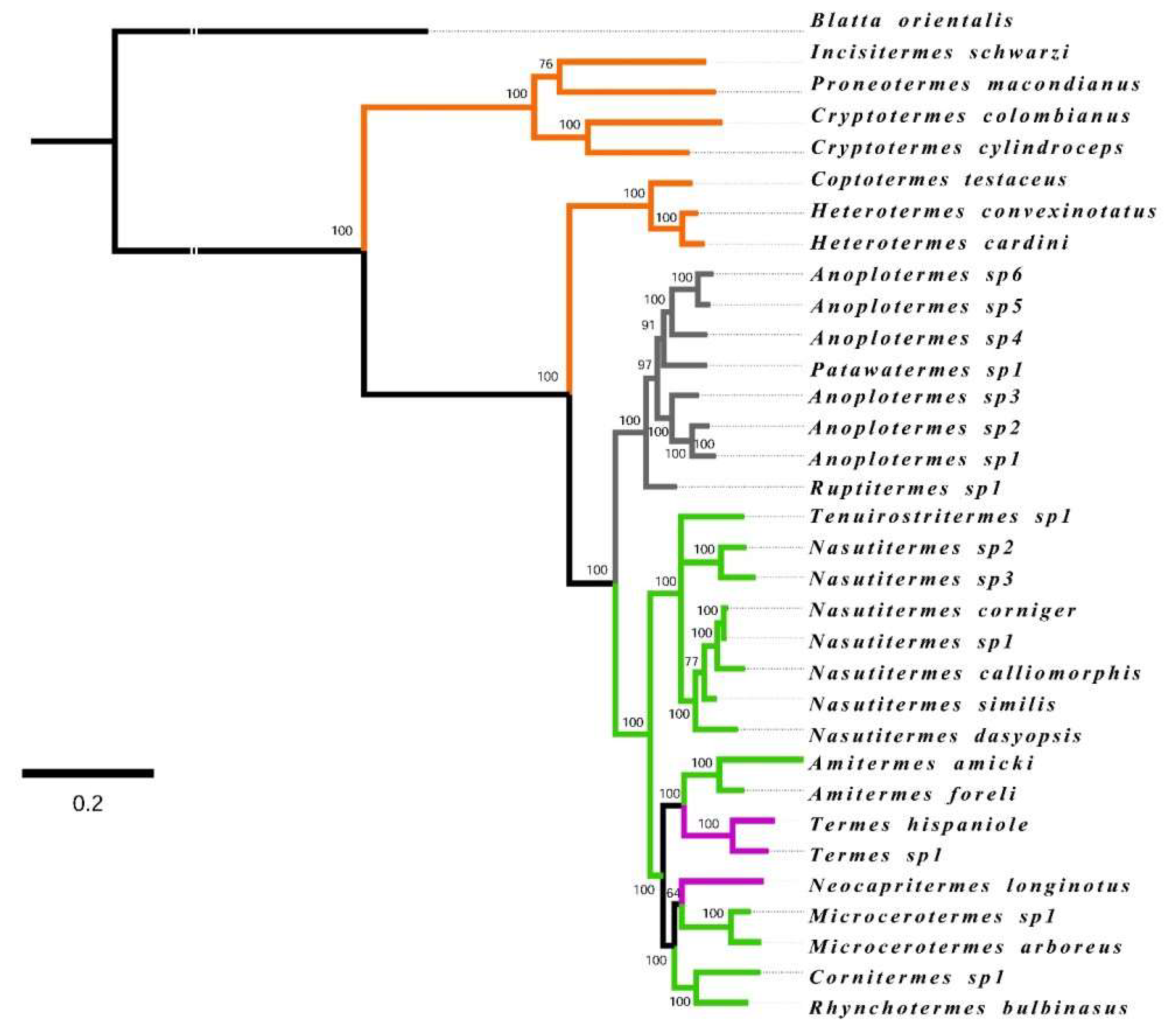
2.3. Phylogenetic Community Analyses
2.4. Mapping Food Niche Traits on Phylogeny
3. Results
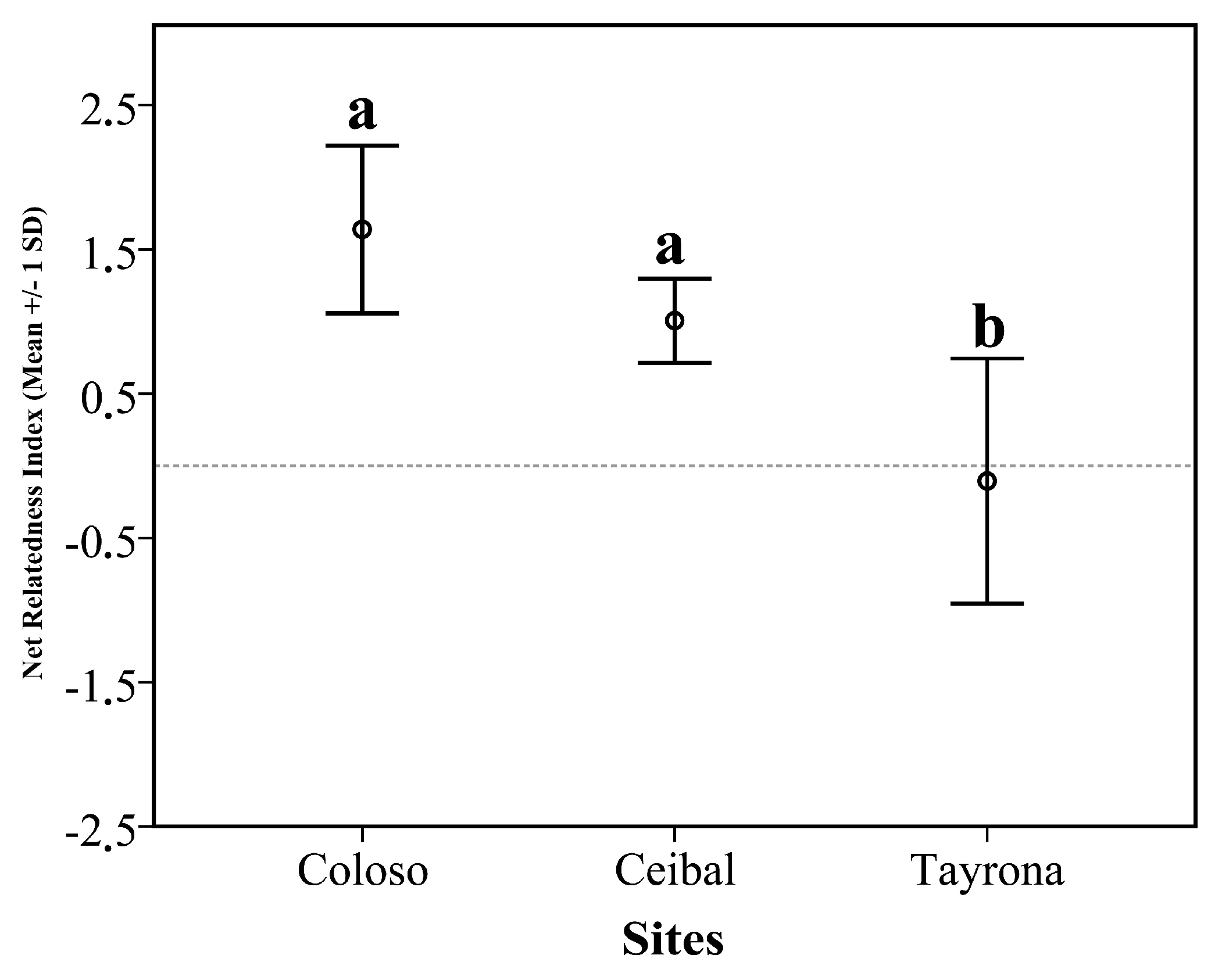
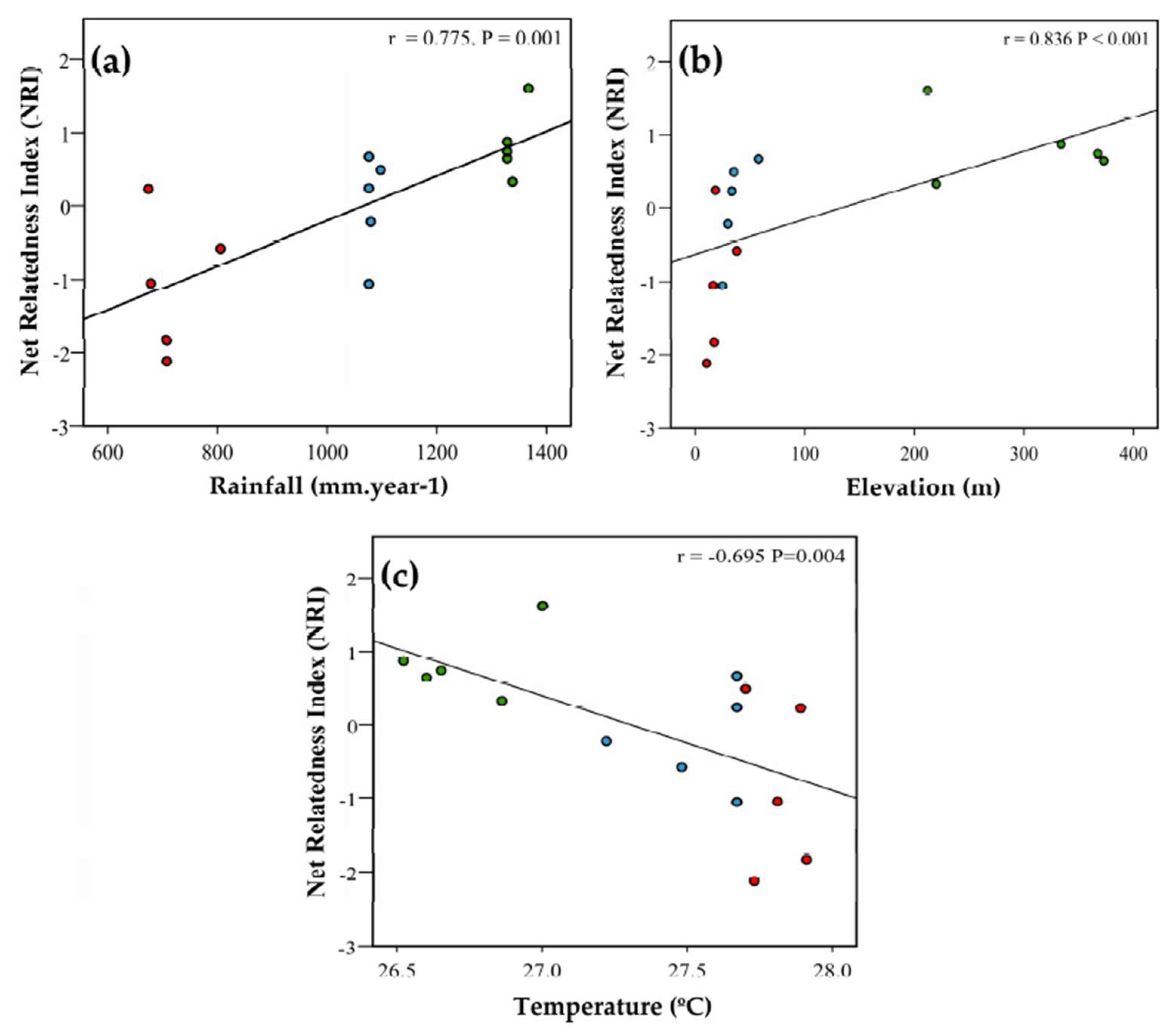
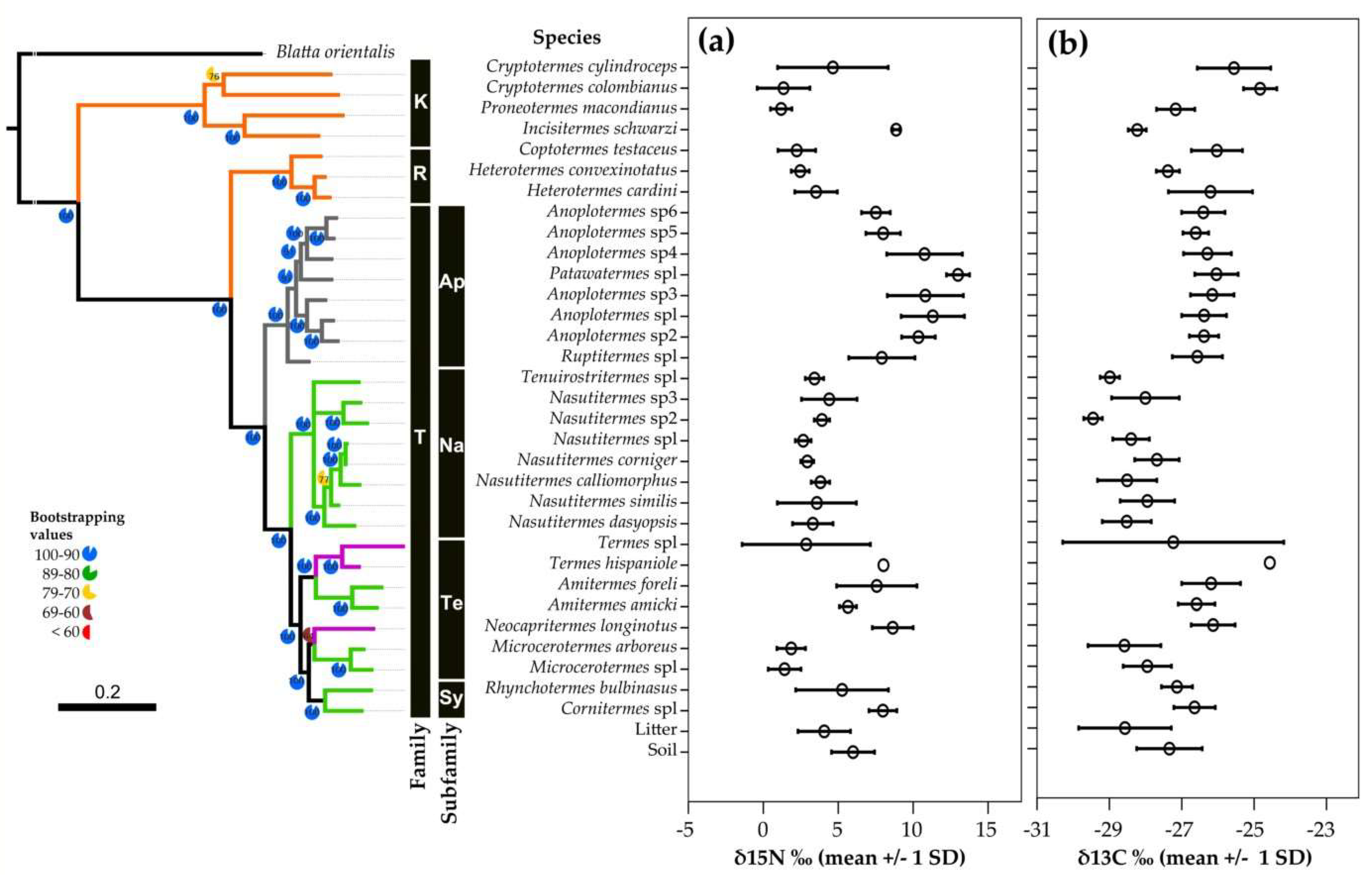
3.1. Phylogenetic Community Structure
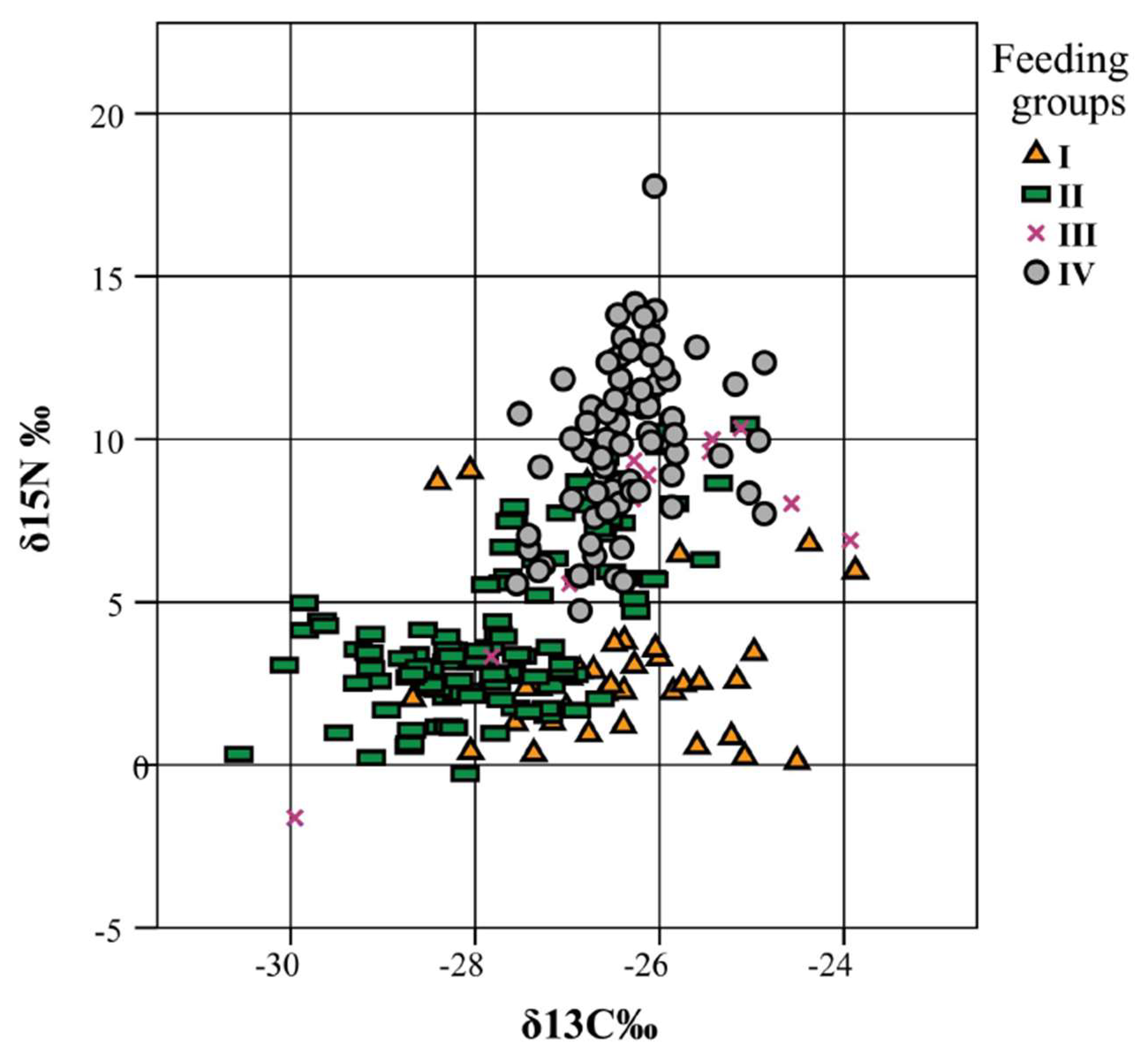
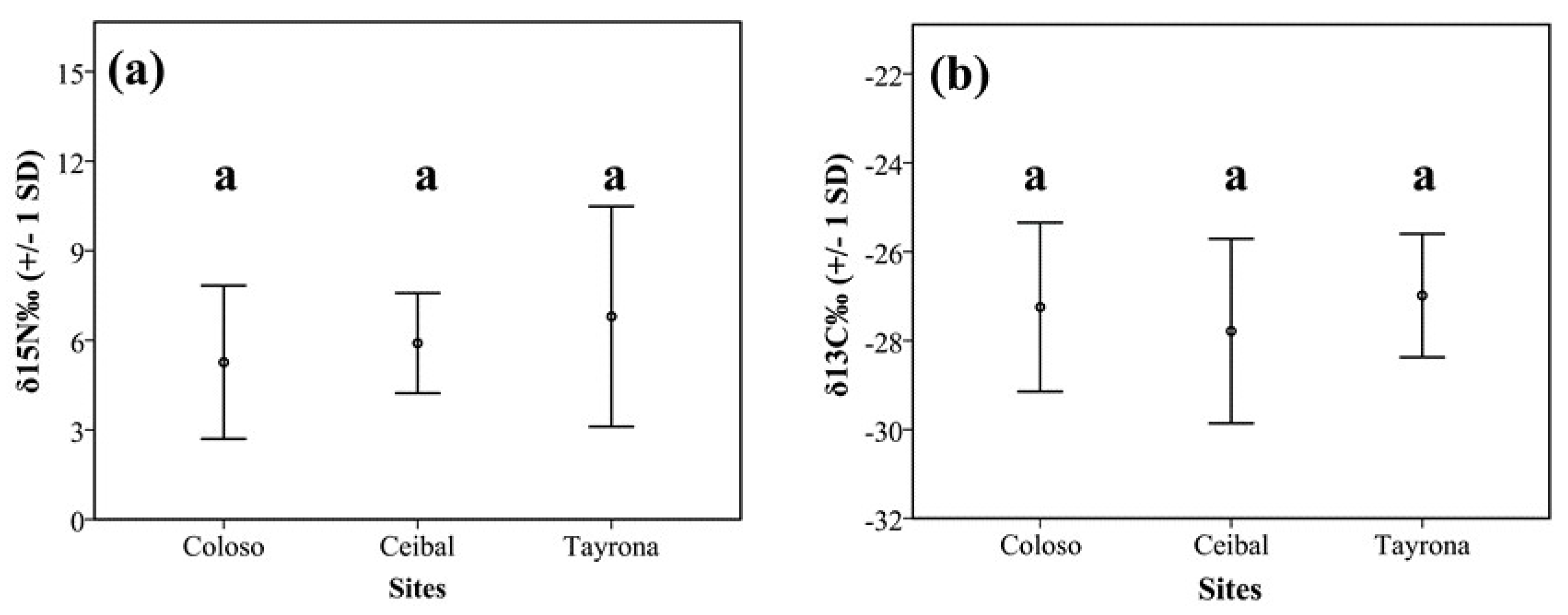
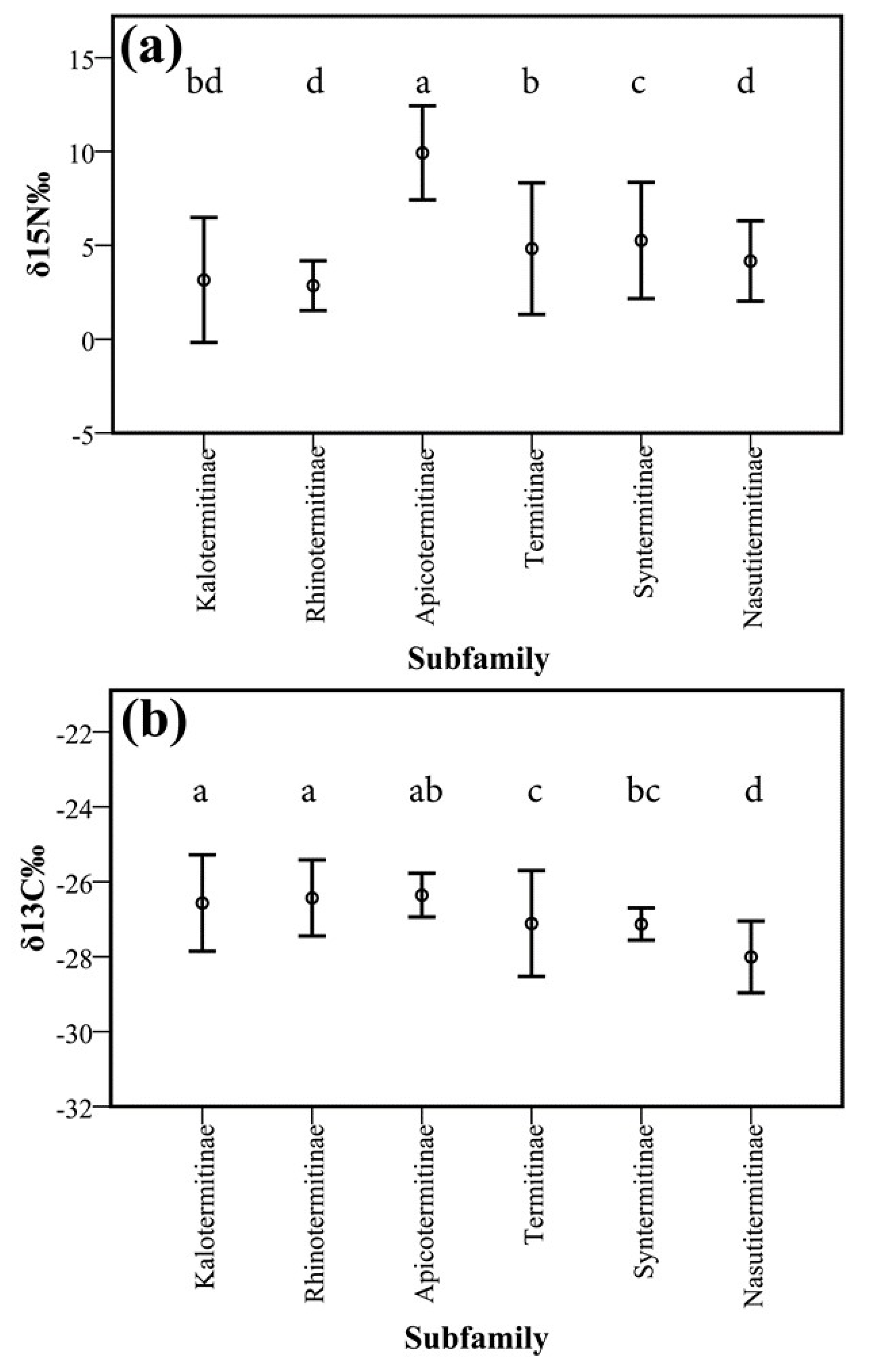
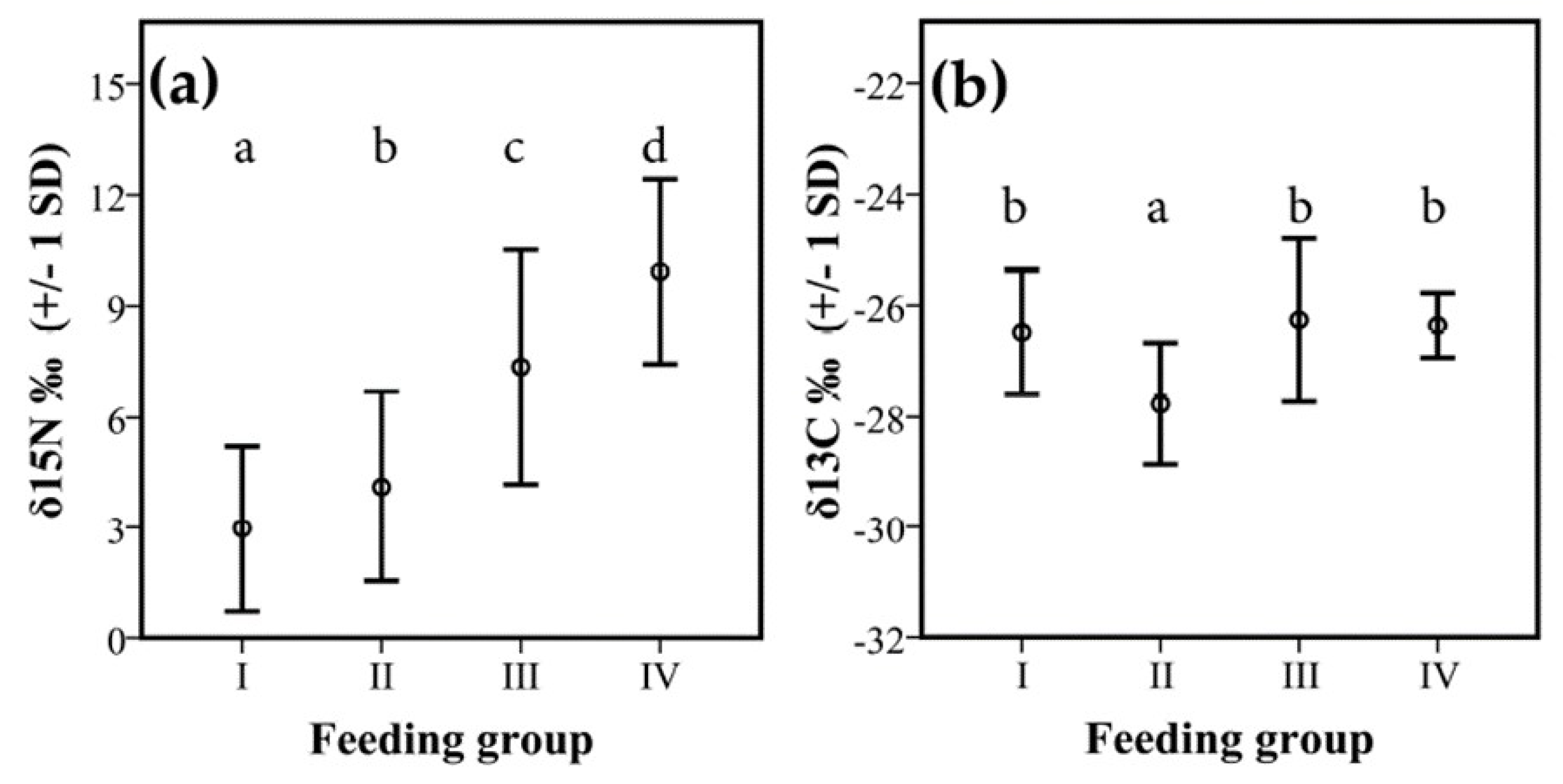
3.2. Isotopes Stable Analyses
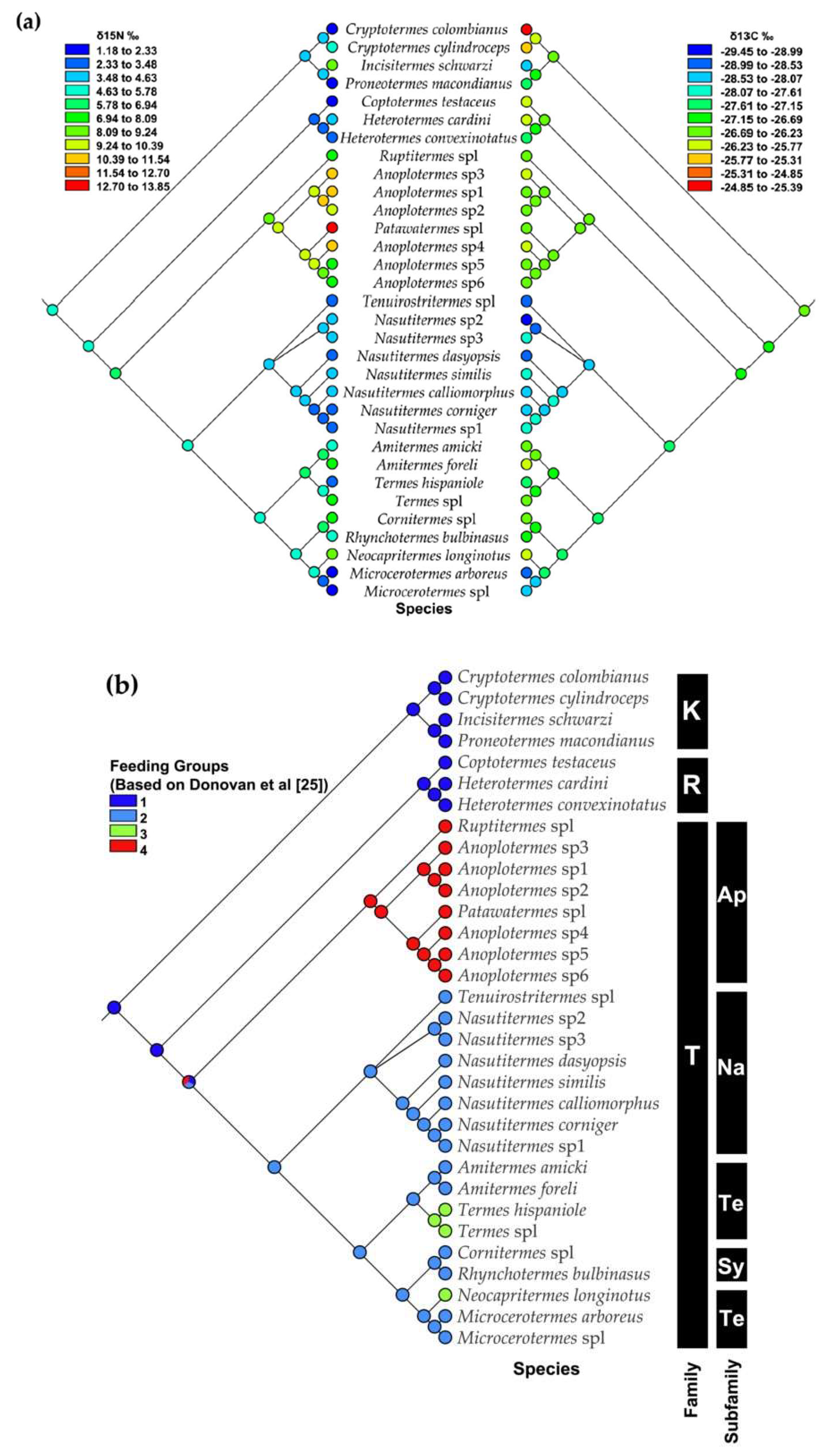
3.3. Mapping Food Niche Traits on Phylogeny
4. Discussion
4.1. Mechanisms Structuring Termite Assemblages
4.2. Food Niche: Isotopes and Termite Feeding Groups
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Species | GenBank | ||
|---|---|---|---|
| Accession COII | Accession 12S | Accession 16S | |
| Blatta orientalis | DQ874267.1 | DQ87403.1 | U17774.1 |
| Amitermes amicki | MH090825 | MH090861 | MH090885 |
| Amitermes foreli | MH090826 | MH090860 | MH090886 |
| Anoplotermes sp1 | MH090827 | MH090876 | MH090887 |
| Anoplotermes sp2 | MH090828 | MH090881 | MH090888 |
| Anoplotermes sp3 | MH090829 | MH090878 | MH090889 |
| Anoplotermes sp4 | MH090831 | MH090880 | MH090891 |
| Anoplotermes sp5 | MH090832 | MH090877 | MH090892 |
| Anoplotermes sp6 | MH090833 | MH090879 | MH090893 |
| Coptotermes testaceus | MH090834 | MH090857 | MH090894 |
| Cornitermes sp1 | MH090835 | MH090866 | MH090895 |
| Cryptotermes colombianus | KU510330 | KX267100 | KX267099 |
| Cryptotermes cylindroceps | MH090836 | MH090856 | MH090896 |
| Heterotermes cardini | MH090837 | MH090859 | MH090897 |
| Heterotermes convexinotatus | MH090838 | MH090858 | MH090898 |
| Incisitermes schwarzi | MH090839 | MH090855 | MH090899 |
| Microcerotermes arboreus | MH090840 | MH090872 | MH090900 |
| Microcerotermes sp1 | MH090841 | MH090871 | MH090901 |
| Nasutitermes corniger | MH090846 | MH090882 | MH090906 |
| Nasutitermes dasyopsis | MH090843 | MH090869 | MH090903 |
| Nasutitermes similis | MH090844 | MH090873 | MH090904 |
| Nasutitermes callimorphus | MH090845 | MH090870 | MH090905 |
| Nasutitermes sp1 | MH090842 | MH090868 | MH090902 |
| Nasutitermes sp2 | MH090848 | MH090862 | MH090908 |
| Nasutitermes sp3 | MH090849 | MH090863 | MH090909 |
| Neocapritermes longinotus | MH090847 | MH090867 | MH090907 |
| Patawatermes sp1 | MH090830 | MH090874 | MH090890 |
| Proneotermes macondianus | KX267098 | KX267095 | KX267092 |
| Rhynchotermes bulbinasus | MH090850 | MH090865 | MH090910 |
| Ruptitermes sp1 | MH090851 | MH090875 | MH090911 |
| Tenuirostritermes sp1 | MH090852 | MH090864 | MH090912 |
| Termes hispaniolae | MH090853 | MH090883 | MH090913 |
| Termes sp1 | MH090854 | MH090884 | MH090914 |
| Species | F | SF | FG | CO1 | CO2 | CO3 | CO4 | CO5 | CE1 | CE2 | CE3 | CE4 | CE5 | TA1 | TA2 | TA3 | TA4 | TA5 | TOT |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cryptotermes colombianus | K | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | |
| Cryptotermes cylindroceps | K | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 2 | 0 | 3 | 7 | |
| Incisitermes schwarzi | K | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 2 | |
| Proneotermes macondianus | K | I | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 9 | |
| Coptotermes testaceus | R | I | 1 | 0 | 0 | 5 | 0 | 4 | 0 | 0 | 6 | 0 | 0 | 0 | 0 | 0 | 0 | 16 | |
| Heterotermes cardini | R | I | 5 | 6 | 6 | 1 | 7 | 16 | 12 | 5 | 0 | 3 | 20 | 2 | 8 | 4 | 11 | 106 | |
| Heterotermes convexinotatus | R | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 8 | 0 | 0 | 0 | 0 | 0 | 0 | 8 | |
| Anoplotermes sp1 | T | Ap | IV | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 9 | 2 | 10 | 0 | 0 | 0 | 0 | 0 | 23 |
| Anoplotermes sp2 | T | Ap | IV | 2 | 5 | 7 | 6 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 21 |
| Anoplotermes sp3 | T | Ap | IV | 1 | 0 | 0 | 7 | 19 | 0 | 0 | 3 | 0 | 12 | 1 | 1 | 1 | 0 | 0 | 45 |
| Anoplotermes sp4 | T | Ap | IV | 0 | 3 | 2 | 2 | 0 | 0 | 0 | 1 | 0 | 4 | 1 | 0 | 3 | 1 | 1 | 18 |
| Anoplotermes sp5 | T | Ap | IV | 0 | 0 | 0 | 0 | 3 | 1 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 6 |
| Anoplotermes sp6 | T | Ap | IV | 2 | 15 | 1 | 8 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 29 |
| Patawatermes sp1 | T | Ap | IV | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 4 | 0 | 0 | 1 | 0 | 0 | 6 |
| Ruptitermes sp1 | T | Ap | IV | 0 | 0 | 1 | 0 | 2 | 1 | 1 | 0 | 0 | 0 | 6 | 5 | 8 | 1 | 4 | 29 |
| Nasutitermes corniger | T | Na | II | 16 | 13 | 0 | 3 | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 36 |
| Nasutitermes dasyopsis | T | Na | II | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 2 | 1 | 5 | 21 | 1 | 5 | 0 | 9 | 47 |
| Nasutitermes similis | T | Na | II | 6 | 0 | 0 | 10 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 17 |
| Nasutitermes callimorphus | T | Na | II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 18 | 0 | 11 | 0 | 0 | 0 | 0 | 0 | 29 |
| Nasutitermes sp1 | T | Na | II | 0 | 0 | 28 | 18 | 35 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 81 |
| Nasutitermes sp2 | T | Na | II | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Nasutitermes sp3 | T | Na | II | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 4 | 0 | 0 | 2 | 0 | 8 |
| Tenuisrostritermes sp1 | T | Na | II | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Amitermes amicki | T | Te | II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 2 | 1 | 0 | 5 |
| Amitermes foreli | T | Te | II | 0 | 0 | 1 | 3 | 0 | 6 | 5 | 0 | 8 | 24 | 7 | 0 | 3 | 1 | 1 | 59 |
| Microcerotermes arboreus | T | Te | II | 0 | 0 | 0 | 0 | 0 | 15 | 0 | 19 | 29 | 46 | 0 | 0 | 0 | 0 | 0 | 109 |
| Microcerotermes sp1 | T | Te | II | 8 | 12 | 1 | 16 | 22 | 6 | 37 | 6 | 47 | 21 | 35 | 24 | 17 | 11 | 22 | 285 |
| Neocapritermes longinotus | T | Te | III | 3 | 1 | 2 | 6 | 1 | 3 | 5 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 24 |
| Termes hispaniolae | T | Te | III | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 |
| Termes sp1 | T | Te | III | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 2 |
| Cornitermes sp1 | T | Sy | II | 7 | 21 | 16 | 6 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 53 |
| Rhynchotermes bulbinasus | T | Sy | II | 0 | 0 | 0 | 0 | 0 | 0 | 11 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 14 |
| Number of species | 32 | 11 | 9 | 10 | 17 | 14 | 9 | 9 | 9 | 7 | 13 | 11 | 6 | 12 | 8 | 7 | |||
| Number of occurrences | 54 | 77 | 65 | 98 | 104 | 53 | 74 | 70 | 101 | 146 | 99 | 34 | 55 | 22 | 51 | 1103 |
| Plot | Taxa | NRI |
|---|---|---|
| Coloso_1 | 11 | 1.456 |
| Coloso_2 | 9 | 1.411 |
| Coloso_3 | 10 | 1.594 |
| Coloso_4 | 17 | 1.282 |
| Coloso_5 | 14 | 2.452 |
| Ceibal_1 | 9 | 1.18 |
| Ceibal_2 | 9 | 1.313 |
| Ceibal_3 | 9 | 0.009 |
| Ceibal_4 | 7 | 0.886 |
| Ceibal_5 | 13 | 0.738 |
| Tayrona_1 | 11 | −0.821 |
| Tayrona_2 | 6 | 0.798 |
| Tayrona_3 | 12 | 0.412 |
| Tayrona_4 | 7 | −0.576 |
| Tayrona_5 | 7 | −0.340 |
| NRI | Sum of Squares | df | Mean Square | F | p-Value |
|---|---|---|---|---|---|
| Between sites | 7.80 | 2 | 3.900 | 15.76 | <0.001 |
| Within sites | 2.97 | 12 | 0.248 | ||
| Total | 10.77 | 14 |
| (I) Site | (J) Site | Mean Difference (I–J) | Std. Error | p-Value |
|---|---|---|---|---|
| Colosó | Ceibal | 0.63 | 0.31 | 0.152 |
| Tayrona | 1.74 | 0.31 | <0.001 | |
| Ceibal | Tayrona | 1.11 | 0.31 | 0.011 |
| Plot | Taxa | NRI |
|---|---|---|
| Coloso_1 | 11 | 0.408 |
| Coloso_2 | 9 | 0.549 |
| Coloso_3 | 10 | 0.71 |
| Coloso_4 | 17 | −0.856 |
| Coloso_5 | 14 | 1.446 |
| Ceibal_1 | 9 | 1.023 |
| Ceibal_2 | 9 | 1.382 |
| Ceibal_3 | 9 | −0.219 |
| Ceibal_4 | 7 | 0.834 |
| Ceibal_5 | 13 | 0.659 |
| Tayrona_1 | 11 | −0.818 |
| Tayrona_2 | 6 | 0.828 |
| Tayrona_3 | 12 | 0.449 |
| Tayrona_4 | 7 | −0.522 |
| Tayrona_5 | 7 | −0.336 |
| NRI | Sum of Squares | df | Mean Square | F | p-Value |
|---|---|---|---|---|---|
| Between sites | 1.714 | 2 | 0.857 | 1.68 | 0.227 |
| Within sites | 6.113 | 12 | 0.509 | ||
| Total | 7.826 | 14 |
| Rainfall | Elevation | Temperature | |
|---|---|---|---|
| NRI | 0.354 (p = 0.195) | 0.147 (p = 0.600) | −0.144 (p = 0.609) |
| Variable | Estimate | Standard Error | df | t-Value | p-Value |
|---|---|---|---|---|---|
| Intercept | 0.369 | 0.192 | 9 | 1.92 | 0.087 |
| Rainfall | 0.657 | 0.393 | 9 | 1.67 | 0.129 |
| Temperature | 0.490 | 0.593 | 9 | 0.82 | 0.430 |
| Elevation | 0.044 | 0.478 | 9 | 0.09 | 0.928 |
| Species | n | Delta Nitro | Delta Carbon | ||
|---|---|---|---|---|---|
| Mean | +/− SD | Mean | +/− SD | ||
| Amitermes amicki | 5 | 5.65 | 0.57 | −26.59 | 0.51 |
| Amitermes foreli | 10 | 7.57 | 2.68 | −26.19 | 0.81 |
| Anoplotermes sp1 | 11 | 11.31 | 2.11 | −26.38 | 0.62 |
| Anoplotermes sp2 | 10 | 10.35 | 1.12 | −26.38 | 0.41 |
| Anoplotermes sp3 | 10 | 10.81 | 2.54 | −26.16 | 0.60 |
| Anoplotermes sp4 | 11 | 10.76 | 2.53 | −26.29 | 0.66 |
| Anoplotermes sp5 | 6 | 8.00 | 1.15 | −26.61 | 0.36 |
| Anoplotermes sp6 | 10 | 7.51 | 0.96 | −26.40 | 0.60 |
| Coptotermes testaceus | 8 | 2.23 | 1.26 | −26.03 | 0.71 |
| Cornitermes sp1 | 10 | 7.98 | 0.93 | −26.65 | 0.57 |
| Cryptotermes colombianus | 2 | 1.35 | 1.76 | −24.83 | 0.46 |
| Cryptotermes cylindroceps | 4 | 4.64 | 3.69 | −25.56 | 1.02 |
| Heterotermes cardini | 11 | 3.52 | 1.42 | −26.21 | 1.16 |
| Heterotermes convexinotatus | 6 | 2.47 | 0.60 | −27.38 | 0.32 |
| Incisitermes schwarzi | 2 | 8.86 | 0.24 | −28.23 | 0.25 |
| Microcerotermes arboreus | 10 | 1.86 | 0.96 | −28.58 | 1.01 |
| Microcerotermes sp1 | 11 | 1.42 | 1.09 | −27.95 | 0.67 |
| Nasutitermes calliomorphus | 8 | 3.81 | 0.62 | −28.51 | 0.82 |
| Nasutitermes corniger | 9 | 2.93 | 0.43 | −27.69 | 0.62 |
| Nasutitermes dasyopsis | 7 | 3.30 | 1.35 | −28.52 | 0.68 |
| Nasutitermes similis | 7 | 3.57 | 2.64 | −27.95 | 0.75 |
| Nasutitermes sp1 | 9 | 2.66 | 0.53 | −28.40 | 0.51 |
| Nasutitermes sp2 | 2 | 3.91 | 0.52 | −29.45 | 0.26 |
| Nasutitermes sp3 | 8 | 4.40 | 1.86 | −28.00 | 0.93 |
| Neocapritermes longinotus | 10 | 8.64 | 1.36 | −26.13 | 0.61 |
| Patawatermes sp1 | 6 | 12.99 | 0.77 | −26.04 | 0.60 |
| Proneotermes macondianus | 7 | 1.19 | 0.72 | −27.17 | 0.53 |
| Rhinchotermes bulbinasus | 6 | 5.26 | 3.10 | −27.13 | 0.43 |
| Ruptitermes sp1 | 10 | 7.91 | 2.21 | −26.57 | 0.69 |
| Tenuirostritermes sp1 | 3 | 3.42 | 0.62 | −28.99 | 0.27 |
| Termes sp1 | 3 | 8.02 | - | −24.57 | - |
| Termes hispaniole | 1 | 2.87 | 4.28 | −27.23 | 3.06 |
| Litter | 75 | 4.0653 | 1.7535 | −28.56 | 1.2804 |
| Soil | 15 | 5.98 | 1.4387 | −27.34 | 0.9068 |
| Source | df | F | p-Value | |
|---|---|---|---|---|
| Intercept | Hypothesis | 1 | 376.28 | <0.001 |
| Error | 27 | |||
| Sites | Hypothesis | 2 | 4.78 | 0.030 |
| Error | 12 | |||
| Feeding | Hypothesis | 1 | 23.80 | <0.001 |
| Error | 212 | |||
| Subfamily | Hypothesis | 3 | 12.22 | <0.001 |
| Error | 212 | |||
| Plots | Hypothesis | 12 | 3.32 | <0.001 |
| Error | 212 | |||
| (I) Sites | (J) Sites | Mean Difference (I–J) | Std. Error | p-Value |
|---|---|---|---|---|
| CE | CO | −0.99 | 0.34 | 0.013 |
| PT | −1.41 | 0.43 | 0.004 | |
| CO | PT | −0.42 | 0.42 | 0.580 |
| Feeding Groups | Mean Difference (I–J) | Std. Error | p-Value | |
|---|---|---|---|---|
| I | II | −1.15 | 0.44 | 0.047 |
| III | −4.39 | 0.73 | <0.001 | |
| IV | −6.96 | 0.46 | <0.001 | |
| II | III | −3.24 | 0.67 | <0.001 |
| IV | −5.81 | 0.36 | <0.001 | |
| III | IV | −2.57 | 0.69 | 0.001 |
| (I) Subfamily | (J) Subfamily | Mean Difference (I–J) | Std. Error | p-Value |
|---|---|---|---|---|
| Kalotermitinae | Rhinotermitinae | 0.30 | 0.75 | 0.999 |
| Apicotermitinae | −6.77 | 0.65 | <0.001 | |
| Termitinae | −1.67 | 0.68 | 0.143 | |
| Syntermitinae | −3.80 | 0.83 | <0.001 | |
| Nasutitermitinae | −0.28 | 0.68 | 0.998 | |
| Rhinotermitinae | Apicotermitinae | −7.07 | 0.53 | <0.001 |
| Termitinae | −1.97 | 0.57 | 0.008 | |
| Syntermitinae | −4.10 | 0.74 | <0.001 | |
| Nasutitermitinae | −0.58 | 0.56 | 0.903 | |
| Apicotermitinae | Termitinae | 5.10 | 0.42 | <0.001 |
| Syntermitinae | 2.97 | 0.64 | <0.001 | |
| Nasutitermitinae | 6.49 | 0.42 | <0.001 | |
| Termitinae | Syntermitinae | −2.14 | 0.66 | 0.018 |
| Nasutitermitinae | 1.38 | 0.46 | 0.031 | |
| Syntermitinae | Nasutitermitinae | 3.52 | 0.66 | <0.001 |
| Source | df | F | p-Value | |
|---|---|---|---|---|
| Intercept | Hypothesis | 1 | 57,708.37 | <0.001 |
| Error | 37 | |||
| Sites | Hypothesis | 2 | 5.32 | 0.022 |
| Error | 12 | |||
| Feeding | Hypothesis | 1 | 9.34 | 0.003 |
| Error | 212 | |||
| Subfamily | Hypothesis | 3 | 14.49 | <0.001 |
| Error | 212 | |||
| Plots | Hypothesis | 12 | 2.13 | 0.016 |
| Error | 212 | |||
| (I) Sites | (J) Sites | Mean Difference (I–J) | Std. Error | p-Value |
|---|---|---|---|---|
| CE | CO | −0.12 | 0.12 | 0.560 |
| PT | −0.91 | 0.15 | <0.001 | |
| CO | PT | −0.79 | 0.15 | <0.001 |
| Feeding Groups | Mean Difference (I–J) | Std. Error | p-Value | |
|---|---|---|---|---|
| I | II | 1.28 | 0.15 | <0.001 |
| III | −0.23 | 0.25 | 0.809 | |
| IV | −0.13 | 0.16 | 0.863 | |
| II | III | −1.51 | 0.23 | <0.001 |
| IV | −1.41 | 0.12 | <0.001 | |
| III | IV | 0.10 | 0.24 | 0.974 |
| (I) Subfamily | (J) Subfamily | Mean Difference (I–J) | Std. Error | p-Value |
|---|---|---|---|---|
| Kalotermitinae | Rhinotermitinae | −0.14 | 0.27 | 0.996 |
| Apicotermitinae | −0.21 | 0.23 | 0.944 | |
| Termitinae | 0.55 | 0.24 | 0.211 | |
| Syntermitinae | 0.26 | 0.29 | 0.950 | |
| Nasutitermitinae | 1.70 | 0.24 | <0.001 | |
| Rhinotermitinae | Apicotermitinae | −0.07 | 0.19 | 0.999 |
| Termitinae | 0.68 | 0.20 | 0.010 | |
| Syntermitinae | 0.40 | 0.26 | 0.656 | |
| Nasutitermitinae | 1.83 | 0.20 | <0.001 | |
| Apicotermitinae | Termitinae | 0.76 | 0.15 | <0.001 |
| Syntermitinae | 0.47 | 0.23 | 0.298 | |
| Nasutitermitinae | 1.91 | 0.15 | <0.001 | |
| Termitinae | Syntermitinae | −0.29 | 0.23 | 0.827 |
| Nasutitermitinae | 1.15 | 0.16 | <0.001 | |
| Syntermitinae | Nasutitermitinae | 1.44 | 0.23 | <0.001 |
Appendix B
References
- Webb, C.; Ackerly, D.; McPeek, M.; Donoghue, J. Phylogenies and community ecology. Annu. Rev. Ecol. Syst. 2002, 33, 475–505. [Google Scholar] [CrossRef]
- Kraft, N.; Cornwell, W.; Webb, C.; Ackerly, D. Trait evolution, community assembly, and the phylogenetic structure of ecological communities. Am. Nat. 2007, 170, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Emerson, B.; Gillespie, R. Phylogenetic analysis of community assembly and structure over space and time. Trends Ecol. Evol. 2008, 23, 619–630. [Google Scholar] [CrossRef]
- Cavender-Bares, J.; Kozak, K.; Fine, P.; Kembel, S. The merging of community ecology and phylogenetic biology. Ecol. Lett. 2009, 12, 693–715. [Google Scholar] [CrossRef]
- Webb, C.; Ackerly, D.; Kembel, S. Phylocom: Software for the analysis of phylogenetic community structure and trait evolution. Bioinformatics 2008, 24, 2098–2100. [Google Scholar] [CrossRef] [PubMed]
- Weiher, E.; Freund, D.; Bunton, T.; Stefanski, A.; Lee, T.; Bentivenga, S. Advances, challenges and a developing synthesis of ecological community assembly theory. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2011, 366, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Hubbell, S. The Unified Neutral Theory of Biodiversity and Biogeography; Princeton University Press: Princeton, NJ, USA, 2001; p. 392. ISBN 9780691021287. [Google Scholar]
- Diamond, J. Assembly of Species Communities. In Ecology and Evolution of Communities; Cody, M.L., Diamond, J., Eds.; Belknap Press of Harvard University Press: Cambridge, MA, USA, 1975; pp. 342–444. ISBN 9780674224445. [Google Scholar]
- Tilman, D. Resource Competition and Community Structure; Princeton University Press: Princeton, NJ, USA, 1982; p. 296. ISBN 9780691083025. [Google Scholar]
- Hebert, P.; Cywinska, A.; Ball, S.; Dewaard, J. Biological identification through DNA barcodes. Proc. R. Soc. Lond. B 2003, 270, 313–321. [Google Scholar] [CrossRef]
- Delgado-Salinas, A.; Dexter, K.; Linares-Palomino, R.; Oliveira-Filho, A.; Prado, D.; Pullan, M.; Quintana, C.; Riina, R.; Rodríguez, M.G.M.; Weintritt, J.; et al. Plant diversity patterns in neotropical dry forests and their conservation implications. Science 2016, 353, 1383–1387. [Google Scholar] [CrossRef]
- Hausberger, J.; Korb, J. The impact of anthropogenic disturbance on assembly patterns of termite communities. Biotropica 2016, 48, 356–364. [Google Scholar] [CrossRef]
- Schyra, J.; Korb, J. Termite communities along a disturbance gradient in a West African savanna. Insects 2019, 10, 17. [Google Scholar] [CrossRef]
- Miles, L.; Newton, A.; DeFries, R.; Ravilious, C.; May, I.; Blyth, S.; Kapos, V.; Gordon, J. A global overview of the conservation status of tropical dry forests. J. Biogeogr. 2006, 33, 491–505. [Google Scholar] [CrossRef]
- Portillo-Quintero, C.; Sánchez, G. Extent and conservation of tropical dry forests in the Americas. Biol. Conserv. 2010, 143, 144–155. [Google Scholar] [CrossRef]
- Instituto de Investigación Alexander von Humboldt. El Bosque Seco Tropical en Colombia; Pizano, C., García, H., Eds.; Instituto de Investigación Alexander von Humboldt: Bogotá, Colombia, 2014; p. 353. ISBN 978-958-8343-97-6. [Google Scholar]
- Bignell, D.; Eggleton, P. Termites in ecosystems. In Termites: Evolution, Sociality, Symbioses, Ecology; Abe, T., Bignell, D., Higashi, M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000; pp. 363–387. ISBN 978-94-017-3223-9. [Google Scholar]
- Jones, D.; Eggleton, P. Global Biogeography of Termites: A Modern Synthesis; Bignell, D., Rosin, Y., Lo, N., Eds.; Springer: New York, NY, USA, 2010; pp. 477–498. ISBN 978-90-481-3977-4. [Google Scholar]
- Bodot, P. Destruction of the nests of termites, Bellicositermes natalensis Hay., by an ant, Dorylus (Typhlopone) dentifrons Wasmann. Compt. Rend. Hebd. Séances Acad. Sci. 1961, 253, 3053–3054. [Google Scholar]
- Petrakova, L.; Lıznarova, E.; Pekar, S.; Haddad, C.; Sentenska, L.; Symondson, W. Discovery of a monophagous true predator, a specialist termite-eating spider (Araneae: Ammoxenidae). Sci. Rep. 2015, 5, 14013. [Google Scholar] [CrossRef]
- Mirco, S.; Ketterl, J.; Di-Bernardo, M.; Kwet, A. Ants and termites are the diet of the microhylid frog Elachistocleis ovalis (Schneider, 1799) at an Araucaria forest in Rio Grande do Sul, Brazil. Herpetol. Bull. 2002, 79, 14–17. [Google Scholar]
- Abensperg-Traun, M.; Steven, D. Ant- and termite-eating in Australian mammals and lizards: A comparison. Aust. J. Ecol. 1997, 22, 9–17. [Google Scholar] [CrossRef]
- Mcgrew, W.; Marchant, L. Chimpanzees, tools and termites: Hand preference or handedness? Curr. Anthropol. 1992, 33, 114–119. [Google Scholar] [CrossRef]
- Wood, T.G.; Sands, W.A. The role of termites in ecosystems. In Production Ecology of Ants and Termites; Brian, M.V., Ed.; Cambridge University Press: Cambridge, UK, 1978; pp. 245–292. ISBN 0521107148. [Google Scholar]
- Collins, N.M. Termite populations and their role in litter removal in Malaysian rain forests. In Tropical Rain Forest: Ecology and Management; Sutton, S.L., Whitmore, T.C., Chadwick, A.C., Eds.; Blackwell Science: Oxford, UK, 1983; pp. 311–325. ISBN 0632011424. [Google Scholar]
- Casalla, R.; Korb, J. Termite diversity in Neotropical dry forests of Colombia and the potential role of rainfall in structuring termite diversity. Biotropica 2019, 51, 165–177. [Google Scholar] [CrossRef]
- Donovan, S.E.; Eggleton, P.; Bignell, D.E. Gut content analysis and a new feeding group classification of termites. Ecol. Entmol. 2001, 26, 356–366. [Google Scholar] [CrossRef]
- Eggleton, P.; Tayasu, I. Feeding groups, lifetypes and the global ecology of termites. Ecol. Res. 2001, 16, 941–960. [Google Scholar] [CrossRef]
- Dahlsjö, C.; Parr, C.; Malhi, Y.; Meir, P.; Eggleton, P. Describing termite assemblage structure in a Peruvian lowland tropical rain forest: A comparison of two alternative methods. Insectes Soc. 2014, 62, 141–150. [Google Scholar] [CrossRef]
- Tayasu, I.; Abe, T.; Eggleton, P.; Bignell, D. Nitrogen and carbon isotope ratios in termites: An indicator of trophic habit along the gradient from wood-feeding to soil-feeding. Ecol. Entomol. 1997, 22, 343–351. [Google Scholar] [CrossRef]
- Florencio, F.D.; Marins, A.; Rosa, S.C.; Cristaldo, P.F.; Albano Araújo, A.P.; Silva, I.R.; DeSouza, O. Diet segregation between cohabiting builder and inquiline termite species. PLoS ONE 2013, 8, e66535. [Google Scholar] [CrossRef]
- Bourguignon, T.; Šobotník, J.; Lepoint, G.; Martin, J.; Roisin, Y. Niche differentiation among neotropical soldierless soil-feeding termites revealed by stable isotope ratios. Soil Biol. Biochem. 2009, 41, 2038–2043. [Google Scholar] [CrossRef]
- Boutton, T.; Arshad, M.; Tieszen, L. Stable isotope analysis of termite food habits in East African grasslands. Oecologia 1983, 59, 1–6. [Google Scholar] [CrossRef]
- Tayasu, I.; Nakamura, T.; Oda, H.; Hyodo, F.; Takematsu, Y.; Abe, T. Termite ecology in a dry evergreen forest in Thailand in terms of stable (δ13C and δ15N) and radio (14C, 137Cs and 210Pb) isotopes. Ecol. Res. 2002, 17, 195–206. [Google Scholar] [CrossRef]
- Lepage, M.; Abbadie, L.; Marriotti, A. Food habits of sympatric termite species (Isoptera, Macrotermitinae) as determined by stable carbon isotope analysis in a Guinean savanna (Lamto, CBte d’voire). J. Trop. Ecol. 1993, 9, 303–311. [Google Scholar]
- Tayasu, I.; Hyodo, F.; Takematsu, Y.; Sugimoto, A.; Inoue, T.; Kirtibutr, N.; Abe, T. Stable isotope ratios and uric acid preservation in termites belonging to three feeding habits in Thailand. Isot. Environ. Health Stud. 2000, 36, 259–272. [Google Scholar] [CrossRef]
- Tayasu, I.; Hyodo, F.; Abe, T. Caste-specific N and C isotope ratios in fungus-growing termites with special reference to uric acid preservation and their nutritional interpretation. Ecol. Entomol. 2002, 27, 355–361. [Google Scholar] [CrossRef]
- Instituto Nacional de los Recursos Naturales Renovables y del Ambiente. Acuerdo 0028 del 6 de Julio 1983. Por el Cual se Declara Área de Reserva Forestal Protectora, la Serranía de Coraza y Montes de María (Serranía de San Jacinto), Ubicada en Jurisdicción de los Municipios de Toluviejo, Colosó y Chalán (Departamento de Sucre); Instituto Nacional de los Recursos Naturales Renovables y del Ambiente: Bogotá, Colombia, 1983; 6p. [Google Scholar]
- Corporación Autónoma Regional Del Canal del Dique. Acuerdo No 0004 27 Nov 2013. Declaratoria Parque Natural Regional Bosque Seco El Ceibal Mono Tití; Corporación Autónoma Regional Del Canal del Dique: Santa Catalina, Bolívar, Colombia, 2013; 60p. [Google Scholar]
- Instituto Colombiano para la Reforma Agraria. Resolución Nº 191 Declaratoria Parque Nacional Natural Tayrona; Instituto Colombiano para la Reforma Agraria: Bogota, Colombia, 1964; 2p. [Google Scholar]
- Carbonó, E. Terrestrial vegetation at Neguanje cove, Tayrona National Natural Park (Magdalena, Colombia). Caldasia 2010, 32, 235–256. [Google Scholar]
- Gentry, A. Diversity and floristic composition of Neotropical dry forests. In Seasonally Dry Tropical Forests; Bullock, S.H., Mooney, H., Medina, E., Eds.; Cambridge University Press: Cambridge, UK, 1995; pp. 146–194. ISBN 9780511753398. [Google Scholar]
- Jones, D.; Eggleton, P. Sampling termite assemblages in tropical forests: Testing a rapid biodiversity assessment protocol. J. Appl. Ecol. 2000, 37, 191–203. [Google Scholar] [CrossRef]
- Hausberger, B.; Korb, J. A phylogenetic community approach for studying termite communities in a West African savannah. Biol. Lett. 2015, 11, 20150625. [Google Scholar] [CrossRef]
- Tayasu, I.; Sugimoto, A.; Wada, E.; Abe, T. Xylophagous termites depending on atmospheric nitrogen. Naturwissenschaften 1994, 81, 229–231. [Google Scholar] [CrossRef]
- Tayasu, I.; Inoue, T.; Miller, L.; Sugimoto, A.; Takeichi, S.; Abe, T. Confirmation of soil-feeding termites (Isoptera; Termitidae; Termitinae) in Australia using stable isotope ratios. Funct. Ecol. 1998, 12, 536–542. [Google Scholar] [CrossRef]
- Pansu, M.; Gautheyrou, J. Hand Book of Soil Analysis; Springer-Verlag: Berlin, Germany, 2006; 993p. [Google Scholar]
- Osorio, N. Muestra de Suelos; Universidad Nacional de Colombia: Medellín, Colombia, 2012; pp. 1–4. [Google Scholar]
- Ronquist, F.; Huelsenbeck, J. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar] [CrossRef]
- Gotelli, N.; Entsminger, G. Swap algorithms in null model analysis. Ecology 2003, 84, 532–535. [Google Scholar] [CrossRef]
- Maddison, W. Squared-change parsimony reconstructions of ancestral states for continuous-valued characters on a phylogenetic tree. Syst. Zool. 1991, 40, 304–314. [Google Scholar] [CrossRef]
- Maddison, W.; Maddison, D.R. Mesquite: A Modular System for Evolutionary Analysis. Version 3.51. 2018. Available online: http://www.mesquiteproject.org (accessed on 15 November 2018).
- Allen, K.; Dupuy, J.; Gei, M.; Hulshof, C.; Medvigy, D.; Pizano, C.; Salgado-Negret, B.; Smith, B.M.; Trierweiler, A.; Van Bloem, S.J.; et al. Will seasonally dry tropical forests be sensitive or resistant to future changes in rainfall regimes? Environ. Res. Lett. 2017, 12, 23001. [Google Scholar] [CrossRef]
- Olascuaga-Vargas, D.; Mercado-Gómez, J.; Sanchez-Montaño, L. Analysis of successional vegetation in a tropical dry forest fragment in Toluviejo-Sucre (Colombia). Colomb. For. 2015, 19, 23–40. [Google Scholar] [CrossRef]
- Rodríguez, G. Inventario Florístico de un Bosque seco Tropical (Bs-T) en la Hacienda “El Ceibal”, Santa Catalina, Bolívar, con Énfasis en las Especies Asociadas a la Dieta del Tití Cabeciblanco (Saguinus oedipus). Bachelor’s Thesis, Universidad Nacional de Colombia, Bogotá, Colombia, 2001. [Google Scholar]
- Rodríguez, G.; Banda-R., K.; Reyes, S.; Estupiñán, A. Lista comentada de las plantas vasculares de bosques secos prioritarios para la conservación en los departamentos de Atlántico y Bolívar (Caribe colombiano). Biota Colomb. 2012, 13, 7–39. [Google Scholar]
- Korb, J.; Linsenmair, K.E. The causes of spatial patterning of mounds of a fungus-growing termite: Results from nearest neighbour analysis and ecological studies. Oecologia 2001, 127, 324–333. [Google Scholar] [CrossRef]
- Korb, J.; Linsenmair, K.E. Resource availabiliy and distribution patterns, indicators of competition between Macrotermes bellicosus and other macro-detritores in the Comoé National Park, Cote d’Ivoire. Afr. J. Ecol. 2001, 39, 257–265. [Google Scholar] [CrossRef]
- Leponce, M.; Roisin, Y.; Pasteels, J.M. Intraspecific interactions in a community of arboreal nesting termites (Isoptera: Termitidae). J. Insect Behav. 1996, 9, 799–817. [Google Scholar] [CrossRef]
- Bourguignon, T.; Leponce, M.; Roisin, Y. Beta-diversity of termite assemblages among primary French Guiana rain forests. Biotropica 2011, 43, 473–479. [Google Scholar] [CrossRef]
- Korb, J.; Foster, K.R. Ecological competition favours cooperation in termite societies. Ecol. Lett. 2010, 13, 754–760. [Google Scholar] [CrossRef]
- Valladares, C. Ensamblaje de Comunidades de Termitas (Blattodea: Isoptera) en dos Bosques Tropicales Amazónicos de Ecuador, Parque Nacional. Yasuní y Parque Nacional Podocarpus. Master’s Thesis, Universidad Técnica de Loja, Loja, Ecuador, 2016. [Google Scholar]
- Palin, O.; Eggleton, P.; Malhi, Y.; Girardin, C.; Rozas-Dávila, A.; Parr, C. Termite diversity along an Amazon–Andes elevation gradient, Perú. Biotropica 2011, 43, 100–107. [Google Scholar] [CrossRef]
- Davies, R.; Hernández, L.; Eggleton, P.; Didham, R.; Fagan, L.; Winchester, N. Environmental and spatial influences upon species composition of a termite assemblage across neotropical forest islands. J. Trop. Ecol. 2003, 19, 509–524. [Google Scholar] [CrossRef]
- Dosso, K.; Deligne, J.; Yeó, K.; Konate, S.; Linsenmair, K. Changes in the termite assemblage across a sequence of land-use systems in the rural area around lamto reserve in central cote d’ivoire. J. Insect Conserv. 2013, 17, 1047–1057. [Google Scholar] [CrossRef]
- Gathorne-Hardy, F.; Syaukani, F.; Eggleton, P. The effects of altitude and rainfall on the composition of the termites (Isoptera) of the Leuser ecosystem (Sumatra, Indonesia). J. Trop. Ecol. 2001, 17, 379–393. [Google Scholar] [CrossRef]
- Vasconcellos, A.; Bandeira, A.; Moura, F.; Araujo, V.; Gusmão, M.; Constantino, R. Termite assemblages in three habitats under different disturbance regimes in the semi-arida Caatinga of NE Brazil. J. Arid Environ. 2010, 74, 298–302. [Google Scholar] [CrossRef]
- Viana, A.; Souza, V.; Reis, Y.; Costa, A. Termite assemblages in dry tropical forests of Northeastern Brazil: Are termites bioindicators of environmental disturbances? Sociobiology 2014, 61, 324–331. [Google Scholar]
- Su, N.; Scheffrahn, R. Intra-and interspecific competition of the Formosan and the Eastern subterranean termite: Evidence from field observations (Isoptera: Rhinotermitidae). Sociobiology 1988, 14, 157–164. [Google Scholar]
- Leponce, M.; Roisin, Y.; Pasteels, J. Structure and dynamics of the arboreal termite community in New Guinean coconut plantations. Biotropica 1997, 29, 193–203. [Google Scholar] [CrossRef]
- Schyra, J.; Scheu, S.; Korb, J. Cryptic niche differentiation in West African savannah termites as indicated by stable isotopes: Niche differentiation in termites. Ecol. Entomol. 2018. [Google Scholar] [CrossRef]
- Goldstein, G.; Drake, D.; Alpha, C.; Melcher, P.; Heraux, J.; Azocar, A. Growth and photosynthetic responses of Scaevola sericea, a Hawaiian coastal shrub, to substrate salinity and salt spray. Int. J. Plant. Sci. 1996, 157, 171–179. [Google Scholar] [CrossRef]
- Diefendorf, A.; Mueller, K.; Wing, S.; Koch, P.; Freeman, K. Global patterns in leaf 13C discrimination and implications for studies of past and future climate. Proc. Natl. Acad. Sci. USA 2010, 107, 5738–5743. [Google Scholar] [CrossRef]
- Tayasu, I. Use of carbon and nitrogen isotope ratios in termite research. Ecol. Res. 1998, 13, 377–387. [Google Scholar] [CrossRef]

| Variable | Estimate | Standard Error | df | t-Value | p-Value |
|---|---|---|---|---|---|
| Intercept | 0.787 | 0.142 | 9 | 5.55 | <0.001 |
| Rainfall | 0.885 | 0.290 | 9 | 3.05 | 0.014 |
| Temperature | 0.395 | 0.437 | 9 | 0.90 | 0.391 |
| Elevation | 0.263 | 0.352 | 9 | 0.74 | 0.475 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casalla Daza, R.; Korb, J. Phylogenetic Community Structure and Niche Differentiation in Termites of the Tropical Dry Forests of Colombia. Insects 2019, 10, 103. https://doi.org/10.3390/insects10040103
Casalla Daza R, Korb J. Phylogenetic Community Structure and Niche Differentiation in Termites of the Tropical Dry Forests of Colombia. Insects. 2019; 10(4):103. https://doi.org/10.3390/insects10040103
Chicago/Turabian StyleCasalla Daza, Robin, and Judith Korb. 2019. "Phylogenetic Community Structure and Niche Differentiation in Termites of the Tropical Dry Forests of Colombia" Insects 10, no. 4: 103. https://doi.org/10.3390/insects10040103
APA StyleCasalla Daza, R., & Korb, J. (2019). Phylogenetic Community Structure and Niche Differentiation in Termites of the Tropical Dry Forests of Colombia. Insects, 10(4), 103. https://doi.org/10.3390/insects10040103






