Symbiotic Plant Biomass Decomposition in Fungus-Growing Termites
Abstract
1. Introduction
1.1. Plant Substrate Use as Main Nutrient Source
1.2. Termites Have Relied on Symbiotic Digestion of Lignocellulose for Millions of Years
2. The Tripartite Fungus-Growing Termite Symbiosis
2.1. The Symbiosis Between Fungus-Growing Termites and Termitomyces
2.2. The Symbiosis between Fungus-Growing Termites and Gut Bacteria
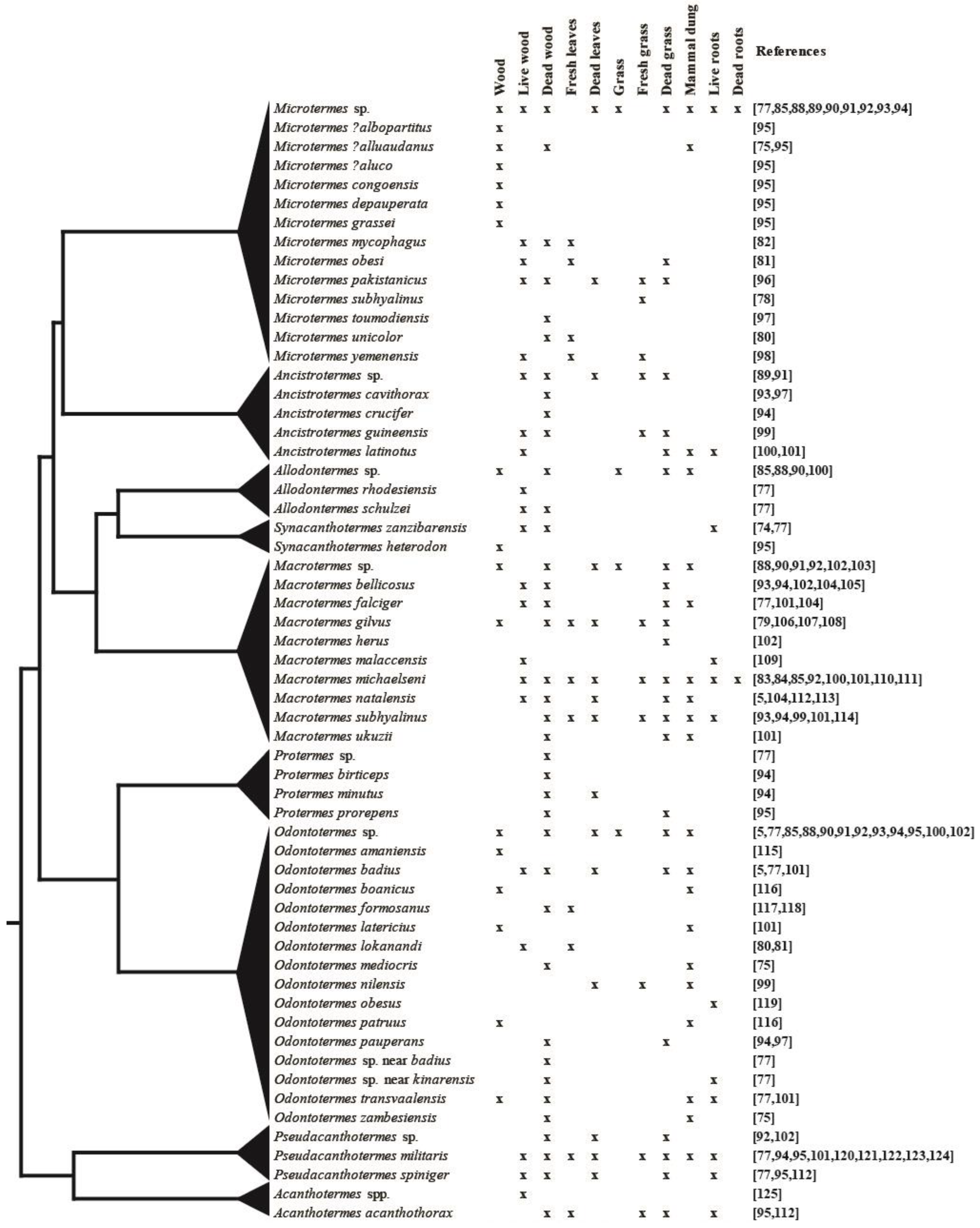
2.3. Substrate Use by Different Fungus-Growing Termite Species
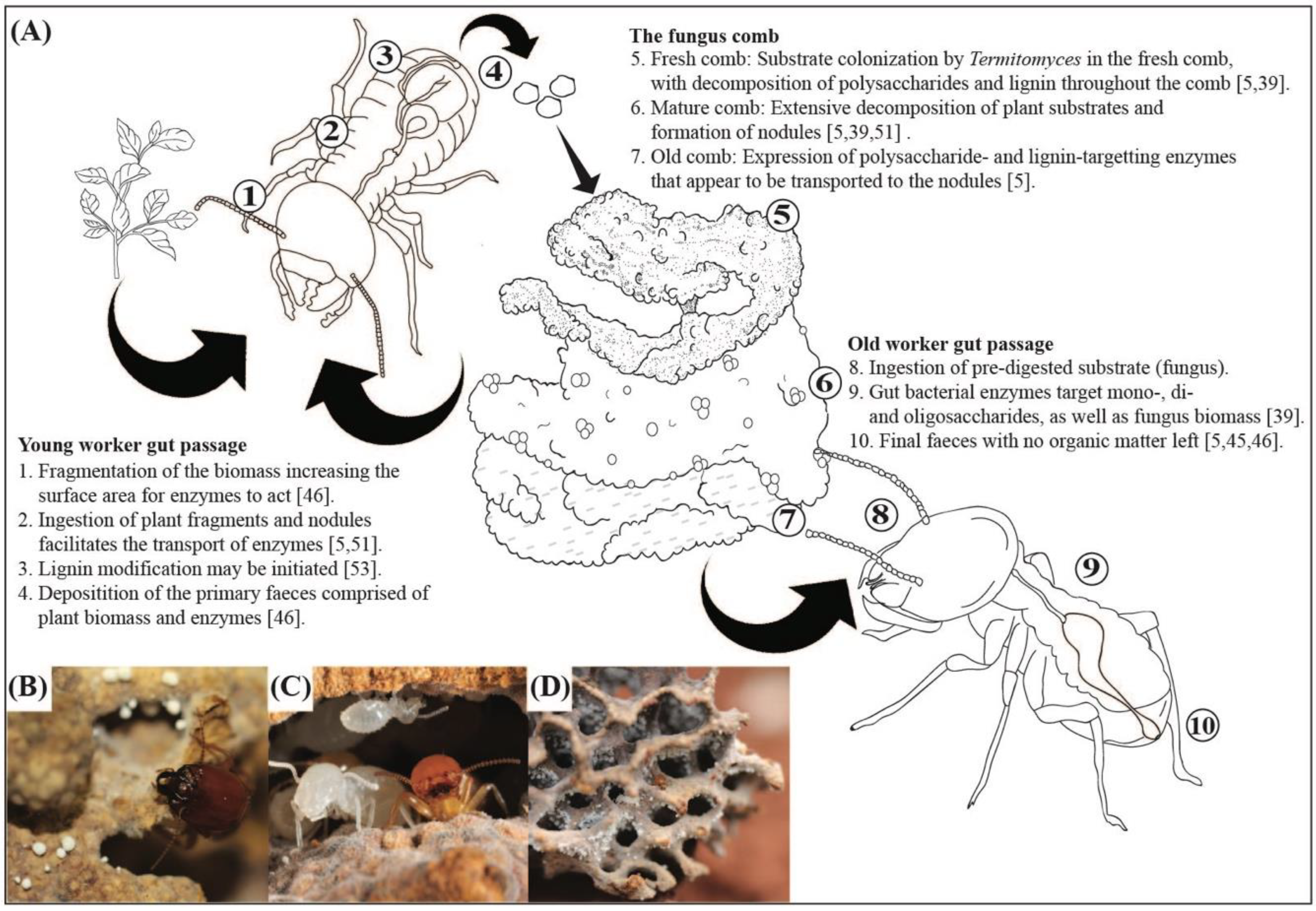
2.4. Plant Biomass Processing and Breakdown
3. Research Avenues to Improve Our Understanding of the Evolution of Ancient Symbiotic Plant Biomass Decomposition
3.1. How has 30 Million Years of Evolution Altered Symbiotic Contributions to Plant-Biomass Decomposition?
3.2. Improving Our Understanding of the Link between Enzyme Targets and the Producing Organisms
3.3. How Variable Is Substrate Use across Termite Species
3.4. How Flexible Is Enzyme Production in Fungus-Growing Termite-Associated Symbionts?
3.5. How Do Caste Roles and Caste-Specific Symbionts Interact to Affect Decomposition?
3.6. Do Differences in Substrate Use Align with the Interaction Specificity between Termite Host and Symbionts?
3.7. Does Fenton Chemistry Play a Role in Lignin Depolymerization?
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pauly, M.; Keegstra, K. Cell-wall carbohydrates and their modification as a resource for biofuels. Plant J. 2008, 54, 559–568. [Google Scholar] [CrossRef] [PubMed]
- McNeil, M.; Darvill, A.G.; Fry, S.C.; Albersheim, P. Structure and functional of the primary cell walls of plants. Annu. Rev. Biochem. 1984, 53, 625–663. [Google Scholar] [CrossRef] [PubMed]
- Rancour, D.M.; Marita, J.M.; Hatfield, R.D. Cell wall composition throughout development for the model grass Brachypodium distachyon. Front. Plant Sci. 2012, 3, 266. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, R.C. Wood sugar analysis by anion chromatography. J. Wood Chem. Technol. 1991, 14, 495–501. [Google Scholar] [CrossRef]
- Da Costa, R.R.; Hu, H.; Pilgaard, B.; Sabine, S.M.; Schückel, J.; Pedersen, K.S.K.; Kračun, S.K.; Busk, P.K.; Harholt, J.; Sapountzis, P.; et al. Enzyme activities at different stages of plant biomass decomposition in three species of fungus-growing termites. Appl. Environ. Microbiol. 2018, 84, e01815-17. [Google Scholar] [CrossRef]
- Burton, R.A.; Fincher, G.B. (1,3;1,4)-β-D-glucans in cell walls of the Poaceae, lower plants, and fungi: A tale of two linkages. Mol. Plant 2009, 2, 873–882. [Google Scholar] [CrossRef]
- Taylor, N.G. Cellulose biosynthesis and deposition in higher plants. New Phytol. 2008, 178, 239–252. [Google Scholar] [CrossRef]
- Ishii, S. Cell Wall Cementing materials of grass leaves. Plant Physiol. 1984, 76, 959–961. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, H.; Wang, C.; Xu, J.R. Comparative analysis of fungal genomes reveals different plant cell wall degrading capacity in fungi. BMC Genom. 2013, 14, 274. [Google Scholar] [CrossRef]
- Smant, G.; Stokkermans, J.P.W.G.; Yan, Y.; de Boer, J.M.; Baum, T.J.; Wang, X.; Hussey, R.S.; Gommers, F.J.; Henrissat, B.; Davis, E.L.; et al. Endogenous cellulases in animals: Isolation of -1,4-endoglucanase genes from two species of plant-parasitic cyst nematodes. Proc. Natl. Acad. Sci. USA 1998, 95, 4906–4911. [Google Scholar] [CrossRef]
- Suen, G.; Scott, J.J.; Aylward, F.O.; Adams, S.A.; Tringe, S.G.; Pinto-Tómas, A.A.; Foster, C.E.; Pauly, M.; Weimer, P.J.; Barry, K.W.; et al. An insect herbivore microbiome with high plant biomass-degrading capacity. PLoS Genet. 2010, 6, e1001129. [Google Scholar] [CrossRef] [PubMed]
- Leschine, S.B. Cellulose degradation in anaerobic environments. Annu. Rev. Microbiol. 1995, 49, 399–426. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.; Denman, S.E.; Hughes, R.J.; Geier, M.S.; Crowley, T.M.; Chen, H.; Haring, V.R.; Moore, R.J. Intestinal microbiota associated with differential feed conversion efficiency in chickens. Appl. Microbiol. Biotechnol. 2012, 96, 1361–1369. [Google Scholar] [CrossRef] [PubMed]
- Brulc, J.M.; Antonopoulos, D.A.; Berg, M.E.; Wilson, M.K.; Yannarell, A.C.; Dinsdale, E.A.; Edwards, R.E.; Frank, E.D.; Emerson, J.B.; Wacklin, P.; et al. Gene-centric metagenomics of the fiber-adherent bovine rumen microbiome reveals forage specific glycoside hydrolases. Proc. Natl. Acad. Sci. USA 2009, 106, 6–11. [Google Scholar] [CrossRef]
- Kambhampati, S.; Eggleton, P. Taxonomy and phylogenetics of Termites. In Termites: Evolution, Sociality, Symbioses and Ecology; Abe, T., Bignell, D.E., Higashi, M., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 1–23. [Google Scholar]
- Eggleton, P. An introduction to termites: Biology, taxonomy and functional morphology. In Biology of Termites: A Modern Synthesis; Bignell, D., Roisin, Y., Lo, N., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 1–26. [Google Scholar]
- Inward, D.J.G.; Vogler, A.P.; Eggleton, P. A comprehensive phylogenetic analysis of termites (Isoptera) illuminates key aspects of their evolutionary biology. Mol. Phylogenet. Evol. 2007, 44, 953–967. [Google Scholar] [CrossRef]
- Krishna, K.; Grimaldi, D.A.; Krishna, V. Introduction. In Treatise on the Isoptera of the World; Krishna, K., Grimaldi, D.A., Krishna, V., Engel, M.S., Eds.; American Museum of History: New York, NY, USA, 2013; pp. 1–200. ISBN 1530-7085. [Google Scholar]
- Bourguignon, T.; Lo, N.; Cameron, S.L.; Šobotník, J.; Hayashi, Y.; Shigenobu, S.; Watanabe, D.; Roisin, Y.; Miura, T.; Evans, T.A. The evolutionary history of termites as inferred from 66 mitochondrial genomes. Mol. Biol. Evol. 2015, 32, 406–421. [Google Scholar] [CrossRef]
- Bourguignon, T.; Lo, N.; Sobotnik, J.; Ho, S.Y.W.; Iqbal, N.; Coissac, E.; Lee, M.; Jendryka, M.M.; Sillam-Dussès, D.; Krizkova, B.; et al. Mitochondrial phylogenomics resolves the global spread of higher termites, ecosystem engineers of the tropics. Mol. Biol. Evol. 2017, 34, 597. [Google Scholar] [CrossRef]
- Bignell, D.E. Introduction to symbiosis. In Termites: Evolution, Sociality, Symbioses, Ecology, Part 2; Abe, T., Bignell, D.E., Higashi, M., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 189–208. [Google Scholar]
- Donovan, S.E.; Eggleton, P.; Bignell, D.E. Gut content analysis and a new feeding group classification of termites. Ecol. Entomol. 2001, 26, 356–366. [Google Scholar] [CrossRef]
- Scrivener, A.M.; Slaytor, M.; Rose, H.A. Symbiont-independent digestion of cellulose and starch in Panesthia cribrata Saussure, an Australian wood-eating cockroach. J. Insect Physiol. 1989, 35, 935–941. [Google Scholar] [CrossRef]
- Cragg, M.S.; Beckham, G.T.; Bruce, N.C.; Bugg, T. D. H.; Distel, D. L.; Dupree, P.; Etxabe, A.G.; Goodell, B.S.; Jellison, J.; McGeehan, J.E.; et al. Lignocellulose degradation mechanisms across the Tree of Life. Curr. Opin. Chem. Biol. 2015, 29, 108–119. [Google Scholar] [CrossRef]
- Brune, A. Symbiotic digestion of lignocellulose in termite guts. Nat. Rev. Microbiol. 2014, 12, 168–180. [Google Scholar] [CrossRef]
- Breznak, J.A.; Brune, A. Role of microorganisms in the digestion of lignocellulose by termites. Annu. Rev. Entomol. 1994, 39, 453–487. [Google Scholar] [CrossRef]
- Lo, N.; Eggleton, P. Termite phylogenetics and co-cladogenesis with symbionts. In Biology of Termites: A Modern Synthesis; Bignell, D.E., Roisin, Y., Lo, N., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 27–50. [Google Scholar]
- Brune, A.; Dietrich, C. The gut microbiota of termites: Digesting the diversity in the light of ecology and evolution. Annu. Rev. Microbiol. 2015. [Google Scholar] [CrossRef]
- Bignell, D.E.; Eggleton, P. Termites in Ecosystems. In Termites: Evolution, Sociality, Symbioses, Ecology; Abe, T., Bignell, D.E., Higashi, M., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 363–387. [Google Scholar]
- Ohkuma, M.; Brune, A. Diversity, structure, and evolution of the termite gut microbial community. In Biology of Termites: A Modern Synthesis; Bignell, D.E., Roisin, Y., Lo, N., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 413–438. ISBN 9789048139767. [Google Scholar]
- Abdul Rahman, N.; Parks, D.H.; Willner, D.L.; Engelbrektson, A.L.; Goffredi, S.K.; Warnecke, F.; Scheffrahn, R.H.; Hugenholtz, P. A molecular survey of Australian and North American termite genera indicates that vertical inheritance is the primary force shaping termite gut microbiomes. Microbiome 2015, 3, 5. [Google Scholar] [CrossRef] [PubMed]
- Brune, A.; Ohkuma, M. Role of the termite gut microbiota in symbiotic digestion. In Biology of Termites: A Modern Synthesis; Bignell, D.E., Roisin, Y., Lo, N., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 439–475. [Google Scholar]
- Hongoh, Y. Toward the functional analysis of uncultivable, symbiotic microorganisms in the termite gut. Cell. Mol. Life Sci. 2011, 68, 1311–1325. [Google Scholar] [CrossRef] [PubMed]
- Mikaelyan, A.; Köhler, T.; Lampert, N.; Rohland, J.; Boga, H.; Meuser, K.; Brune, A. Classifying the bacterial gut microbiota of termites and cockroaches: A curated phylogenetic reference database (DictDb). Syst. Appl. Microbiol. 2015, 38, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Roberts, E.M.; Todd, C.N.; Aanen, D.K.; Nobre, T.; Hilbert-Wolf, H.L.; O’Connor, P.M.; Tapanila, L.; Mtelela, C.; Stevens, N.J. Oligocene termite nests with in situ fungus gardens from the Rukwa rift basin, Tanzania, support a paleogene African origin for insect agriculture. PloS ONE 2016, 11, 1–17. [Google Scholar] [CrossRef]
- Korb, J.; Aanen, D.K. The evolution of uniparental transmission of fungal symbionts in fungus-growing termites (Macrotermitinae). Behav. Ecol. Sociobiol. 2003, 53, 65–71. [Google Scholar] [CrossRef]
- Aanen, D.K.; Eggleton, P.; Rouland-Lefevre, C.; Guldberg-Frøslev, T.; Rosendahl, S.; Boomsma, J.J. The evolution of fungus-growing termites and their mutualistic fungal symbionts. Proc. Natl. Acad. Sci. USA 2002. [Google Scholar] [CrossRef]
- Kirk, P.M.; Cannon, P.F.; David, J.C.; Stalpers, J.A. Ainsworth & Bigby’s Dictionary of the Fungi; Kirk, P.M., Cannon, P.F., David, J.C., Stalpers, J.A., Eds.; CAB Int.: Wallingford, UK, 2011. [Google Scholar]
- Poulsen, M.; Hu, H.; Li, C.; Chen, Z.; Xu, L.; Otani, S.; Nygaard, S.; Nobre, T.; Klaubauf, S.; Schindler, P.M.; et al. Complementary symbiont contributions to plant decomposition in a fungus-farming termite. Proc. Natl. Acad. Sci. USA 2014, 111, 14500–14505. [Google Scholar] [CrossRef]
- Makonde, H.M.; Boga, H.I.; Osiemo, Z.; Mwirichia, R.; Mackenzie, L.M.; Göker, M.; Klenk, H.P. 16S-rRNA-based analysis of bacterial diversity in the gut of fungus-cultivating termites (Microtermes and Odontotermes species). Antonie Leeuwenhoek 2013, 104, 869–883. [Google Scholar] [CrossRef]
- Hongoh, Y.; Ekpornprasit, L.; Inoue, T.; Moriya, S.; Trakulnaleamsai, S.; Ohkuma, M.; Noparatnaraporn, N.; Kudo, T. Intracolony variation of bacterial gut microbiota among castes and ages in the fungus-growing termite Macrotermes gilvus. Mol. Ecol. 2006, 15, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Otani, S.; Zhukova, M.; Koné, N.A.; da Costa, R.R.; Sapountzis, P.; Poulsen, M. Gut microbial compositions mirror caste-specific diets in a major lineage of social insects. Environ. Microbiol. Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M. Towards an integrated understanding of the consequences of fungus domestication on the fungus-growing termite gut microbiota. Environ. Microbiol. 2015, 17, 2562–2572. [Google Scholar] [CrossRef]
- Sands, W.A. The initiation of fungus comb construction in laboratory colonies of Ancistrotermes guineensis (Silvestri). Insectes Soc. 1960, 7, 251–263. [Google Scholar] [CrossRef]
- Sieber, R. Establishment of fungus comb in laboratory colonies of Macrotermes michaelseni and Odontotermes montanus (Isoptera, Macrotermitinae). Insectes Soc. 1983, 30, 204–209. [Google Scholar] [CrossRef]
- Sieber, R.; Leuthold, R.H. Behavioural elements and their meaning in incipient laboratory colonies of the fungus-growing termite Macrotermes michaelseni (Isoptera: Macrotermitinae). Insectes Soc. 1981, 28, 371–382. [Google Scholar] [CrossRef]
- Johnson, R.A.; Thomas, R.J.; Wood, T.G.; Swift, M.J. The inoculation of the fungus comb in newly founded colonies of some species of the Macrotermitinae (Isoptera) from Nigeria. J. Nat. Hist. 1981, 15, 751–756. [Google Scholar] [CrossRef]
- Aanen, D.K.; De Fine Licht, H.H.; Debets, A.J.M.; Kerstes, N.A.G.; Hoekstra, R.F.; Boomsma, J.J. High symbiont relatedness stabilizes mutualistic cooperation in fungus-growing termites. Science 2009, 326, 1103–1106. [Google Scholar] [CrossRef]
- Darlington, J.P.E.C. The underground passages and storage pits used in foraging by a nest of the termite Macrotermes michaelseni in Kajiado, Kenya. J. Zool. 1982, 198, 237–247. [Google Scholar] [CrossRef]
- De Fine Licht, H.H.; Boomsma, J.J.; Aanen, D.K. Asymmetric interaction specificity between two sympatric termites and their fungal symbionts. Ecol. Entomol. 2007. [Google Scholar] [CrossRef]
- Nobre, T.; Rouland-Lefèvre, C.; Aanen, D.K. Comparative biology of fungus cultivation in termites and ants. In Biology of Termites: A Modern Synthesis; Bignell, D.E., Roisin, Y., Lo, N., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 193–210. ISBN 9789048139767. [Google Scholar]
- Rouland-lefèvre, C.; Bignell, D.E. Cultivation of symbiotic fungi by termites of the subfamily Macrotermitinae. In Symbiosis; Seckbach, J., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; pp. 733–756. [Google Scholar]
- Li, H.; Yelle, D.J.; Li, C.; Yang, M.; Ke, J.; Zhang, R.; Liu, Y.; Zhu, N.; Liang, S.; Mo, X.; et al. Lignocellulose pretreatment in a fungus-cultivating termite. Proc. Natl. Acad. Sci. USA 2017, 114, 4709–4714. [Google Scholar] [CrossRef]
- De Fine Licht, H.H.; Boomsma, J.J.; Aanen, D.K. Presumptive horizontal symbiont transmission in the fungus-growing termite Macrotermes natalensis. Mol. Ecol. 2006, 64, 2055–2069. [Google Scholar] [CrossRef] [PubMed]
- Aanen, D.K. As you reap, so shall you sow: Coupling of harvesting and inoculating stabilizes the mutualism between termites and fungi. Biol. Lett. 2006, 2, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Aanen, D.K.; Ros, V.I.D.; De Fine Licht, H.H.; Mitchell, J.; De Beer, Z.W.; Slippers, B.; Rouland-LeFèvre, C.; Boomsma, J.J. Patterns of interaction specificity of fungus-growing termites and Termitomyces symbionts in South Africa. BMC Evol. Biol. 2007, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, R.R.; Vreeburg, S.M.E.; Shik, J.Z.; Aanen, D.K.; Poulsen, M. Can interaction specificity in the fungus-farming termite symbiosis be explained by nutritional requirements of the fungal crop? Fungal Ecol. 2018. [Google Scholar] [CrossRef]
- Nobre, T.; Aanen, D.K. Fungiculture or termite husbandry? The ruminant hypothesis. Insects 2012, 3, 307–323. [Google Scholar] [CrossRef] [PubMed]
- Nobre, T.; Eggleton, P.; Aanen, D.K. Vertical transmission as the key to the colonization of Madagascar by fungus-growing termites? Proc. R. Soc. B Biol. Sci. 2010, 277, 359–365. [Google Scholar] [CrossRef]
- Bright, M.; Bulgheresi, S. A complex journey: Transmission of microbial symbionts. Nat. Rev. Microbiol. 2010, 8, 218–230. [Google Scholar] [CrossRef]
- Visser, B.; Le Lann, C.; den Blanken, F.J.; Harvey, J.A.; van Alphen, J.J.M.; Ellers, J. Loss of lipid synthesis as an evolutionary consequence of a parasitic lifestyle. Proc. Natl. Acad. Sci. USA 2010, 11, 8677–8682. [Google Scholar] [CrossRef]
- Ellers, J.; Toby Kiers, E.; Currie, C.R.; Mcdonald, B.R.; Visser, B. Ecological interactions drive evolutionary loss of traits. Ecol. Lett. 2012, 15, 1071–1082. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Zhang, L.; Zhou, H.; Zhang, M.; Yan, X.; Wang, Q.; Long, Y.; Xie, L.; Wang, S.; Huang, Y.; et al. Metagenomic Insights into metabolic capacities of the gut microbiota in a fungus-cultivating termite (Odontotermes yunnanensis). PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Yan, X.; Zhang, M.; Xie, L.; Wang, Q.; Huang, Y.; Zhou, X.; Wang, S.; Zhou, Z. Microbiome of fungus-growing termites: A new reservoir for lignocellulase genes. Appl. Environ. Microbiol. 2011, 77, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Um, S.; Fraimout, A.; Sapountzis, P.; Oh, D.C.; Poulsen, M. The fungus-growing termite Macrotermes natalensis harbors bacillaene-producing Bacillus sp. that inhibit potentially antagonistic fungi. Sci. Rep. 2013, 3, 3250. [Google Scholar] [CrossRef] [PubMed]
- Breznak, J.A. Intestinal microbiota of termites and other xylophagous Insects. Annu. Rev. Microbiol. 1982, 36, 323. [Google Scholar] [CrossRef]
- Sapountzis, P.; de Verges, J.; Rousk, K.; Cilliers, M.; Vorster, B.J.; Poulsen, M. Potential for nitrogen fixation in the fungus-growing termite symbiosis. Front. Microbiol. 2016, 7, 1–5. [Google Scholar] [CrossRef]
- Hu, H.; da Costa, R.R.; Pilgaard, B.; Busk, P.K.; Schiøtt, M.; Lange, L.; Poulsen, M. Fungiculture in termites is associated with a mycolytic gut bacteria community. mSphere 2019. accepted pending minor revision. [Google Scholar]
- Otani, S.; Mikaelyan, A.; Nobre, T.; Hansen, L.H.; Koné, N.A.; Sørensen, S.J.; Aanen, D.K.; Boomsma, J.J.; Brune, A.; Poulsen, M. Identifying the core microbial community in the gut of fungus-growing termites. Mol. Ecol. 2014, 23, 4631–4644. [Google Scholar] [CrossRef]
- Hongoh, Y.; Deevong, P.; Inoue, T.; Moriya, S.; Trakulnaleamsai, S.; Ohkuma, M.; Vongkaluang, C.; Noparatnaraporn, N.; Kudo, T. Intra- and interspecific comparisons of bacterial diversity and community structure support coevolution of gut microbiota and termite host. Appl. Environ. Microbiol. 2005, 71, 6590–6599. [Google Scholar] [CrossRef]
- Hongoh, Y. Diversity and genomes of uncultured microbial symbionts in the termite gut. Biosci. Biotechnol. Biochem. 2010, 74, 1145–1151. [Google Scholar] [CrossRef]
- Dietrich, C.; Köhler, T.; Brune, A. The cockroach origin of the termite gut microbiota: Patterns in bacterial community structure reflect major evolutionary events. Appl. Environ. Microbiol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Dietrich, C.; Zhu, N.; Mikaelyan, A.; Ma, B.; Pi, R.; Liu, Y.; Yang, M.; Brune, A.; Mo, J. Age polyethism drives community structure of the bacterial gut microbiota in the fungus-cultivating termite Odontotermes formosanus. Environ. Microbiol. 2016, 18, 1440–1451. [Google Scholar] [CrossRef] [PubMed]
- Wood, T.G.; Sands, W.A. The role of termites in ecosystems. In Production Ecology of Ants and Termites; Brian, M.V., Ed.; Cambridge Univ. Press: Cambridge, UK, 1978; pp. 245–292. [Google Scholar]
- Buxton, R.D. Termites and the turnover of dead wood in an arid tropical environment. Oecologia 1981, 51, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Ashton, L.A.; Griffiths, H.G.; Parr, C.L.; Evans, T.A.; Didham, R.K.; Hasan, F.; Teh, Y.A.; Tin, H.S.; Vairappan, C.S.; Eggleton, P. Termites mitigate the ecosystem-wide effects of drought in tropical rainforest. Science 2019, 177, 174–177. [Google Scholar] [CrossRef]
- Nkunika, P.O.Y. A survey of the termite species associated with Eucalyptus plantations in Zambia. Zamb. J. Sci. Technol. 1980, 5, 33–38. [Google Scholar]
- Mora, P.; Rouland, C.; Renoux, J. Foraging, nesting and damage caused by Microtermes subhyalinus (Isoptera: Termitidae) in a sugarcane plantation in the Central African Republic. Bull. Entomol. Res. 1996, 86, 387. [Google Scholar] [CrossRef]
- Acda, M.N. Foraging populations and territories of the tropical subterranean termite Macrotermes gilvus (Isoptera: Macrotermitinae). Sociobiology 2004, 43, 169–177. [Google Scholar]
- Sattar, A.; Misbah, M.; Salihah, Z.; Khatoon, R. Foraging activity of Microtermes unicolor Synder and Odontotermes lokanandi Chatterjee and Thakur (Termitidae, Isoptera) in Peshawar, Pakistan. Suranaree J. Sci. Technol. 2007, 15, 69–74. [Google Scholar]
- Sattar, A.; Naeem, M.; ul-Haq, E. Impact of environmental factors on the population dynamics, density and foraging activities of Odontotermes lokanandi and Microtermes obesi in Islamabad. SpringerPlus 2013, 2, 1–7. [Google Scholar] [CrossRef]
- Iqbal, N.; Saeed, S.; Evans, T.A.; Kwon, Y.J. Foraging activity and population estimation of Microtermes mycophagus Desneux (Isoptera: Termitidae: Macrotermitinae) in Multan, Punjab, Pakistan. Entomol. Res. 2015, 45, 51–57. [Google Scholar] [CrossRef]
- Lepage, M.G. L’impact des populations récoltantes de Macrotermes michaelseni (Sjostedt) (Isoptera, Macrotermitinae) dans un écosystème semi-aride (Kajiado-Kenya)—I—L’activité de récolte et son déterminisme. Insectes Soc. 1981, 28, 297–308. [Google Scholar] [CrossRef]
- Boutton, T.W.; Arshad, M.A.; Tieszen, L.L. Stable isotope analysis of termite food habits in East African grasslands. Oecologia 1983, 59, 1–6. [Google Scholar] [CrossRef]
- Dangerfield, J.M.; Schuurman, G. Foraging by fungus-growing termites (Isoptera: Termitidae, Macrotermitinae) in the Okavango Delta, Botswana. J. Trop. Ecol. 2000, 16, 717–731. [Google Scholar] [CrossRef]
- Janusz, G.; Pawlik, A.; Sulej, J.; Świderska-Burek, U.; Jarosz-Wilkołazka, A.; Paszczyński, A. Lignin degradation: Microorganisms, enzymes involved, genomes analysis and evolution. FEMS Microbiol. Rev. 2017, 41, 941–962. [Google Scholar] [CrossRef] [PubMed]
- Johjima, T.; Taprab, Y.; Noparatnaraporn, N.; Kudo, T.; Ohkuma, M. Large-scale identification of transcripts expressed in a symbiotic fungus (Termitomyces) during plant biomass degradation. Appl. Microbiol. Biotechnol. 2006, 73, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Schuurman, G. Foraging and distribution patterns in a termite assemblage dominated by fungus-growing species in semi-arid northern Botswana. J. Trop. Ecol. 2006, 22, 277–287. [Google Scholar] [CrossRef]
- N’Dri, A.B.; Gignoux, J.; Konaté, S. Food preferences and foraging strategies of wood-feeding termites in a West African savanna. Curr. Sci. 2018, 114, 186–192. [Google Scholar] [CrossRef]
- Coaton, W.; Sheasby, J. Preliminary report on a survey of the termites (Isoptera) of South West Africa. State Mus. 1972, 2, 1–129. [Google Scholar]
- Longhurst, C.; Howse, P.E. The use of kairomones by Megaponera foetens (Fab.) (Hymenoptera: Formicidae) in the detection of its termite prey. Anim. Behav. 1978, 2013–2018. [Google Scholar] [CrossRef]
- Abe, T. Termite community in the grassland of Kenya with special reference to their feeding habits. In Social Insects and the Environment, Proceedings of the 11th International Congress of IUSSI, Bangalore, India, 5–8 August 1990; International Union for the Study of Social Insects: Bangalore, India, 1990; pp. 207–208. [Google Scholar]
- Collins, N.M. The role of termites in the decomposition of wood and leaf litter in the Southern Guinea savanna of Nigeria. Oecologia 1981, 51, 389–399. [Google Scholar] [CrossRef]
- Wood, A.T.G.; Johnson, R.A.; Bacchus, S.; Shittu, M.O.; Anderson, J.M. Abundance and Distribution of Termites (Isoptera) in a Riparian Forest in the Southern Guinea Savanna Vegetation Zone of Nigeria. Assoc. Trop. Biol. Conserv. 1982, 14, 25–39. [Google Scholar] [CrossRef]
- Eggleton, P.; Bignell, D.E.; Sands, W.A.; Waite, B.; Wood, T.G.; Lawton, J.H. The species richness of termites (Isoptera) under differing levels of forest disturbance in the Mbalmayo Forest Reserve, southern Cameroon. J. Trop. Ecol. 1995, 11, 85–98. [Google Scholar] [CrossRef]
- Lee, C.Y.; Ngee, P.S.; Lee, L.C. Foraging populations and territories of a mound-building subterranean termite, Microtermes pakistanicus (Isoptera: Macrotermitinae). Sociobiology 2003, 41, 307–316. [Google Scholar]
- Jouquet, P.; Boulain, N.; Gignoux, J.; Lepage, M. Association between subterranean termites and grasses in a West African savanna: Spatial pattern analysis shows a significant role for Odontotermes n. pauperans. Appl. Soil Ecol. 2004, 27, 99–107. [Google Scholar] [CrossRef]
- Al-Hemyari, A.A. Factors affecting foraging activity of Microtermes yemenensis (Isoptera: Termitidae). Ann. Agric. Sci. 1994, 32, 987–995. [Google Scholar]
- Rouland, C.; Lepage, M.; Chotte, J.L.; Diouf, M.; Ndiaye, D.; Ndiaye, S.; Seugé, C.; Brauman, A. Experimental manipulation of termites (Isoptera, Macrotermitinae) foraging patterns in a Sahelo-Sudanese savanna: Effect of litter quality. Insectes Soc. 2003, 50, 309–316. [Google Scholar] [CrossRef]
- Schuurman, G. Decomposition rates and termite assemblage composition in semiarid Africa. Ecology 2005, 86, 1236–1249. [Google Scholar] [CrossRef]
- Mitchell, B.L. Report on a survey of the termites of Zimbabwe. Occas. Pap. Natl. Mus. South. Rhod. 1980, 6, 187–323. [Google Scholar]
- Mugerwa, S.; Nyangito, M.; Mpairwe, D.; Nderitu, J. Effect of biotic and abiotic factors on composition and foraging intensity of subterranean termites. Afr. J. Environ. Sci. Technol. 2011, 5, 579–588. [Google Scholar] [CrossRef]
- Ouédraogo, E.; Mando, A.; Brussaard, L. Soil macrofaunal-mediated organic resource disappearance in semi-arid West Africa. Appl. Soil Ecol. 2004, 27, 259–267. [Google Scholar] [CrossRef]
- Sands, W.A. Observations on termites destructive to trees and crops. North. Reg. Min. Agric. Samaru Res. Bull. 1962, 26, 1–14. [Google Scholar]
- Korb, J.; Linsenmair, K.E. Evaluation of predation risk in the collectively foraging termite Macrotermes bellicosus. Insectes Soc. 2002, 49, 264–269. [Google Scholar] [CrossRef]
- Johjima, T.; Inoue, T.; Ohkuma, M.; Noparatnaraporn, N.; Kudo, T. Chemical Analysis of Food Processing by the Fungus-Growing Termite Macrotermes gilvus. Sociobiology 2003, 42, 815–824. [Google Scholar]
- Bathellier, J. Contribution à l’etude systematique et biologique des Termites de l’Indochine. Faune Colon. Fran 1927, 1, 125–365. [Google Scholar]
- Kalshoven, L.G.E. Observations on Macrotermes gilvus Holmgr. In Java-3 Accumulations of finely cut vegetable matter in the nests. Insectes Soc. 1956, 3, 455–461. [Google Scholar] [CrossRef]
- Santoso, E.T.H. Pest and disease control of timber estates in East Kalimantan. J. Res. Dev. 1991, 7, 14–17. [Google Scholar]
- Lepage, M. Foraging and food consumption of Macrotermes subhyalinus. In Proceedings of the VIIIth International Congress IUSSI, Wageningen, The Netherlands, 5–9 September 1977; pp. 249–252. [Google Scholar]
- Lepage, M. L’ impacte des populations récoltantes des Macrotermes michaelseni (Sjostedt) (Isoptera: Macrotermitinae) dans un écosystème semi-aride (Kaijaido–Kenya) II. Nourriture récoltée, comparison avec les grands herbivores. Insectes Soc. 1981, 28, 309–319. [Google Scholar] [CrossRef]
- Grassé, P.P.; Noirot, C. La transmission des flagellés symbiotiques et les aliments des termites. Bull. Biol. Fr. Belg. 1945, 79, 273–292. [Google Scholar]
- Meyer, V.W.; Crewe, R.M.; Braack, L.E.O. Estimates of food consumption by the fungus-growing termite Macrotermes natalensis in a South African savanna-woodland. S. Afr. J. Sci. 2003, 99, 207–208. [Google Scholar]
- Lepage, M. L’impact des populations récoltantes de Macrotermes michaelseni (Sjöstedt) (Isoptera: Macrotermitinae) dans un écosystème semi-aride (Kajiado-Kenya) II—La nourriture récoltée, comparaison avec les grands herbivores. Insectes Soc. 1978, 28, 309–319. [Google Scholar]
- Burchard, I. On the removal of herbivore dung by Odontotermes spp. in Africa. Sociobiology 1989, 15, 261. [Google Scholar]
- Kemp, P.B. The termites of north-eastern Tanganyika: Their distribution and biology. Bull. Entomol. Res. 1955, 46, 112–135. [Google Scholar] [CrossRef]
- Soleymaninejadian, E.; Ji, B.Z.; Liu, S.W.; Yang, J.J.; Wang, H.J.; Ding, F. Polyethism in Odontotermes formosanus Shiraki. Int. J. Biol. Med. Res. 2014, 5, 4231–4238. [Google Scholar]
- Chiu, C.I.; Yeh, H.T.; Li, P.L.; Kuo, C.Y.; Tsai, M.J.; Li, H.F. Foraging Phenology of the Fungus-Growing Termite Odontotermes formosanus (Blattodea: Termitidae). Environ. Entomol. 2018, 47, 1509–1516. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.U.; Javed, H.; Ayyaz, M. Evaluation of Different Groundnut Arachis hypogea L. Cultivars Against Termites, Odontotermes obesus (Rambur) in Rawalpindi, Pakistan. Turk. J. Agric. Food Sci. Technol. 2015, 3, 448–452. [Google Scholar] [CrossRef]
- Wheatley, P.E.; Crowe, T.J. Pest Handbook: The Recognition and Control of the More Important Pests of Agriculture in Kenya; Government Printer: Nairobi, Kenya, 1967.
- Benjamen, D.M. Insects and mites on tea in Africa and adjacent islands. East Afr. Agric. J. 1968, 33, 345–357. [Google Scholar] [CrossRef]
- Togola, A.; Kotoklo, E.A.; Nwilene, F.E.; Amevoin, K.; Glitho, I.A.; Oyetunji, O.E.; Kiepe, P. Specific diversity and damage of termites on upland rice in Benin. J. Entomol. 2012, 9, 352–360. [Google Scholar] [CrossRef]
- Sands, W.A. Termites as pests of tropical food crops. Pest Artic. News Summ. 1973, 19, 167–177. [Google Scholar] [CrossRef]
- Dibangou, V.; Mora, P.; Celini, L.; Rouland-LeFèvre, C. Spatial distribution and density of fungus-growing termite Pseudacanthotermes militaris (Isoptera: Macrotermitinae) in the Congo Republic. Asian J. Biol. Sci. 2012, 5, 406–416. [Google Scholar] [CrossRef]
- Nair, K.S.S. Tropical Forest Insect Pests: Ecology, Impact, and Management; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Taprab, Y.; Johjima, T.; Maeda, Y.; Moriya, S.; Trakulnaleamsai, S.; Noparatnaraporn, N.; Ohkuma, M.; Kudo, T. Symbiotic fungi produce laccases potentially involved in phenol degradation in fungus combs of fungus-growing termites in Thailand. Appl. Environ. Microbiol. 2005, 71, 7696–7704. [Google Scholar] [CrossRef]
- Geier, M.S.; Filley, T.R.; Hatcher, P.G.; Hoover, K.; Carlson, J.E.; Jimenez-Gasco, M.M.; Nakagawa-Izumi, A.; Sleighter, R.L.; Tien, M. Lignin degradation in wood-feeding insects. Proc. Natl. Acad. Sci. USA 2008, 105, 12932–12937. [Google Scholar] [CrossRef]
- Tarmadi, D.; Tobimatsu, Y.; Yamamura, M.; Miyamoto, T.; Miyagawa, Y.; Umezawa, T.; Yoshimura, T. NMR studies on lignocellulose deconstructions in the digestive system of the lower termite Coptotermes formosanus Shiraki. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Li, H.; Chevrette, M.G.; Zhang, L.; Cao, L.; Zhou, H.; Zhou, X.; Zhou, Z.; Pope, P.B.; Currie, C.R.; et al. Functional metagenomics reveals abundant polysaccharide-degrading gene clusters and cellobiose utilization pathways within gut microbiota of a wood-feeding higher termite. ISME J. 2019, 13, 104–117. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, G.; Mikaelyan, A.; Fukui, C.; Matsuura, Y.; Watanabe, H.; Fujishima, M.; Brune, A. Fiber-associated spirochetes are major agents of hemicellulose degradation in the hindgut of wood-feeding higher termites. Proc. Natl. Acad. Sci. USA 2018, 115, 201810550. [Google Scholar] [CrossRef] [PubMed]
- Busk, P.K.; Pilgaard, B.; Lezyk, M.J.; Meyer, A.S.; Lange, L. Homology to peptide pattern for annotation of carbohydrate-active enzymes and prediction of function. BMC Bioinform. 2017, 18, 214. [Google Scholar] [CrossRef] [PubMed]
- Busk, P.K.; Lange, L. Function-based classification of carbohydrate-active enzymes by recognition of short, conserved peptide motifs. Appl. Environ. Microbiol. 2013, 79, 3380–3391. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, P.F.; Willerslev, E. Environmental DNA—An emerging tool in conservation for monitoring past and present biodiversity. Biol. Conserv. 2015, 183, 4–18. [Google Scholar] [CrossRef]
- De Fine Licht, H.H.; Schiøtt, M.; Mueller, U.G.; Boomsma, J.J. Evolutionary transitions in enzyme activity of ant fungus gardens. Evolution 2010, 64, 2055–2069. [Google Scholar] [CrossRef]
- Kračun, S.K.; Schückel, J.; Westereng, B.; Hygesen, L.G.; Monrad, R.N.; Eijsink, V.G.; Willats, W.G.T. A new generation of versatile chromogenic substrates for high-throughput analysis of biomass-degrading enzymes. Biotechnol. Biofuels 2018, 8, 70. [Google Scholar] [CrossRef]
- Sahadevan, L.D.M.; Misra, C.S.; Thankamani, V. Characterization of lignin-degrading enzymes (LDEs) from a dimorphic novel fungus and identification of products of enzymatic breakdown of lignin. 3 Biotech 2016, 6, 1–16. [Google Scholar] [CrossRef]
- Barbehenn, R.; Dodick, T.; Poopat, U.; Spencer, B. Fenton-type reactions and iron concentrations in the midgut fluids of tree-feeding caterpillars. Arch. Insect Biochem. Physiol. 2005, 60, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Franco Cairo, J.P.L.; Carazzolle, M.F.; Leonardo, F.C.; Mofatto, L.S.; Brenelli, L.B.; Gonçalves, T.A.; Uchima, C.A.; Domingues, R.R.; Alvarez, T.M.; Tramontina, R.; et al. Expanding the knowledge on lignocellulolytic and redox enzymes of worker and soldier castes from the lower termite Coptotermes gestroi. Front. Microbiol. 2016, 7, 1518. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Costa, R.R.; Hu, H.; Li, H.; Poulsen, M. Symbiotic Plant Biomass Decomposition in Fungus-Growing Termites. Insects 2019, 10, 87. https://doi.org/10.3390/insects10040087
da Costa RR, Hu H, Li H, Poulsen M. Symbiotic Plant Biomass Decomposition in Fungus-Growing Termites. Insects. 2019; 10(4):87. https://doi.org/10.3390/insects10040087
Chicago/Turabian Styleda Costa, Rafael R., Haofu Hu, Hongjie Li, and Michael Poulsen. 2019. "Symbiotic Plant Biomass Decomposition in Fungus-Growing Termites" Insects 10, no. 4: 87. https://doi.org/10.3390/insects10040087
APA Styleda Costa, R. R., Hu, H., Li, H., & Poulsen, M. (2019). Symbiotic Plant Biomass Decomposition in Fungus-Growing Termites. Insects, 10(4), 87. https://doi.org/10.3390/insects10040087




