Attraction of Female Aedes aegypti (L.) to Aphid Honeydew
Abstract
1. Introduction
2. Materials and Methods
2.1. Rearing of Experimental Mosquitoes
2.2. Rearing of Plants and Aphids
2.3. General Design of Y-Tube Behavioural Experiments
2.4. Attractiveness of Aphid-Infested and Honeydew-Soiled Plants
2.5. Attractiveness of Mechanically-Injured Plants
2.6. Attractiveness of Plants in the Presence of Non-Feeding Aphids
2.7. Honeydew Collection and Odorant Analysis
2.8. Preparation and Testing of Synthetic Honeydew Odorant Blends
2.9. Captures of Mosquitoes in Traps Baited with Synthetic Honeydew Odorant Blends
2.10. Statistical Analyses
3. Results
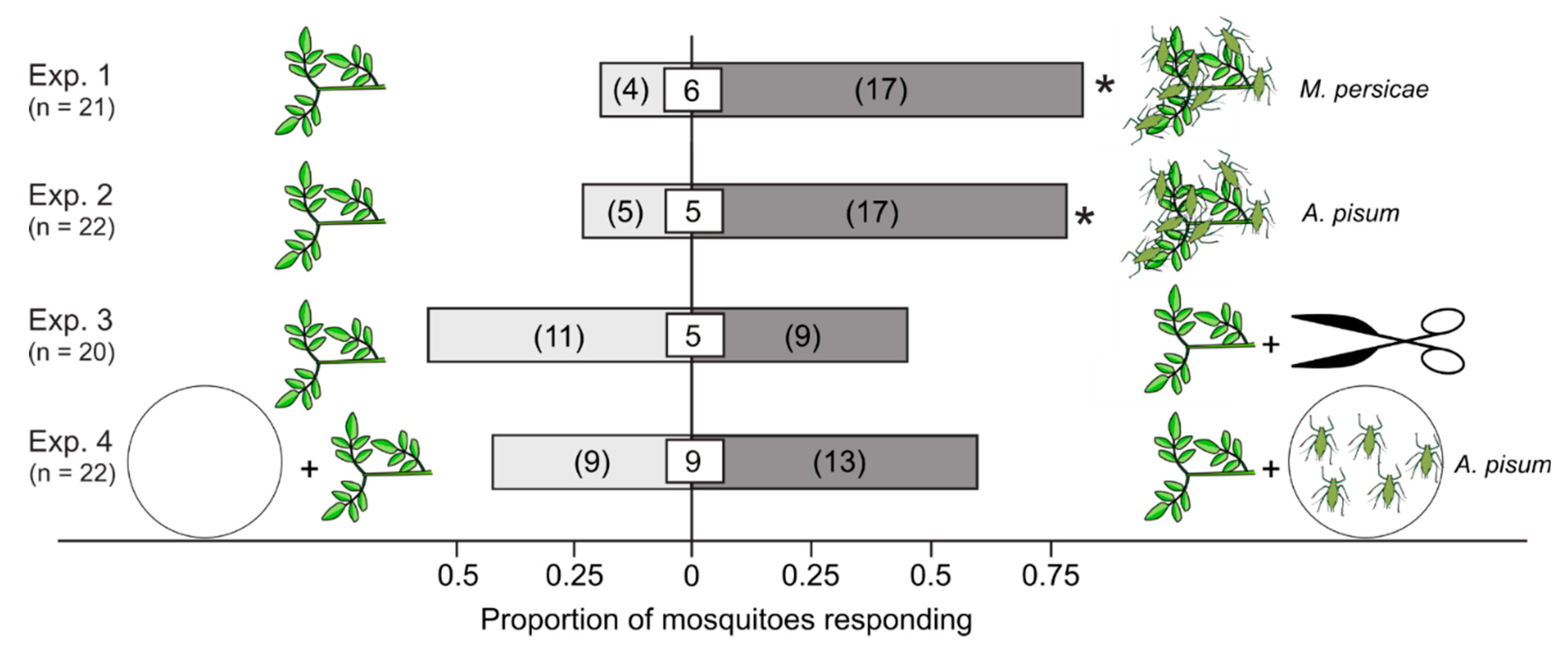
3.1. Attractiveness of Plants that Were Aphid-Infested, Mechanically Injured, or Paired with Non-Feeding Aphids
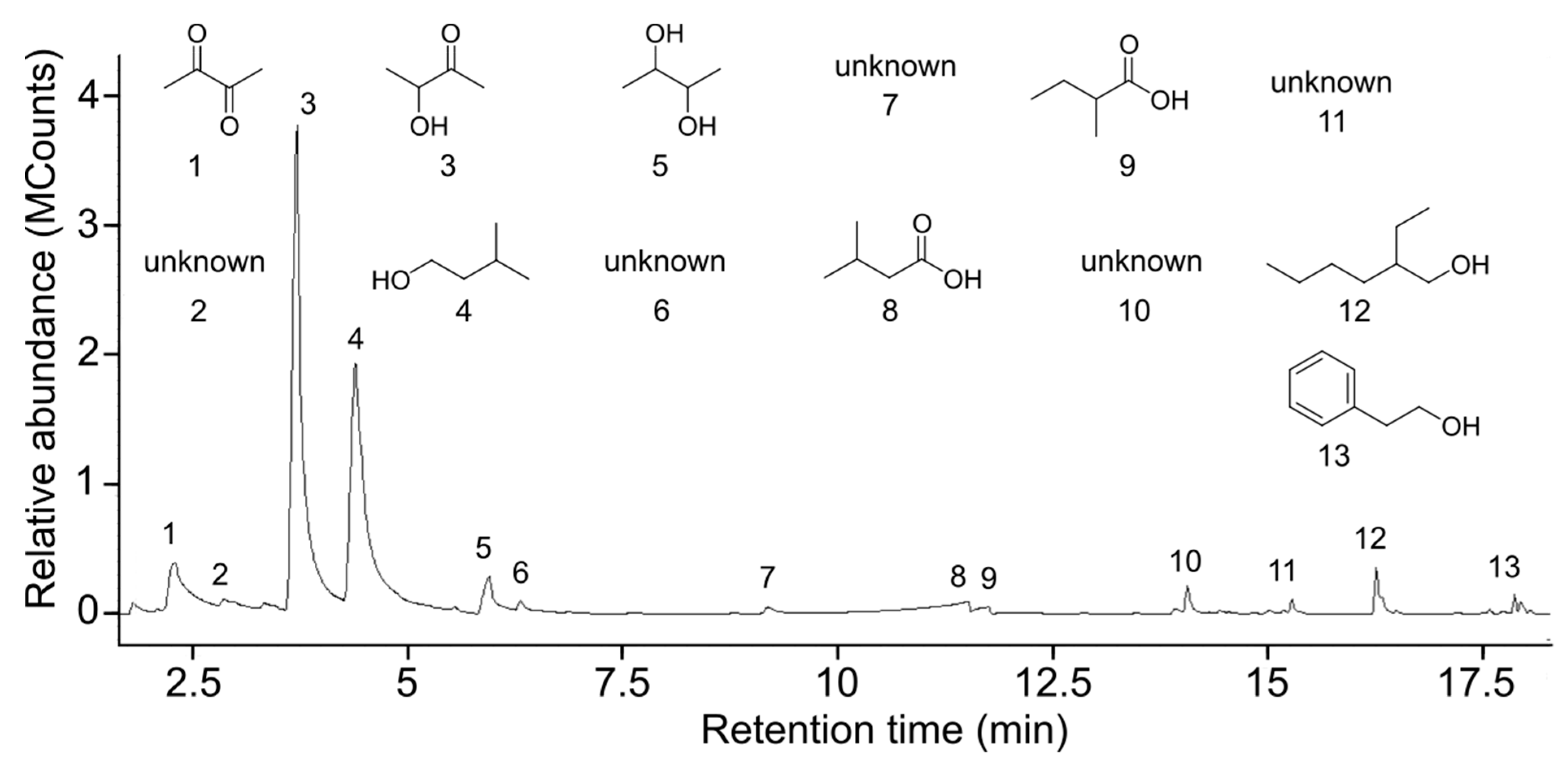
3.2. Analyses of Honeydew Headspace Odorants
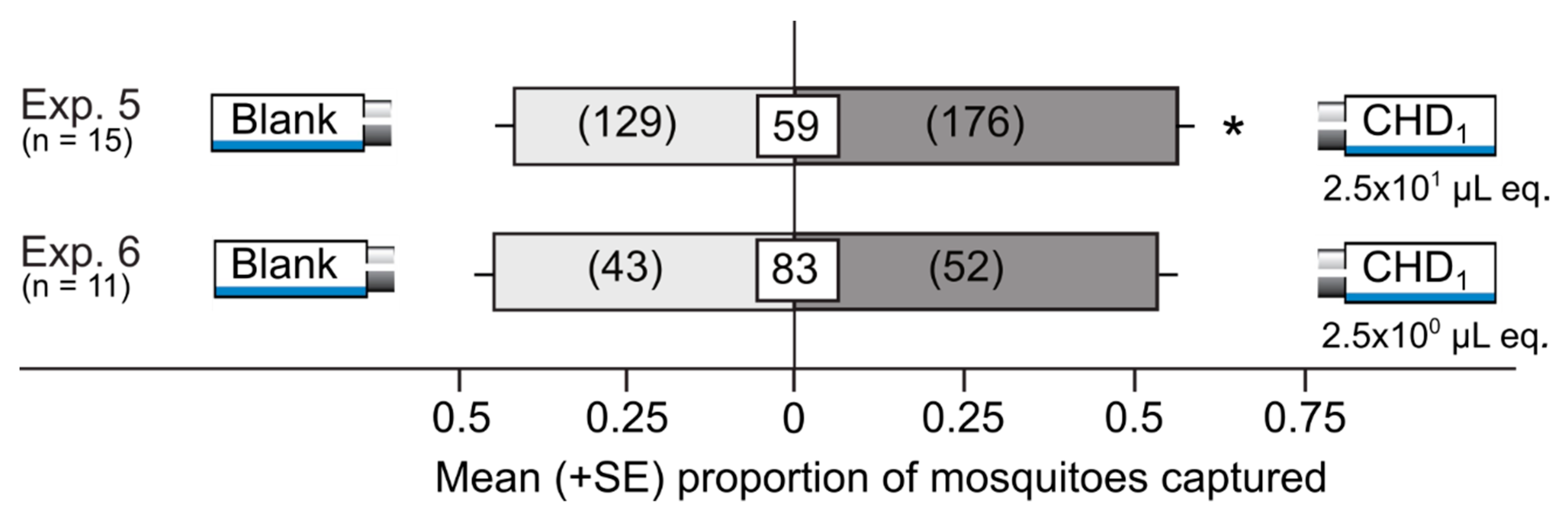
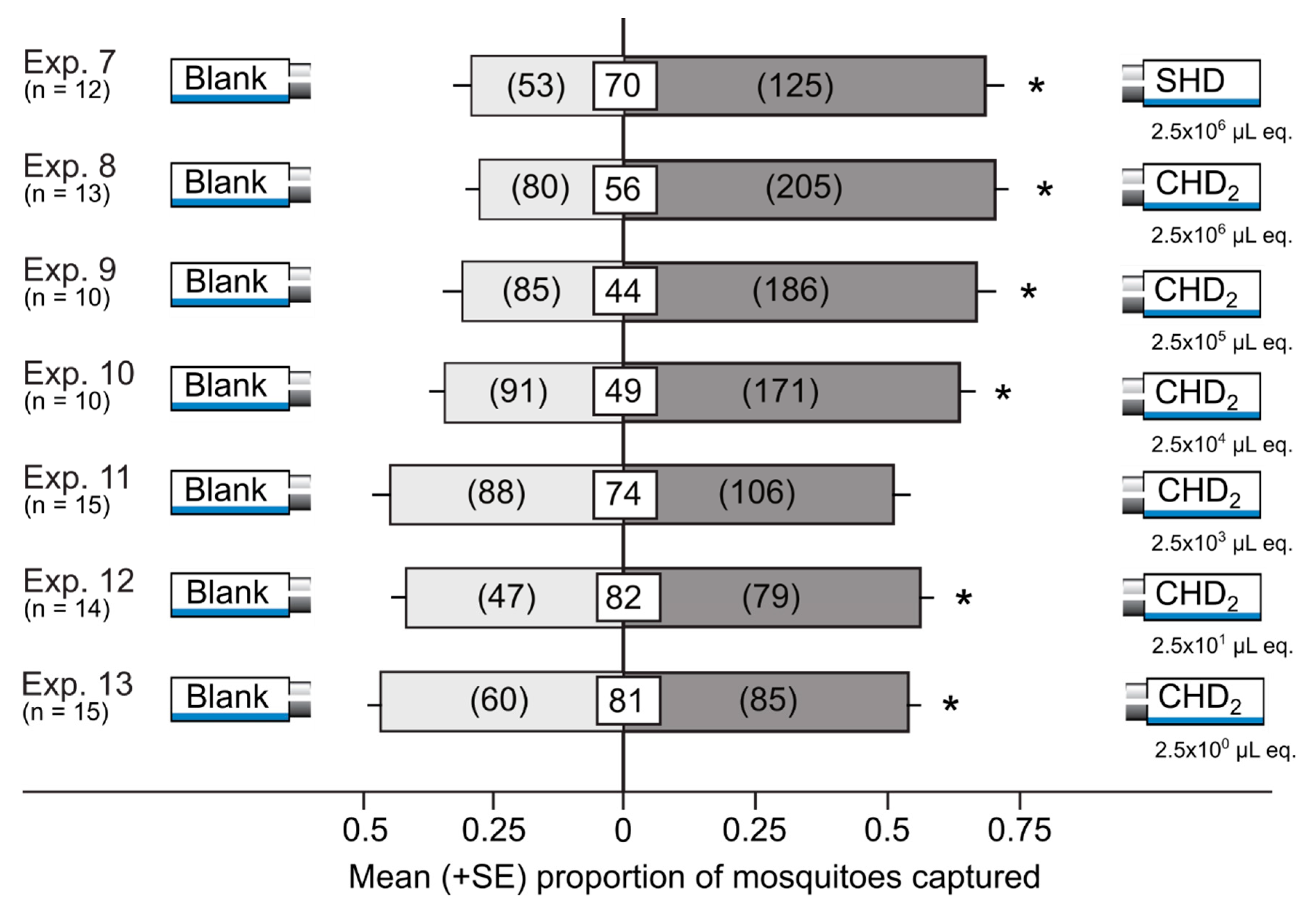
3.3. Attractiveness of Synthetic Honeydew Odorant Blends in Y-tube Olfactometers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Auclair, J. Aphid feeding and nutrition. Annu. Rev. Entomol. 1963, 8, 439–490. [Google Scholar] [CrossRef]
- Douglas, A.E. Honeydew. In Encyclopedia of Insects; Elsevier Inc.: Amsterdam, The Netherlands, 2009; pp. 461–463. [Google Scholar]
- Janzen, T.A.; Hunter, F.F. Honeydew sugars in wild-caught female deer flies (Diptera: Tabanidae). J. Med. Entomol. 1998, 35, 685–689. [Google Scholar] [CrossRef] [PubMed]
- Ossowski, A.; Hunter, F. Distribution patterns, body size, and sugar-feeding habits of two species of Chrysops (Diptera: Tabanidae). Can. Entomol. 2000, 132, 213–221. [Google Scholar] [CrossRef]
- Burgin, G.; Hunter, F. Evidence of honeydew feeding in black flies (Diptera: Simuliidae). Can. Entomol. 1997, 129, 859–869. [Google Scholar] [CrossRef]
- Burgin, S.G.; Hunter, F.F. Nectar versus honeydew as sources of sugar for male and female black flies (Diptera: Simuliidae). J. Med. Entomol. 1997, 34, 605–608. [Google Scholar] [CrossRef] [PubMed]
- MacVicker, J.; Moore, J.; Molyneux, D.; Maroli, M. Honeydew sugars in wild-caught Italian phlebotomine sandlies (Diptera: Culicidae) as detected by high performance liquid chromatography. Bull. Entomol. Res. 1990, 80, 339–344. [Google Scholar] [CrossRef]
- Haegar, J. The non-blood feeding habits of Aedes taeniorhynchus on Sanibel Island, Florida. Mosq. News 1955, 15, 21–26. [Google Scholar]
- Gary, R.; Foster, W. Anopheles gambiae feeding and survival on honeydew and extra-floral nectar of peridomestic plants. Med. Vet. Entomol. 2004, 18, 102–107. [Google Scholar] [CrossRef]
- Russell, C.; Hunter, F. Analysis of nectar and honeydew feeding in Aedes and Ochlerotatus mosquitoes. J. Am. Mosq. Control Assoc. 2002, 18, 86–90. [Google Scholar]
- Burkett, D.; Kline, D.; Carlson, D. Sugar meal composition of five North Central Florida mosquito species (Diptera: Culicidae) as determined by gas chromatography. J. Med. Entomol. 1999, 36, 462–467. [Google Scholar] [CrossRef]
- Foster, W.A. Mosquito sugar feeding and reproductive energetics. Annu. Rev. Entomol. 1995, 40, 443–474. [Google Scholar] [CrossRef] [PubMed]
- Stone, C.M.; Taylor, R.M.; Roitberg, B.D. Sugar deprivation reduces insemination of Anopheles gambiae (Diptera: Culicidae), despite daily recruitment of adults, and predicts decline in model populations. J. Med. Entomol. 2009, 46, 1327–1337. [Google Scholar] [CrossRef] [PubMed]
- Nayar, J.K.; Van Handel, E. The fuel for sustained mosquito flight. J. Insect Physiol. 1971, 17, 471–481. [Google Scholar] [CrossRef]
- Van Handel, E. The obese mosquito. J. Physiol. 1965, 181, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Foster, W.A.; Takken, W. Nectar-related vs. human-related volatiles: Behavioural response and choice by female and male Anopheles gambiae (Diptera: Culicidae) between emergence and first feeding. Bull. Entomol. Res. 2004, 94, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Grant, A.J.; O’Connell, R.J. Age-related changes in female mosquito carbon dioxide detection. J. Med. Entomol. 2007, 44, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Bohbot, J.D.; Durand, N.F.; Vinyard, B.T.; Dickens, J.C. Functional development of the octenol response in Aedes aegypti. Front. Physiol. 2013, 4, 39. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.E. Development of lactic acid-receptor sensitivity and host-seeking behaviour in newly emerged female Aedes aegypti mosquitoes. J. Insect Physiol. 1984, 30, 211–215. [Google Scholar] [CrossRef]
- Gu, W.; Müller, G.; Schlein, Y.; Novak, R.J.; Beier, J.C. Natural plant sugar sources of Anopheles mosquitoes strongly impact malaria transmission potential. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Stone, C.M.; Witt, A.B.R.; Walsh, G.C.; Foster, W.A.; Murphy, S.T. Would the control of invasive alien plants reduce malaria transmission? A review. Parasit. Vectors 2018, 11, 76. [Google Scholar] [CrossRef]
- Clements, A. The Biology of Mosquitoes Volume 2: Sensory Reception and Behaviour; CABI Publishing: Wallingford, UK, 1999. [Google Scholar]
- Van Rijn, P.C.J.; Kooijman, J.; Wäckers, F.L. The contribution of floral resources and honeydew to the performance of predatory hoverflies (Diptera: Syrphidae). Biol. Control 2013, 67, 32–38. [Google Scholar] [CrossRef]
- Foster, W.A. Phytochemicals as population sampling lures. J. Am. Mosq. Control Assoc. 2008, 24, 138–146. [Google Scholar] [CrossRef]
- Nyasembe, V.; Torto, B. Volatile phytochemicals as mosquito semiochemicals. Phytochem. Lett. 2014, 8, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.Y.; Roitberg, B.D.; Shani, A.; Raworth, D.A.; Lee, G.H. Olfactory response by the aphidophagous gall midge, Aphidoletes aphidimyza to honeydew from green peach aphid, Myzus persicae. Entomol. Exp. Appl. 2004, 111, 37–45. [Google Scholar] [CrossRef]
- Leroy, P.D.; Heuskin, S.; Sabri, A.; Verheggen, F.J.; Farmakidis, J.; Lognay, G.; Thonart, P.; Wathelet, J.P.; Brostaux, Y.; Haubruge, E. Honeydew volatile emission acts as a kairomonal message for the Asian lady beetle Harmonia axyridis (Coleoptera: Coccinellidae). Insect Sci. 2012, 19, 498–506. [Google Scholar] [CrossRef]
- Brown, R.L.; El-sayed, A.M.; Unelius, C.R.; Beggs, J.R.; Suckling, D.M. Invasive Vespula wasps utilize kairomones to exploit honeydew produced by sooty scale insects, Ultracoelostoma. J. Chem. Ecol. 2015, 41, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Forrest, J.; Dixon, A. Sugar, organic acid, phenolic acid and plant growth regulator content of extracts of honeydew of the aphid Myzus persicae and of its host plant, Raphanus sativus. Ann. Appl. Biol. 1974, 78, 65–73. [Google Scholar] [CrossRef]
- Pozo, M.I.; Lievens, B.; Jacquemyn, H. Impact of microorganisms on nectar chemistry, pollinator attraction and plant fitness. In Nectar: Production, Chemical Composition and Benefits to Animals and Plants; Peck, R., Ed.; Nova Publishers: New York, NY, USA, 2014; pp. 1–45. ISBN 978-1-63463-679-7. [Google Scholar]
- Stadler, B.; Müller, T. Aphid honeydew and its effect on the phyllosphere microflora of Picea abies (L) Karst. Oecologia 1996, 108, 771–776. [Google Scholar] [CrossRef]
- Leroy, P.D.; Sabri, A.; Heuskin, S.; Thonart, P.; Lognay, G.; Verheggen, F.J.; Francis, F.; Brostaux, Y.; Felton, G.W.; Haubruge, E. Microorganisms from aphid honeydew attract and enhance the efficacy of natural enemies. Nat. Commun. 2011, 2, 348. [Google Scholar] [CrossRef]
- Álvarez-Pérez, S.; Herrera, C.M.; de Vega, C. Zooming-in on floral nectar: A first exploration of nectar-associated bacteria in wild plant communities. FEMS Microbiol. Ecol. 2012, 80, 591–602. [Google Scholar] [CrossRef]
- Fridman, S.; Izhaki, I.; Gerchman, Y.; Halpern, M. Bacterial communities in floral nectar. Environ. Microbiol. Rep. 2012, 4, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Verhulst, N.O.; Beijleveld, H.; Knols, B.G.; Takken, W.; Schraa, G.; Bouwmeester, H.J.; Smallegange, R.C. Cultured skin microbiota attracts malaria mosquitoes. Malar. J. 2009, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Verhulst, N.O.; Andriessen, R.; Groenhagen, U.; Kiss, G.B.; Schulz, S.; Takken, W.; van Loon, J.J.A.; Schraa, G.; Smallegange, R.C. Differential attraction of malaria mosquitoes to volatile blends produced by human skin bacteria. PLoS ONE 2010, 5, e15829. [Google Scholar] [CrossRef] [PubMed]
- Busula, A.O.; Takken, W.; de Boer, J.G.; Mukabana, W.R.; Verhulst, N.O. Variation in host preferences of malaria mosquitoes is mediated by skin bacterial volatiles. Med. Vet. Entomol. 2017, 31, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Takken, W.; Verhulst, N.O. Chemical signaling in mosquito–host interactions: The role of human skin microbiota. Curr. Opin. Insect Sci. 2017, 20, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Ponnusamy, L.; Xu, N.; Nojima, S.; Wesson, D.M.; Schal, C.; Apperson, C.S. Identification of bacteria and bacteria-associated chemical cues that mediate oviposition site preferences by Aedes aegypti. Proc. Natl. Acad. Sci. USA 2008, 105, 9262–9267. [Google Scholar] [CrossRef]
- Müller, G.C.; Xue, R.; Schlein, Y. Differential attraction of Aedes albopictus in the field to flowers, fruits and honeydew. Acta Trop. 2011, 118, 45–49. [Google Scholar] [CrossRef]
- Pialoux, G.; Gaüzère, B.A.; Jauréguiberry, S.; Strobel, M. Chikungunya, an epidemic arbovirosis. Lancet Infect. Dis. 2007, 7, 319–327. [Google Scholar] [CrossRef]
- Hayes, E.B. Zika virus outside Africa. Emerg. Infect. Dis. 2009, 15, 2007–2010. [Google Scholar] [CrossRef]
- Jansen, C.C.; Beebe, N.W. The dengue vector Aedes aegypti: What comes next. Microbes Infect. 2010, 12, 272–279. [Google Scholar] [CrossRef]
- Monath, T.P. Yellow fever: An update. Lancet Infect. Dis. 2001, 1, 11–20. [Google Scholar] [CrossRef]
- Derstine, N.T.; Ohler, B.; Jimenez, S.I.; Landolt, P.; Gries, G. Evidence for sex pheromones and inbreeding avoidance in select North American yellowjacket species. Entomol. Exp. Appl. 2017, 164, 35–44. [Google Scholar] [CrossRef]
- Landolt, P.; Tumlinson, J.; Alborn, D. Attraction of Colorado potato beetle (Coleoptera: Chrysomelidae) to damaged and chemically induced potato plants. Environ. Entomol. 1999, 28, 973–978. [Google Scholar] [CrossRef]
- Van Den Dool, H.; Kratz, P. A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. [Google Scholar] [CrossRef]
- Adams, R. Identification of Essential Oils by Ion Trap Mass Spectroscopy, 1st ed.; Academic Press: San Diego, CA, USA, 1989. [Google Scholar]
- Boullis, A.; Blanchard, S.; Francis, F.; Verheggen, F. Elevated CO2 concentrations impact the semiochemistry of aphid honeydew without having a cascade effect on an aphid predator. Insects 2018, 9, 47. [Google Scholar] [CrossRef] [PubMed]
- Sunderland, K.D.; Vickerman, G.P. Aphid feeding by some polyphagous predators in relation to aphid density in cereal fields. J. Appl. Ecol. 1980, 17, 389–396. [Google Scholar] [CrossRef]
- Elliott, N.; Kieckhefer, R.W. Response by coccinellids to spatial variation in cereal aphid density. Popul. Ecol. 2000, 42, 81–90. [Google Scholar] [CrossRef]
- Peach, D.A.H.; Gries, R.; Huimin, Z.; Young, N.; Gries, G. Multimodal floral cues guide mosquitoes to tansy inflorescences. Sci. Rep. in review.
- Hare, J.D. Ecological role of volatiles produced by plants in response to damage by herbivorous insects. Annu. Rev. Entomol. 2011, 56, 161–180. [Google Scholar] [CrossRef]
- Aljbory, Z.; Chen, M.S. Indirect plant defense against insect herbivores: A review. Insect Sci. 2018, 25, 2–23. [Google Scholar] [CrossRef]
- Allmann, S.; Baldwin, I. Insects betray themselves in nature to predators by the rapid isomerization of green leaf volatiles. Nature 2010, 439, 1075–1078. [Google Scholar] [CrossRef] [PubMed]
- Schwartzberg, E.G.; Böröczky, K.; Tumlinson, J.H. Pea aphids, Acyrthosiphon pisum, suppress induced plant volatiles in broad bean, Vicia faba. J. Chem. Ecol. 2011, 37, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Hung, K.Y.; Michailides, T.J.; Millar, J.G.; Wayadande, A. House fly (Musca domestica L.) attraction to insect honeydew. PLoS ONE 2015, 10, e0124746. [Google Scholar] [CrossRef] [PubMed]
- Peach, D.A.H.; Department of Biological Sciences, Simon Fraser University, Burnaby, BC, Canada. Unpublished data. 2018.
- Fischer, C.Y.; Lognay, G.C.; Detrain, C.; Heil, M.; Grigorescu, A.; Sabri, A.; Thonart, P.; Haubruge, E.; Verheggen, F.J. Bacteria may enhance species association in an ant–aphid mutualistic relationship. Chemoecology 2015, 25, 223–232. [Google Scholar] [CrossRef]
- Downes, W.; Dahlem, G. Keys to the evolution of Diptera: Role of Homoptera. Environ. Entomol. 1987, 16, 847–854. [Google Scholar] [CrossRef]
- Schulz, S.; Dickschat, J.S. Bacterial volatiles: The smell of small organisms. Nat. Prod. Rep. 2007, 24, 814–842. [Google Scholar] [CrossRef]
- Pringle, E.G.; Novo, A.; Ableson, I.; Barbehenn, R.V.; Vannette, R.L. Plant-derived differences in the composition of aphid honeydew and their effects on colonies of aphid-tending ants. Ecol. Evol. 2014, 4, 4065–4079. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.K.; Shingleton, A.W. Host plant and ants influence the honeydew sugar composition of aphids. Funct. Ecol. 2001, 15, 544–550. [Google Scholar] [CrossRef]
- Stone, C.; Foster, W. Plant-sugar feeding and vectorial capacity. In Ecology of Parasite-Vector Interactions; Takken, W., Koenraadt, C.J.M., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2013; pp. 35–79. ISBN 9789086861880. [Google Scholar]
- Ebrahimi, B.; Jackson, B.T.; Guseman, J.L.; Przybylowicz, C.M.; Stone, C.M.; Foster, W.A. Alteration of plant species assemblages can decrease the transmission potential of malaria mosquitoes. J. Appl. Ecol. 2017, 55, 841–851. [Google Scholar] [CrossRef]
| Exp. | Treatment 1,2,3,4,5 | Control | Details | Design | N |
|---|---|---|---|---|---|
| Attraction of Mosquitoes to Plants Aphid-Infested, Mechanically Injured, or Paired with Non-Feeding Aphids | |||||
| 1 | M. persicae-infested V. faba | V. faba | Mean of 31 aphids per plant | Y-tubes | 21 |
| 2 | A. pisum-infested V. faba | V. faba | Mean of 103 aphids per plant | Y-tubes | 22 |
| 3 | V. faba (injured) | V. faba | Experimentally injured plant | Y-tubes | 20 |
| 4 | V. faba + A. pisum | V. faba | 100 A. pisum in Petri dish | Y-tubes | 22 |
| Attraction of Mosquitoes to Synthetic Honeydew Odorants | |||||
| 5 | CHD1 | Solvents | 2.5 × 101 µL honeydew equiv. | Delta traps | 15 |
| 6 | CHD1 | Solvents | 2.5 × 100 µL honeydew equiv. | Delta traps | 11 |
| 7 | SHD | Solvents | 2.5 × 106 µL honeydew equiv. | Delta traps | 12 |
| 8 | CHD2 | Solvents | 2.5 × 106 µL honeydew equiv. | Delta traps | 13 |
| 9 | CHD2 | Solvents | 2.5 × 105 µL honeydew equiv. | Delta traps | 10 |
| 10 | CHD2 | Solvents | 2.5 × 104 µL honeydew equiv. | Delta traps | 10 |
| 11 | CHD2 | Solvents | 2.5 × 103 µL honeydew equiv. | Delta traps | 15 |
| 12 | CHD2 | Solvents | 2.5 × 101 µL honeydew equiv. | Delta traps | 14 |
| 13 | CHD2 | Solvents | 2.5 × 100 µL honeydew equiv. | Delta traps | 15 |
| Attraction of mosquitoes to odorants from honeydew-dwelling microbes | |||||
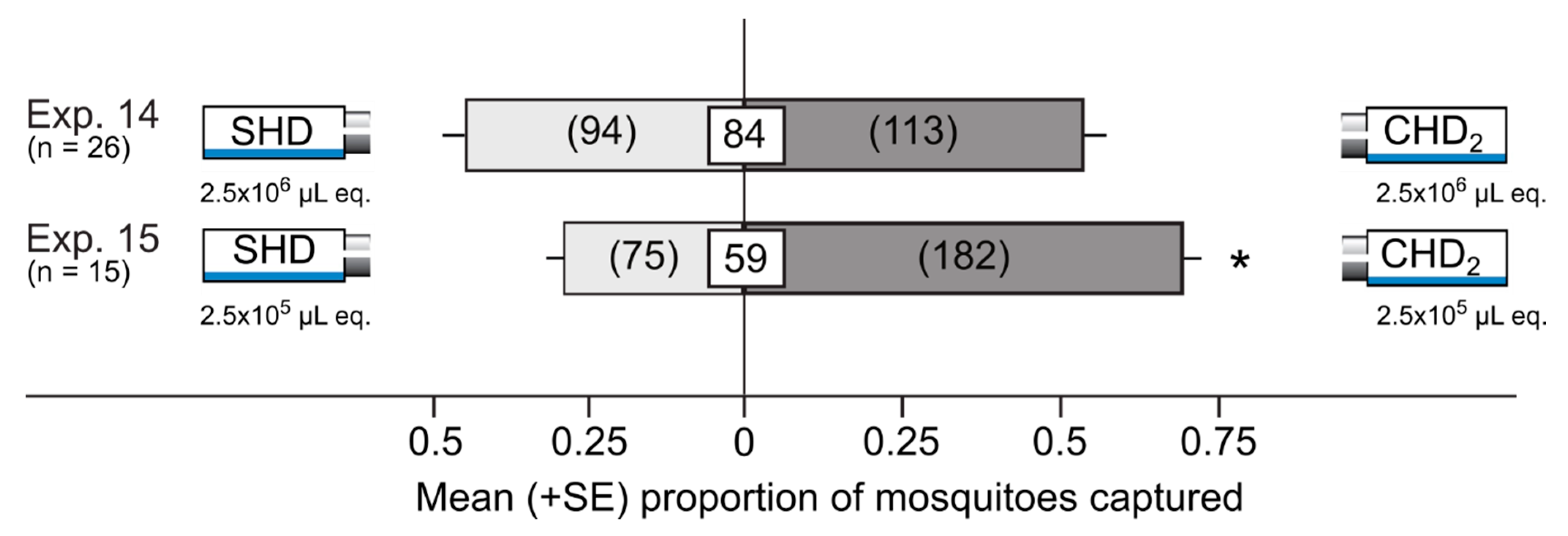
| 14 | CHD2 | SHD | 2.5 × 106 µL honeydew equiv. | Delta traps | 26 |
| 15 | CHD2 | SHD | 2.5×105 µL honeydew equiv. | Delta traps | 15 |
| Odorants | Purity (%) | CHD1 (%) | CHD2 (%) | SHD (%) |
|---|---|---|---|---|
| Propanone 1 | 99.8 | - | 9.25 | 24.62 |
| 2,3-Butanedione 2 | 86 | 7.70 | 2.31 | 40.54 |
| 2,3-Butanediol 1 | 98 | 3.49 | - | - |
| 3-Methylbutanal 1 | 97 | - | 14.01 | - |
| 2-Methylbutanal 1 | >99 | - | 12.92 | - |
| 3-Hydroxybutanone 1 | 98 | 46.38 | 0.78 | 4.77 |
| 3-Methyl-3-buten-1-ol 1 | 97 | - | 0.89 | 5.64 |
| 3-Methyl-1-butanol 3 | 98.5 | 36.82 | 12.32 | - |
| 2-Methyl-2-buten-1-ol 5 | 83 | - | 14.41 | - |
| 3-Methyl-2-butenal 6 | 88 | - | 10.73 | - |
| Butanoic acid 1 | 99 | - | 6.24 | 24.43 |
| 3-Methylbutanoic acid 1 | 99 | 3.07 | 4.56 | - |
| 2-Methylbutanoic acid 1 | 98 | 0.63 | 6.73 | - |
| 2,5-Dimethylpyrazine 1 | 99 | - | 0.31 | - |
| Limonene 1 | 90 | - | 2.81 | - |
| Benzeneethanol 1 | 99 | - | 1.73 | - |
| 2-Ethylhexanol 1 | 99 | 1.57 | - | - |
| 2-Phenylethyl alcohol 4 | 98 | 0.35 | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peach, D.A.H.; Gries, R.; Young, N.; Lakes, R.; Galloway, E.; Alamsetti, S.K.; Ko, E.; Ly, A.; Gries, G. Attraction of Female Aedes aegypti (L.) to Aphid Honeydew. Insects 2019, 10, 43. https://doi.org/10.3390/insects10020043
Peach DAH, Gries R, Young N, Lakes R, Galloway E, Alamsetti SK, Ko E, Ly A, Gries G. Attraction of Female Aedes aegypti (L.) to Aphid Honeydew. Insects. 2019; 10(2):43. https://doi.org/10.3390/insects10020043
Chicago/Turabian StylePeach, Daniel A. H., Regine Gries, Nathan Young, Robyn Lakes, Erin Galloway, Santosh Kumar Alamsetti, Elton Ko, Amy Ly, and Gerhard Gries. 2019. "Attraction of Female Aedes aegypti (L.) to Aphid Honeydew" Insects 10, no. 2: 43. https://doi.org/10.3390/insects10020043
APA StylePeach, D. A. H., Gries, R., Young, N., Lakes, R., Galloway, E., Alamsetti, S. K., Ko, E., Ly, A., & Gries, G. (2019). Attraction of Female Aedes aegypti (L.) to Aphid Honeydew. Insects, 10(2), 43. https://doi.org/10.3390/insects10020043





