Recruiting on the Spot: A Biodegradable Formulation for Lacewings to Trigger Biological Control of Aphids
Abstract
1. Introduction
2. Materials and Methods
2.1. Volatile Releasing Formulations
2.2. Measurement of Volatile Release
2.3. Attraction Longevity
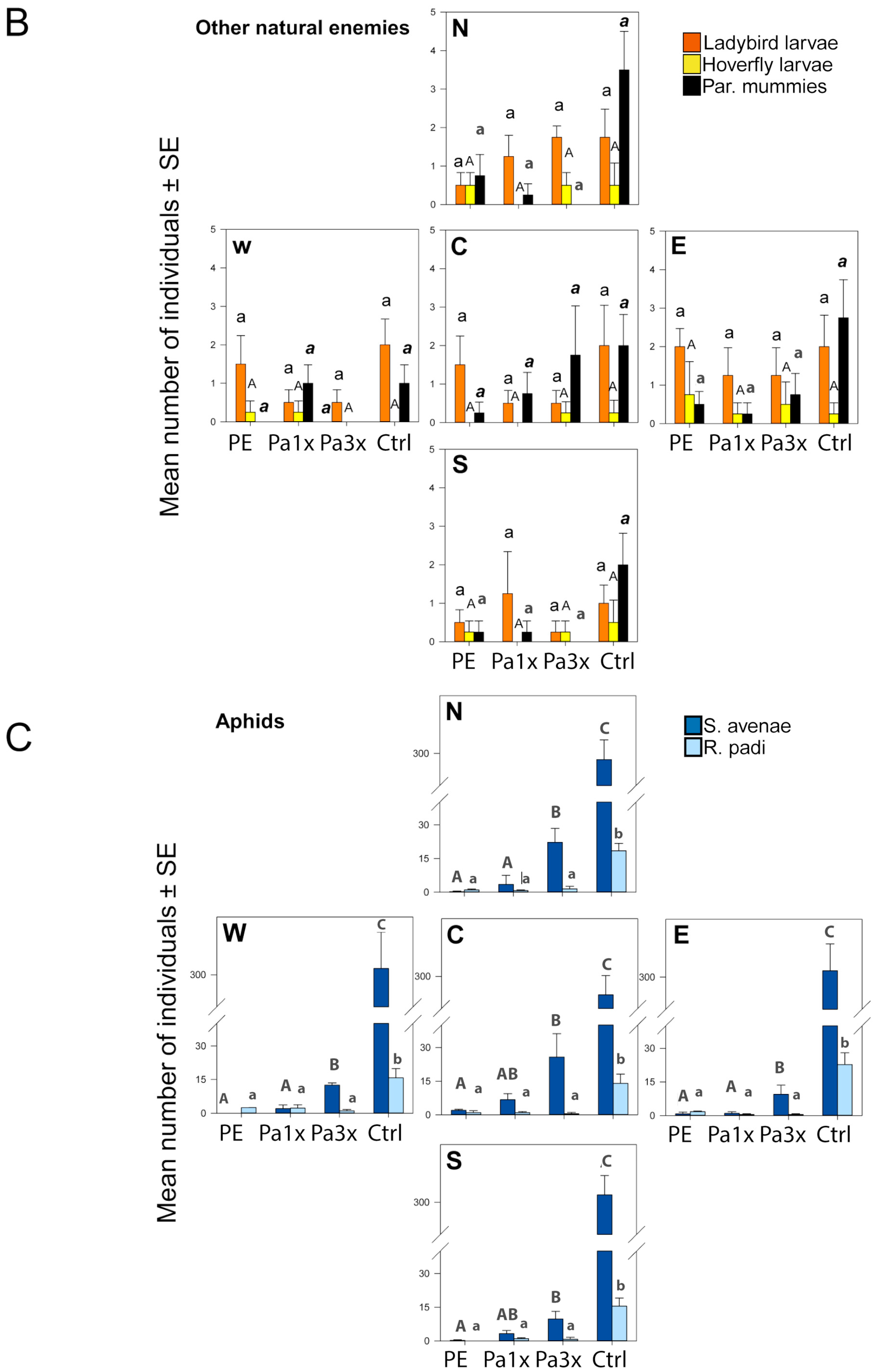
2.4. Measurement of Biological Control
2.5. Data Analysis
3. Results
3.1. Measurement of Volatile Release
3.2. Attraction Longevity
3.3. Measurement of Biological Control
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ripple, W.J.; Wolf, C.; Newsome, T.M.; Galetti, M.; Alamgir, M.; Crist, E.; Mahmoud, M.I.; Laurance, W.F. World Scientists’ Warning to Humanity: A Second Notice. Bioscience 2017, 67, 1026–1028. [Google Scholar] [CrossRef]
- Geiger, F.; Bengtsson, J.; Berendse, F.; Weisser, W.W.; Emmerson, M.; Morales, M.B.; Ceryngier, P.; Liira, J.; Tscharntke, T.; Winqvist, C.; et al. Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic Appl. Ecol. 2010, 11, 97–105. [Google Scholar] [CrossRef]
- Tittonell, P. Ecological intensification of agriculture-sustainable by nature. Curr. Opin. Environ. Sustain. 2014, 8, 53–61. [Google Scholar] [CrossRef]
- Letourneau, D.K.; Armbrecht, I.; Rivera, B.S.; Montoya, J.; Carmona, E.J.; Daza, M.C.; Escobar, S.; Galindo, V.; Gutiérrez, C.; López, S.D.; et al. Does plant diversity benefit agroecosystems? A synthetic review. Ecol. Appl. 2015, 21, 9–21. [Google Scholar] [CrossRef]
- Scarano, F.R. Ecosystem-based adaptation to climate change: Concept, scalability and a role for conservation science. Perspect. Ecol. Conserv. 2017, 15, 65–73. [Google Scholar] [CrossRef]
- Batáry, P.; Dicks, L.V.; Kleijn, D.; Sutherland, W.J. The role of agri-environment schemes in conservation and environmental management. Conserv. Biol. 2015, 29, 1006–1016. [Google Scholar] [CrossRef] [PubMed]
- Rowen, E.; Kaplan, I.; Rowen, E. Eco-evolutionary factors drive induced plant volatiles: A meta-analysis. New Phytol. 2016, 210, 284–294. [Google Scholar] [CrossRef]
- Ninkovic, V.; Markovic, D.; Dahlin, I. Decoding neighbour volatiles in preparation for future competition and implications for tritrophic interactions. Perspect. Plant Ecol. Evol. Syst. 2016, 23, 11–17. [Google Scholar] [CrossRef]
- Heil, M.; Ton, J. Long-distance signalling in plant defence. Trends Plant Sci. 2008, 13, 264–272. [Google Scholar] [CrossRef]
- Braasch, J.; Kaplan, I. Over what distance are plant volatiles bioactive? Estimating the spatial dimensions of attraction in an arthropod assemblage. Entomol. Exp. Appl. 2012, 145, 115–123. [Google Scholar] [CrossRef]
- Turlings, T.C.J.; Erb, M. Tritrophic interactions mediated by herbivore-induced plant volatiles: mechanisms, ecological relevance, and application potential. Annu. Rev. Entomol. 2018, 63, 433–452. [Google Scholar] [CrossRef] [PubMed]
- Peñaflor, M.F.G.V.; Bento, J.M.S. Herbivore-Induced Plant Volatiles to Enhance Biological Control in Agriculture. Neotrop. Entomol. 2013, 42, 331–343. [Google Scholar] [CrossRef]
- Silva, D.B.; Weldegergis, B.T.; Van Loon, J.J.A.; Bueno, V.H.P. Qualitative and Quantitative Differences in Herbivore-Induced Plant Volatile Blends from Tomato Plants Infested by Either Tuta absoluta or Bemisia tabaci. J. Chem. Ecol. 2017, 43, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Landolt, P.J.; Alfaro, J.F. Trapping Lacanobia subjuncta, Xestia c-nigrum, and Mamestra configurata (Lepidoptera: Noctuidae) with acetic acid and 3-Methyl-1-butanol in controlled release dispensers. Environ. Entomol. 2000, 30, 656–662. [Google Scholar] [CrossRef]
- Knight, A.L.; Hilton, R.; Basoalto, E.; Stelinski, L.L. Use of glacial acetic acid to enhance bisexual monitoring of tortricid pests with kairomone lures in pome fruits. Environ. Entomol. 2014, 43, 1628–1640. [Google Scholar] [CrossRef]
- Jones, V.P.; Horton, D.R.; Mills, N.J.; Unruh, T.R.; Baker, C.C.; Melton, T.D.; Milickzy, E.; Steffan, S.A.; Shearer, P.W.; Amarasekare, K.G. Evaluating plant volatiles for monitoring natural enemies in apple, pear and walnut orchards. Biol. Control 2016, 102, 53–65. [Google Scholar] [CrossRef]
- El-Sayed, A.M.; Knight, A.L.; Byers, J.A.; Judd, G.J.R.; Suckling, D.M. Caterpillar-induced plant volatiles attract conspecific adults in nature. Sci. Rep. 2016, 6, 1–14. [Google Scholar] [CrossRef]
- Gadino, A.N.; Walton, V.M.; Lee, J.C. Evaluation of methyl salicylate lures on populations of Typhlodromus pyri (Acari: Phytoseiidae) and other natural enemies in western Oregon vineyards. Biol. Control 2012, 63, 48–55. [Google Scholar] [CrossRef]
- Aqueel, M.A.; Collins, C.M.; Raza, A.B.; Ahmad, S.; Tariq, M.; Leather, S.R. Effect of plant nutrition on aphid size, prey consumption, and life history characteristics of green lacewing. Insect Sci. 2014, 21, 74–82. [Google Scholar] [CrossRef]
- Tóth, M.; Szentkirályi, F.; Vuts, J.; Letardi, A.; Tabilio, M.R.; Jaastad, G.; Knudsen, G.K. Optimization of a phenylacetaldehyde-based attractant for common green lacewings (Chrysoperla carnea s.l.). J. Chem. Ecol. 2009, 35, 449–458. [Google Scholar] [CrossRef]
- Pickett, J.A.; Khan, Z.R. Plant volatile-mediated signalling and its application in agriculture: Successes and challenges. New Phytol. 2016, 212, 856–870. [Google Scholar] [CrossRef] [PubMed]
- Koczor, S.; Knudsen, G.K.; Hatleli, L.; Szentkirályi, F.; Tóth, M. Manipulation of oviposition and overwintering site choice of common green lacewings with synthetic lure (Neuroptera: Chrysopidae). J. Appl. Entomol. 2015, 139, 201–206. [Google Scholar] [CrossRef]
- Teixeira, L.A.F.; Mason, K.; Mafra-Neto, A.; Isaacs, R. Mechanically-applied wax matrix (SPLAT-GBM) for mating disruption of grape berry moth (Lepidoptera: Tortricidae). Crop Prot. 2010, 29, 1514–1520. [Google Scholar] [CrossRef]
- Tasin, M.; Bäckman, A.C.; Bengtsson, M.; Varela, N.; Ioriatti, C.; Witzgall, P. Wind tunnel attraction of grapevine moth females, Lobesia botrana, to natural and artificial grape odour. Chemoecology 2006, 16, 87–92. [Google Scholar] [CrossRef]
- R Core Team R. A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Bates, D.; Machler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Lenth, R.V. Least-Squares Means: The R Package lsmeans. J. Stat. Softw. 2016, 69. [Google Scholar] [CrossRef]
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression, 2nd ed.; Sage: Thousand Oaks, CA, USA, 2011. [Google Scholar]
- Koczor, S.; Szentkirályi, F.; Birkett, M.A.; Pickett, J.A.; Voigt, E.; Tóth, M. Attraction of Chrysoperla carnea complex and Chrysopa spp. lacewings (Neuroptera: Chrysopidae) to aphid sex pheromone components and a synthetic blend of floral compounds in Hungary. Pest Manag. Sci. 2010, 66, 1374–1379. [Google Scholar] [CrossRef]
- Schorkopf, D.L.P.; Spanoudis, C.G.; Mboera, L.E.G.; Mafra-Neto, A.; Ignell, R.; Dekker, T. Combining attractants and larvicides in biodegradable matrices for sustainable mosquito vector control. PLoS Negl. Trop. Dis. 2016, 10, e0005043. [Google Scholar] [CrossRef]
- Pettersson, J.; Smart, L.E. Winter host component reduces coloniza- tion by bird-cherry-oat aphid, Rhopalosiphum padi (L.) (Homop-tera, Aphididae), and other aphids in cereal fields. J. Chem. Ecol. 1994, 20, 2565–2574. [Google Scholar] [CrossRef]
- Bernasconi, M.L.; Turlings, T.C.J.; Ambrosetti, L.; Bassetti, P.; Dorn, S. Herbivore-induced emissions of maize volatiles repel the corn leaf aphid, Rhopalosiphum maidis. Entomol. Exp. Appl. 1998, 87, 133–142. [Google Scholar] [CrossRef]
- Snoeren, T.A.L.; Mumm, R.; Poelman, E.H.; Yang, Y.; Pichersky, E.; Dicke, M. The herbivore-induced plant volatile methyl salicylate negatively affects attraction of the parasitoid Diadegma semiclausum. J. Chem. Ecol. 2010, 36, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Ruzicka, Z. Further evidence of oviposition-deterring allomone in chrysopids (Neuroptera: Chrysopidae ) Larvae of a eurytopic Nearctic species Chrysopa oculata Say (Neuroptera: Chrysopi-dae) mark substrates they walk over with an oviposition-deterring pheromon. Eur. J. Entomol. 1998, 95, 35–39. [Google Scholar]
- Jones, V.P.; Steffan, S.A.; Wiman, N.G.; Horton, D.R.; Miliczky, E.; Zhang, Q.H.; Baker, C.C. Evaluation of herbivore-induced plant volatiles for monitoring green lacewings in Washington apple orchards. Biol. Control 2011, 56, 98–105. [Google Scholar] [CrossRef]
- Seagraves, M.P. Lady beetle oviposition behavior in response to the trophic environment. Biol. Control 2009, 51, 313–322. [Google Scholar] [CrossRef]
- Yazdani, M.; Baker, G. A plant volatile-based attractant formulation is not attractive to Diadegma semiclausum (Hymenoptera: Ichneumonidae). Austral Entomol. 2018, 57, 359–364. [Google Scholar] [CrossRef]
- Klug, T.; Meyh, R. Intraguild predation on the aphid parasitoid Lysiphlebus fabarum (Marshall) (Hymenoptera: Aphidiidae): Mortality risks and behavioral decisions made under the threats of predation. Biol. Control 2002, 25, 239–248. [Google Scholar]
- Porcel, M.; Andersson, G.K.S.; Pålsson, J.; Tasin, M. Organic management in apple orchards: Higher impacts on biological control than on pollination. J. Appl. Ecol. 2018, 55, 2779–2789. [Google Scholar] [CrossRef]
- Orre, G.U.S.; Wratten, S.D.; Jonsson, M.; Hale, R.J. Effects of an herbivore-induced plant volatile on arthropods from three trophic levels in brassicas. Biol. Control 2010, 53, 62–67. [Google Scholar] [CrossRef]
- Simpson, M.; Gurr, G.M.; Simmons, A.T.; Wratten, S.D.; James, D.G.; Leeson, G.; Nicol, H.I.; Orre-Gordon, G.U.S. Attract and reward: Combining chemical ecology and habitat manipulation to enhance biological control in field crops. J. Appl. Ecol. 2011, 48, 580–590. [Google Scholar] [CrossRef]
- Hatt, S.; Boeraeve, F.; Artru, S.; Dufrêne, M.; Francis, F. Spatial diversification of agroecosystems to enhance biological control and other regulating services: An agroecological perspective. Sci. Total Environ. 2018, 621, 600–611. [Google Scholar] [CrossRef]
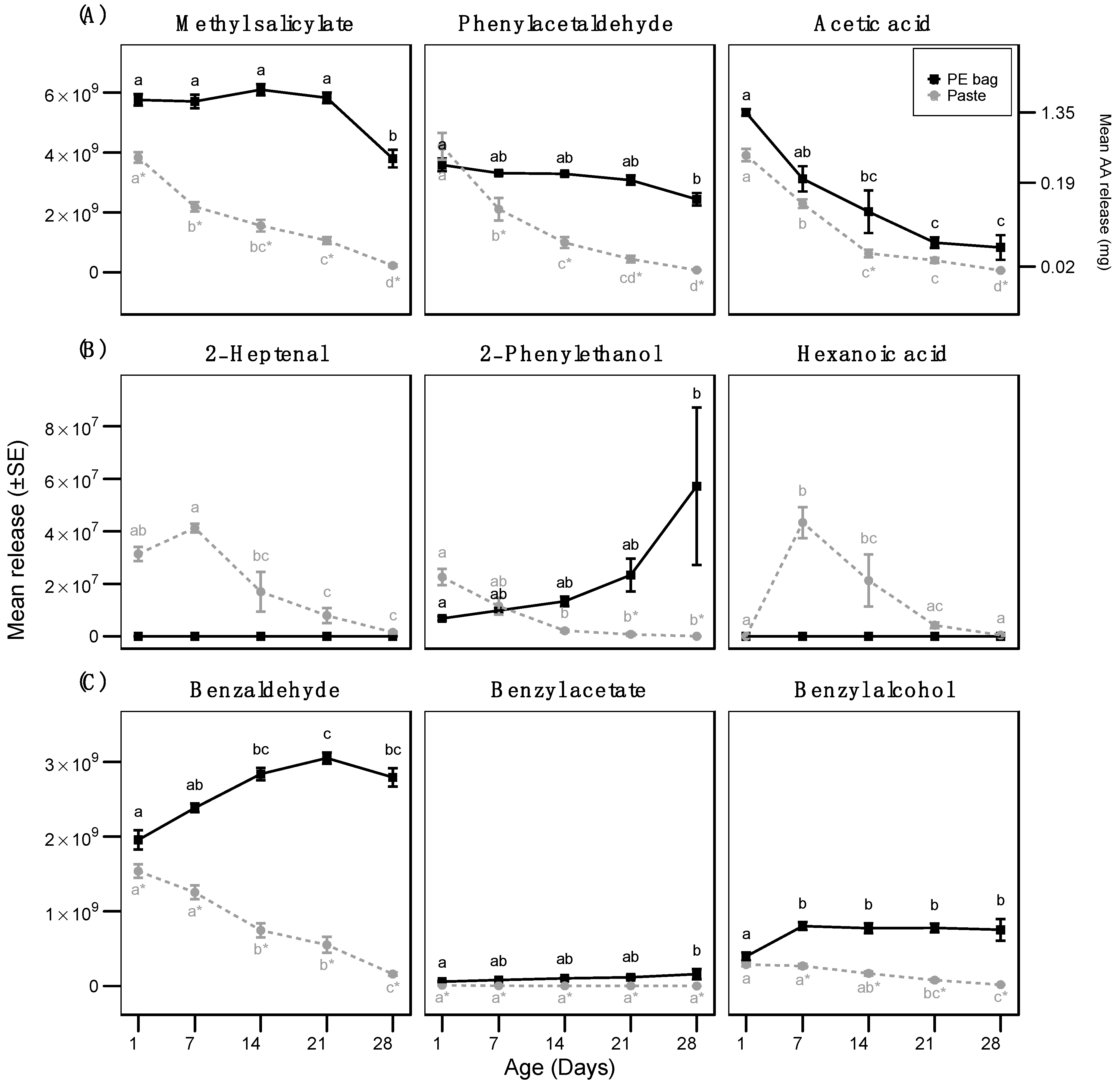
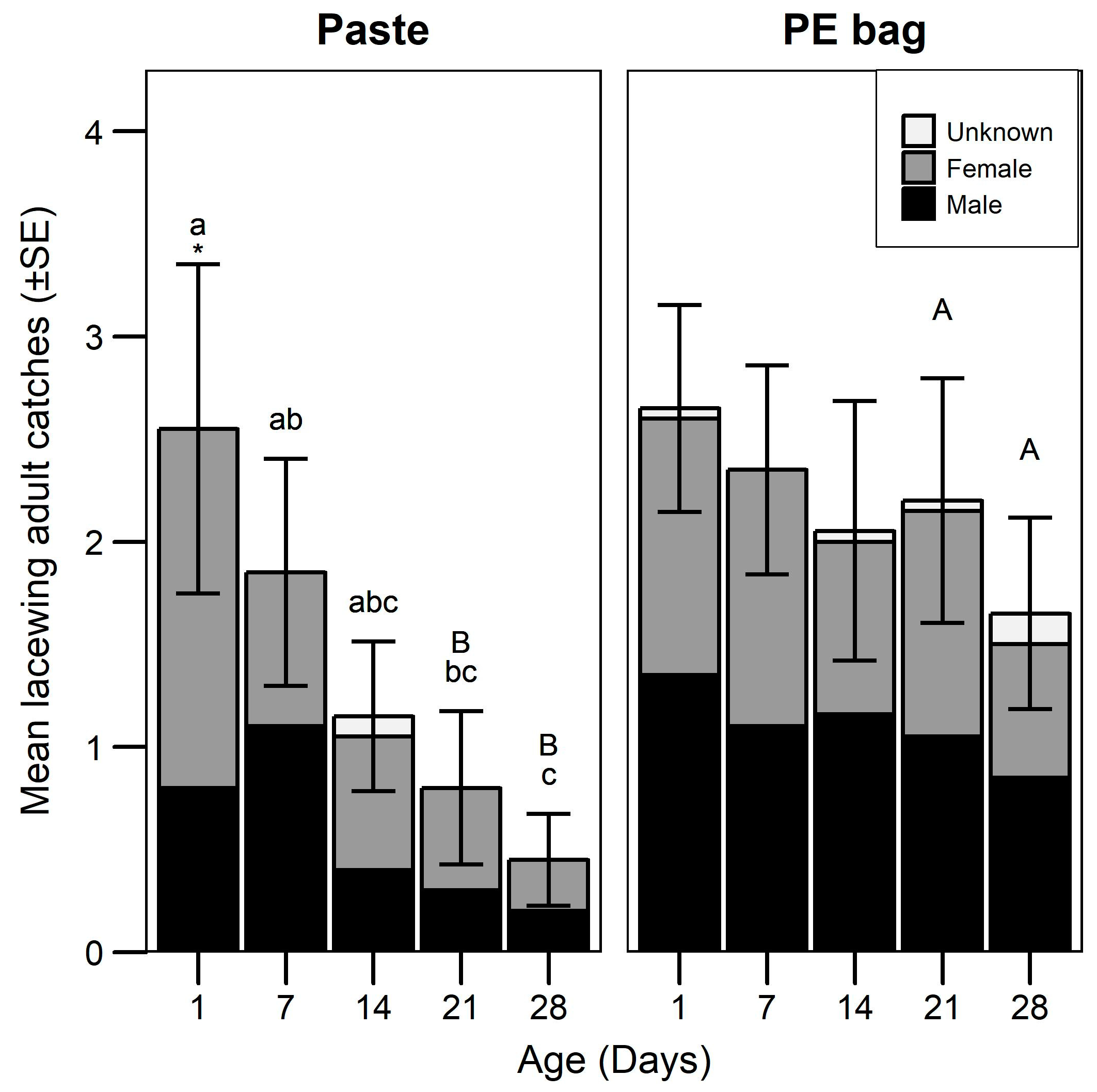
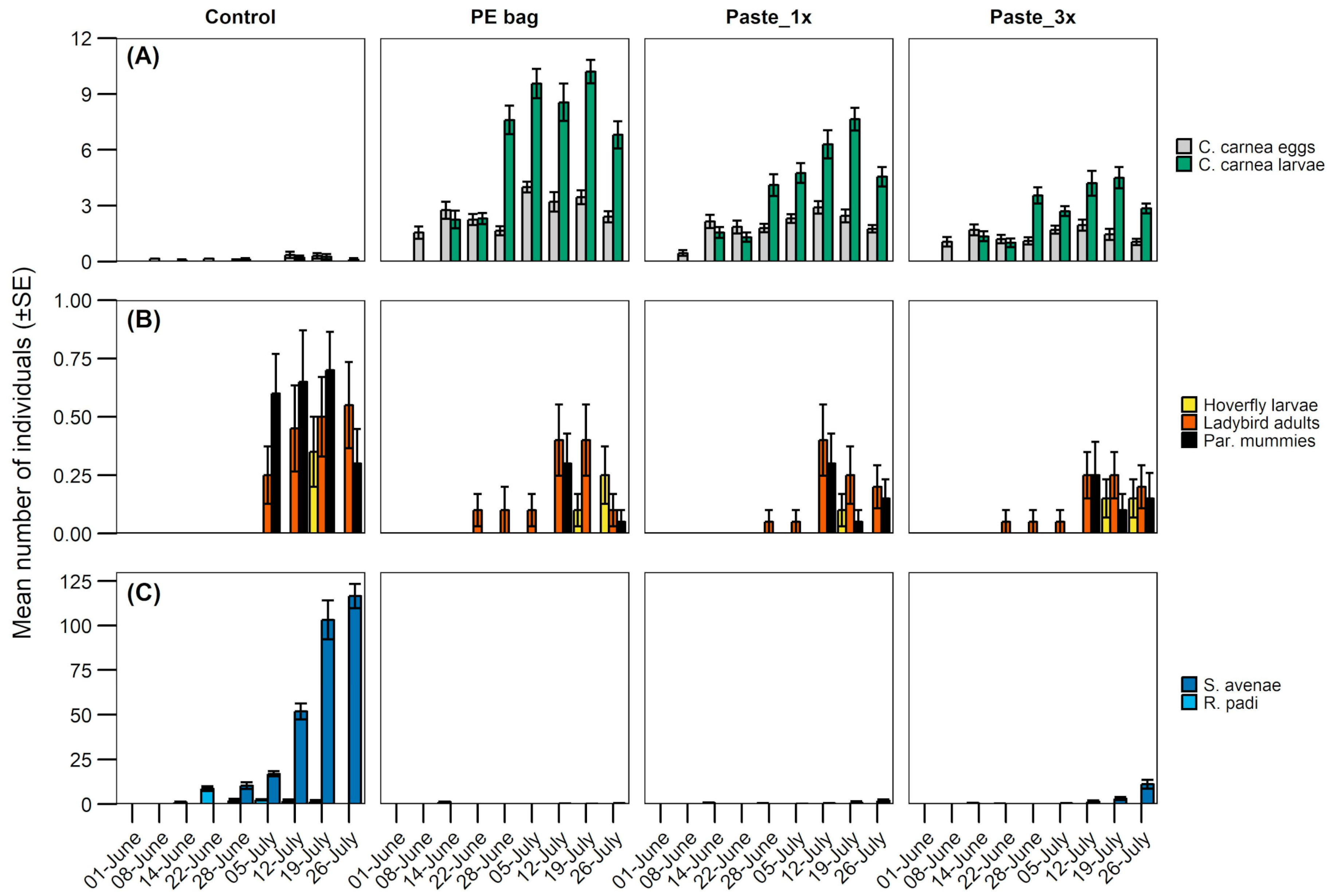
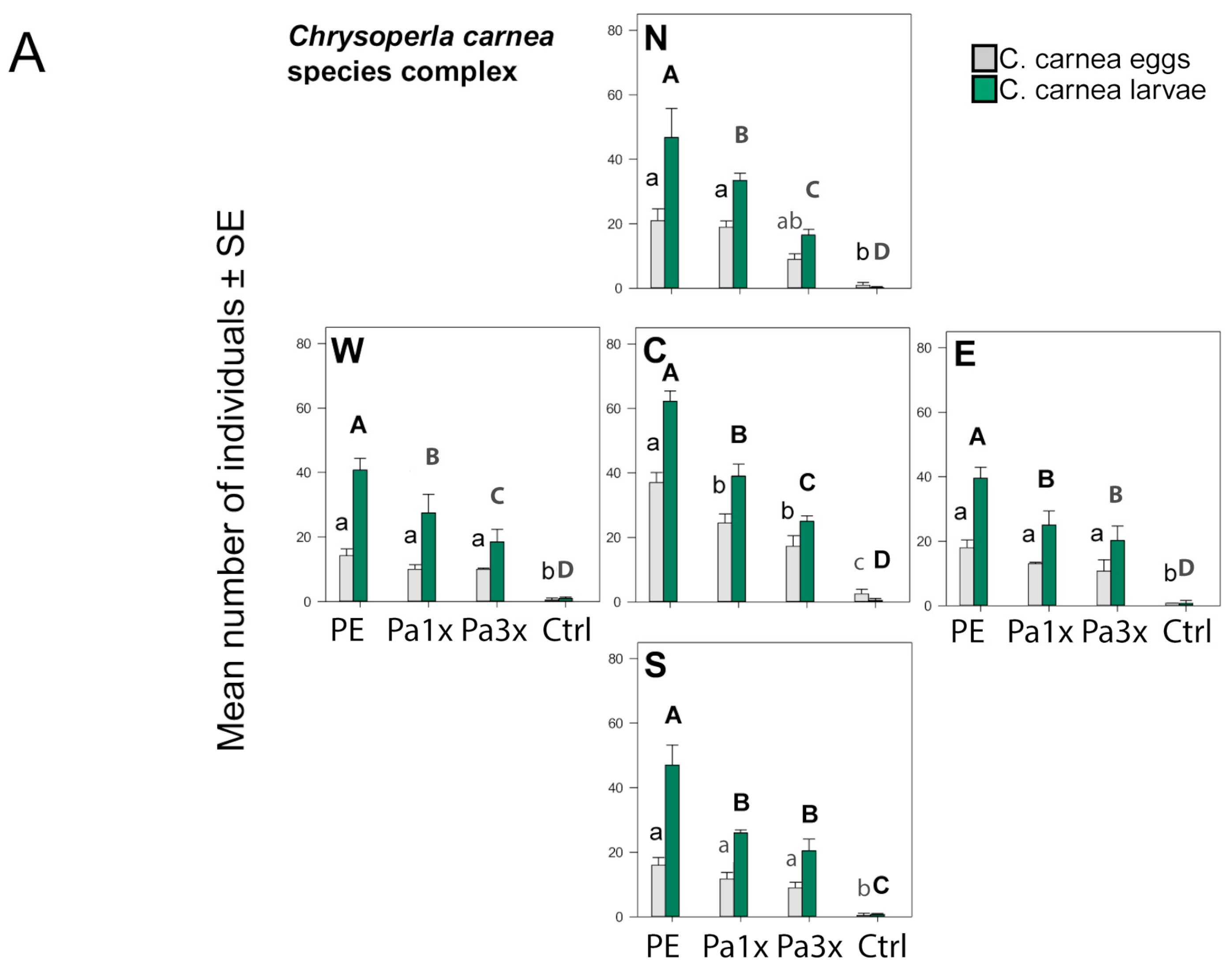
| Experiment | Model | Statistical Test (Distribution) | Fixed Factor/s | χ2./F | df. | p-Value |
|---|---|---|---|---|---|---|
| Apple (volatile release) | Methyl salicylate | LRM (Gaussian) | Formulation | 967.2 | 1 | <0.001 |
| Age | 59.9 | 4 | <0.001 | |||
| Formulation:Age | 18.1 | 4 | <0.001 | |||
| Phenylacetaldehyde | LRM (Gaussian) | Formulation | 120.6 | 1 | <0.001 | |
| Age | 40.0 | 4 | <0.001 | |||
| Formulation:Age | 17.6 | 4 | <0.001 | |||
| Acetic acid | GLS (Gaussian) | Formulation | 24.8 | 1 | <0.001 | |
| Age | 678.9 | 4 | <0.001 | |||
| Formulation:Age | 2.2 | 4 | 0.7 | |||
| 2-heptenal | LRM (Gaussian) | Age | 17.9 | 4 | <0.001 | |
| 2-phenyletahnol | LRM (Gaussian) | Formulation | 16.8 | 1 | <0.001 | |
| Age | 0.6 | 4 | 0.641 | |||
| Formulation:Age | 8.1 | 4 | <0.001 | |||
| Benzaldehyde | LRM (Gaussian) | Formulation | 820.7 | 1 | <0.001 | |
| Age | 17.3 | 4 | <0.001 | |||
| Formulation:Age | 60.4 | 4 | <0.001 | |||
| Benzyl acetate | LRM (Gaussian) | Formulation | 226.4 | 1 | <0.001 | |
| Age | 0.3 | 4 | 0.906 | |||
| Formulation:Age | 4.3 | 4 | <0.001 | |||
| Benzyl alcohol | LRM (Gaussian) | Formulation | 316.6 | 1 | <0.001 | |
| Age | 7.9 | 4 | <0.001 | |||
| Formulation:Age | 18.0 | 4 | <0.001 | |||
| Hexanoic acid | LRM (Gaussian) | Age | 18.2 | 4 | <0.001 | |
| Apple (trap catches) | Trap position | GLMM (Negative binomial) | Position | 2.4 | 5 | 0.795 |
| Formulation | 0.3 | 1 | 0.602 | |||
| Positoin:Formulation | 2.5 | 5 | 0.777 | |||
| Age vs. Blank (PE bag) | GLMM (Negative binomial) | Age | 23.2 | 5 | <0.001 | |
| Age vs. Blank (Paste) | GLMM (Negative binomial) | Age | 30.6 | 5 | <0.001 | |
| Lacewing catches | GLMM (Negative binomial) | Formulation | 12.2 | 1 | <0.001 | |
| Age | 20.0 | 4 | <0.001 | |||
| Formulation:Age | 8.3 | 4 | 0.082 | |||
| Sex ratio between formulations | GLMM (Binomial) | Formulation | 2.5 | 1 | 0.109 | |
| Age | 2.5 | 4 | 0.645 | |||
| Formulation:Age | 6.2 | 4 | 0.187 | |||
| Sex ratio age (Paste) | GLMM (Binomial) | Age | 7.1 | 4 | 0.133 | |
| Wasp (PE bag vs. Blank) | Fisher’s exact test | Treatment | 9.2 | - | 0.007 | |
| Wasp (Paste vs. Blank) | Fisher’s exact test | Treatment | 10.9 | - | 0.003 | |
| Wasp (PE bag vs. Paste) | Fisher’s exact test | Treatment | 1.2 | - | 0.377 | |
| Barley | Lacewing eggs | (GLMM) Poisson | Treatment | 137.4 | 3 | <0.001 |
| Sector | 574.4 | 4 | <0.001 | |||
| Date | 96.5 | 1 | <0.001 | |||
| Treatment:Sector | 14.3 | 12 | 0.279 | |||
| Lacewing larvae | GLMM (Negative binomial) | Treatment | 408.9 | 3 | <0.001 | |
| Sector | 44.1 | 4 | <0.001 | |||
| Date | 759.7 | 1 | <0.001 | |||
| Treatment:Sector | 11.4 | 12 | 0.499 | |||
| Hoverfly larvae | (GLMM) Poisson | Treatment | 2.1 | 3 | 0.989 | |
| Sector | 2.2 | 4 | 0.994 | |||
| Date | 19.0 | 1 | <0.001 | |||
| Treatment:Sector | 1.6 | 12 | 0.999 | |||
| Ladybird | (GLMM) Poisson | Treatment | 5.6 | 3 | 0.133 | |
| Sector | 5.6 | 4 | 0.429 | |||
| Date | 67.7 | 1 | <0.001 | |||
| Treatment:Sector | 12.0 | 12 | 0.444 | |||
| Parasitoid mummies | (GLMM) Poisson | Treatment | 30.1 | 3 | <0.001 | |
| Sector | 7.7 | 4 | 0.171 | |||
| Date | 50.4 | 1 | <0.001 | |||
| Treatment:Sector | 9.1 | 12 | 0.697 | |||
| S. avenae | GLMM (Negative binomial) | Treatment | 628.6 | 3 | <0.001 | |
| Sector | 3.8 | 4 | 0.441 | |||
| Date | 455.8 | 1 | <0.001 | |||
| Treatment:Sector | 17.5 | 12 | 0.130 | |||
| R. padi | GLMM (Negative binomial) | Treatment | 120.0 | 3 | <0.001 | |
| Sector | 2.6 | 4 | 0.631 | |||
| Date | 13.0 | 1 | <0.001 | |||
| Treatment:Sector | 6.0 | 12 | 0.914 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pålsson, J.; Thöming, G.; Silva, R.; Porcel, M.; Dekker, T.; Tasin, M. Recruiting on the Spot: A Biodegradable Formulation for Lacewings to Trigger Biological Control of Aphids. Insects 2019, 10, 6. https://doi.org/10.3390/insects10010006
Pålsson J, Thöming G, Silva R, Porcel M, Dekker T, Tasin M. Recruiting on the Spot: A Biodegradable Formulation for Lacewings to Trigger Biological Control of Aphids. Insects. 2019; 10(1):6. https://doi.org/10.3390/insects10010006
Chicago/Turabian StylePålsson, Joakim, Gunda Thöming, Rodrigo Silva, Mario Porcel, Teun Dekker, and Marco Tasin. 2019. "Recruiting on the Spot: A Biodegradable Formulation for Lacewings to Trigger Biological Control of Aphids" Insects 10, no. 1: 6. https://doi.org/10.3390/insects10010006
APA StylePålsson, J., Thöming, G., Silva, R., Porcel, M., Dekker, T., & Tasin, M. (2019). Recruiting on the Spot: A Biodegradable Formulation for Lacewings to Trigger Biological Control of Aphids. Insects, 10(1), 6. https://doi.org/10.3390/insects10010006







