In Vitro Wear Testing of a CoCr-UHMWPE Finger Prosthesis with Hydroxyapatite Coated CoCr Stems
Abstract
:1. Introduction

2. Methods

3. Results
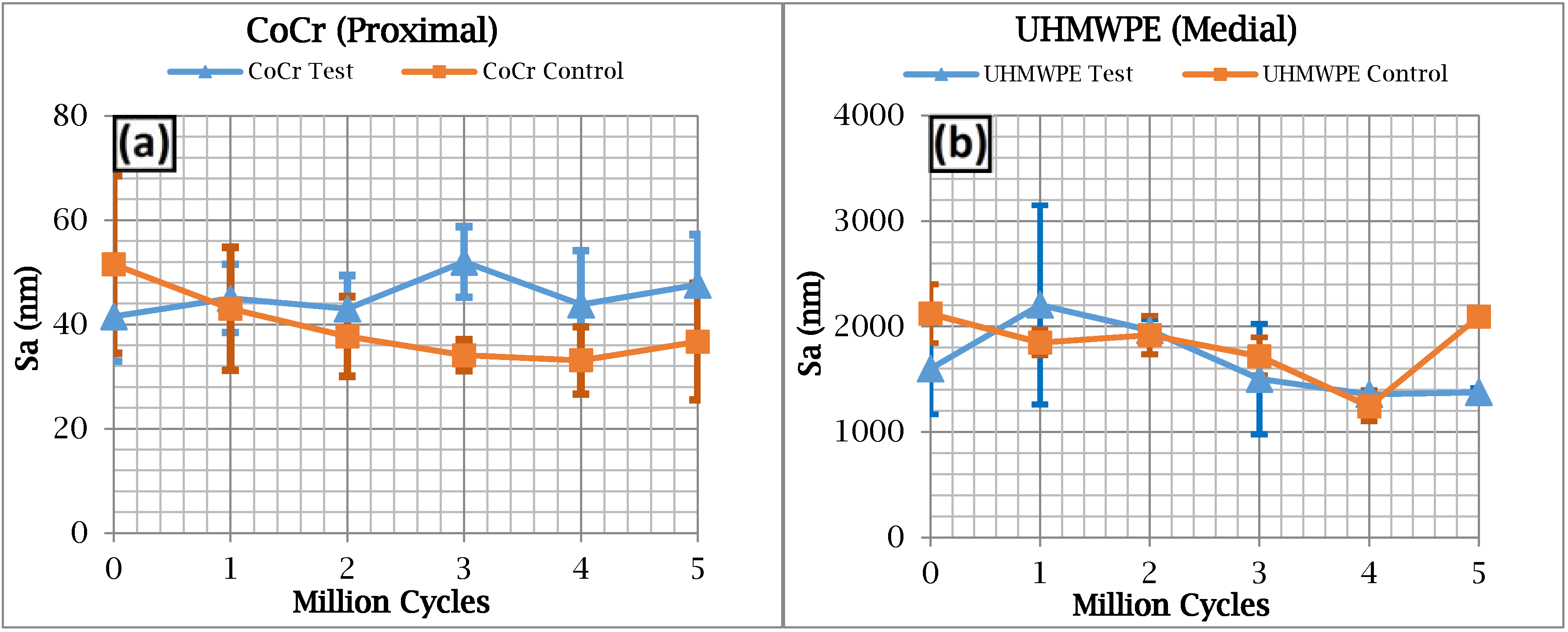
3.1. Surface Analysis


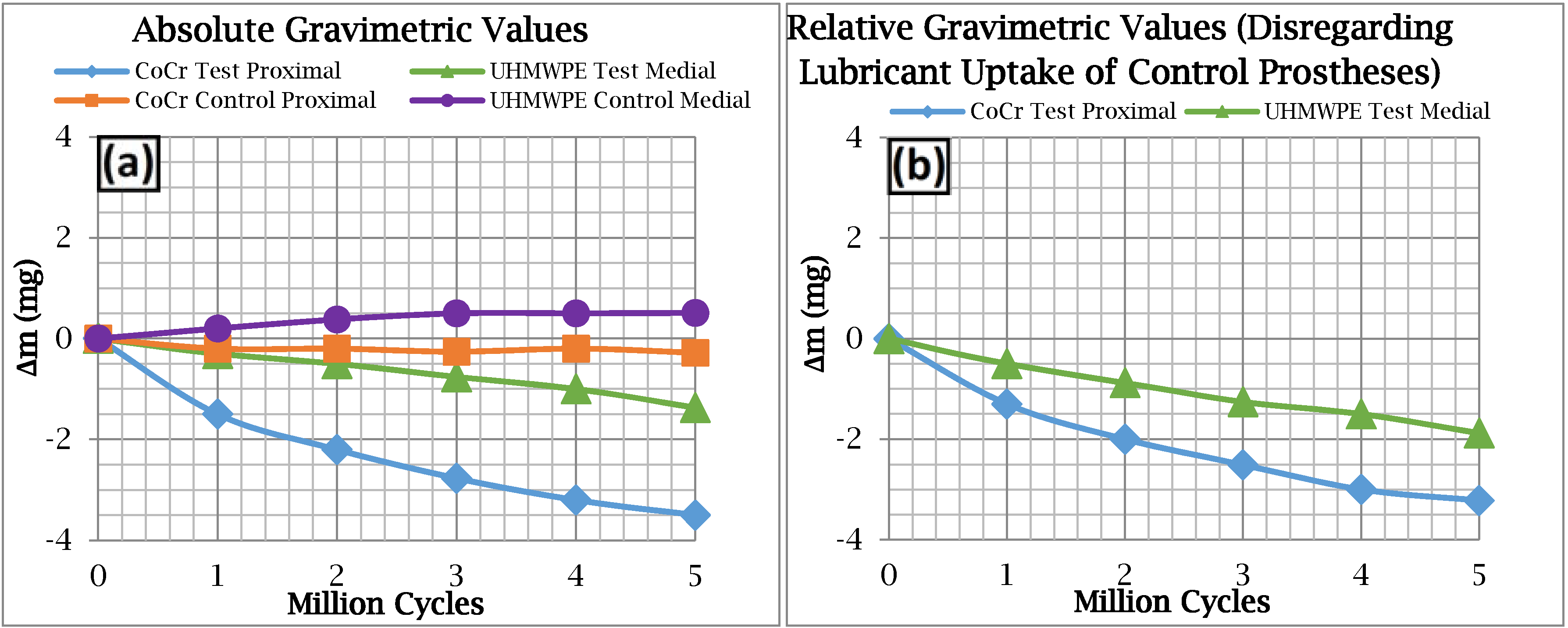
3.2. Gravimetric Analysis
3.3. Micrographic Analysis of the Stems


4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lawrence, J.S.; Bremner, J.M.; Bier, F. Osteo-arthrosis. Prevalence in the population and relationship between symptoms and x-ray changes. Ann. Rheum. Dis. 1966, 25, 1–24. [Google Scholar] [PubMed]
- Tsai, C.-L.; Liu, T.-K. Osteoarthritis in women: Its relationship to estrogen and current trends. Life Sci. 1992, 50, 1739–1744. [Google Scholar] [CrossRef]
- Linscheid, R.L. Implant arthroplasty of the hand: Retrospective and prospective considerations. J. Hand Surg. Am. 2000, 25, 796–816. [Google Scholar] [PubMed]
- Ashworth, C.R.; Hansraj, K.K.; Todd, A.O.; Dukhram, K.M.; Ebramzadeh, E.; Boucree, J.B.; Hansraj, M.S. Swanson proximal interphalangeal joint arthroplasty in patients with rheumatoid arthritis. Clin. Orthop. Relat. Res. 1997, 34–37. [Google Scholar] [CrossRef]
- Bales, J.G.; Wall, L.B.; Stern, P.J. Long-term results of swanson silicone arthroplasty for proximal interphalangeal joint osteoarthritis. J. Hand Surg. Am. 2014, 39, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Unsworth, A. The design of a finger wear simulator and preliminary results. Proc. Inst. Mech. Eng. H. 2000, 214, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Unsworth, A. A literature review of “failures” of the swanson finger prosthesis in the metacarpophalangeal joint. Hand Surg. 2002, 7, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Takigawa, S.; Meletiou, S.; Sauerbier, M.; Cooney, W.P. Long-term assessment of swanson implant arthroplasty in the proximal interphalangeal joint of the hand. J. Hand Surg. Am. 2004, 29, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Broadbent, M.; Birch, A.; Trail, I. A short-term review of the finsbury proximal interphalangeal joint replacement. In Proceedings of the British Society for Surgery of the Hand, Autumn Scientific Meeting, Nottingham, UK, 12 November 2009.
- Watts, A.; Trail, I. Anatomical small joint replacement of the hand. Available online: http://www.boneandjoint.org.uk/content/focus/anatomical-small-joint-replacement-hand (accessed on 9 February 2015).
- Daecke, W.; Kaszap, B.; Martini, A.K.; Hagena, F.W.; Rieck, B.; Jung, M. A prospective, randomized comparison of 3 types of proximal interphalangeal joint arthroplasty. J. Hand Surg. Am. 2012, 37, 1770–1779. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, B.R. Proximal interphalangeal joint surface replacement arthroplasty. Hand Surg. 2001, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, B.R.; Fitzgerald, M.; Smith, K.R.; Currie, L.J. Cemented versus uncemented surface replacement arthroplasty of the proximal interphalangeal joint with a mean 5-year follow-up. J. Hand Surg. Am. 2008, 33, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.M.; Linscheid, R.L.; Cooney, W.P.; Baker, V.; Heckman, M.G. Long-term outcomes of proximal interphalangeal joint surface replacement arthroplasty. J. Bone Jt. Surg. Am. 2012, 94, 1120–1128. [Google Scholar] [CrossRef]
- Schindele, S.F.; Hensler, S.; Audigé, L.; Marks, M.; Herren, D.B. A modular surface gliding implant (capflex-pip) for proximal interphalangeal joint osteoarthritis: A prospective case series. J. Hand Surg. Am. 2015, 40, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Selier, J.G. American Society for Surgery of the Hand: Essentials of Hand Surger; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2002. [Google Scholar]
- Joyce, T.J.; Unsworth, A. A test procedure for artificial finger joints. Proc. Inst. Mech. Eng. H. 2002, 216, 105–110. [Google Scholar] [CrossRef] [PubMed]
- British Standards Institute. BS ISO 14242-3: Implants for Surgery. Wear of Total Hip-Joint Prostheses; British Standards Institute: London, UK, 2009. [Google Scholar]
- British Standards Institute. BS ISO 14243-3: Implants for Surgery. Wear of Total Knee-Joint Prostheses; British Standards Institute: London, UK, 2014. [Google Scholar]
- Joyce, T.J. Prediction of lubrication regimes in two-piece metacarpophalangeal prostheses. Med. Eng. Phys. 2007, 29, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Rieker, C.; Unsworth, A. Comparative in vitro wear testing of peek and uhmwpe capped metacarpophalangeal prostheses. Biomed. Mater. Eng. 2006, 16, 1–10. [Google Scholar] [PubMed]
- Naylor, A.; Bone, M.C.; Unsworth, A.; Talwalkar, S.C.; Trail, I.A.; Joyce, T.J. Evaluating the wear and surface roughness of pyrocarbon finger prostheses tested in vitro. In Proceedings of the 10th Anniversary Bath Biomechanics Symposium: Current Issues and Future Opportunities in Orthopaedic Research, Bath, UK, 15 September 2014.
- Naylor, A.; Bone, M.C.; Unsworth, A.; Trail, I.A.; Talwalkar, S.C.; Joyce, T.J. In vitro testing of pyrolytic carbon proximal interphalangeal prostheses. In Proceedings of the British Society for Surgery of the Hand, Spring Scientific Meeting, Gateshead, UK, 1–2 May 2014.
- Joyce, T.J.; Unsworth, A. Neuflex metacarpophalangeal prostheses tested in vitro. Proc. Inst. Mech. Eng. H. 2005, 219, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Unsworth, A. The wear of artificial finger joints using different lubricants in a new finger wear simulator. Wear 2001, 250, 199–205. [Google Scholar] [CrossRef]
- Rempel, D.; Dennerlein, J.; Mote, C.D., Jr.; Armstrong, T. A method of measuring fingertip loading during keyboard use. J. Biomech. 1994, 27, 1101–1104. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.R.; Unsworth, A.; Haslock, I. A microcomputer controlled hand assessment system used for clinical measurement. Eng. Med. 1985, 14, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Weightman, B.; Amis, A.A. Finger joint force predictions related to design of joint replacements. J. Biomed. Eng. 1982, 4, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Katti, K.S. Biomaterials in total joint replacement. Colloids Surf. B Biointerfaces 2004, 39, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Szycher, M. Biocompatible Polymers, Metals, and Composites; Technomic Pub. Co.: Lancaster, OA, USA, 1983. [Google Scholar]
- Joyce, T.J.; Langton, D.J.; Jameson, S.S.; Nargol, A.V.F. Tribological analysis of failed resurfacing hip prostheses and comparison with clinical data. Proc. Inst. Mech. Eng. J. 2009, 223, 317–323. [Google Scholar] [CrossRef]
- Joyce, T.J. Wear testing of a djoa finger prosthesis in vitro. J. Mater. Sci. Mater. Med. 2010, 21, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Unsworth, A. The influence of bovine serum lubricant on the wear of cross-linked polyethylene finger prostheses. J. Appl. Biomater. Biomech. JABB 2004, 2, 136–142. [Google Scholar]
- Bone, M.C.; Cunningham, J.L.; Lord, J.; Giddins, G.; Field, J.; Joyce, T.J. Analysis of failed van straten lpm proximal interphalangeal prostheses. J. Hand Surg. Eur. Vol. 2013, 38, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Cortés, P.; Pajares-López, M.; Robles-Molina, M.J.; Gómez-Sánchez, R.; Toledo-Romero, M.A.; de Torres-Urrea, J. Two-year outcomes of elektra prosthesis for trapeziometacarpal osteoarthritis: A longitudinal cohort study. J. Hand Surg. Eur. Vol. 2012, 37, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Hobby, J.L.; Edwards, S.; Field, J.; Giddins, G. A report on the early failure of the LPM proximal interphalangeal joint replacement. J. Hand Surg. Eur. Vol. 2008, 33, 526–527. [Google Scholar] [CrossRef] [PubMed]
- Kaszap, B.; Daecke, W.; Jung, M. High frequency failure of the moje thumb carpometacarpal joint arthroplasty. J. Hand Surg. Eur. Vol. 2012, 37, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Middleton, A.; Lakshmipathy, R.; Irwin, L.R. Failures of the rm finger prosthesis joint replacement system. J. Hand Surg. Eur. Vol. 2011, 36, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J. Snapping the fingers. J. Hand Surg. Br. 2003, 28, 566–567. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naylor, A.; Talwalkar, S.C.; Trail, I.A.; Joyce, T.J. In Vitro Wear Testing of a CoCr-UHMWPE Finger Prosthesis with Hydroxyapatite Coated CoCr Stems. Lubricants 2015, 3, 244-255. https://doi.org/10.3390/lubricants3020244
Naylor A, Talwalkar SC, Trail IA, Joyce TJ. In Vitro Wear Testing of a CoCr-UHMWPE Finger Prosthesis with Hydroxyapatite Coated CoCr Stems. Lubricants. 2015; 3(2):244-255. https://doi.org/10.3390/lubricants3020244
Chicago/Turabian StyleNaylor, Andrew, Sumedh C. Talwalkar, Ian A. Trail, and Thomas J. Joyce. 2015. "In Vitro Wear Testing of a CoCr-UHMWPE Finger Prosthesis with Hydroxyapatite Coated CoCr Stems" Lubricants 3, no. 2: 244-255. https://doi.org/10.3390/lubricants3020244
APA StyleNaylor, A., Talwalkar, S. C., Trail, I. A., & Joyce, T. J. (2015). In Vitro Wear Testing of a CoCr-UHMWPE Finger Prosthesis with Hydroxyapatite Coated CoCr Stems. Lubricants, 3(2), 244-255. https://doi.org/10.3390/lubricants3020244






