Researching Mitigation of Alcohol Binge Drinking in Polydrug Abuse: KCNK13 and RASGRF2 Gene(s) Risk Polymorphisms Coupled with Genetic Addiction Risk Severity (GARS) Guiding Precision Pro-Dopamine Regulation
Abstract
1. Introduction
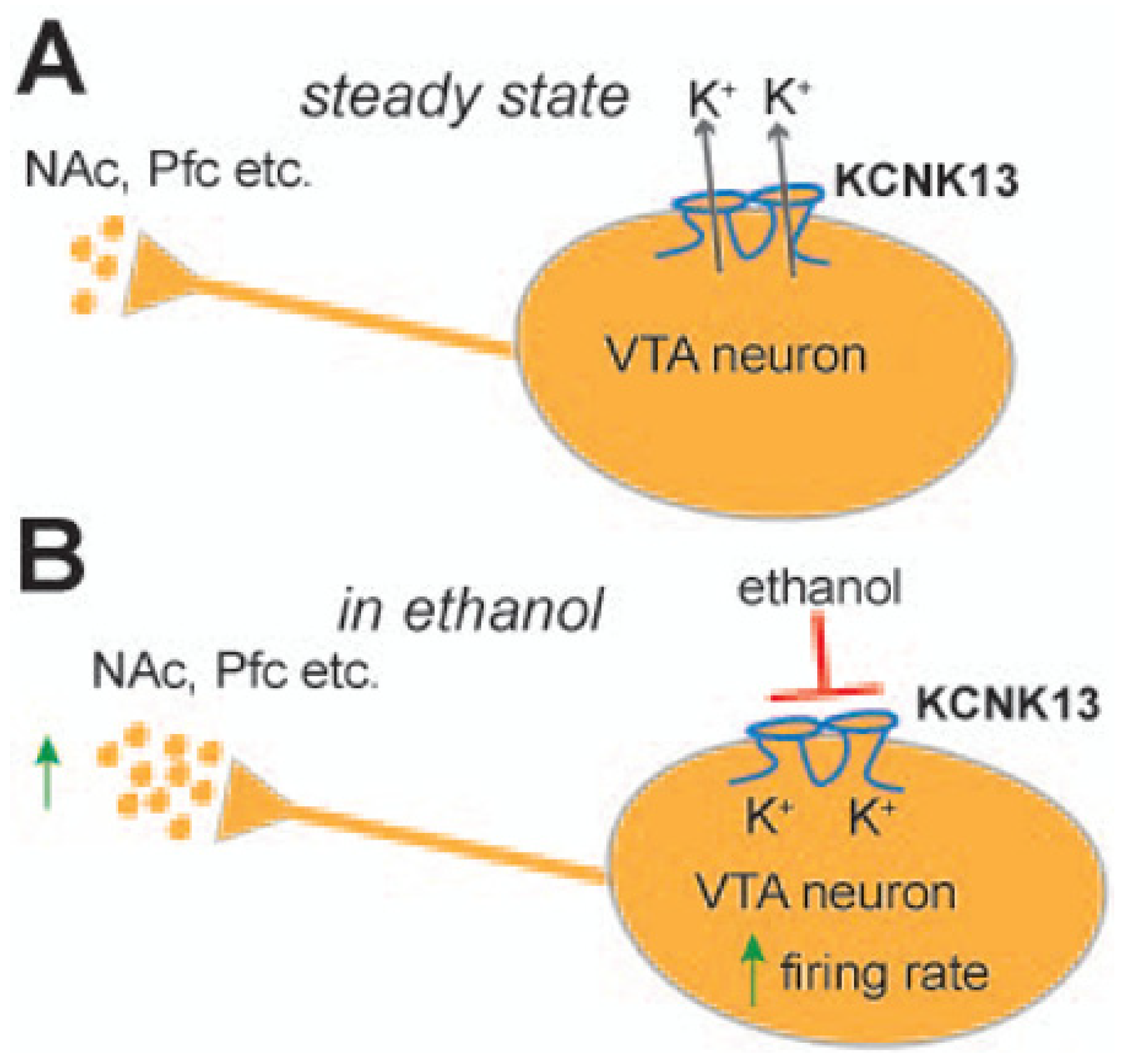
1.1. KCNK13 and Binge Drinking
1.2. Ras-Specific Guanine-Nucleotide Releasing Factor 2 (RASGRF2) and Binge Drinking
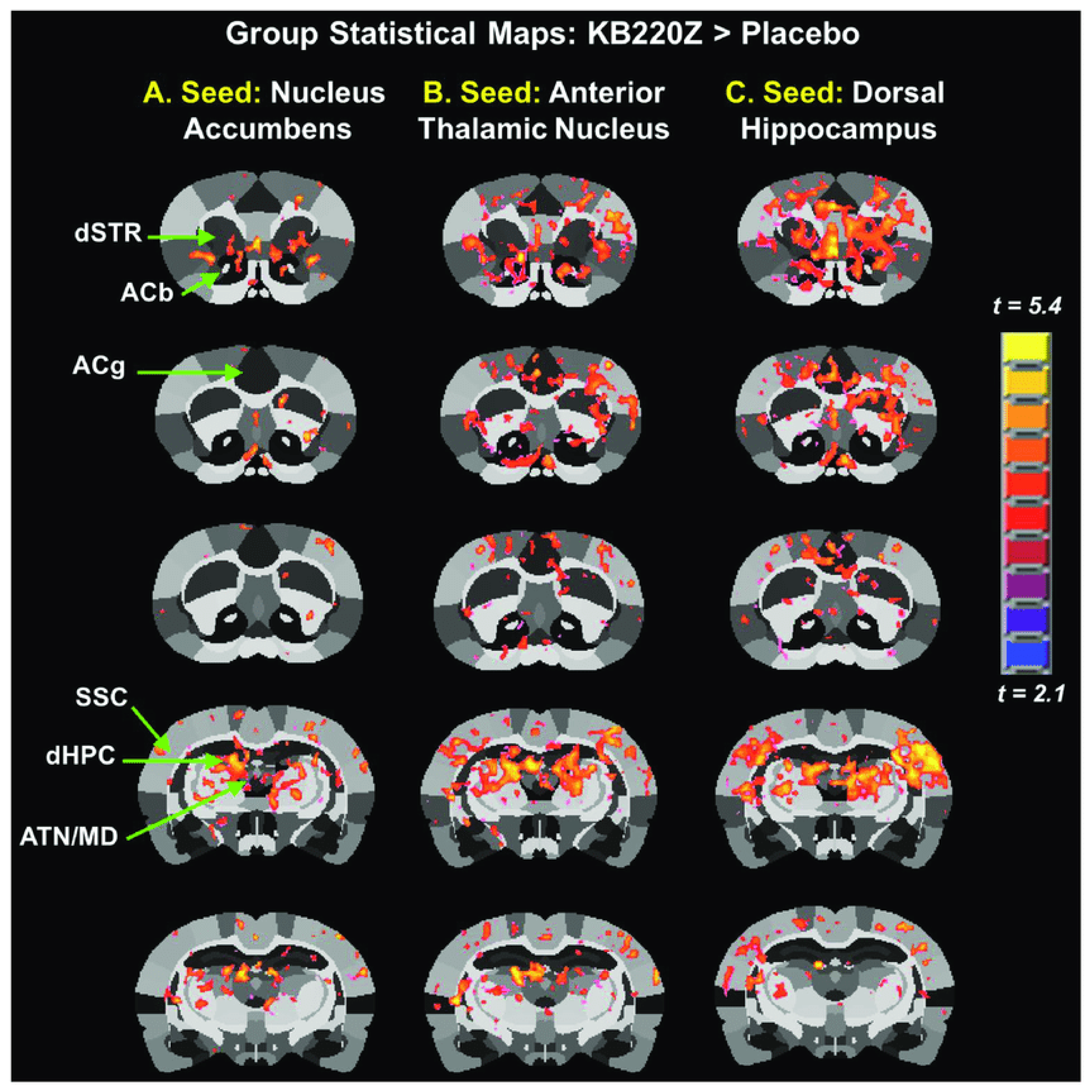
1.3. Precision Addiction Management for Binge Drinking Behavior
1.4. Determinism versus Free Will
2. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grucza, R.A.; Sher, K.J.; Kerr, W.C.; Krauss, M.J.; Lui, C.K.; McDowell, Y.E.; Hartz, S.; Virdi, G.; Bierut, L.J. Trends in Adult Alcohol Use and Binge Drinking in the Early 21st-Century United States: A Meta-Analysis of 6 National Survey Series. Alcohol. Clin. Exp. Res. 2018, 42, 1939–1950. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Sheridan, P.J.; Wood, R.C.; Braverman, E.R.; Chen, T.J.; Cull, J.G.; Comings, D.E. The D2 dopamine receptor gene as a determinant of reward deficiency syndrome. J. R. Soc. Med. 1996, 89, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Solanki, N.; Abijo, T.; Galvao, C.; Darius, P.; Blum, K.; Gondré-Lewis, M.C. Administration of a putative pro-dopamine regulator, a neuronutrient, mitigates alcohol intake in alcohol-preferring rats. Behav. Brain Res. 2020, 385, 112563. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Bowirrat, A.; Braverman, E.R.; Baron, D.; Cadet, J.L.; Kazmi, S.; Elman, I.; Thanos, P.K.; Badgaiyan, R.D.; Downs, W.B.; et al. Reward Deficiency Syndrome (RDS): A Cytoarchitectural Common Neurobiological Trait of All Addictions. Int. J. Environ. Res. Public Health 2021, 18, 11529. [Google Scholar] [CrossRef]
- Geller, I. Ethanol Preference in the Rat as a Function of Photoperiod. Science 1971, 173, 456–459. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Merritt, J.H.; Reiter, R.J.; Wallace, J.E. A possible relationship between the pineal gland and ethanol preference in the rat. Curr. Ther. Res. 1973, 15, 25–30. [Google Scholar]
- Reiter, R.J.; Blum, K.; Wallace, J.E.; Merritt, J.H. Effect of the Pineal Gland on Alcohol Consumption by Congenitally Blind Male Rats. Q. J. Stud. Alcohol 1973, 34, 937–939. [Google Scholar] [CrossRef]
- Reiter, R.; Blum, K.; Wallace, J.; Merritt, J. Pineal gland: Evidence for an influence on ethanol preference in male syrian hamsters. Comp. Biochem. Physiol. Part A Physiol. 1974, 47, 11–16. [Google Scholar] [CrossRef]
- Koob, G.F.; Volkow, N.D. Neurocircuitry of Addiction. Neuropsychopharmacology 2010, 35, 217–238, Erratum in Neuropsychopharmacology 2010, 35, 1051. [Google Scholar] [CrossRef]
- Waszkiewicz, N.; Galińska-Skok, B.; Nestsiarovich, A.; Kułak-Bejda, A.; Wilczyńska, K.; Simonienko, K.; Kwiatkowski, M.; Konarzewska, B. Neurobiological Effects of Binge Drinking Help in Its Detection and Differential Diagnosis from Alcohol Dependence. Dis. Markers 2018, 2018, 5623683. [Google Scholar] [CrossRef]
- Blum, K.; Gondré-Lewis, M.C.; Modestino, E.J.; Lott, L.; Baron, D.; Siwicki, D.; McLaughlin, T.; Howeedy, A.; Krengel, M.H.; Oscar-Berman, M.; et al. Understanding the Scientific Basis of Post-traumatic Stress Disorder (PTSD): Precision Behavioral Management Overrides Stigmatization. Mol. Neurobiol. 2019, 56, 7836–7850. [Google Scholar] [CrossRef] [PubMed]
- Palm, S.; Momeni, S.; Lundberg, S.; Nylander, I.; Roman, E. Risk-assessment and risk-taking behavior predict potassium- and amphetamine-induced dopamine response in the dorsal striatum of rats. Front. Behav. Neurosci. 2014, 8, 236. [Google Scholar] [CrossRef] [PubMed]
- Piantadosi, P.T.; Halladay, L.R.; Radke, A.K.; Holmes, A. Advances in understanding meso-cortico-limbic-striatal systems mediating risky reward seeking. J. Neurochem. 2021, 157, 1547–1571. [Google Scholar] [CrossRef] [PubMed]
- Gondré-Lewis, M.C.; Bassey, R.; Blum, K. Pre-clinical models of reward deficiency syndrome: A behavioral octopus. Neurosci. Biobehav. Rev. 2020, 115, 164–188. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Gondré-Lewis, M.C.; Baron, D.; Thanos, P.K.; Braverman, E.R.; Neary, J.; Elman, I.; Badgaiyan, R.D. Introducing Precision Addiction Management of Reward Deficiency Syndrome, the Construct That Underpins All Addictive Behaviors. Front. Psychiatry 2018, 9, 548. [Google Scholar] [CrossRef] [PubMed]
- Philippart, F.; Khaliq, Z.M. Gi/o protein-coupled receptors in dopamine neurons inhibit the sodium leak channel NALCN. eLife 2018, 7, e40984. [Google Scholar] [CrossRef]
- Goldman, D.E. Potential, impedance, and rectification in membranes. J. Gen. Physiol. 1943, 27, 37–60. [Google Scholar] [CrossRef]
- Hodgkin, A.L.; Katz, B. The effect of sodium ions on the electrical activity of the giant axon of the squid. J. Physiol. 1949, 108, 37–77. [Google Scholar] [CrossRef]
- Siegelbaum, S.A.; Camardo, J.S.; Kandel, E.R. Serotonin and cyclic AMP close single K+ channels in Aplysia sensory neurones. Nature 1982, 299, 413–417. [Google Scholar] [CrossRef]
- Shen, K.Z.; North, R.A.; Surprenant, A. Potassium channels opened by noradrenaline and other transmitters in excised membrane patches of guinea-pig submucosal neurons. J. Physiol. 1990, 22, 173–187. [Google Scholar] [CrossRef]
- Buckler, K.J. A novel oxygen-sensitive potassium current in rat carotid body type I cells. J. Physiol. 1997, 498, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Wagner, P.G.; Dekin, M.S. cAMP modulates an S-type K+ channel coupled to GABAB receptors in mammalian respiratory neurons. Neuroreport 1997, 8, 1667–1670. [Google Scholar] [CrossRef] [PubMed]
- Ketchum, K.A.; Joiner, W.J.; Sellers, A.J.; Kaczmarek, L.K.; Goldstein, S.A.N. A new family of outwardly rectifying potassium channel proteins with two pore domains in tandem. Nature 1995, 376, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, S.A.N.; Price, L.A.; Rosenthal, D.N.; Pausch, M.H. ORK1, a potassium-selective leak channel with two pore domains cloned from Drosophila melanogaster by expression in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1996, 93, 13256–13261. [Google Scholar] [CrossRef] [PubMed]
- Rajan, S.; Wischmeyer, E.; Karschin, C.; Preisig-Müller, R.; Grzeschik, K.-H.; Daut, J.; Karschin, A.; Derst, C. THIK-1 and THIK-2, a Novel Subfamily of Tandem Pore Domain K+ Channels. J. Biol. Chem. 2001, 276, 7302–7311. [Google Scholar] [CrossRef]
- You, C.; Savarese, A.; Vandegrift, B.J.; He, D.; Pandey, S.C.; Lasek, A.W.; Brodie, M.S. Ethanol acts on KCNK13 potassium channels in the ventral tegmental area to increase firing rate and modulate binge–like drinking. Neuropharmacology 2018, 144, 29–36. [Google Scholar] [CrossRef]
- You, C.; Vandegrift, B.J.; Brodie, M.S. KCNK13 potassium channels in the ventral tegmental area of rats are important for excitation of VTA neurons by ethanol. Alcohol. Clin. Exp. Res. 2021, 45, 1348–1358. [Google Scholar] [CrossRef]
- Moran, M.; Blum, K.; Ponce, J.V.; Lott, L.; Gondré–Lewis, M.C.; Badgaiyan, S.; Brewer, R.; Downs, B.W.; Fynman, P.; Weingarten, A.; et al. High Genetic Addiction Risk Score (GARS) in Chronically Prescribed Severe Chronic Opioid Probands Attending Multi-pain Clinics: An Open Clinical Pilot Trial. Mol. Neurobiol. 2021, 58, 3335–3346. [Google Scholar] [CrossRef]
- Dreher, J.-C.; Kohn, P.; Kolachana, B.; Weinberger, D.R.; Berman, K.F. Variation in dopamine genes influences responsivity of the human reward system. Proc. Natl. Acad. Sci. USA 2009, 106, 617–622. [Google Scholar] [CrossRef]
- Wise, R.A.; Robble, M.A. Dopamine and Addiction. Annu. Rev. Psychol. 2020, 71, 79–106. [Google Scholar] [CrossRef]
- Stollstorff, M.; Foss-Feig, J.; Cook, E.H., Jr.; Stein, M.A.; Gaillard, W.D.; Vaidya, C.J. Neural response to working memory load varies by dopamine transporter genotype in children. NeuroImage 2010, 53, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-Y.; Lee, S.-Y.; Chen, S.L.; Chang, Y.-H.; Chen, S.-H.; Chu, C.-H.; Huang, S.-Y.; Tzeng, N.-S.; Wang, C.-L.; Yeh, P.-H.; et al. The ADH1B and DRD2 gene polymorphism may modify the protective effect of the ALDH2 gene against heroin dependence. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 43, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-S.; Lee, S.-Y.; Chen, S.-L.; Chang, Y.-H.; Wang, T.-Y.; Lin, S.-H.; Wang, C.-L.; Huang, S.-Y.; Lee, I.; Chen, P.; et al. Role of DRD2 and ALDH2 genes in bipolar II disorder with and without comorbid anxiety disorder. Eur. Psychiatry 2014, 29, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Worbe, Y.; Palminteri, S.; Savulich, G.; Daw, N.D.; Fernandezegea, E.; Robbins, T.; Voon, V. Valence-dependent influence of serotonin depletion on model-based choice strategy. Mol. Psychiatry 2015, 21, 624–629. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Peng, G.-S.; Wang, M.-F.; Tsao, T.-P.; Yin, S.-J. Polymorphism of ethanol-metabolism genes and alcoholism: Correlation of allelic variations with the pharmacokinetic and pharmacodynamic consequences. Chem. Interact. 2009, 178, 2–7. [Google Scholar] [CrossRef]
- Thompson, M.D.; Kenna, G.A. Variation in the Serotonin Transporter Gene and Alcoholism: Risk and Response to Pharmacotherapy. Alcohol Alcohol. 2016, 51, 164–171. [Google Scholar] [CrossRef]
- Uhl, G.R.; Koob, G.F.; Cable, J. The neurobiology of addiction. Ann. N. Y. Acad. Sci. 2019, 1451, 5–28. [Google Scholar] [CrossRef]
- Ursini, G.; Punzi, G.; Chen, Q.; Marenco, S.; Robinson, J.F.; Porcelli, A.; Hamilton, E.G.; Mitjans, M.; Maddalena, G.; Begemann, M.; et al. Convergence of placenta biology and genetic risk for schizophrenia. Nat. Med. 2018, 24, 792–801. [Google Scholar] [CrossRef]
- Stern, D.L. The genetic causes of convergent evolution. Nat. Rev. Genet. 2013, 14, 751–764. [Google Scholar] [CrossRef]
- Deracinois, B.; Duban-Deweer, S.; Pottiez, G.; Cecchelli, R.; Karamanos, Y.; Flahaut, C. TNAP and EHD1 Are Over-Expressed in Bovine Brain Capillary Endothelial Cells after the Re-Induction of Blood-Brain Barrier Properties. PLoS ONE 2012, 7, e48428. [Google Scholar] [CrossRef]
- Easton, A.C.; Rotter, A.; Lourdusamy, A.; Desrivières, S.; Fernández-Medarde, A.; Biermann, T.; Fernandes, C.; Santos, E.; Kornhuber, J.; Schumann, G.; et al. Rasgrf2 controls dopaminergic adaptations to alcohol in mice. Brain Res. Bull. 2014, 109, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Easton, A.C.; Rotter, A.; Lourdusamy, A.; Desrivières, S.; Fernández-Medarde, A.; Biermann, T.; Fernandes, C.; Santos, E.; Kornhuber, J.; Schumann, G.; et al. Rasgrf2 controls noradrenergic involvement in the acute and subchronic effects of alcohol in the brain. Psychopharmacology 2014, 231, 4199–4209. [Google Scholar] [CrossRef] [PubMed]
- McCall, N.M.; De Velasco, E.M.F.; Wickman, K. GIRK Channel Activity in Dopamine Neurons of the Ventral Tegmental Area Bidirectionally Regulates Behavioral Sensitivity to Cocaine. J. Neurosci. 2019, 39, 3600–3610. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Ikeda, K.; Kojima, H.; Niki, H.; Yano, R.; Yoshioka, T.; Kumanishi, T. Ethanol opens G-protein-activated inwardly rectifying K+ channels. Nat. Neurosci. 1999, 2, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Lewohl, J.M.; Wilson, W.R.; Mayfield, R.D.; Brozowski, S.J.; Morrisett, R.A.; Harris, R.A. G-protein-coupled inwardly rectifying potassium channels are targets of alcohol action. Nat. Neurosci. 1999, 2, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Herman, M.A.; Sidhu, H.; Stouffer, D.G.; Kreifeldt, M.; Le, D.; Cates-Gatto, C.; Munoz, M.B.; Roberts, A.J.; Parsons, L.H.; Roberto, M.; et al. GIRK3 gates activation of the mesolimbic dopaminergic pathway by ethanol. Proc. Natl. Acad. Sci. USA 2015, 112, 7091–7096. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Meng, Q.; Xia, Y.; Ding, C.; Wang, L.; Dai, R.; Cheng, L.; Gunaratne, P.; Gibbs, R.A.; Min, S.; et al. The transcription factor POU3F2 regulates a gene coexpression network in brain tissue from patients with psychiatric disorders. Sci. Transl. Med. 2018, 10, eaat8178. [Google Scholar] [CrossRef]
- Appel, S.B.; Liu, Z.; McElvain, M.A.; Brodie, M.S. Ethanol Excitation of Dopaminergic Ventral Tegmental Area Neurons Is Blocked by Quinidine. J. Pharmacol. Exp. Ther. 2003, 306, 437–446. [Google Scholar] [CrossRef]
- Nimitvilai, S.; You, C.; Arora, D.; McElvain, M.A.; Vandegrift, B.; Brodie, M.; Woodward, J.J. Differential Effects of Toluene and Ethanol on Dopaminergic Neurons of the Ventral Tegmental Area. Front. Neurosci. 2016, 10, 434. [Google Scholar] [CrossRef]
- Okamoto, T.; Harnett, M.T.; Morikawa, H. Hyperpolarization-Activated Cation Current (Ih) Is an Ethanol Target in Midbrain Dopamine Neurons of Mice. J. Neurophysiol. 2006, 95, 619–626. [Google Scholar] [CrossRef]
- Rivera-Meza, M.; Quintanilla, M.E.; Bustamante, D.; Delgado, R.; Buscaglia, M.; Herrera-Marschitz, M. Overexpression of Hyperpolarization-Activated Cyclic Nucleotide-Gated Channels into the Ventral Tegmental Area Increases the Rewarding Effects of Ethanol in UChB Drinking Rats. Alcohol. Clin. Exp. Res. 2014, 38, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, S.A.N.; Bockenhauer, D.; O’Kelly, I.; Zilberberg, N. Potassium leak channels and the KCNK family of two-p-domain subunits. Nat. Rev. Neurosci. 2001, 2, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, C.A.; Karkhanis, A.N.; Holleran, K.M.; Melchior, J.R.; Jones, S.R. Cross-Species Alterations in Synaptic Dopamine Regulation After Chronic Alcohol Exposure. Handb. Exp. Pharmacol. 2018, 248, 213–238. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Febo, M.; Badgaiyan, R.D.; Baron, D.; Thanos, P.K.; Colon-Perez, L.M.; Demotrovics, Z.; Gold, M.S. Dopamine homeostasis brain functional connectivity in reward deficiency syndrome. Front. Biosci. 2017, 22, 669–691. [Google Scholar] [CrossRef]
- Elman, I.; Borsook, D. Common Brain Mechanisms of Chronic Pain and Addiction. Neuron 2016, 89, 11–36. [Google Scholar] [CrossRef]
- Stacey, D.; Lourdusamy, A.; Ruggeri, B.; Maroteaux, M.; Jia, T.; Cattrell, A.; Nymberg, C.; Banaschewski, T.; Bhattacharyya, S.; Band, H.; et al. A translational systems biology approach in both animals and humans identifies a functionally related module of accumbal genes involved in the regulation of reward processing and binge drinking in males. J. Psychiatry Neurosci. 2016, 41, 192–202. [Google Scholar] [CrossRef]
- Blum, K.; Noble, E.P.; Sheridan, P.J.; Montgomery, A.; Ritchie, T.; Jagadeeswaran, P.; Nogami, H.; Briggs, A.H.; Cohn, J.B. Allelic as-sociation of human dopamine D2 receptor gene in alcoholism. JAMA 1990, 263, 2055–2060. [Google Scholar] [CrossRef]
- Robinson, M.J.; Burghardt, P.R.; Patterson, C.M.; Nobile, C.W.; Akil, H.; Watson, S.J.; Berridge, K.C.; Ferrario, C.R. Individual Differences in Cue-Induced Motivation and Striatal Systems in Rats Susceptible to Diet-Induced Obesity. Neuropsychopharmacology 2015, 40, 2113–2123. [Google Scholar] [CrossRef]
- Tian, X.; Gotoh, T.; Tsuji, K.; Lo, E.H.; Huang, S.; Feig, L.A. Developmentally regulated role for Ras-GRFs in coupling NMDA glutamate receptors to Ras, Erk and CREB. EMBO J. 2004, 23, 1567–1575. [Google Scholar] [CrossRef]
- Fasano, S.; D’Antoni, A.; Orban, P.C.; Valjent, E.; Putignano, E.; Vara, H.; Pizzorusso, T.; Giustetto, M.; Yoon, B.; Soloway, P.; et al. Ras-Guanine Nucleotide-Releasing Factor 1 (Ras-GRF1) Controls Activation of Extracellular Signal-Regulated Kinase (ERK) Signaling in the Striatum and Long-Term Behavioral Responses to Cocaine. Biol. Psychiatry 2009, 66, 758–768. [Google Scholar] [CrossRef]
- Girault, J.-A.; Valjent, E.; Caboche, J.; Hervé, D. ERK2: A logical AND gate critical for drug-induced plasticity? Curr. Opin. Pharmacol. 2007, 7, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Baik, I.; Cho, N.H.; Kim, S.H.; Han, B.-G.; Shin, C. Genome-wide association studies identify genetic loci related to alcohol consumption in Korean men. Am. J. Clin. Nutr. 2011, 93, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Heath, A.C.; Whitfield, J.B.; Martin, N.G.; Pergadia, M.L.; Goate, A.M.; Lind, P.A.; McEvoy, B.P.; Schrage, A.J.; Grant, J.D.; Chou, Y.-L.; et al. A Quantitative-Trait Genome-Wide Association Study of Alcoholism Risk in the Community: Findings and Implications. Biol. Psychiatry 2011, 70, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Schumann, G.; Coin, L.J.; Lourdusamy, A.; Charoen, P.; Berger, K.H.; Stacey, D.; Desrivières, S.; Aliev, F.A.; Khan, A.A.; Amin, N.; et al. Genome-wide association and genetic functional studies identify autism susceptibility candidate 2 gene (AUTS2) in the regulation of alcohol consumption. Proc. Natl. Acad. Sci. USA 2011, 108, 7119–7124. [Google Scholar] [CrossRef]
- Stacey, D.; Bilbao, A.; Maroteaux, M.; Jia, T.; Easton, A.C.; Longueville, S.; Nymberg, C.; Banaschewski, T.; Barker, G.J.; Büchel, C.; et al. RASGRF2 regulates alcohol-induced reinforcement by influencing mesolimbic dopamine neuron activity and dopamine release. Proc. Natl. Acad. Sci. USA 2012, 109, 21128–21133. [Google Scholar] [CrossRef]
- Fasano, S.; Brambilla, R. Ras?ERK Signaling in Behavior: Old Questions and New Perspectives. Front. Behav. Neurosci. 2011, 5, 79. [Google Scholar] [CrossRef]
- Pandey, S.C.; Sakharkar, A.J.; Tang, L.; Zhang, H. Potential role of the adolescent alcohol exposure-induced amygdaloid his-tone modifications in anxiety and alcohol intake at adulthood. Neurobiol. Dis. 2015, 82, 607–619. [Google Scholar] [CrossRef]
- Zhang, H.; Kyzar, E.J.; Bohnsack, J.P.; Kokare, D.M.; Teppen, T.; Pandey, S.C. Adolescent alcohol exposure epigenetically regulates CREB signaling in the adult amygdala. Sci. Rep. 2018, 8, 10376. [Google Scholar] [CrossRef]
- Haass-Koffler, C.L.; Swift, R.M.; Leggio, L. Noradrenergic targets for the treatment of alcohol use disorder. Psychopharmacology 2018, 235, 1625–1634. [Google Scholar] [CrossRef]
- Giovannitti, J.A.; Thoms, S.M.; Crawford, J.J. Alpha-2 Adrenergic Receptor Agonists: A Review of Current Clinical Applications. Anesthesia Prog. 2015, 62, 31–38. [Google Scholar] [CrossRef]
- Elman, I.; Borsook, D. The failing cascade: Comorbid post traumatic stress- and opioid use disorders. Neurosci. Biobehav. Rev. 2019, 103, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Excessive Drinking is Draining the, U.S. Economy. 2020. Available online: https://www.cdc.gov/alcohol/features/excessive-drinking.html (accessed on 11 March 2022).
- Zhao, G.; Wu, C.; Houston, R.J.; Creager, W. The Effects of Binge Drinking and Socio-Economic Status on Sober Driving Behavior. Traffic Inj. Prev. 2010, 11, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Flowers, N.T.; Naimi, T.S.; Brewer, R.D.; Elder, R.W.; Shults, R.A.; Jiles, R. Patterns of Alcohol Consumption and Alcohol-Impaired Driving in the United States. Alcohol. Clin. Exp. Res. 2008, 32, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Naimi, T.S.; Nelson, D.E.; Brewer, R.D. Driving After Binge Drinking. Am. J. Prev. Med. 2009, 37, 314–320. [Google Scholar] [CrossRef]
- Park, J.-Y.; Wu, L.-T. Trends and correlates of driving under the influence of alcohol among different types of adult substance users in the United States: A national survey study. BMC Public Health 2019, 19, 509–511. [Google Scholar] [CrossRef]
- Utter, G.H.; Young, J.B.; Theard, L.A.; Cropp, D.M.; Mohar, C.J.; Eisenberg, D.; Schermer, C.R.; Owens, L.J. The effect on problematic drinking behavior of a brief motivational interview shortly after a first arrest for driving under the influence of alcohol: A randomized trial. J. Trauma Acute Care Surg. 2014, 76, 661–671. [Google Scholar] [CrossRef]
- Nochajski, T.H.; Stasiewicz, P.R. Relapse to driving under the influence (DUI): A review. Clin. Psychol. Rev. 2006, 26, 179–195. [Google Scholar] [CrossRef]
- Shaffer, H.J.; Nelson, S.E.; LaPlante, D.A.; LaBrie, R.A.; Albanese, M.; Caro, G. The epidemiology of psychiatric disorders among repeat DUI offenders accepting a treatment-sentencing option. J. Consult. Clin. Psychol. 2007, 75, 795–804. [Google Scholar] [CrossRef]
- Febo, M.; Blum, K.; Badgaiyan, R.D.; Perez, P.D.; Colon-Perez, L.; Thanos, P.K.; Ferris, C.F.; Kulkarni, P.; Giordano, J.; Baron, D.; et al. Enhanced functional connectivity and volume between cognitive and reward centers of naïve rodent brain produced by pro-dopaminergic agent KB220Z. PLoS ONE 2017, 12, e0174774. [Google Scholar] [CrossRef]
- Szücs, A.; Berton, F.; Sanna, P.P.; Francesconi, W. Excitability of jcBNST Neurons Is Reduced in Alcohol-Dependent Animals during Protracted Alcohol Withdrawal. PLoS ONE 2012, 7, e42313. [Google Scholar] [CrossRef]
- Brown, R.J.; Blum, K.; Trachtenberg, M.C. Neurodynamics of Relapse Prevention: A Neuronutrient Approach to Outpatient DUI Offenders. J. Psychoact. Drugs 1990, 22, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Blum, K.; Chen, A.L.; Chen, T.J.; Braverman, E.R.; Reinking, J.; Blum, S.H.; Cassel, K.; Downs, B.W.; Waite, R.L.; Williams, L.; et al. Activation instead of blocking mesolimbic dopaminergic reward circuitry is a preferred modality in the long term treatment of reward deficiency syndrome (RDS): A commentary. Theor. Biol. Med Model. 2008, 5, 24. [Google Scholar] [CrossRef] [PubMed]
- Borsook, D.; Linnman, C.; Faria, V.; Strassman, A.; Becerra, L.; Elman, I. Reward deficiency and anti-reward in pain chronification. Neurosci. Biobehav. Rev. 2016, 68, 282–297. [Google Scholar] [CrossRef] [PubMed]
- Elman, I.; Borsook, D.; Volkow, N.D. Pain and suicidality: Insights from reward and addiction neuroscience. Prog. Neurobiol. 2013, 109, 1–27. [Google Scholar] [CrossRef]
- Blum, K.; Berman, M.O.; Badgaiyan, R.D.; Braverman, E.R.; Gold, M.S. Hypothesizing Darkness Induced Alcohol Intake Linked to Dopaminergic Regulation of Brain Function. Psychology 2014, 05, 282–288. [Google Scholar] [CrossRef][Green Version]
- Blum, K.; Oscar-Berman, M.; Demetrovics, Z.; Barh, D.; Gold, M.S. Genetic Addiction Risk Score (GARS): Molecular Neurogenetic Evidence for Predisposition to Reward Deficiency Syndrome (RDS). Mol. Neurobiol. 2014, 50, 765–796. [Google Scholar] [CrossRef]
- Kenneth, B.; Edward, M.J.; Marjorie, G.L.C.; David, B.; Bruce, S.; Panayotis, T.K.; William, D.B.; Davis, S.; Lisa, L.; Eric, B.R.; et al. Pro-Dopamine Regulator (KB220) A Fifty Year Sojourn to Combat Reward Deficiency Syndrome (RDS): Evidence Based Bibliography (Annotated). CPQ Neurol. Psychol. 2018, 1, 2. [Google Scholar]
- Blum, K.; Chen, A.L.; Thanos, P.K.; Febo, M.; Demetrovics, Z.; Dushaj, K.; Kovoor, A.; Baron, D.; Smith, D.E.; Lll, A.K.R.; et al. Genetic addiction risk score GARS trade a predictor of vulnerability to opioid dependence. Front. Biosci. 2018, 10, 175–196. [Google Scholar] [CrossRef]
- Blum, K.; Lott, L.; Siwicki, D.; Fried, L.; Hauser, M.; Simpatico, T.; Baron, D.; Howeedy, A.; Badgaiyan, R.D. Genetic Addiction Risk Score (GARS™) as a Predictor of Substance Use Disorder: Iden-tifying predisposition not diagnosis. Curr. Trends Med. Diagn Methods 2018, 1, 1. [Google Scholar] [CrossRef]
- Blum, K.; Modestino, E.J.; Lott, L.; Siwicki, D.; Baron, D.; Howeedy, A.; Badgaiyan, R.D. Introducing “Precision Addiction Management (PAM®)” as an Adjunctive Genetic Guided Therapy for Abusable Drugs in America. Open Access J. Behav. Sci. Psychol. 2018, 1, 1–4. [Google Scholar]
- Blum, K. Promoting Precision Addiction Management (PAM) to Combat the Global Opioid Crisis. Biomed. J. Sci. Tech. Res. 2018, 2, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Baron, D.; Blum, K.; Chen, A.; Gold, M.; Badgaiyan, R.D. Conceptualizing Addiction from an Osteopathic Perspective: Dopamine Homeostasis. J. Osteopat. Med. 2018, 118, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Abijo, T.; Blum, K.; Gondré-Lewis, M.C. Neuropharmacological and Neurogenetic Correlates of Opioid Use Disorder (OUD) As A Function of Ethnicity: Relevance to Precision Addiction Medicine. Curr. Neuropharmacol. 2020, 18, 578–595. [Google Scholar] [CrossRef] [PubMed]
- Madigan, M.A.; Badgaiyan, R.D.; Baron, D.; Blum, K. Precision Behavioral Management (PBM) A Novel Approach to Combat Post-Traumatic Stress Disorder (PTSD). SOJ Psychol. 2018, 5, 2. [Google Scholar]
- Fried, L.; Modestino, E.J.; Siwicki, D.; Lott, L.; Thanos, P.K.; Baron, D.; Badgaiyan, R.D.; Ponce, J.V.; Giordano, J.; Downs, W.B.; et al. Hypodopaminergia and “Precision Behavioral Management” (PBM): It is a Generational Family Affair. Curr. Pharm. Biotechnol. 2020, 21, 528–541. [Google Scholar] [CrossRef]
- Bau, C.H.; Almeida, S.; Hutz, M.H. The TaqI A1 allele of the dopamine D2 receptor gene and alcoholism in Brazil: Association and interaction with stress and harm avoidance on severity prediction. Am. J. Med. Genet. 2000, 96, 302–306. [Google Scholar] [CrossRef]
- Failure of Prison Rehabilitation (from Critical Issues in Criminal Justice, 1979, by R G Iacovetta and Dae H Chang—See NCJ-63717). Available online: https://www.ojp.gov/ncjrs/virtual-library/abstracts/failure-prison-rehabilitation-critical-issues-criminal-justice-1979 (accessed on 11 March 2022).
- Bohnsack, J.P.; Zhang, H.; Wandling, G.M.; He, D.; Kyzar, E.J.; Lasek, A.W.; Pandey, S.C. Targeted epigenomic editing ameliorates adult anxiety and excessive drinking after adolescent alcohol exposure. Sci. Adv. 2022, 8, eabn2748. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blum, K.; Brodie, M.S.; Pandey, S.C.; Cadet, J.L.; Gupta, A.; Elman, I.; Thanos, P.K.; Gondre-Lewis, M.C.; Baron, D.; Kazmi, S.; et al. Researching Mitigation of Alcohol Binge Drinking in Polydrug Abuse: KCNK13 and RASGRF2 Gene(s) Risk Polymorphisms Coupled with Genetic Addiction Risk Severity (GARS) Guiding Precision Pro-Dopamine Regulation. J. Pers. Med. 2022, 12, 1009. https://doi.org/10.3390/jpm12061009
Blum K, Brodie MS, Pandey SC, Cadet JL, Gupta A, Elman I, Thanos PK, Gondre-Lewis MC, Baron D, Kazmi S, et al. Researching Mitigation of Alcohol Binge Drinking in Polydrug Abuse: KCNK13 and RASGRF2 Gene(s) Risk Polymorphisms Coupled with Genetic Addiction Risk Severity (GARS) Guiding Precision Pro-Dopamine Regulation. Journal of Personalized Medicine. 2022; 12(6):1009. https://doi.org/10.3390/jpm12061009
Chicago/Turabian StyleBlum, Kenneth, Mark S. Brodie, Subhash C. Pandey, Jean Lud Cadet, Ashim Gupta, Igor Elman, Panayotis K. Thanos, Marjorie C. Gondre-Lewis, David Baron, Shan Kazmi, and et al. 2022. "Researching Mitigation of Alcohol Binge Drinking in Polydrug Abuse: KCNK13 and RASGRF2 Gene(s) Risk Polymorphisms Coupled with Genetic Addiction Risk Severity (GARS) Guiding Precision Pro-Dopamine Regulation" Journal of Personalized Medicine 12, no. 6: 1009. https://doi.org/10.3390/jpm12061009
APA StyleBlum, K., Brodie, M. S., Pandey, S. C., Cadet, J. L., Gupta, A., Elman, I., Thanos, P. K., Gondre-Lewis, M. C., Baron, D., Kazmi, S., Bowirrat, A., Febo, M., Badgaiyan, R. D., Braverman, E. R., Dennen, C. A., & Gold, M. S. (2022). Researching Mitigation of Alcohol Binge Drinking in Polydrug Abuse: KCNK13 and RASGRF2 Gene(s) Risk Polymorphisms Coupled with Genetic Addiction Risk Severity (GARS) Guiding Precision Pro-Dopamine Regulation. Journal of Personalized Medicine, 12(6), 1009. https://doi.org/10.3390/jpm12061009












