AI-Driven Cell Tracking to Enable High-Throughput Drug Screening Targeting Airway Epithelial Repair for Children with Asthma
Abstract
1. Introduction
2. Materials and Methods
2.1. Datasets
2.1.1. Wound Repair Dataset
2.1.2. CTMC Dataset
2.2. Algorithm
2.3. Performance Evaluation
3. Results and Discussion
3.1. Dataset Comparison
3.2. Wound Repair Dataset
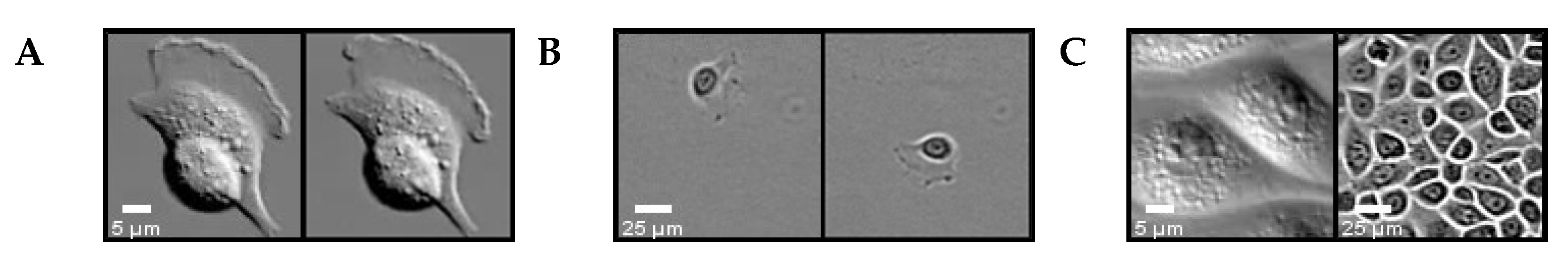
3.2.1. Cell Detection
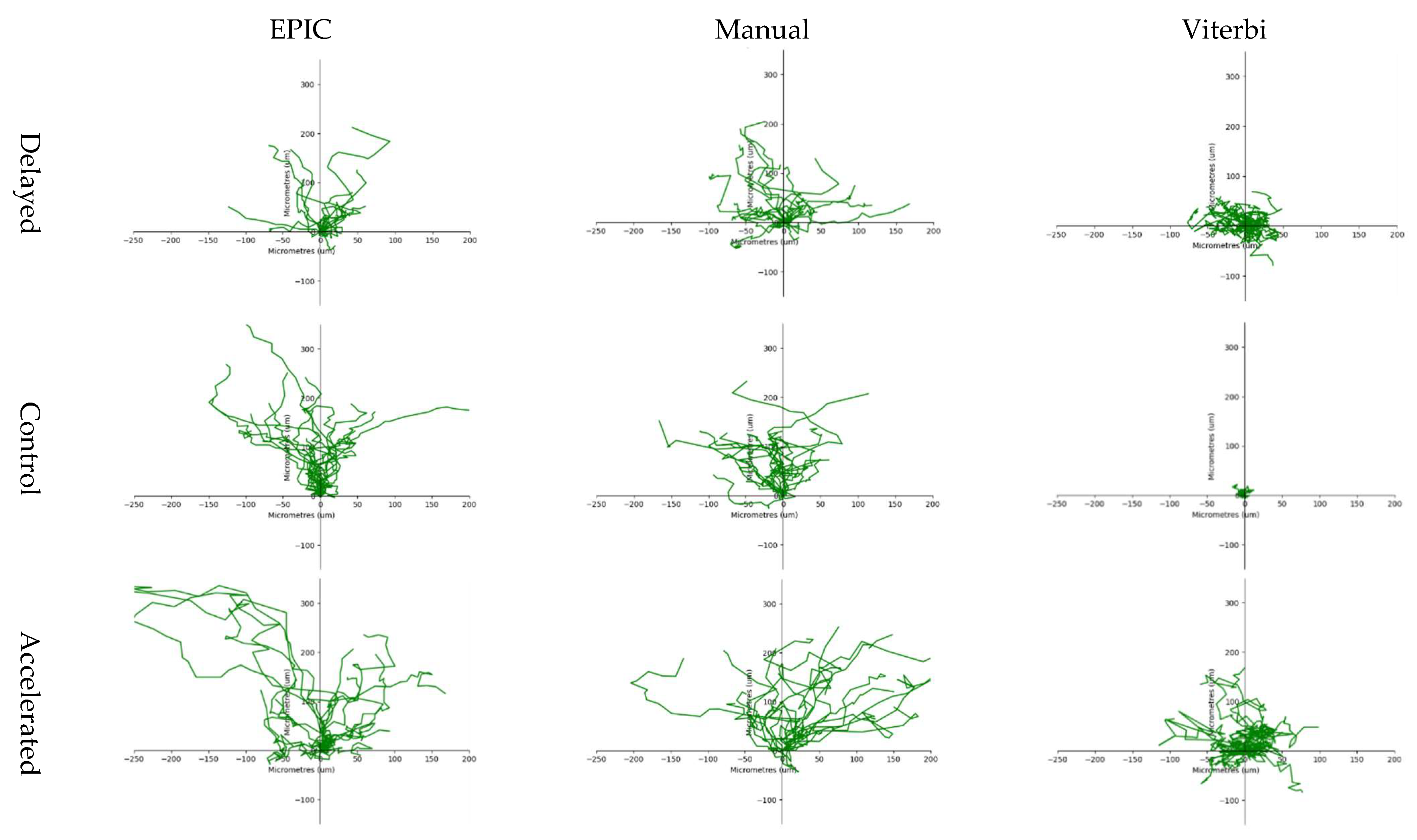
3.2.2. Cell Tracking
3.2.3. Automated Leading Edge Identification (EPIC)
3.2.4. Runtimes
3.3. CTMC Dataset
3.4. Software Features and Report
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allahverdian, S. Basic Mechanism of Airway Epithelial Repair: Role of IL-13 and EGFR Glycosylation. Ph.D. Thesis, University of British Columbia, Vancouver, BC, Canada, 2008. [Google Scholar]
- Kicic, A.; Hallstrand, T.S.; Sutanto, E.N.; Stevens, P.T.; Kobor, M.S.; Taplin, C.; Paré, P.D.; Beyer, R.P.; Stick, S.M.; Knight, D.A. Decreased fibronectin production significantly contributes to dysregulated repair of asthmatic epithelium. Am. J. Respir. Crit. Care Med. 2010, 181, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Kicic, A.; Sutanto, E.N.; Stevens, P.T.; Knight, D.A.; Stick, S.M. Intrinsic biochemical and functional differences in bronchial epithelial cells of children with asthma. Am. J. Respir. Crit. Care Med. 2006, 174, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-Y.; Lo, K.-Y.; Sun, Y.-S. A microfluidics-based wound-healing assay for studying the effects of shear stresses, wound widths, and chemicals on the wound-healing process. Sci. Rep. 2019, 9, 20016. [Google Scholar] [CrossRef] [PubMed]
- Iosifidis, T.; Sutanto, E.N.; Buckley, A.G.; Coleman, L.; Gill, E.E.; Lee, A.H.; Ling, K.M.; Hillas, J.; Looi, K.; Garratt, L.W.; et al. Aberrant cell migration contributes to defective airway epithelial repair in childhood wheeze. JCI Insight 2020, 5, e133125. [Google Scholar] [CrossRef]
- Ranzato, E.; Martinotti, S.; Burlando, B. Wound healing properties of jojoba liquid wax: An in vitro study. J. Ethnopharmacol. 2011, 134, 443–449. [Google Scholar] [CrossRef]
- Pinto, B.I.; Tabor, A.J.; Stearns, D.M.; Diller, R.B.; Kellar, R.S. A Bench-Top In Vitro Wound Assay to Demonstrate the Effects of Platelet-Rich Plasma and Depleted Uranium on Dermal Fibroblast Migration. Appl. In Vitro Toxicol. 2016, 2, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Pinto, B.I.; Cruz, N.D.; Lujan, O.R.; Propper, C.R.; Kellar, R.S. In Vitro Scratch Assay to Demonstrate Effects of Arsenic on Skin Cell Migration. JoVE 2019, 144, e58838. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.-C.; Park, A.; Guan, J.-L. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Meijering, E.; Dzyubachyk, O.; Smal, I. Methods for cell and particle tracking. Methods Enzymol. 2012, 504, 183–200. [Google Scholar]
- Cordelières, F.P.; Petit, V.; Kumasaka, M.; Debeir, O.; Letort, V.; Gallagher, S.J.; Larue, L. Automated Cell Tracking and Analysis in Phase-Contrast Videos (iTrack4U): Development of Java Software Based on Combined Mean-Shift Processes. PLoS ONE 2013, 8, e81266. [Google Scholar] [CrossRef]
- Sacan, A.; Ferhatosmanoglu, H.; Coskun, H. CellTrack: An open-source software for cell tracking and motility analysis. Bioinformatics 2008, 24, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Aow Yong, L.Y.; Sulong, G. Automated cell migration tracking technique: A review. J. Teknol. 2015, 75. [Google Scholar] [CrossRef]
- Emami, N.; Sedaei, Z.; Ferdousi, R. Computerized cell tracking: Current methods, tools and challenges. Vis. Inform. 2021, 5, 1–13. [Google Scholar] [CrossRef]
- Ulman, V.; Maška, M.; Magnusson, K.E.G.; Ronneberger, O.; Haubold, C.; Harder, N.; Matula, P.; Matula, P.; Svoboda, D.; Radojevic, M.; et al. An objective comparison of cell-tracking algorithms. Nat. Methods 2017, 14, 1141–1152. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-F.; Gajda, J.; Sloan, T.F.W.; Rares, A.; Shen, A.Q. Usiigaci: Instance-aware cell tracking in stain-free phase contrast microscopy enabled by machine learning. SoftwareX 2019, 9, 230–237. [Google Scholar] [CrossRef]
- Al-Zaben, N.; Medyukhina, A.; Dietrich, S.; Marolda, A.; Hünniger, K.; Kurzai, O.; Figge, M.T. Automated tracking of label-free cells with enhanced recognition of whole tracks. Sci. Rep. 2019, 9, 3317. [Google Scholar] [CrossRef] [PubMed]
- Piccinini, F.; Kiss, A.; Horvath, P. CellTracker (not only) for dummies. Bioinformatics 2016, 32, 955–957. [Google Scholar] [CrossRef]
- Hayashida, J.; Bise, R. Cell tracking with deep learning for cell detection and motion estimation in low-frame-rate. In Medical Image Computing and Computer Assisted Intervention–MICCAI 2019; Springer: Berlin/Heidelberg, Germany, 2019; pp. 397–405. [Google Scholar]
- Bise, R.; Kanade, T.; Yin, Z.; Huh, S. Automatic cell tracking applied to analysis of cell migration in wound healing assay. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Conference, Boston, MA, USA, 30 August–3 September 2011; Volume 2011, pp. 6174–6179. [Google Scholar]
- Van Valen, D.A.; Kudo, T.; Lane, K.M.; Macklin, D.N.; Quach, N.T.; DeFelice, M.M.; Maayan, I.; Tanouchi, Y.; Ashley, E.A.; Covert, M.W. Deep Learning Automates the Quantitative Analysis of Individual Cells in Live-Cell Imaging Experiments. PLoS Comput. Biol. 2016, 12, e1005177. [Google Scholar] [CrossRef]
- Hayashida, J.; Nishimura, K.; Bise, R. MPM: Joint Representation of Motion and Position Map for Cell Tracking. In Proceedings of the 2020 IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR), Seattle, WA, USA, 13–19 June 2020; pp. 3822–3831. [Google Scholar]
- Nishimura, K.; Hayashida, J.; Wang, C.; Ker, D.F.E.; Bise, R. Weakly-supervised cell tracking via backward-and-forward propagation. In Computer Vision–ECCV 2020; Vedaldi, A., Bischof, H., Brox, T., Frahm, J.-M., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 104–121. [Google Scholar]
- Hernandez, D.E.; Chen, S.W.; Hunter, E.E.; Steager, E.B.; Kumar, V. Cell Tracking with Deep Learning and the Viterbi Algorithm. In Proceedings of the 2018 International Conference on Manipulation, Automation and Robotics at Small Scales (MARSS), Nagoya, Japan, 4–8 July 2018; pp. 1–6. [Google Scholar]
- DuChez, B.J. Automated Tracking of Cell Migration with Rapid Data Analysis. Curr. Protoc. Cell Biol. 2017, 76, 12–16. [Google Scholar] [CrossRef]
- Fu, K.S.; Mui, J.K. A survey on image segmentation. Pattern Recognit. 1981, 13, 3–16. [Google Scholar] [CrossRef]
- Hilsenbeck, O.; Schwarzfischer, M.; Skylaki, S.; Schauberger, B.; Hoppe, P.S.; Loeffler, D.; Kokkaliaris, K.D.; Hastreiter, S.; Skylaki, E.; Filipczyk, A.; et al. Software tools for single-cell tracking and quantification of cellular and molecular properties. Nat. Biotechnol. 2016, 34, 703–706. [Google Scholar] [CrossRef] [PubMed]
- Zhi, X.-H.; Meng, S.; Shen, H.-B. High density cell tracking with accurate centroid detections and active area-based tracklet clustering. Neurocomputing 2018, 295, 86–97. [Google Scholar] [CrossRef]
- Hilsenbeck, O.; Schwarzfischer, M.; Loeffler, D.; Dimopoulos, S.; Hastreiter, S.; Marr, C.; Theis, F.J.; Schroeder, T. fastER: A user-friendly tool for ultrafast and robust cell segmentation in large-scale microscopy. Bioinformatics 2017, 33, 2020–2028. [Google Scholar] [CrossRef] [PubMed]
- Kasprowicz, R.; Suman, R.; O’Toole, P. Characterising live cell behaviour: Traditional label-free and quantitative phase imaging approaches. Int. J. Biochem. Cell Biol. 2017, 84, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Das, N.; Das, I.; Maulik, U. Understanding Deep Learning Techniques for Image Segmentation. ACM Comput. Surv. 2019, 52, 1–35. [Google Scholar] [CrossRef]
- Moen, E.; Borba, E.; Miller, G.; Schwartz, M.; Bannon, D.; Koe, N.; Camplisson, I.; Kyme, D.; Pavelchek, C.; Price, T. Accurate cell tracking and lineage construction in live-cell imaging experiments with deep learning. bioRxiv. 2019, 803205. [Google Scholar] [CrossRef]
- Lugagne, J.-B.; Lin, H.; Dunlop, M.J. DeLTA: Automated cell segmentation, tracking, and lineage reconstruction using deep learning. PLoS Comput. Biol. 2020, 16, e1007673. [Google Scholar] [CrossRef]
- Dai, J.; He, K.; Sun, J. BoxSup: Exploiting Bounding Boxes to Supervise Convolutional Networks for Semantic Segmentation. In Proceedings of the 2015 IEEE International Conference on Computer Vision (ICCV), Washington, DC, USA, 7–13 December 2015; pp. 1635–1643. [Google Scholar]
- Hu, R.; Dollár, P.; He, K.; Darrell, T.; Girshick, R. Learning to Segment Every Thing. In Proceedings of the 2018 IEEE/CVF Conference on Computer Vision and Pattern Recognition, Salt Lake City, UT, USA, 18–23 June 2018; pp. 4233–4241. [Google Scholar]
- Anjum, S.; Gurari, D. CTMC: Cell Tracking with Mitosis Detection Dataset Challenge. In Proceedings of the 2020 IEEE/CVF Conference on Computer Vision and Pattern Recognition Workshops (CVPRW), Seattle, WA, USA, 14–19 June 2020; pp. 4228–4237. [Google Scholar]
- Dendorfer, P.; Osep, A.; Milan, A.; Schindler, K.; Cremers, D.; Reid, I.; Roth, S.; Leal-Taixé, L. MOTChallenge: A Benchmark for Single-Camera Multiple Target Tracking. Int. J. Comput. Vis. 2021, 129, 845–881. [Google Scholar] [CrossRef]
- Luo, W.; Xing, J.; Milan, A.; Zhang, X.; Liu, W.; Kim, T.-K. Multiple object tracking: A literature review. Artif. Intell. 2021, 293, 103448. [Google Scholar] [CrossRef]
- Ciaparrone, G.; Luque Sánchez, F.; Tabik, S.; Troiano, L.; Tagliaferri, R.; Herrera, F. Deep learning in video multi-object tracking: A survey. Neurocomputing 2020, 381, 61–88. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, P.; Jiang, Y.; Yu, D.; Weng, F.; Yuan, Z.; Luo, P.; Liu, W.; Wang, X. ByteTrack: Multi-Object Tracking by Associating Every Detection Box. arXiv 2022, arXiv:211006864. [Google Scholar] [CrossRef]
- Chu, P.; Wang, J.; You, Q.; Ling, H.; Liu, Z. TransMOT: Spatial-Temporal Graph Transformer for Multiple Object Tracking. arXiv 2021, arXiv:210400194. [Google Scholar] [CrossRef]
- He, K.; Gkioxari, G.; Dollár, P.; Girshick, R. Mask R-CNN. In Proceedings of the 2017 IEEE International Conference on Computer Vision (ICCV), Venice, Italy, 22–29 October 2017; pp. 2980–2988. [Google Scholar]
- Chen, Y.; Song, Y.; Zhang, C.; Zhang, F.; O’Donnell, L.; Chrzanowski, W.; Cai, W. Celltrack R-CNN: A Novel End-To-End Deep Neural Network For Cell Segmentation And Tracking in Microscopy Images. In Proceedings of the 2021 IEEE 18th International Symposium on Biomedical Imaging (ISBI), Nice, France, 13–16 April 2021; pp. 779–782. [Google Scholar]
- Kisantal, M.; Wojna, Z.; Murawski, J.; Naruniec, J.; Cho, K. Augmentation for Small Object Detection; Aircc Publishing Corporation: Chennai, India, 2019. [Google Scholar]
- Chen, Y.; Quelhas, P.; Campilho, A. Low frame rate cell tracking: A Delaunay graph matching approach. In Proceedings of the 2011 IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Chicago, IL, USA, 30 March–2 April 2011; pp. 1015–1018. [Google Scholar]
- Zhou, Z.; Wang, F.; Xi, W.; Chen, H.; Gao, P.; He, C. Joint Multi-frame Detection and Segmentation for Multi-cell Tracking. In Image and Graphics; Zhao, Y., Barnes, N., Chen, B., Westermann, R., Kong, X., Lin, C., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 435–446. [Google Scholar]
- Ling, H.; Wu, Y.; Blasch, E.; Chen, G.; Lang, H.; Bai, L. Evaluation of visual tracking in extremely low frame rate wide area motion imagery. In Proceedings of the 14th International Conference on Information Fusion, Chicago, IL, USA, 5–8 July 2011; pp. 1–8. [Google Scholar]
- Barry, D.J.; Durkin, C.H.; Abella, J.V.; Way, M. Open source software for quantification of cell migration, protrusions, and fluorescence intensities. J. Cell Biol. 2015, 209, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Xu, S.; Wei, L.; Zhang, X.; Wang, X. CellTracker: An automated toolbox for single-cell segmentation and tracking of time-lapse microscopy images. Bioinformatics 2021, 37, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Winter, M.; Mankowski, W.; Wait, E.; Temple, S.; Cohen, A.R. LEVER: Software tools for segmentation, tracking and lineaging of proliferating cells. Bioinformatics 2016, 32, 3530–3531. [Google Scholar] [CrossRef][Green Version]
- Dosovitskiy, A.; Beyer, L.; Kolesnikov, A.; Weissenborn, D.; Zhai, X.; Unterthiner, T.; Dehghani, M.; Minderer, M.; Heigold, G.; Gelly, S.; et al. An Image is Worth 16 × 16 Words: Transformers for Image Recognition at Scale. In Proceedings of the International Conference on Learning Representations, Virtual Event, Austria, 3–7 May 2021. [Google Scholar]
- Gwatimba, A.; Ho, J.; Iosifidis, T.; Karpievitch, Y.V. Rainbow: Automated air-liquid interface cell culture analysis using deep optical flow. J. Open Source Softw. 2022, 7, 4080. [Google Scholar] [CrossRef]
- Zabner, J.; Karp, P.; Seiler, M.; Phillips, S.L.; Mitchell, C.J.; Saavedra, M.; Welsh, M.; Klingelhutz, A.J. Development of cystic fibrosis and noncystic fibrosis airway cell lines. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2003, 284, L844–L854. [Google Scholar] [CrossRef]
- Looi, K.; Troy, N.M.; Garratt, L.W.; Iosifidis, T.; Bosco, A.; Buckley, A.G.; Ling, K.M.; Martinovich, K.M.; Kicic-Starcevich, E.; Shaw, N.C.; et al. Effect of human rhinovirus infection on airway epithelium tight junction protein disassembly and transepithelial permeability. Null 2016, 42, 380–395. [Google Scholar] [CrossRef]
- Maška, M.; Ulman, V.; Svoboda, D.; Matula, P.; Matula, P.; Ederra, C.; Urbiola, A.; España, T.; Venkatesan, S.; Balak, D.M.; et al. A benchmark for comparison of cell tracking algorithms. Bioinformatics 2014, 30, 1609–1617. [Google Scholar] [CrossRef]
- Roth, M.; Bäuml, M.; Nevatia, R.; Stiefelhagen, R. Robust multi-pose face tracking by multi-stage tracklet association. In Proceedings of the 21st International Conference on Pattern Recognition (ICPR2012), Tsukuba, Japan, 11–15 November 2012; pp. 1012–1016. [Google Scholar]
- Wang, G.; Wang, Y.; Zhang, H.; Gu, R.; Hwang, J.-N. Exploit the Connectivity: Multi-Object Tracking with TrackletNet. In Proceedings of the 27th ACM International Conference on Multimedia, Nice, France, 21–25 October 2019; Association for Computing Machinery: New York, NY, USA, 2019; pp. 482–490. [Google Scholar]
- Ma, C.; Yang, C.; Yang, F.; Zhuang, Y.; Zhang, Z.; Jia, H.; Xie, X. Trajectory Factory: Tracklet Cleaving and Re-Connection by Deep Siamese Bi-GRU for Multiple Object Tracking. In Proceedings of the 2018 IEEE International Conference on Multimedia and Expo (ICME), San Diego, CA, USA, 23–27 July 2018; pp. 1–6. [Google Scholar]
- Magnusson, K.E.G.; Jaldén, J.; Gilbert, P.M.; Blau, H.M. Global Linking of Cell Tracks Using the Viterbi Algorithm. IEEE Trans. Med. Imaging 2015, 34, 911–929. [Google Scholar] [CrossRef]
- Magnusson, K. klasma/BaxterAlgorithms. 2021. Available online: https://github.com/klasma/BaxterAlgorithms (accessed on 17 September 2021).
- Wojke, N.; Bewley, A.; Paulus, D. Simple online and realtime tracking with a deep association metric. In Proceedings of the 2017 IEEE International Conference on Image Processing (ICIP), Beijing, China, 17–20 September 2017; pp. 3645–3649. [Google Scholar]
- Cordelières, F. Manual Tracking. 2004. Available online: https://imagej.nih.gov/ij/plugins/manual-tracking.html (accessed on 19 October 2021).
- Milan, A.; Leal-Taixe, L.; Reid, I.; Roth, S.; Schindler, K. MOT16: A Benchmark for Multi-Object Tracking. arXiv 2016, arXiv:160300831. [Google Scholar]




| EPIC | DeepSORT | Viterbi | ||||
|---|---|---|---|---|---|---|
| Experiment (Replicate) | Total Cell Tracks | Sampled Cell Tracks | Total Cell Tracks | Sampled Cell Tracks | Total Cell Tracks | Sampled Cell Tracks |
| Accelerated (A) | 127 | 20 | 0 | 0 | 2046 | 20 |
| Accelerated (B) | 236 | 20 | 0 | 0 | 2008 | 20 |
| Accelerated (C) | 211 | 20 | 0 | 0 | 2019 | 20 |
| Control (A) | 783 | 20 | 0 | 0 | 0 | 0 |
| Control (B) | 539 | 20 | 0 | 0 | 37 | 4 |
| Control (C) | 586 | 20 | 0 | 0 | 0 | 0 |
| Delayed (A) | 146 | 20 | 0 | 0 | 1883 | 20 |
| Delayed (B) | 725 | 20 | 0 | 0 | 2726 | 20 |
| Delayed (C) | 1006 | 20 | 0 | 0 | 13 | 2 |
| EPIC | DeepSORT | Viterbi | |
|---|---|---|---|
| Total Running Time | 20 min | 30 h | 3 h |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gwatimba, A.; Rosenow, T.; Stick, S.M.; Kicic, A.; Iosifidis, T.; Karpievitch, Y.V. AI-Driven Cell Tracking to Enable High-Throughput Drug Screening Targeting Airway Epithelial Repair for Children with Asthma. J. Pers. Med. 2022, 12, 809. https://doi.org/10.3390/jpm12050809
Gwatimba A, Rosenow T, Stick SM, Kicic A, Iosifidis T, Karpievitch YV. AI-Driven Cell Tracking to Enable High-Throughput Drug Screening Targeting Airway Epithelial Repair for Children with Asthma. Journal of Personalized Medicine. 2022; 12(5):809. https://doi.org/10.3390/jpm12050809
Chicago/Turabian StyleGwatimba, Alphons, Tim Rosenow, Stephen M. Stick, Anthony Kicic, Thomas Iosifidis, and Yuliya V. Karpievitch. 2022. "AI-Driven Cell Tracking to Enable High-Throughput Drug Screening Targeting Airway Epithelial Repair for Children with Asthma" Journal of Personalized Medicine 12, no. 5: 809. https://doi.org/10.3390/jpm12050809
APA StyleGwatimba, A., Rosenow, T., Stick, S. M., Kicic, A., Iosifidis, T., & Karpievitch, Y. V. (2022). AI-Driven Cell Tracking to Enable High-Throughput Drug Screening Targeting Airway Epithelial Repair for Children with Asthma. Journal of Personalized Medicine, 12(5), 809. https://doi.org/10.3390/jpm12050809






