Personalized Self-Monitoring of Energy Balance through Integration in a Web-Application of Dietary, Anthropometric, and Physical Activity Data
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Protocol
2.2. Wearables and Devices
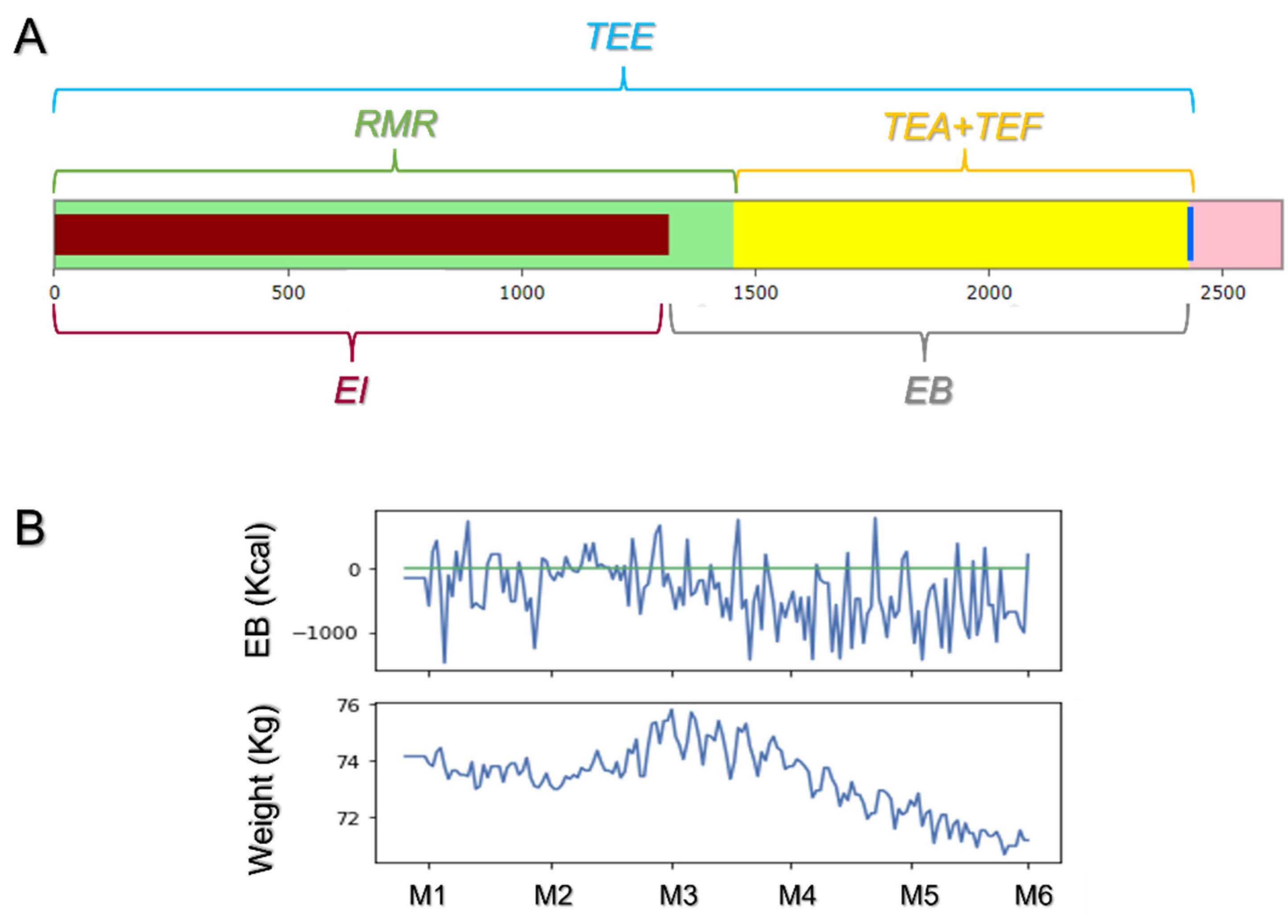
2.3. Web-App Development and Estimation of Personalized Energy Balance (EB)
- (a)
- Data Collection
- Data from wearables were fetched through the ZEPP API®.
- Food and other activities not included in the Smartband (home activities, music playing, driving, etc.) were provided in-person through a digital diary.
- (b)
- Data StorageFetched data underwent anonymization and storage into a NoSQL database (MongoDB®).
- (c)
- Data Analysis and Visualization
2.4. Statistics
3. Results
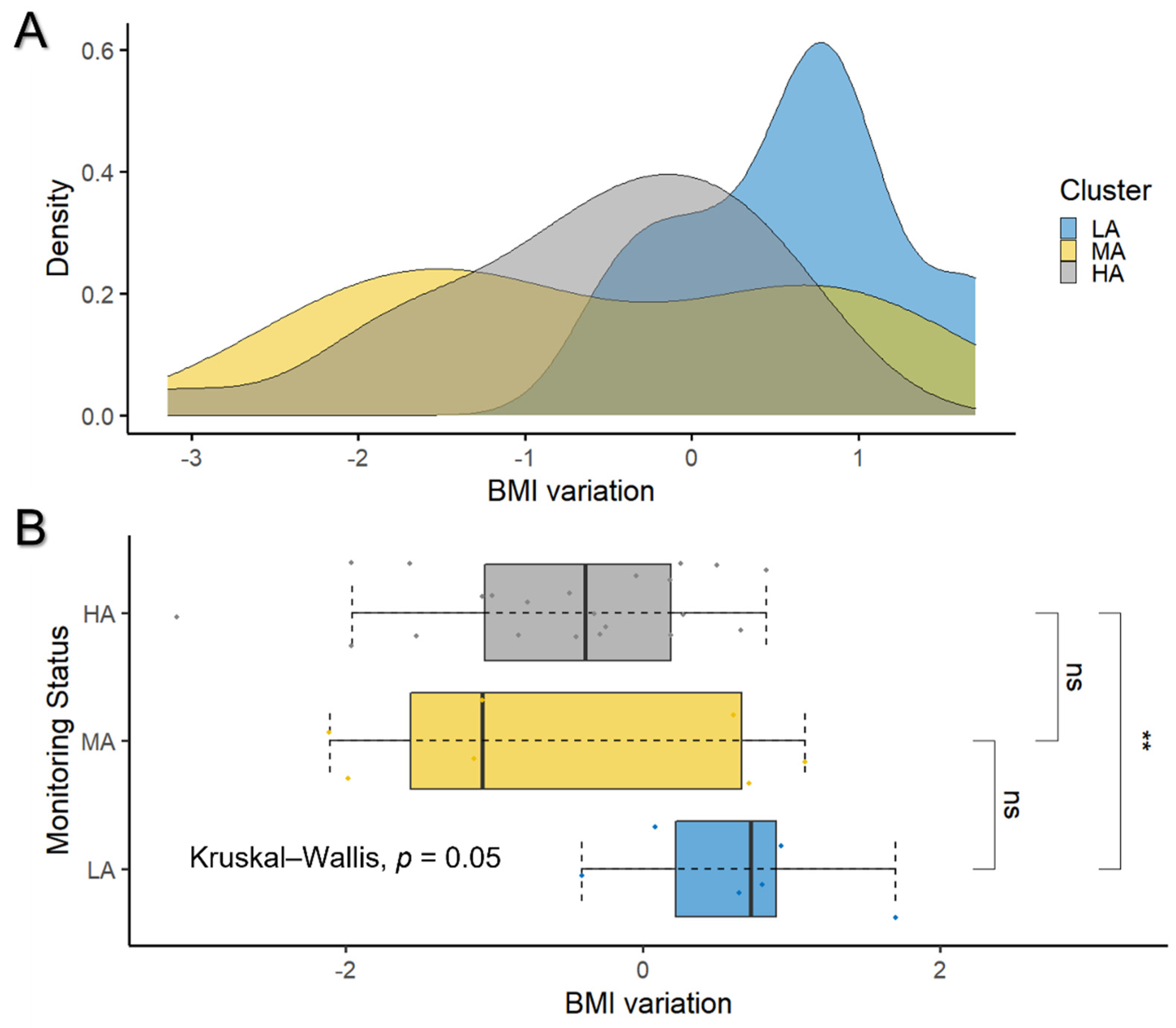
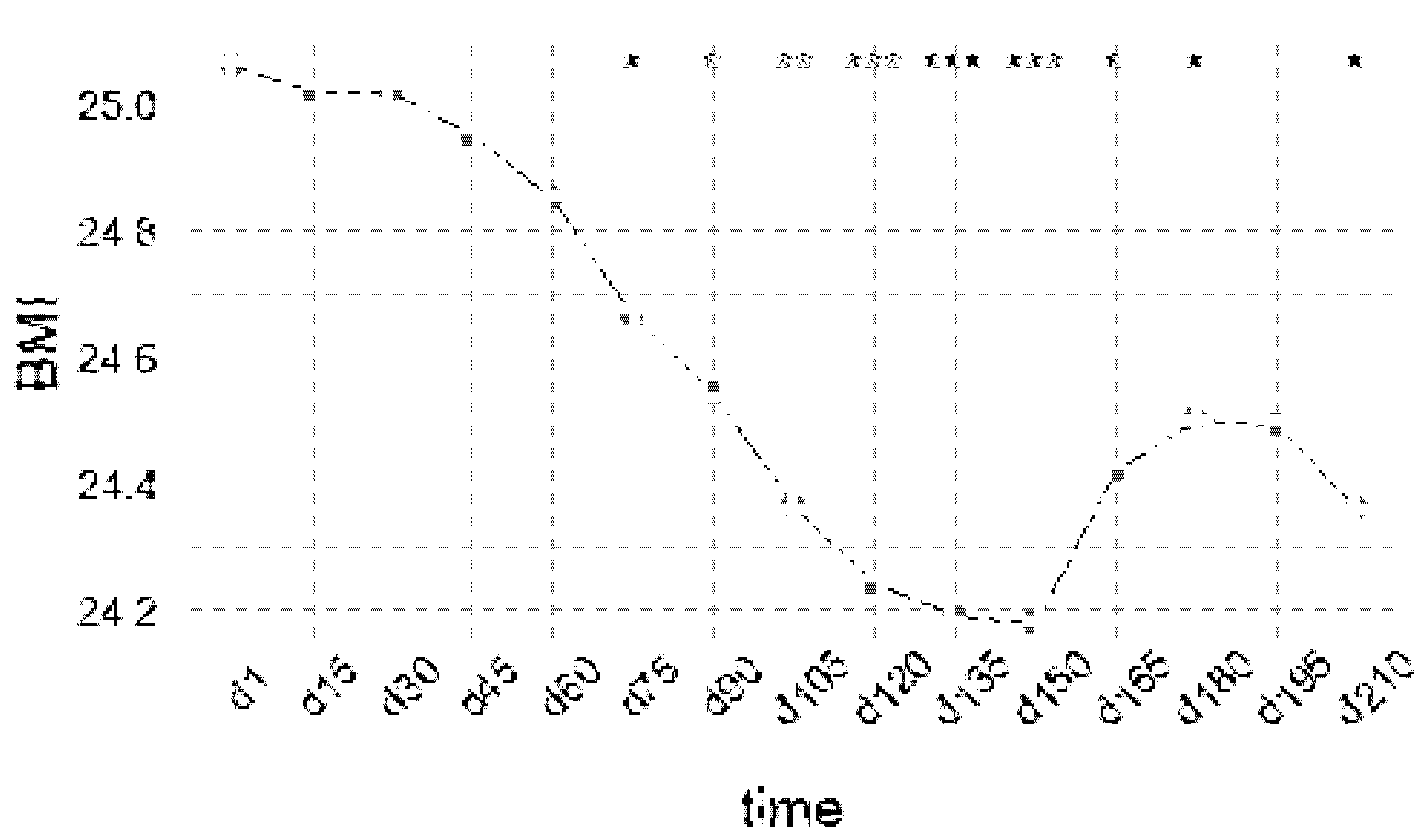
3.1. Overall Effect of Self-Monitoring on BMI Variation
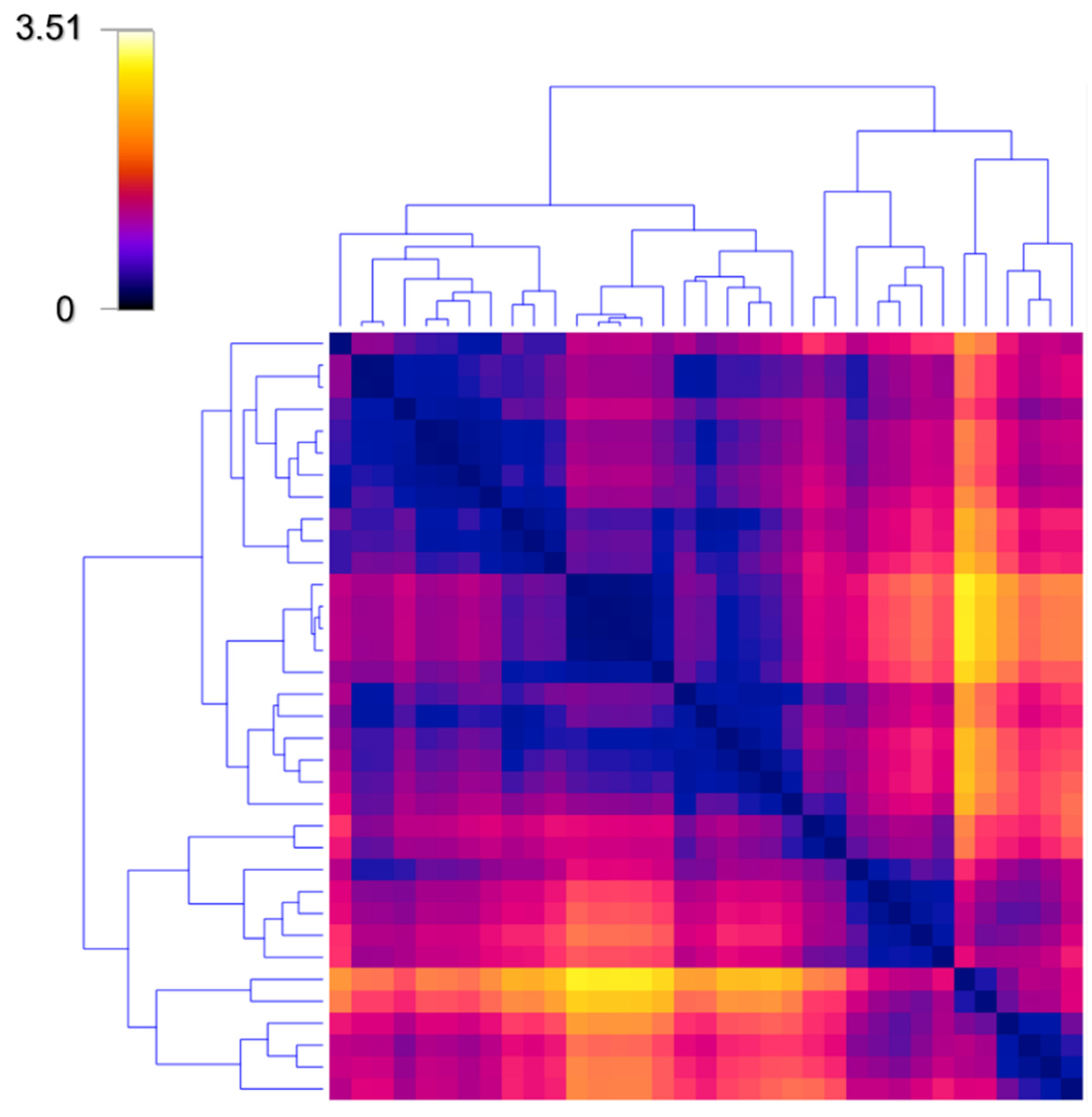
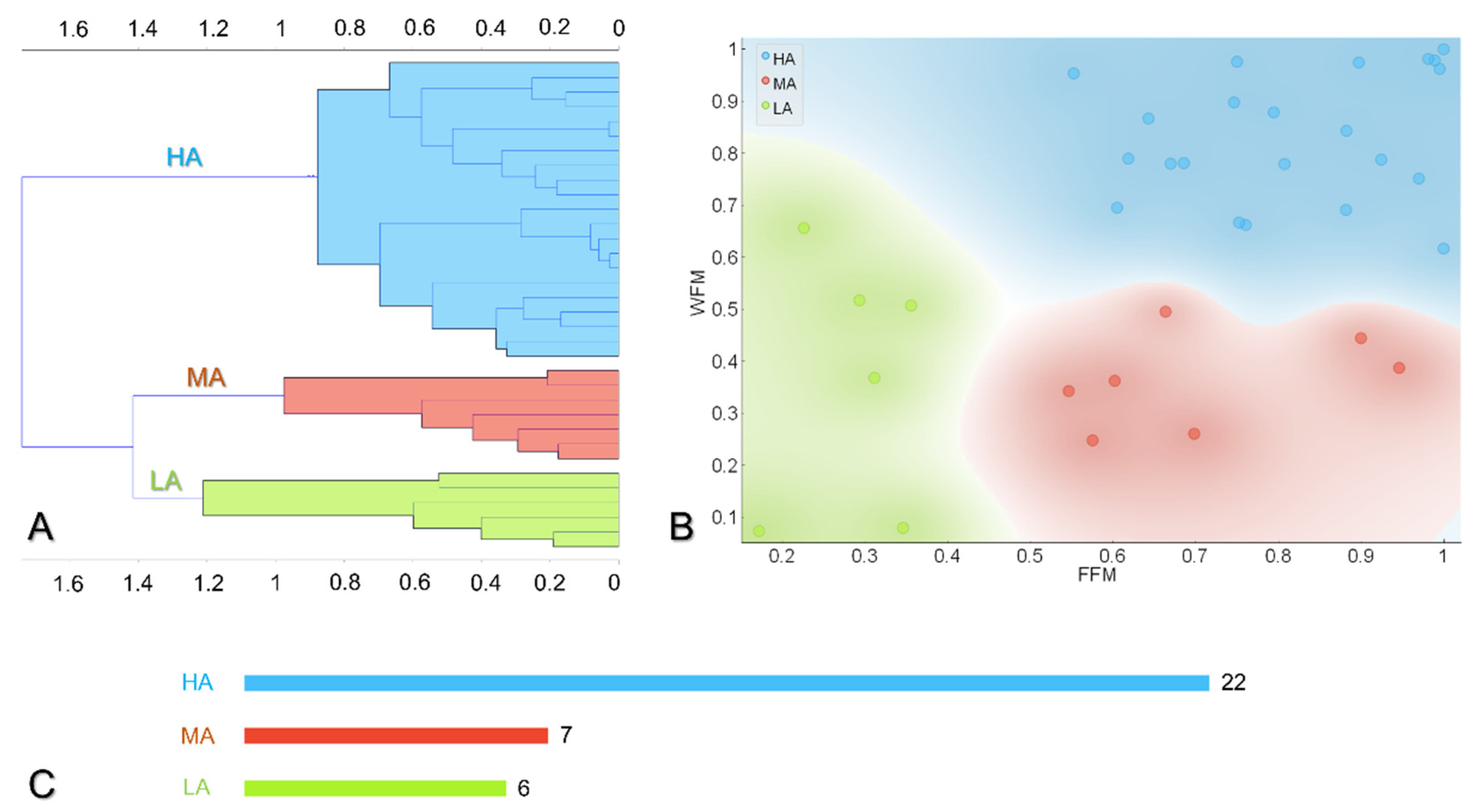
3.2. Hierarchical Clustering Reveals Three Main Behaviors towards Digital Self-Monitoring
3.3. Description of the Subgroups
3.4. Determination of the Average Time and Optimal Adherence Required to Achieve a Significant Weight Loss
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- K Y, S.K.; R Bhat, P.K.; Sorake, C.J. Double trouble: A pandemic of obesity and COVID-19. Lancet Gastroenterol. Hepatol. 2021, 6, 608. [Google Scholar] [CrossRef]
- Gallus, S.; Lugo, A.; Murisic, B.; Bosetti, C.; Boffetta, P.; La Vecchia, C. Overweight and Obesity in 16 European Countries. Eur. J. Nutr. 2015, 54, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Burke, L.E.; Wang, J.; Sevick, M.A. Self-Monitoring in Weight Loss: A Systematic Review of the Literature. J. Am. Diet. Assoc. 2011, 111, 92–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, R.; Kassavou, A.; Sutton, S. Does Self-Monitoring Diet and Physical Activity Behaviors Using Digital Technology Support Adults with Obesity or Overweight to Lose Weight? A Systematic Literature Review with Meta-Analysis. Obes. Rev. 2021, 22, e13306. [Google Scholar] [CrossRef] [PubMed]
- Smart Wearable Devices in Cardiovascular Care: Where We Are and How to Move Forward-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/33664502/ (accessed on 21 February 2022).
- Hao, Y.; Ma, X.-K.; Zhu, Z.; Cao, Z.-B. Validity of Wrist-Wearable Activity Devices for Estimating Physical Activity in Adolescents: Comparative Study. JMIR Mhealth Uhealth 2021, 9, e18320. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.Y.; Ravussin, E. Analysis of Energy Metabolism in Humans: A Review of Methodologies. Mol. Metab. 2016, 5, 1057–1071. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.L.; Hopkins, C.M.; Brooks, T.L.; Bennett, G.G. Comparing Self-Monitoring Strategies for Weight Loss in a Smartphone App: Randomized Controlled Trial. JMIR Mhealth Uhealth 2019, 7, e12209. [Google Scholar] [CrossRef] [PubMed]
- Harvey, J.; Krukowski, R.; Priest, J.; West, D. Log Often, Lose More: Electronic Dietary Self-Monitoring for Weight Loss. Obesity 2019, 27, 380–384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, M.L.; Wakayama, L.N.; Bennett, G.G. Self-Monitoring via Digital Health in Weight Loss Interventions: A Systematic Review Among Adults with Overweight or Obesity. Obesity 2021, 29, 478–499. [Google Scholar] [CrossRef] [PubMed]
- Bianchetti, G.; Spirito, M.D.; Maulucci, G. Unsupervised Clustering of Multiparametric Fluorescent Images Extends the Spectrum of Detectable Cell Membrane Phases with Sub-Micrometric Resolution. Biomed. Opt. Express 2020, 11, 5728–5744. [Google Scholar] [CrossRef] [PubMed]
- Köhn, H.-F.; Hubert, L.J. Hierarchical Cluster Analysis. In Wiley StatsRef: Statistics Reference Online; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015; pp. 1–13. ISBN 978-1-118-44511-2. [Google Scholar]
- Payne, J.E.; Turk, M.T.; Kalarchian, M.A.; Pellegrini, C.A. Adherence to Mobile-App-Based Dietary Self-Monitoring—Impact on Weight Loss in Adults. Obes. Sci. Pract. 2021. [Google Scholar] [CrossRef]
- Maulucci, G.; Cohen, O.; Daniel, B.; Sansone, A.; Petropoulou, P.I.; Filou, S.; Spyridonidis, A.; Pani, G.; De Spirito, M.; Chatgilialoglu, C.; et al. Fatty Acid-Related Modulations of Membrane Fluidity in Cells: Detection and Implications. Free Radical. Res. 2016, 50, S40–S50. [Google Scholar] [CrossRef] [Green Version]
- Bianchetti, G.; Viti, L.; Scupola, A.; Di Leo, M.; Tartaglione, L.; Flex, A.; De Spirito, M.; Pitocco, D.; Maulucci, G. Erythrocyte Membrane Fluidity as a Marker of Diabetic Retinopathy in Type 1 Diabetes Mellitus. Eur. J. Clin. Investig. 2021, 51, e13455. [Google Scholar] [CrossRef]
- Cordelli, E.; Maulucci, G.; De Spirito, M.; Rizzi, A.; Pitocco, D.; Soda, P. A Decision Support System for Type 1 Diabetes Mellitus Diagnostics Based on Dual Channel Analysis of Red Blood Cell Membrane Fluidity. Comput. Methods Programs Biomed. 2018, 162, 263–271. [Google Scholar] [CrossRef]
- Bianchetti, G.; Azoulay-Ginsburg, S.; Keshet-Levy, N.Y.; Malka, A.; Zilber, S.; Korshin, E.E.; Sasson, S.; De Spirito, M.; Gruzman, A.; Maulucci, G. Investigation of the Membrane Fluidity Regulation of Fatty Acid Intracellular Distribution by Fluorescence Lifetime Imaging of Novel Polarity Sensitive Fluorescent Derivatives. Int. J. Mol. Sci. 2021, 22, 3106. [Google Scholar] [CrossRef] [PubMed]
- Bianchetti, G.; Di Giacinto, F.; Pitocco, D.; Rizzi, A.; Rizzo, G.E.; De Leva, F.; Flex, A.; di Stasio, E.; Ciasca, G.; De Spirito, M.; et al. Red Blood Cells Membrane Micropolarity as a Novel Diagnostic Indicator of Type 1 and Type 2 Diabetes. Anal. Chim. Acta X 2019, 3, 100030. [Google Scholar] [CrossRef]
- Maulucci, G.; Cohen, O.; Daniel, B.; Ferreri, C.; Sasson, S. The Combination of Whole Cell Lipidomics Analysis and Single Cell Confocal Imaging of Fluidity and Micropolarity Provides Insight into Stress-Induced Lipid Turnover in Subcellular Organelles of Pancreatic Beta Cells. Molecules 2019, 24, 3742. [Google Scholar] [CrossRef] [Green Version]
| Monitoring Status Group | Post-Hoc Comparison | |||||||
|---|---|---|---|---|---|---|---|---|
| Variable | Overall (n = 35 1) | LA (n = 6 1) | MA (n = 7 1) | HA (n = 22 1) | p-Value 2 (Anova) | HA-LA | LA-MA | HA-MA |
| Sex | 0.2 | |||||||
| Female | 23/35 (66%) | 2/6 (33%) | 6/7 (86%) | 15/22 (68%) | ||||
| Male | 12/35 (34%) | 4/6 (67%) | 1/7 (14%) | 7/22 (32%) | ||||
| Age | 37 ± 12 | 37 ± 6 | 29 ± 6 | 39 ± 14 | 0.10 | |||
| Duration of the study [d] | 160 ± 53 | 157 ± 58 | 140 ± 50 | 168 ± 54 | 0.3 | |||
| Average Daily Steps | 8320 ± 2648 | 7172 ± 3379 | 8928 ± 2821 | 8440 ± 2418 | 0.4 | |||
| Starting BMI | 24.6 ± 3.8 | 24.02 ± 3.1 | 23.7 ± 3.9 | 25.2 ± 4.0 | 0.5 | |||
| BMI variation | −0.4 ± 1.1 | 0.6 ± 0.7 | −0.6 ± 1.3 | −0.6 ± 0.9 | 0.050 | 0.007 (**) | 0.07 | 0.96 |
| Weight Frequency Monitoring (WFM) | 0.66 ± 0.27 | 0.37 ± 0.24 | 0.6 ± 0.09 | 0.83 ± 0.12 | <0.001 | 0.004 (**) | 0.97 | <0.001 (***) |
| Food Frequency Monitoring (FFM) | 0.70 ± 0.24 | 0.28 ± 0.07 | 0.70 ± 0.16 | 0.81 ± 0.15 | <0.001 | <0.001 (***) | <0.001 (***) | 0.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianchetti, G.; Abeltino, A.; Serantoni, C.; Ardito, F.; Malta, D.; De Spirito, M.; Maulucci, G. Personalized Self-Monitoring of Energy Balance through Integration in a Web-Application of Dietary, Anthropometric, and Physical Activity Data. J. Pers. Med. 2022, 12, 568. https://doi.org/10.3390/jpm12040568
Bianchetti G, Abeltino A, Serantoni C, Ardito F, Malta D, De Spirito M, Maulucci G. Personalized Self-Monitoring of Energy Balance through Integration in a Web-Application of Dietary, Anthropometric, and Physical Activity Data. Journal of Personalized Medicine. 2022; 12(4):568. https://doi.org/10.3390/jpm12040568
Chicago/Turabian StyleBianchetti, Giada, Alessio Abeltino, Cassandra Serantoni, Federico Ardito, Daniele Malta, Marco De Spirito, and Giuseppe Maulucci. 2022. "Personalized Self-Monitoring of Energy Balance through Integration in a Web-Application of Dietary, Anthropometric, and Physical Activity Data" Journal of Personalized Medicine 12, no. 4: 568. https://doi.org/10.3390/jpm12040568
APA StyleBianchetti, G., Abeltino, A., Serantoni, C., Ardito, F., Malta, D., De Spirito, M., & Maulucci, G. (2022). Personalized Self-Monitoring of Energy Balance through Integration in a Web-Application of Dietary, Anthropometric, and Physical Activity Data. Journal of Personalized Medicine, 12(4), 568. https://doi.org/10.3390/jpm12040568











