The Australian Reproductive Genetic Carrier Screening Project (Mackenzie’s Mission): Design and Implementation
Abstract
1. Background
2. Methods
2.1. Funding and Ethics Committee Approval
2.2. Aims
- to assess program outcomes, including, the offer and uptake of RGCS, frequency of increased chance couples, and their reproductive decisions and outcomes;
- to understand the screening experience by measuring psychosocial impacts, including anxiety, factors influencing decision-making, decisional conflict and regret;
- to evaluate the implementation of RGCS by identifying possible barriers and enablers for key stakeholders, including people offered screening, healthcare providers (HCPs), and clinical and laboratory services delivering RGCS;
- to assess the health economic implications of RGCS;
- to understand the bioethical aspects of RGCS and explore the ethical issues arising during the implementation and evaluation of the program.
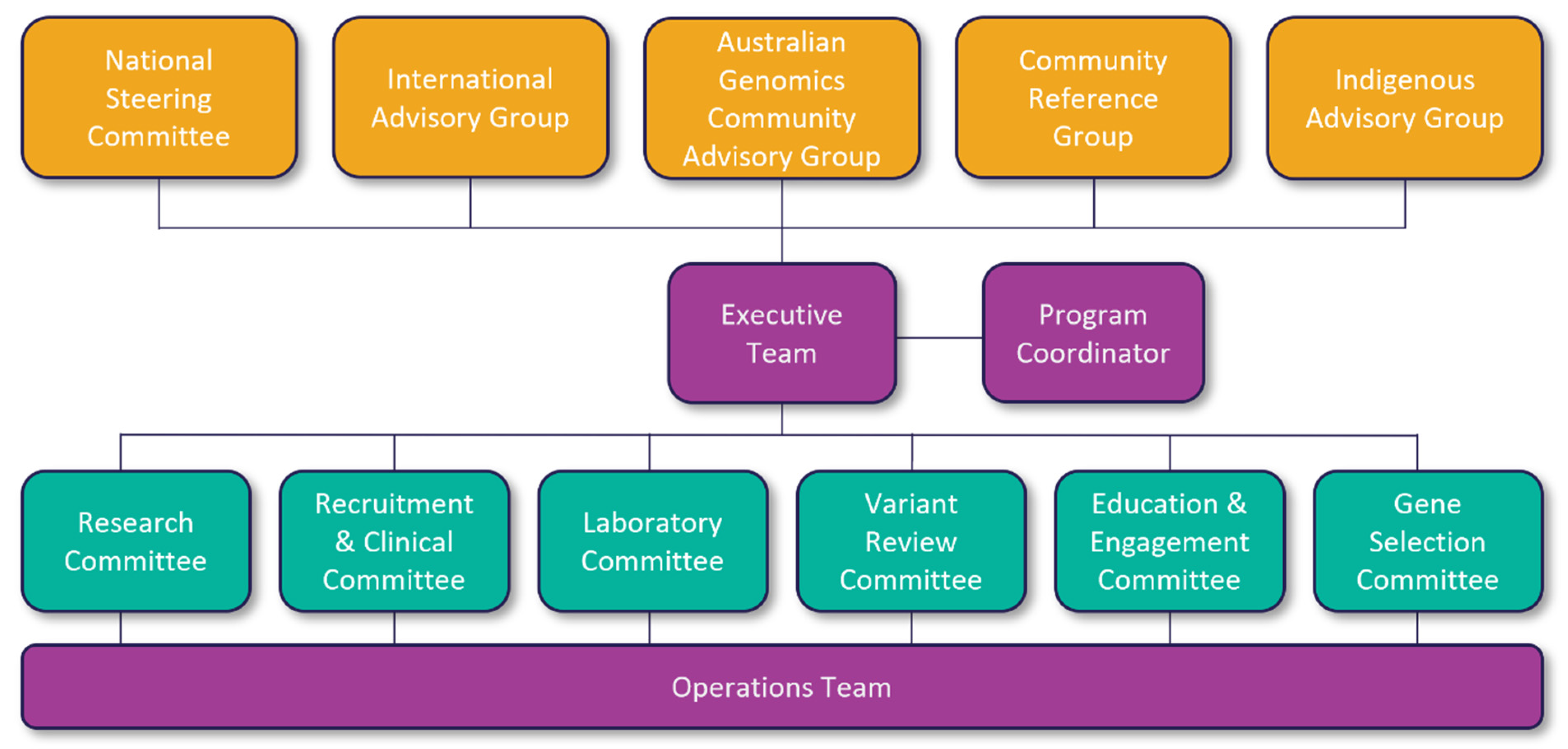
2.3. Study Governance
2.4. Study Design
Evaluation of the Mackenzie’s Mission Reproductive Genetic Carrier Screening Program
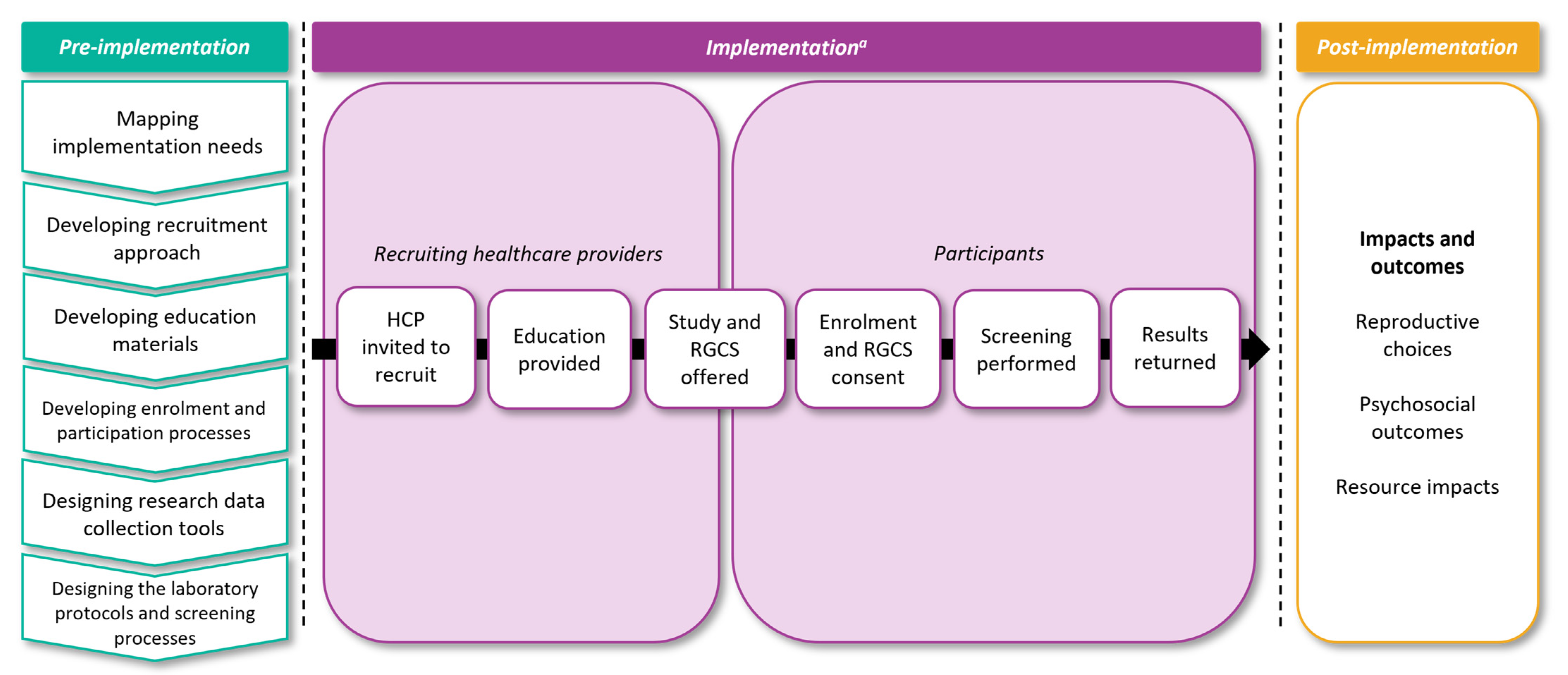
2.5. Phase 1—Pre-Implementation: Study Design and Development
2.5.1. Mapping Implementation Needs
2.5.2. Developing the Recruitment Approach
2.5.3. Developing Education Materials
2.5.4. Developing the Enrollment and Participation Process
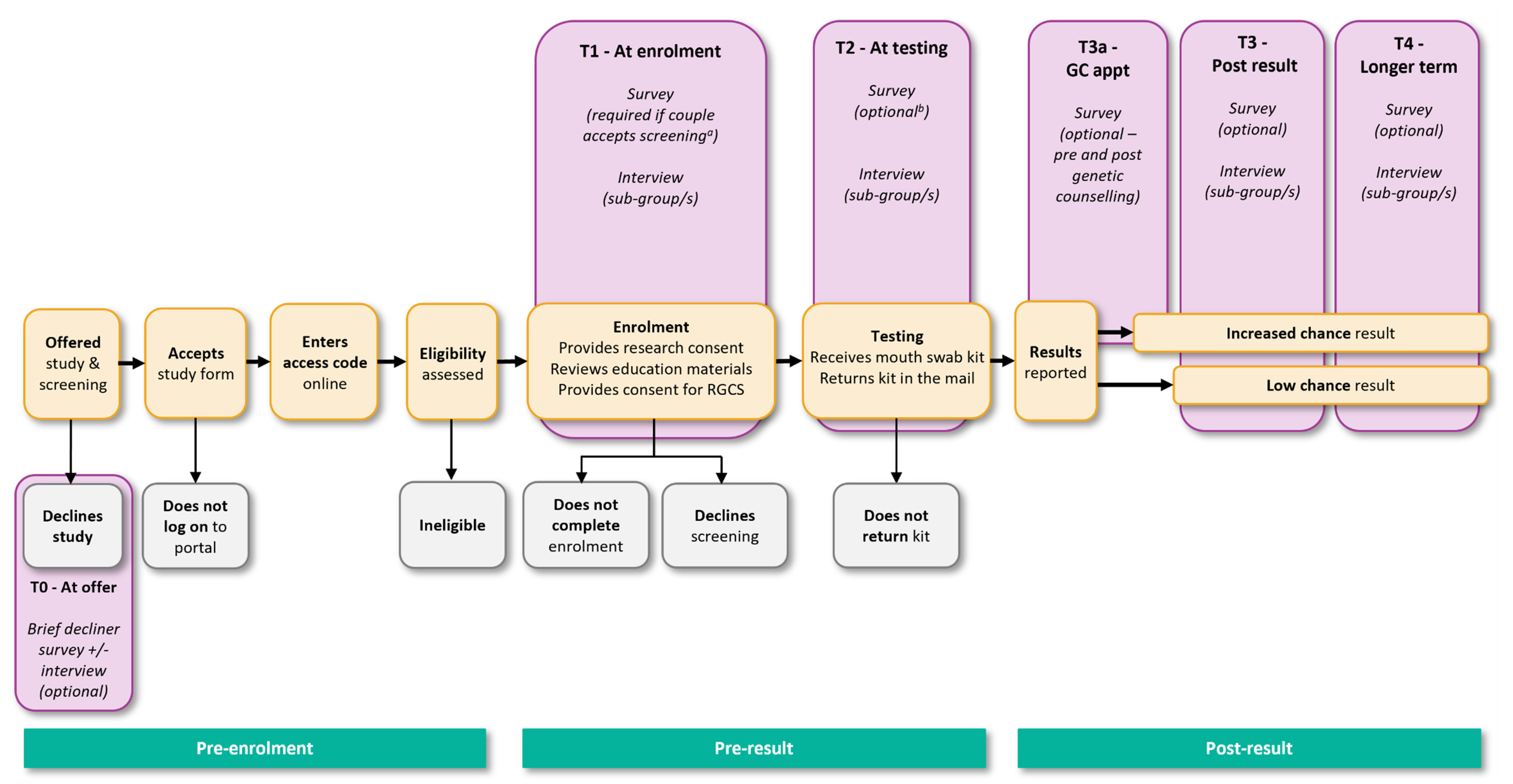
2.5.5. Designing the Data Collection Tools
Data Collection Tools for Recruiting Healthcare Providers
Data Collection Tools for People Offered Screening
| Stage of Participation | Time Point | Data Collection Method | Focus Area | Detail Including Standard Measure Scales |
|---|---|---|---|---|
| Pre-enrollment | T0 At offer | Survey |
|
|
| Interview |
| |||
| Pre result | T1 At enrollment | Survey |
| |
| Interview |
| |||
| T2 At testing | Survey |
|
| |
| Post result | T3a At genetic counseling | Survey |
|
|
| T3 3 months post result | Survey |
|
| |
| Interview |
| |||
| T4 12 months post result | Survey |
|
| |
| Interview |
|
Data Collection Tools for the Study Team
2.5.6. Designing the Reproductive Genetic Carrier Screen and Establishing Screening Processes
2.6. Phase 2—Implementation
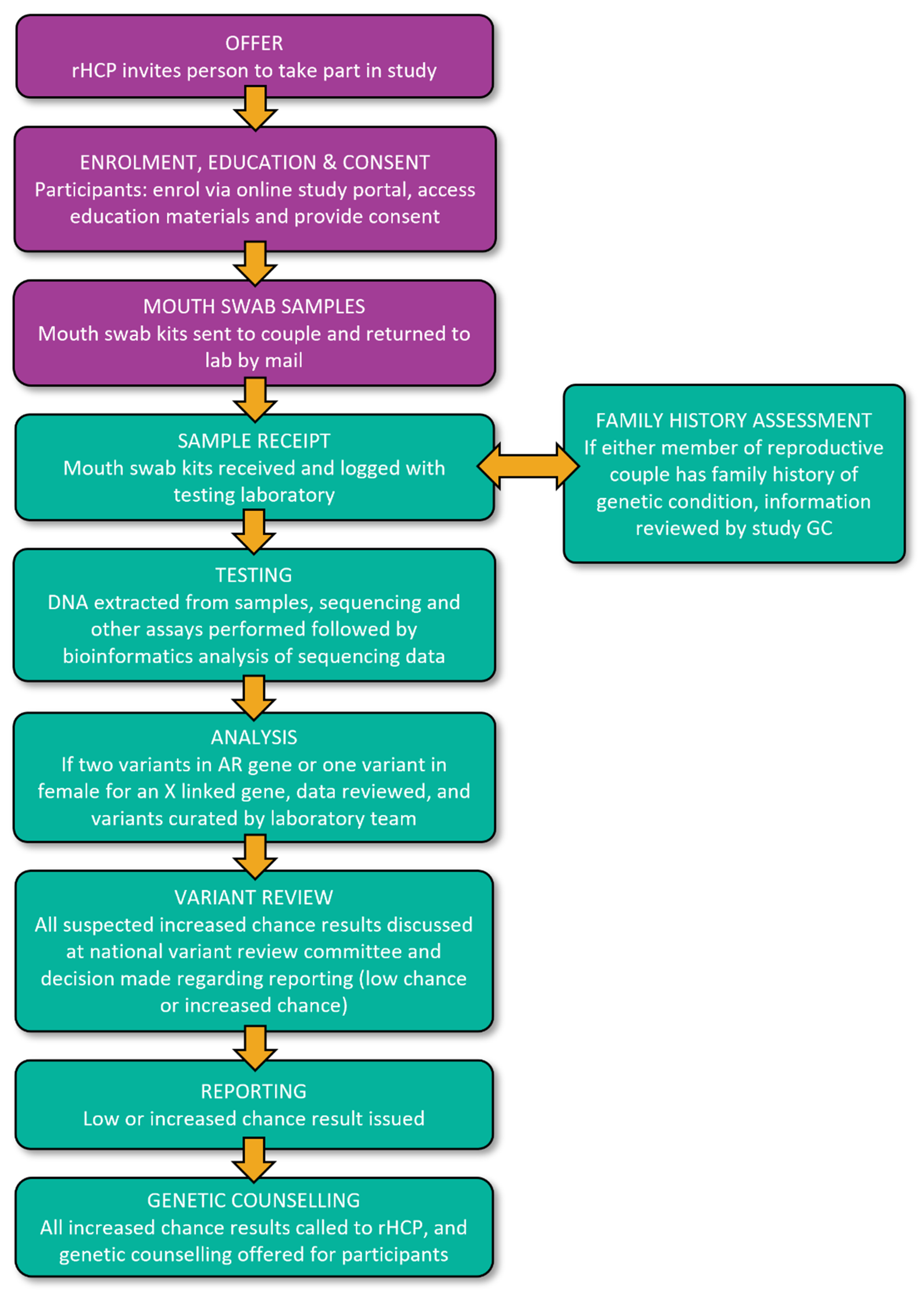
2.6.1. Healthcare Provider Recruitment
2.6.2. Inviting People to Take Part in Mackenzie’s Mission
2.6.3. Enrollment
2.6.4. Laboratory Testing
2.6.5. Management of Results
2.7. Phase 3—Post-Implementation: Assessing Outcomes
2.7.1. Proposed Analysis
Quantitative Analysis
Implementation Science
Health Economic Analysis
Qualitative Analysis
2.7.2. Use and Analysis of Genomic Data
3. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Delatycki, M.B.; Laing, N.; Moore, S.; Emery, J.; Archibald, A.D.; Massie, J.; Kirk, E. Preconception and antenatal carrier screening for genetic conditions: ‘The critical role of general practitioners’. Aust. J. Gen. Pract. 2019, 48, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Delatycki, M.B.; Alkuraya, F.; Archibald, A.D.; Castellani, C.; Cornel, M.; Grody, W.W.; Henneman, L.; Ioannides, A.S.; Kirk, E.; Laing, N.; et al. International perspectives on the implementation of reproductive carrier screening. Prenat. Diagn. 2020, 40, 301–310. [Google Scholar] [CrossRef]
- Beauchamp, K.A.; Johansen Taber, K.A.; Muzzey, D. Clinical impact and cost-effectiveness of a 176-condition expanded carrier screen. Genet. Med. 2019, 21, 1948–1957. [Google Scholar] [CrossRef] [PubMed]
- Ropers, H.H. On the future of genetic risk assessment. J. Community Genet. 2012, 3, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Dive, L.; Newson, A.J. Ethics of reproductive genetic carrier screening: From the clinic to the population. Public Health Ethics 2021, 14, 202–217. [Google Scholar] [CrossRef] [PubMed]
- McClaren, B.J.; Metcalfe, S.A.; Amor, D.J.; Aitken, M.; Massie, J. A case for cystic fibrosis carrier testing in the general population. Med. J. Aust. 2011, 194, 208–209. [Google Scholar] [CrossRef] [PubMed]
- Kraft, S.A.; Duenas, D.; Wilfond, B.S.; Goddard, K.A.B. The evolving landscape of expanded carrier screening: Challenges and opportunities. Genet. Med. 2019, 21, 790–797. [Google Scholar] [CrossRef] [PubMed]
- Westemeyer, M.; Saucier, J.; Wallace, J.; Prins, S.A.; Shetty, A.; Malhotra, M.; Demko, Z.P.; Eng, C.M.; Weckstein, L.; Boostanfar, R. Clinical experience with carrier screening in a general population: Support for a comprehensive pan-ethnic approach. Genet. Med. 2020, 22, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Henneman, L.; Borry, P.; Chokoshvili, D.; Cornel, M.C.; van El, C.G.; Forzano, F.; Hall, A.; Howard, H.C.; Janssens, S.; Kayserili, H.; et al. Responsible implementation of expanded carrier screening. Eur. J. Hum. Genet. 2016, 24, e1–e12. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Committee Opinion No. 690 Summary: Carrier Screening in the Age of Genomic Medicine. Obstet. Gynecol. 2017, 129, 595–596. [Google Scholar] [CrossRef] [PubMed]
- The Royal Australian and New Zealand College of Obstetricians and Gynaecologists. In Genetic Carrier Screening; RANZCOG: Melbourne, VIC, Australia, 2019.
- Beard, C.; Amor, D.; Di Pietro, L.; Archibald, A.D. “I’m Healthy, It’s Not Going To Be Me”: Exploring experiences of carriers identified through a population reproductive genetic carrier screening panel in Australia. Am. J. Med. Genet. A 2016, 170, 2052–2059. [Google Scholar] [CrossRef] [PubMed]
- Plantinga, M.; Birnie, E.; Abbott, K.; Sinke, R.; Lucassen, A.; Schuurmans, J.; Kaplan, S.; Verkerk, M.; Ranchor, A.; van Langen, I. Population-based preconception carrier screening: How potential users from the general population view a test for 50 serious diseases. Eur. J. Hum. Genet. EJHG 2016, 24, 1417–1423. [Google Scholar] [CrossRef]
- Schuurmans, J.; Birnie, E.; Ranchor, A.V.; Abbott, K.M.; Fenwick, A.; Lucassen, A.; Berger, M.Y.; Verkerk, M.; van Langen, I.M.; Plantinga, M. GP-provided couple-based expanded preconception carrier screening in the Dutch general population: Who accepts the test-offer and why? Eur. J. Hum. Genet. 2020, 28, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Ong, R.; Howting, D.; Rea, A.; Christian, H.; Charman, P.; Molster, C.; Ravenscroft, G.; Laing, N.G. Measuring the impact of genetic knowledge on intentions and attitudes of the community towards expanded preconception carrier screening. J. Med. Genet. 2018, 55, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, L.; Delatycki, M.; Massie, J.; Hodgson, J.; Lewis, S. “Suddenly Having two Positive People who are Carriers is a Whole New Thing”—Experiences of Couples Both Identified as Carriers of Cystic Fibrosis Through a Population-Based Carrier Screening Program in Australia. J. Genet. Couns. 2015, 24, 987–1000. [Google Scholar] [CrossRef]
- Tardif, J.; Pratte, A.; Laberge, A.M. Experience of carrier couples identified through a population-based carrier screening pilot program for four founder autosomal recessive diseases in Saguenay-Lac-Saint-Jean. Prenat. Diagn. 2018, 38, 67–74. [Google Scholar] [CrossRef]
- Archibald, A.D.; Hickerton, C.; Jaques, A.; Wake, S.; Cohen, J.; Metcalfe, S. “It’s about having the choice”: Stakeholder perceptions of population-based genetic carrier screening for fragile X syndrome. Am. J. Med. Genet. A 2013, 161, 48–58. [Google Scholar] [CrossRef]
- Metcalfe, S.; Martyn, M.; Ames, A.; Anderson, V.; Archibald, A.; Carter, R.; Cohen, J.; Cotter, M.; Dang, W.; Delatycki, M.; et al. Informed decision making and psychosocial outcomes in pregnant and nonpregnant women offered population fragile X carrier screening. Genet. Med. 2017, 19, 1346–1355. [Google Scholar] [CrossRef]
- Archibald, A.D.; Smith, M.; Burgess, T.; Scarff, K.; Elliott, J.; Hunt, C.; McDonald, Z.; Barns-Jenkins, C.; Holt, C.; Sandoval, K.; et al. Reproductive genetic carrier screening for cystic fibrosis, fragile X syndrome, and spinal muscular atrophy in Australia: Outcomes of 12,000 tests. Genet. Med. 2018, 20, 513–523. [Google Scholar] [CrossRef]
- Leibowitz, R.; Lewis, S.; Massie, J.; Emery, J.D.; Smith, M.; Delatycki, M.B.; Archibald, A.D. Reproductive genetic carrier screening for cystic fibrosis, fragile X syndrome and spinal muscular atrophy: Patterns of community and healthcare provider participation in a Victorian screening program. Aust. J. Prim. Care 2022, in press. [Google Scholar] [CrossRef]
- Robson, S.J.; Caramins, M.; Saad, M.; Suthers, G. Socioeconomic status and uptake of reproductive carrier screening in Australia. Aust. N. Z. J. Obstet. Gynaecol. 2020, 60, 976–979. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie’s Mission. Available online: https://www.mackenziesmission.org.au/ (accessed on 1 June 2022).
- Casella, R. Mackenzie’s Mission; Allen & Unwin: Sydney, NSW, Australia, 2020. [Google Scholar]
- Australian Genomics. Available online: https://www.australiangenomics.org.au/ (accessed on 1 June 2022).
- Dive, L.; Archibald, A.D.; Newson, A.J. Ethical considerations in gene selection for reproductive carrier screening. Hum. Genet. 2021, 141, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Dive, L.; Newson, A.J. Ethical issues in reproductive genetic carrier screening. Med. J. Aust. 2020, 214, 165. [Google Scholar] [CrossRef]
- Dive, L.; Newson, A.J. Reproductive carrier screening: Responding to the eugenics critique. J. Med. Ethics 2021. Published online first. [Google Scholar] [CrossRef] [PubMed]
- Newson, A.J.; Dive, L. Taking seriousness seriously in genomic health. Eur. J. Hum. Genet. 2022, 30, 160–169. [Google Scholar] [CrossRef]
- Newson, A.J.; Dive, L.; Cini, J.; Hurley, E.; Farrar, M.A. Ethical aspects of the changing landscape for spinal muscular atrophy management in Australia. Aust. J. Gen. Pract. 2022, 51, 131–135. [Google Scholar] [CrossRef]
- Best, S.; Long, J.; Theodorou, T.; Hatem, S.; Lake, R.; Archibald, A.D.; Freeman, L.; Braithwaite, J. Health practitioners’ perceptions of the barriers and enablers to the implementation of reproductive genetic carrier screening: A systematic review. Prenat. Diagn. 2021, 41, 708–719. [Google Scholar] [CrossRef] [PubMed]
- Taylor, N.; Parveen, S.; Robins, V.; Slater, B.; Lawton, R. Development and initial validation of the Influences on Patient Safety Behaviours Questionnaire. Implement. Sci. 2013, 8, 81. [Google Scholar] [CrossRef][Green Version]
- Michie, S.; Johnston, M.; Abraham, C.; Lawton, R.; Parker, D.; Walker, A. Making psychological theory useful for implementing evidence based practice: A consensus approach. BMJ Qual. Saf. 2005, 14, 26–33. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Australian Bureau of Statistics. Births, Australia, 2016. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/Previousproducts/3301.0Main%20Features12016?opendocument&tabname=Summary&prodno=3301.0&issue=2016&num=&view= (accessed on 18 April 2022).
- Health, A.I.o. Welfare. In Australia’s Mothers and Babies 2016—In Brief; AIHW: Canberra, Australia, 2018. [Google Scholar]
- Archibald, A.D.; Hickerton, C.; Wake, S.; Jaques, A.; Cohen, J.; Metcalfe, S. “It gives them more options”: Preferences for preconception genetic carrier screening for fragile X syndrome in primary healthcare. J. Community Genet. 2016, 7, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Valente, G.M.; Amor, D.J.; Ioannou, L.J.; Archibald, A.D. Factors influencing medical practitioner participation in population carrier screening for cystic fibrosis. Aust. N. Z. J. Obs. Gynaecol. 2020, 61, 232–238. [Google Scholar] [CrossRef]
- Nagle, C.; Gunn, J.; Bell, R.; Lewis, S.; Meiser, B.; Metcalfe, S.; Ukoumunne, O.C.; Halliday, J. Use of a decision aid for prenatal testing of fetal abnormalities to improve women’s informed decision making: A cluster randomised controlled trial [ISRCTN22532458]. BJOG Int. J. Obstet. Gynaecol. 2008, 115, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Nagle, C.; Lewis, S.; Meiser, B.; Metcalfe, S.; Carlin, J.B.; Bell, R.; Gunn, J.; Halliday, J. Evaluation of a decision aid for prenatal testing of fetal abnormalities: A cluster randomised trial [ISRCTN22532458]. BMC Public Health 2006, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- King, E.; Halliday, J.; Archibald, A.D.; Delatycki, M.; Barlow-Stewart, K.; Newson, A.J.; McClaren, B.J. Development and use of the Australian reproductive genetic carrier screening decision aid. Eur. J. Hum. Genet. 2022, 30, 194–202. [Google Scholar] [CrossRef]
- Haas, M.A.; Teare, H.; Prictor, M.; Ceregra, G.; Vidgen, M.E.; Bunker, D.; Kaye, J.; Boughtwood, T. ‘CTRL’: An online, Dynamic Consent and participant engagement platform working towards solving the complexities of consent in genomic research. Eur. J. Hum. Genet. 2021, 29, 687–698. [Google Scholar] [CrossRef]
- Michie, S.; Van Stralen, M.M.; West, R. The behaviour change wheel: A new method for characterising and designing behaviour change interventions. Implement. Sci. 2011, 6, 42. [Google Scholar] [CrossRef]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.; Vagg, P.R.; Jacobs, G.A. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Alo Alto, CA, USA, 1983. [Google Scholar]
- Marteau, T.; Bekker, H. The development of a six-item short-form of the state scale of the Spielberger State—Trait Anxiety Inventory (STAI). Br. J. Clin. Psychol. 1992, 31, 301–306. [Google Scholar] [CrossRef]
- Hawthorne, G.; Richardson, J.; Osbourne, R. The Assessment of Quality of life (AQoL) instrument: A psychometric measure of Health-Related Quality of Life. Qual. Life Res. 1999, 9, 209–224. [Google Scholar] [CrossRef]
- O’Connor, A. User Manual—Decisional Conflict Scale (16 Item Statement Format); Ottawa Hospital Research Institute: Ottawa, Canada, 1993; (updated 2010). [Google Scholar]
- Grant, P.; Pampaka, M.; Payne, K.; Clarke, A.; McAllister, M. Developing a short-form of the Genetic Counselling Outcome Scale: The Genomics Outcome Scale. Eur. J. Med. Genet. 2018, 62, 324–334. [Google Scholar] [CrossRef] [PubMed]
- McAllister, M.; Wood, A.; Dunn, G.; Shiloh, S.; Todd, C. The Genetic Counseling Outcome Scale: A new patient-reported outcome measure for clinical genetics services. Clin. Genet. 2011, 79, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Marteau, T.M.; Dormandy, E.; Michie, S. A measure of informed choice. Health Expect. 2001, 4, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Martyn, M.; Anderson, V.; Archibald, A.; Carter, R.; Cohen, J.; Delatycki, M.; Donath, S.; Emery, J.; Halliday, J.; Hill, M.; et al. Offering fragile X syndrome carrier screening: A prospective mixed-methods observational study comparing carrier screening of pregnant and non-pregnant women in the general population. BMJ Open 2013, 3, e003660. [Google Scholar] [CrossRef]
- Brehaut, J.C.; O’Connor, A.M.; Wood, T.J.; Hack, T.F.; Siminoff, L.; Gordon, E.; Feldman-Stewart, D. Validation of a decision regret scale. Med. Decis. Mak. Int. J. Soc. Med. Decis. Mak. 2003, 23, 281–292. [Google Scholar] [CrossRef]
- Schuurmans, J.; Birnie, E.; van den Heuvel, L.M.; Plantinga, M.; Lucassen, A.; van der Kolk, D.M.; Abbott, K.M.; Ranchor, A.V.; Diemers, A.D.; van Langen, I.M. Feasibility of couple-based expanded carrier screening offered by general practitioners. Eur. J. Hum. Genet. 2019, 27, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Birnie, E.; Schuurmans, J.; Plantinga, M.; Abbott, K.M.; Fenwick, A.; Lucassen, A.; Berger, M.Y.; Van Langen, I.M.; Ranchor, A.V. Couple-based expanded carrier screening provided by general practitioners to couples in the Dutch general population: Psychological outcomes and reproductive intentions. Genet. Med. 2021, 23, 1761–1768. [Google Scholar] [CrossRef]
- Bell, C.J.; Dinwiddie, D.L.; Miller, N.A.; Hateley, S.L.; Ganusova, E.E.; Mudge, J.; Langley, R.J.; Zhang, L.; Lee, C.C.; Schilkey, F.D.; et al. Carrier testing for severe childhood recessive diseases by next-generation sequencing. Sci. Transl. Med. 2011, 3, 65ra64. [Google Scholar] [CrossRef]
- Lynch, F.L.; Himes, P.; Gilmore, M.J.; Morris, E.M.; Schneider, J.L.; Kauffman, T.L.; Shuster, E.; Reiss, J.A.; Dickerson, J.F.; Leo, M.C. Time costs for genetic counseling in preconception carrier screening with genome sequencing. J. Genet. Couns. 2018, 27, 823–833. [Google Scholar] [CrossRef]
- NATA. Available online: https://nata.com.au/ (accessed on 27 July 2022).
- Kirk, E.P.; Ong, R.; Boggs, K.; Hardy, T.; Righetti, S.; Kamien, B.; Roscioli, T.; Amor, D.J.; Bakshi, M.; Chung, C.W.T.; et al. Gene selection for the Australian Reproductive Genetic Carrier Screening Project (“Mackenzie’s Mission”). Eur. J. Hum. Genet. 2021, 29, 79–87. [Google Scholar] [CrossRef]
- Human Genetics Society of Australasia. Population Based Carrier Screening for Cystic Fibrosis; Human Genetics Society of Australasia: Sydney, NSW, Australia, 2013. [Google Scholar]
- Yrigollen, C.; Durbin-Johnson, B.; Gane, L.; Nelson, D.; Hagerman, R.; Hagerman, P.; Tassone, F. AGG interruptions within the maternal FMR1 gene reduce the risk of offspring with fragile X syndrome. Genet. Med. 2012, 14, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Nolin, S.L.; Glicksman, A.; Ersalesi, N.; Dobkin, C.; Brown, W.T.; Cao, R.U.; Blatt, E.; Sah, S.; Latham, G.J.; Hadd, A.G. Fragile X full mutation expansions are inhibited by one or more AGG interruptions in premutation carriers. Genet. Med. 2015, 17, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Nolin, S.L.; Sah, S.; Glicksman, A.; Sherman, S.L.; Allen, E.; Berry-Kravis, E.; Tassone, F.; Yrigollen, C.; Cronister, A.; Jodah, M. Fragile X AGG analysis provides new risk predictions for 45–69 repeat alleles. Am. J. Med. Genet. Part A 2013, 161, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–423. [Google Scholar] [CrossRef] [PubMed]
- Medical Services Advisory Committee. Guidelines for Preparing Assessments for the MSAC. Available online: http://www.msac.gov.au/internet/msac/publishing.nsf/Content/MSAC-Guidelines (accessed on 19 April 2022).
- Damschroder, L.J.; Aron, D.C.; Keith, R.E.; Kirsh, S.R.; Alexander, J.A.; Lowery, J.C. Fostering implementation of health services research findings into practice: A consolidated framework for advancing implementation science. Implement. Sci. 2009, 4, 50. [Google Scholar] [CrossRef]
- Cane, J.; O’Connor, D.; Michie, S. Validation of the theoretical domains framework for use in behaviour change and implementation research. Implement. Sci. 2012, 7, 37. [Google Scholar] [CrossRef]
- Hawthorne, G.; Korn, S.; Richardson, J. Population norms for the AQoL derived from the 2007 Australian National Survey of Mental Health and Wellbeing. Aust. N. Z. J. Public Health 2013, 37, 7–16. [Google Scholar] [CrossRef]
- Sadedin, S.P.; Dashnow, H.; James, P.A.; Bahlo, M.; Bauer, D.C.; Lonie, A.; Lunke, S.; Macciocca, I.; Ross, J.P.; Siemering, K.R.; et al. Cpipe: A shared variant detection pipeline designed for diagnostic settings. Genome Med. 2015, 7, 1–10. [Google Scholar] [CrossRef]
- Sadedin, S.P.; Ellis, J.A.; Masters, S.L.; Oshlack, A. Ximmer: A system for improving accuracy and consistency of CNV calling from exome data. Gigascience 2018, 7, giy112. [Google Scholar] [CrossRef]
- gnomAD. Available online: https://gnomad.broadinstitute.org/ (accessed on 27 July 2022).
- ClinVar. Available online: https://www.ncbi.nlm.nih.gov/clinvar/ (accessed on 27 July 2022).
- Chen, L.; Hadd, A.; Sah, S.; Filipovic-Sadic, S.; Krosting, J.; Sekinger, E.; Pan, R.; Hagerman, P.J.; Stenzel, T.T.; Tassone, F.; et al. An information-rich CGG repeat primed PCR that detects the full range of fragile X expanded alleles and minimizes the need for southern blot analysis. J. Mol. Diagn. 2010, 12, 589–600. [Google Scholar]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
|
|
|
|
|
|
| Timepoint | Data Collection Tool(s) | Main Focus of Data Collection |
|---|---|---|
| Prior to education to become an rHCP | HCP survey 2 |
|
| Eight to 12 weeks after rHCP education | HCP survey 3 HCP interview guide b |
|
| Toward the end of the study | HCP survey 4 |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Archibald, A.D.; McClaren, B.J.; Caruana, J.; Tutty, E.; King, E.A.; Halliday, J.L.; Best, S.; Kanga-Parabia, A.; Bennetts, B.H.; Cliffe, C.C.; et al. The Australian Reproductive Genetic Carrier Screening Project (Mackenzie’s Mission): Design and Implementation. J. Pers. Med. 2022, 12, 1781. https://doi.org/10.3390/jpm12111781
Archibald AD, McClaren BJ, Caruana J, Tutty E, King EA, Halliday JL, Best S, Kanga-Parabia A, Bennetts BH, Cliffe CC, et al. The Australian Reproductive Genetic Carrier Screening Project (Mackenzie’s Mission): Design and Implementation. Journal of Personalized Medicine. 2022; 12(11):1781. https://doi.org/10.3390/jpm12111781
Chicago/Turabian StyleArchibald, Alison D., Belinda J. McClaren, Jade Caruana, Erin Tutty, Emily A. King, Jane L. Halliday, Stephanie Best, Anaita Kanga-Parabia, Bruce H. Bennetts, Corrina C. Cliffe, and et al. 2022. "The Australian Reproductive Genetic Carrier Screening Project (Mackenzie’s Mission): Design and Implementation" Journal of Personalized Medicine 12, no. 11: 1781. https://doi.org/10.3390/jpm12111781
APA StyleArchibald, A. D., McClaren, B. J., Caruana, J., Tutty, E., King, E. A., Halliday, J. L., Best, S., Kanga-Parabia, A., Bennetts, B. H., Cliffe, C. C., Madelli, E. O., Ho, G., Liebelt, J., Long, J. C., Braithwaite, J., Kennedy, J., Massie, J., Emery, J. D., McGaughran, J., ... The Mackenzie’s Mission Study Team. (2022). The Australian Reproductive Genetic Carrier Screening Project (Mackenzie’s Mission): Design and Implementation. Journal of Personalized Medicine, 12(11), 1781. https://doi.org/10.3390/jpm12111781






