Relatives from Hereditary Breast and Ovarian Cancer and Lynch Syndrome Families Forgoing Genetic Testing: Findings from the Swiss CASCADE Cohort
Abstract
1. Introduction
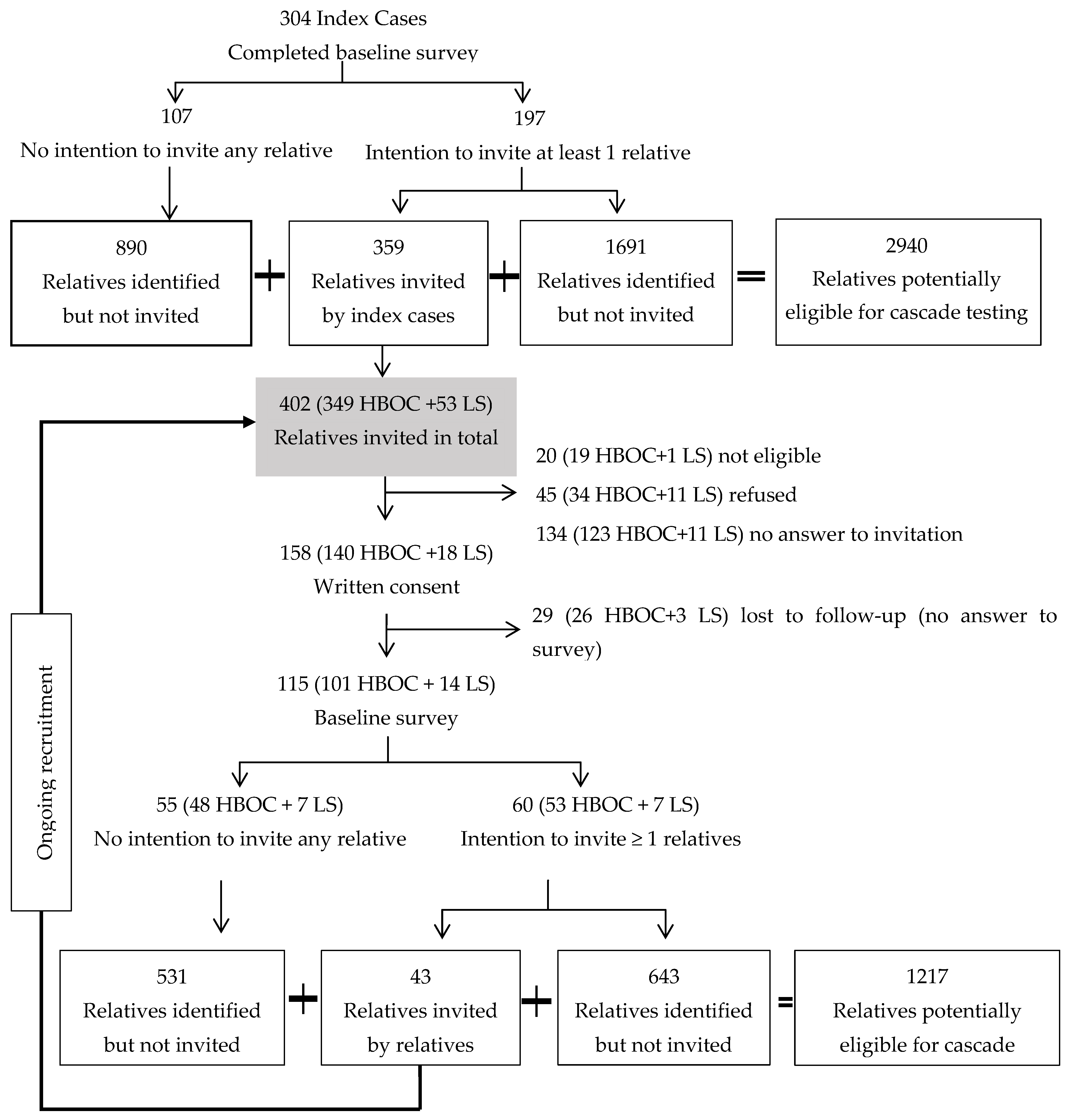
2. Materials and Methods
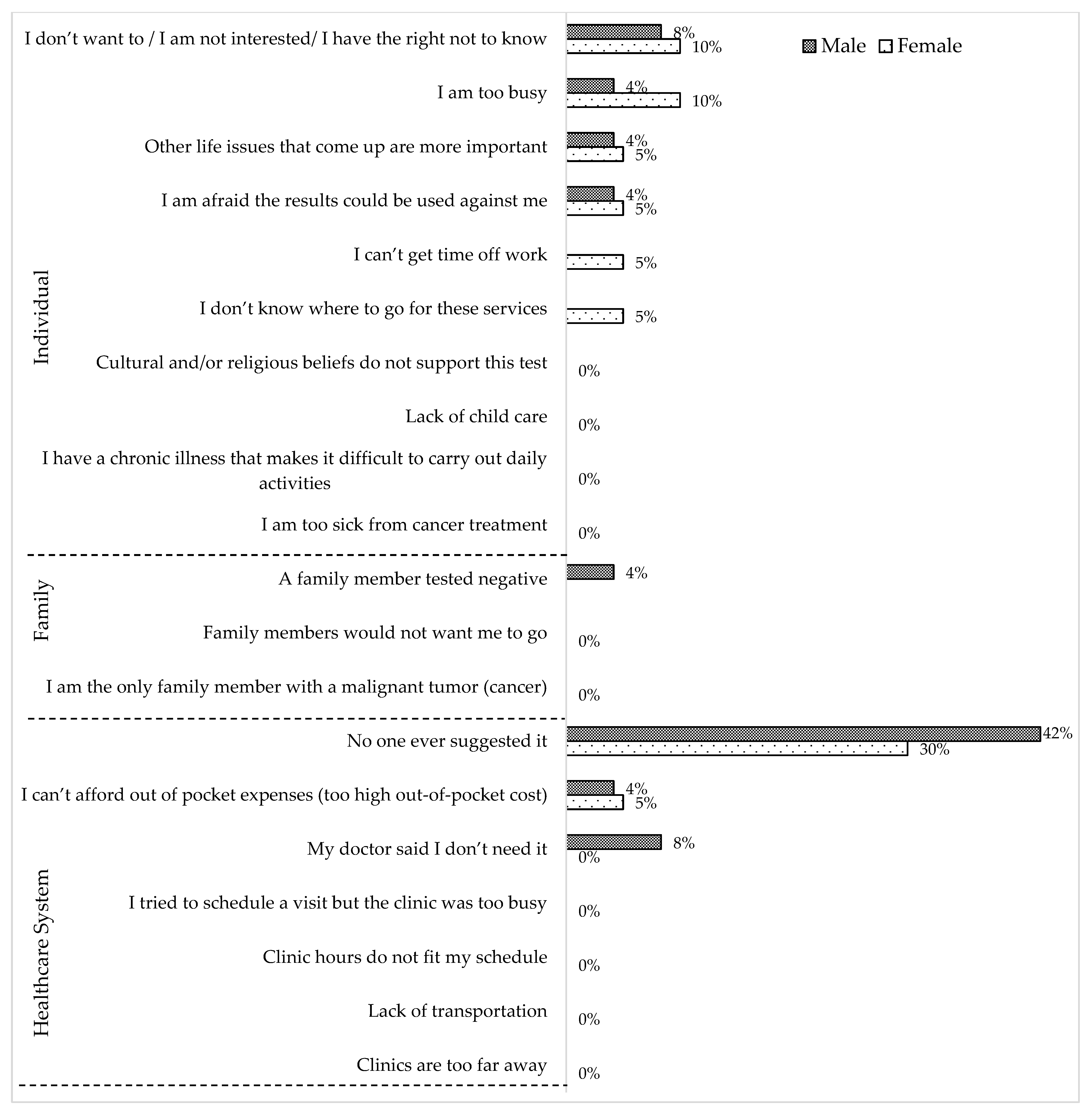
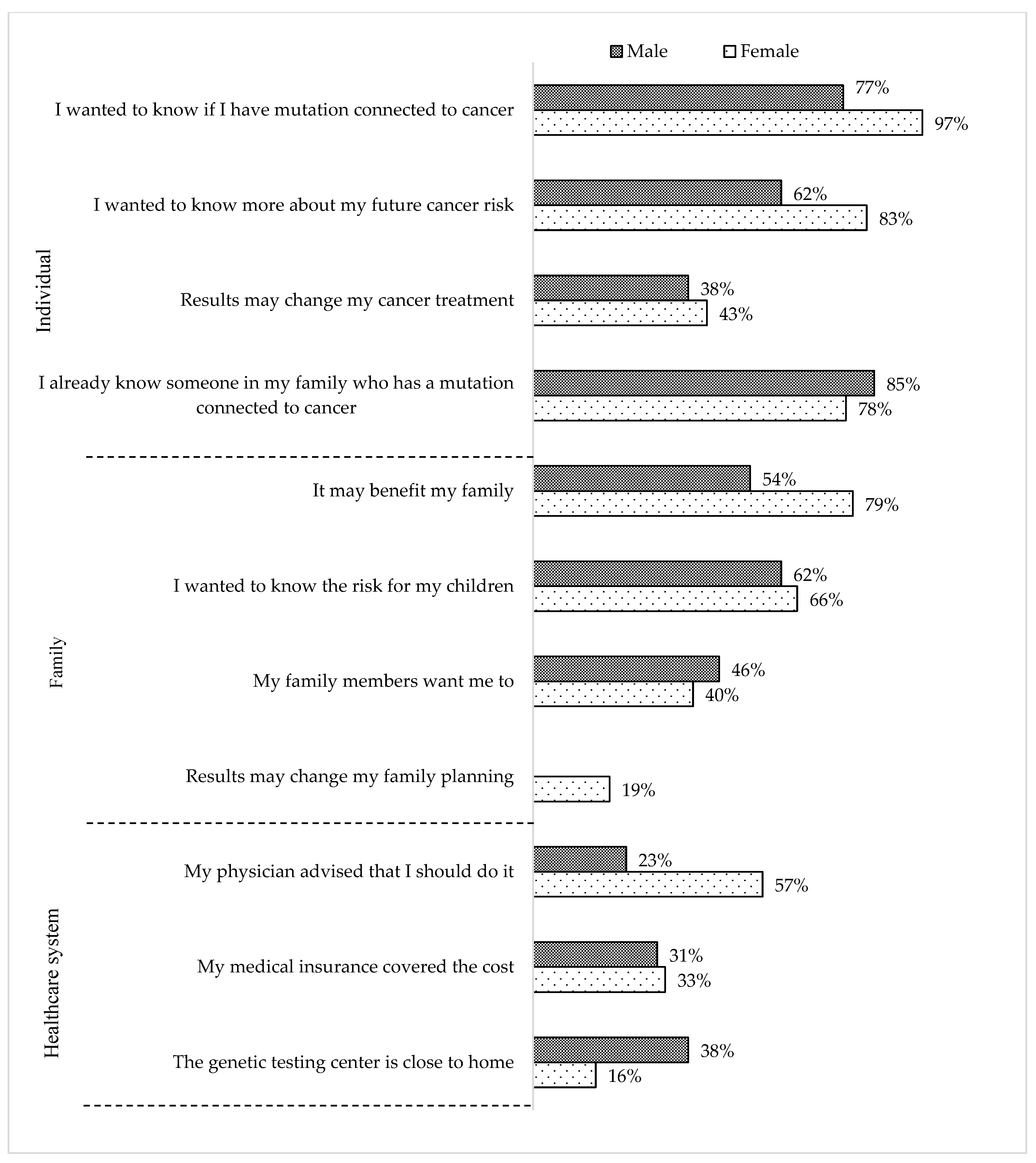
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khoury, M.J.; Dotson, W.D. From genes to public health: Are we ready for DNA-based population screening? Genet. Med. 2021, 23, 996–998. [Google Scholar] [CrossRef]
- Cancer Country Profile 2020: Switzerland. 2020. Available online: https://cdn.who.int/media/docs/default-source/country-profiles/cancer/che-2020.pdf?sfvrsn=5b6b9ced_2&download=true (accessed on 1 October 2022).
- Swiss Cancer Report 2021: Rise in Number of Cancer Patients Coincides with Decline in Mortality. 2021. Available online: https://www.admin.ch/gov/en/start/documentation/media-releases.msg-id-85462.html (accessed on 1 October 2022).
- Lux, M.P.; Fasching, P.A.; Beckmann, M.W. Hereditary breast and ovarian cancer: Review and future perspectives. J. Mol. Med. 2006, 84, 16–28. [Google Scholar] [CrossRef]
- Sinicrope, F.A. Lynch syndrome–associated colorectal cancer. N. Engl. J. Med. 2018, 379, 764–773. [Google Scholar] [CrossRef]
- O’Neill, S.C.; Hamilton, J.G.; Conley, C.C.; Peshkin, B.N.; Sacca, R.; McDonnell, G.A.; Isaacs, C.; Robson, M.E.; Tercyak, K.P. Improving our model of cascade testing for hereditary cancer risk by leveraging patient peer support: A concept report. Hered. Cancer Clin. Pract. 2021, 19, 40. [Google Scholar] [CrossRef]
- Jatoi, I. Risk-reducing options for women with a hereditary breast cancer predisposition. Eur. J. Breast Health 2018, 14, 189. [Google Scholar] [CrossRef]
- Ladabaum, U.; Ford, J.M.; Martel, M.; Barkun, A.N. American Gastroenterological Association technical review on the diagnosis and management of Lynch syndrome. Gastroenterology 2015, 149, 783–813.e20. [Google Scholar] [CrossRef]
- Srinivasan, S.; Won, N.Y.; Dotson, W.D.; Wright, S.T.; Roberts, M.C. Barriers and facilitators for cascade testing in genetic conditions: A systematic review. Eur. J. Hum. Genet. 2020, 28, 1631–1644. [Google Scholar] [CrossRef]
- Allen, C.G.; Roberts, M.; Guan, Y. Exploring predictors of genetic counseling and testing for hereditary breast and ovarian cancer: Findings from the 2015 US National Health Interview Survey. J. Pers. Med. 2019, 9, 26. [Google Scholar] [CrossRef]
- Scott, D.; Friedman, S.; Telli, M.L.; Kurian, A.W. Decision making about genetic testing among women with a personal and family history of breast cancer. JCO Oncol. Pract. 2020, 16, e37–e55. [Google Scholar] [CrossRef]
- Ladd, M.K.; Peshkin, B.N.; Isaacs, C.; Hooker, G.; Willey, S.; Valdimarsdottir, H.; DeMarco, T.; O’Neill, S.; Binion, S.; Schwartz, M.D. Predictors of genetic testing uptake in newly diagnosed breast cancer patients. J. Surg. Oncol. 2020, 122, 134–143. [Google Scholar] [CrossRef]
- Cragun, D.; Bonner, D.; Kim, J.; Akbari, M.; Narod, S.; Gomez-Fuego, A.; Garcia, J.; Vadaparampil, S.; Pal, T. Factors associated with genetic counseling and BRCA testing in a population-based sample of young Black women with breast cancer. Breast Cancer Res. Treat. 2015, 151, 169–176. [Google Scholar] [CrossRef]
- Leenen, C.H.; Den Heijer, M.; van der Meer, C.; Kuipers, E.J.; van Leerdam, M.E.; Wagner, A. Genetic testing for Lynch syndrome: Family communication and motivation. Fam. Cancer 2016, 15, 63–73. [Google Scholar] [CrossRef]
- Childers, K.K.; Maggard-Gibbons, M.; Macinko, J.; Childers, C.P. National distribution of cancer genetic testing in the United States: Evidence for a gender disparity in hereditary breast and ovarian cancer. JAMA Oncol. 2018, 4, 876–879. [Google Scholar] [CrossRef]
- Godard, B.; Pratte, A.; Dumont, M.; Simard-Lebrun, A.; Simard, J. Factors associated with an individual’s decision to withdraw from genetic testing for breast and ovarian cancer susceptibility: Implications for counseling. Genet. Test. 2007, 11, 45–54. [Google Scholar] [CrossRef]
- Gomes, P.; Pietrabissa, G.; Silva, E.R.; Silva, J.; Matos, P.M.; Costa, M.E.; Bertuzzi, V.; Silva, E.; Neves, M.C.; Sales, C.M. Family Adjustment to Hereditary Cancer Syndromes: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 1603. [Google Scholar] [CrossRef]
- de Pinho Rodrigues, A. Influence of Family Communication in the Adjustment to Genetic Cancer Risk: A Review of Reviews. 2020. Available online: https://repositorio-aberto.up.pt/bitstream/10216/131397/2/435327.pdf (accessed on 10 June 2022).
- Mouslim, M.C.; Johnson, R.M.; Dean, L.T. Healthcare system distrust and the breast cancer continuum of care. Breast Cancer Res. Treat. 2020, 180, 33–44. [Google Scholar] [CrossRef]
- Keogh, L.A.; Niven, H.; Rutstein, A.; Flander, L.; Gaff, C.; Jenkins, M. Choosing not to undergo predictive genetic testing for hereditary colorectal cancer syndromes: Expanding our understanding of decliners and declining. J. Behav. Med. 2017, 40, 583–594. [Google Scholar] [CrossRef]
- Foster, C.; Evans, D.G.R.; Eeles, R.; Eccles, D.; Ashley, S.; Brooks, L.; Cole, T.; Cook, J.; Davidson, R.; Gregory, H.; et al. Non-uptake of predictive genetic testing for BRCA1/2 among relatives of known carriers: Attributes, cancer worry, and barriers to testing in a multicenter clinical cohort. Genet. Test. 2004, 8, 23–29. [Google Scholar] [CrossRef]
- Lieberman, S.; Lahad, A.; Tomer, A.; Koka, S.; BenUziyahu, M.; Raz, A.; Levy-Lahad, E. Familial communication and cascade testing among relatives of BRCA population screening participants. Genet. Med. 2018, 20, 1446–1454. [Google Scholar] [CrossRef]
- Caswell-Jin, J.L.; Zimmer, A.D.; Stedden, W.; Kingham, K.E.; Zhou, A.Y.; Kurian, A.W. Cascade genetic testing of relatives for hereditary cancer risk: Results of an online initiative. JNCI-J. Natl. Cancer Inst. 2019, 111, 95–98. [Google Scholar] [CrossRef]
- Courtney, E.; Chok, A.K.-L.; Ang, Z.L.T.; Shaw, T.; Li, S.-T.; Yuen, J.; Ngeow, J. Impact of free cancer predisposition cascade genetic testing on uptake in Singapore. NPJ Genom. Med. 2019, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Frey, M.K.; Kahn, R.M.; Chapman-Davis, E.; Tubito, F.; Pires, M.; Christos, P.; Anderson, S.; Mukherjee, S.; Jordan, B.; Blank, S.V.; et al. Prospective Feasibility Trial of a Novel Strategy of Facilitated Cascade Genetic Testing Using Telephone Counseling. J. Clin. Oncol. 2020, 38, 1389–1397. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.; Cajal, T.Y.; Torres, A.; Darder, E.; Gadea, N.; Velasco, A.; Fortuny, D.; Lopez, C.; Fisas, D.; Brunet, J.; et al. Uptake of predictive testing among relatives of BRCA1 and BRCA2 families: A multicenter study in northeastern Spain. Fam. Cancer 2010, 9, 297–304. [Google Scholar] [CrossRef]
- Whitaker, K.D.; Obeid, E.; Daly, M.B.; Hall, M.J. Cascade Genetic Testing for Hereditary Cancer Risk: An Underutilized Tool for Cancer Prevention. JCO Precis. Oncol. 2021, 5, 1387–1396. [Google Scholar] [CrossRef] [PubMed]
- Bednar, E.M.; Sun, C.C.; McCurdy, S.; Vernon, S.W. Assessing relatives’ readiness for hereditary cancer cascade genetic testing. Genet. Med. 2020, 22, 719–726. [Google Scholar] [CrossRef]
- Griffin, N.E.; Buchanan, T.R.; Smith, S.H.; Leon, A.A.; Meyer, M.F.; Liu, J.X.; Tabak, R.G.; Fuh, K.C.; Thaker, P.H.; Powell, M.A.; et al. Low rates of cascade genetic testing among families with hereditary gynecologic cancer: An opportunity to improve cancer prevention. Gynecol. Oncol. 2020, 156, 140–146. [Google Scholar] [CrossRef]
- Armstrong, N.; Ryder, S.; Forbes, C.; Ross, J.; Quek, R.G. A systematic review of the international prevalence of BRCA mutation in breast cancer. Clin. Epidemiol. 2019, 11, 543–561. [Google Scholar] [CrossRef]
- Abu-Ghazaleh, N.; Kaushik, V.; Gorelik, A.; Jenkins, M.; Macrae, F. Worldwide prevalence of Lynch syndrome in patients with colorectal cancer: Systematic review and meta-analysis. Genet. Med. 2022, 24, 971–985. [Google Scholar] [CrossRef]
- Ayme, A.; Viassolo, V.; Rapiti, E.; Fioretta, G.; Schubert, H.; Bouchardy, C.; Chappuis, P.O.; Benhamou, S. Determinants of genetic counseling uptake and its impact on breast cancer outcome: A population-based study. Breast Cancer Res. Treat. 2014, 144, 379–389. [Google Scholar] [CrossRef]
- De Pietro, C.; Camenzind, P.; Sturny, I.; Crivelli, L.; Edwards-Garavoglia, S.; Spranger, A.; Wittenbecher, F.; Quentin, W. Switzerland Health system review Introduction. Switz. Health Syst. Rev. 2015, 17, 1–18. [Google Scholar]
- Katapodi, M.C.; Viassolo, V.; Caiata-Zufferey, M.; Nikolaidis, C.; Bührer-Landolt, R.; Buerki, N.; Graffeo, R.; Horváth, H.C.; Kurzeder, C.; Rabaglio, M. Cancer predisposition cascade screening for hereditary breast/ovarian cancer and Lynch syndromes in Switzerland: Study protocol. JMIR Res. Protoc. 2017, 6, e184. [Google Scholar] [CrossRef] [PubMed]
- Sarki, M.; Ming, C.; Aissaoui, S.; Burki, N.; Caiata-Zufferey, M.; Erlanger, T.E.; Graffeo-Galbiati, R.; Heinimann, K.; Heinzelmann-Schwarz, V.; Monnerat, C.; et al. Intention to Inform Relatives, Rates of Cascade Testing, and Preference for Patient-Mediated Communication in Families Concerned with Hereditary Breast and Ovarian Cancer and Lynch Syndrome: The Swiss CASCADE Cohort. Cancers 2022, 14, 1636. [Google Scholar] [CrossRef] [PubMed]
- Katapodi, M.C.; Facione, N.C.; Miaskowski, C.; Dodd, M.J.; Waters, C. The influence of social support on breast cancer screening in a multicultural community sample. Oncol. Nurs. Forum. 2002, 29, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Promoting Patient Involvement in Medication Decisions. 2008. Available online: https://archive.ahrq.gov/news/events/conference/2008/Hickam.html (accessed on 16 June 2022).
- Nikolaidis, C.; Duquette, D.; Mendelsohn-Victor, K.E.; Anderson, B.; Copeland, G.; Milliron, K.J.; Merajver, S.D.; Janz, N.K.; Northouse, L.L.; Duffy, S.A. Disparities in genetic services utilization in a random sample of young breast cancer survivors. Genet. Med. 2019, 21, 1363–1370. [Google Scholar] [CrossRef]
- ZipCodeSoft. Postcode Database Switzerland. Switzerland. 2021. Available online: http://www.postcode-distance.com/postcode-database-switzerland (accessed on 20 March 2022).
- The Swiss Group for Clinical Cancer Research (SAKK). Genetic Counselling, Centers in Switzerland. Switzerland. 2021. Available online: https://www.sakk.ch/en/patients/genetic-counseling (accessed on 20 March 2022).
- Das, K.R.; Imon, A. A brief review of tests for normality. Am. J. Theor. Appl. Stat. 2016, 5, 5–12. [Google Scholar]
- Van Buuren, S.; Groothuis-Oudshoorn, K. mice: Multivariate imputation by chained equations in R. J. Stat. Softw. 2011, 45, 1–67. [Google Scholar] [CrossRef]
- Cilluffo, G.; Sottile, G.; La Grutta, S.; Muggeo, V.M. The Induced Smoothed lasso: A practical framework for hypothesis testing in high dimensional regression. Stat. Methods Med. Res. 2020, 29, 765–777. [Google Scholar] [CrossRef]
- Hastie, T.; Qian, J.; Tay, K. An Introduction to Glmnet. 2016. Available online: https://cloud.r-project.org/web/packages/glmnet/vignettes/glmnet.pdf (accessed on 15 February 2022).
- Chappuis, P.O.; Bolliger, B.; Bürki, N.; Buser, K.; Heinimann, K.; Monnerat, C.; Morant, R.; Pagani, O.; Perey, L.; Rabaglio, M. Genetic predisposition to breast and ovarian cancer. Bull. Des Médecins Suisses 2017, 98, 682–684. [Google Scholar]
- Van El, C.G.; Baccolini, V.; Piko, P.; Cornel, M.C. Stakeholder views on active cascade screening for familial hypercholesterolemia. Healthcare 2018, 6, 108. [Google Scholar] [CrossRef]
- Harvey, E.K.; Fogel, C.E.; Peyrot, M.; Christensen, K.D.; Terry, S.F.; McInerney, J.D. Providers’ knowledge of genetics: A survey of 5915 individuals and families with genetic conditions. Genet. Med. 2007, 9, 259–267. [Google Scholar] [CrossRef]
- White, S.; Jacobs, C.; Phillips, J. Mainstreaming genetics and genomics: A systematic review of the barriers and facilitators for nurses and physicians in secondary and tertiary care. Genet. Med. 2020, 22, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Frezzo, T.M.; Rubinstein, W.S.; Dunham, D.; Ormond, K.E. The genetic family history as a risk assessment tool in internal medicine. Genet. Med. 2003, 5, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Beard, C.; Monohan, K.; Cicciarelli, L.; James, P.A. Mainstream genetic testing for breast cancer patients: Early experiences from the Parkville Familial Cancer Centre. Eur. J. Hum. Genet. 2021, 29, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Umans-Eckenhausen, M.A.; Defesche, J.C.; Sijbrands, E.J.; Scheerder, R.L.; Kastelein, J.J. Review of first 5 years of screening for familial hypercholesterolaemia in the Netherlands. Lancet 2001, 357, 165–168. [Google Scholar] [CrossRef]
- Scheinberg, T.; Goodwin, A.; Ip, E.; Linton, A.; Mak, B.; Smith, D.P.; Stockler, M.R.; Strach, M.C.; Tran, B.; Young, A.L. Evaluation of a mainstream model of genetic testing for men with prostate cancer. JCO Oncol. Pract. 2021, 17, e204–e216. [Google Scholar] [CrossRef]
- Bokkers, K.; Vlaming, M.; Engelhardt, E.G.; Zweemer, R.P.; van Oort, I.M.; Kiemeney, L.; Bleiker, E.M.A.; Ausems, M. The Feasibility of Implementing Mainstream Germline Genetic Testing in Routine Cancer Care-A Systematic Review. Cancers 2022, 14, 1059. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.D.; Valdimarsdottir, H.B.; DeMarco, T.A.; Peshkin, B.N.; Lawrence, W.; Rispoli, J.; Brown, K.; Isaacs, C.; O’Neill, S.; Shelby, R.; et al. Randomized trial of a decision aid for BRCA1/BRCA2 mutation carriers: Impact on measures of decision making and satisfaction. Health Psychol. 2009, 28, 11–19. [Google Scholar] [CrossRef]
- White, V.B.; Walsh, K.K.; Foss, K.S.; Amacker-North, L.; Lenarcic, S.; Mcneely, L.; White, R.L., Jr. Genetic testing for hereditary breast cancer: The decision to decline. Am. Surg. 2018, 84, 154–160. [Google Scholar] [CrossRef]
- Salikhanov, I.; Heinimann, K.; Chappuis, P.; Buerki, N.; Graffeo, R.; Heinzelmann, V.; Rabaglio, M.; Taborelli, M.; Wieser, S.; Katapodi, M.C. Swiss cost-effectiveness analysis of universal screening for Lynch syndrome of patients with colorectal cancer followed by cascade genetic testing of relatives. J. Med. Genet. 2021, 59, 824. [Google Scholar] [CrossRef]
| GT (−) n = 44 | GT (+) n = 71 | p Value | ||
|---|---|---|---|---|
| Individual Characteristics | ||||
| Demographics | Age, years | 49 (33.8–67.3) | 47 (35.3–63.8) | 0.26 |
| Male, n (%) | 24 (54.5) | 13 (18.3) | <0.01 a | |
| Female, n (%) | 20 (45.5) | 58 (81.7) | <0.01 | |
| Index case being male, n (%) | 12 (27.3) | 8 (11.3) | 0.05 | |
| HBOC, n (%) | 40 (90.9) | 61(85.9) | 0.62 | |
| LS, n (%) | 4 (9.1) | 10 (14.1) | 0.57 b | |
| More than 12 years of education, n (%) | 25 (56.8) | 45 (63.4) | 0.61 | |
| Employed, n (%) | 27 (61.4) | 43 (60.6) | 0.99 | |
| Married or living with a partner, n (%) | 33 (75.0) | 51 (71.8) | 0.88 | |
| Clinical | Personal history of cancer, n (%) | 3 (6.8) | 20 (28.2) | 0.03 b |
| Costs | Out-of-pocket costs as barrier to medical care, n (%) | 1 (2.3) | 1 (1.4) | 0.99 b |
| Psychological | Perceived risk to get (another) cancer, median (IQR) | 6 (4.5–7.0) | 7 (5.0–8.0) | 0.14 c |
| Relatives’ degree of relationship with the index case d | FDR, n (%) | 30 (68.2) | 48 (67.6) | 0.99 |
| SDR, n (%) | 7 (15.9) | 6 (8.5) | 0.36 | |
| TDR, n (%) | 5 (11.4) | 8 (11.3) | 0.99 | |
| Family characteristics | ||||
| Adults in the family with cancer | Number of adults in the family with cancer, median (IQR) | 1 (1–1.3) | 1 (1–2) | 0.09 a |
| Adult relatives in the family potentially eligible for cascade testing | Number of relatives in the family potentially eligible for cascade testing, median (IQR) | 9.5 (5.8–14.0) | 10 (6–17) | 0.29 a |
| Relatives willing to invite more relatives to the cohort | Number of relatives willing to invite more relatives to the cohort, median (IQR) | 0 (0–0) | 1 (0–4) | <0.01 a |
| Adult relatives tested in the family | Number of relatives tested in the family, median (IQR) | 2 (1–3) | 3 (2–6) | <0.01 a |
| Family support | Family support in illness, median (IQR) | 6.3 (5.8–6.6) | 6.1 (5.5–6.6) | 0.61 b |
| Healthcare system characteristics | ||||
| Coordination of care by specialist, n (%) | 12 (27.3) | 36 (50.7) | 0.02 | |
| ≤2 healthcare providers organize care, n (%) | 36 (81.8) | 68 (95.8) | 0.03 | |
| Shared decision making and patient involvement, median (IQR) | 5.5 (3.5–6.9) | 6.0 (4.0–7.0) | 0.38 c | |
| Distance to the nearest genetic testing center, km, median (IQR) | 10.4 (5.6–15.6.) | 8.9 (3.2–21.8) | 0.85 c | |
| Distance to the index’s case testing center, km, median (IQR) | 27.5 (14.9–69.1) | 21.9 (3.5–63.2) | 0.32 c | |
| OR a | 95% CI | Std. Error | Statistic | p | ||
|---|---|---|---|---|---|---|
| Individual Characteristics | ||||||
| Demographic | Male (ref: female) | 2.79 | 1.10–7.10 | 0.48 | 2.15 | 0.034 |
| Clinical | No cancer diagnosis (ref: having cancer diagnosis) | 4.47 | 1.03–19.42 | 0.75 | 2.00 | 0.048 |
| Family characteristics | ||||||
| Total number of tested relatives in the family | 0.71 | 0.55–0.92 | 0.13 | −2.63 | <0.01 | |
| Healthcare system characteristics | ||||||
| ≤2 healthcare providers organize care (ref: >2 healthcare providers) | 0.23 | 0.04–1.14 | 0.82 | −1.80 | 0.074 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarki, M.; Ming, C.; Aceti, M.; Fink, G.; Aissaoui, S.; Bürki, N.; Graffeo, R.; Heinimann, K.; Caiata Zufferey, M.; Monnerat, C.; et al. Relatives from Hereditary Breast and Ovarian Cancer and Lynch Syndrome Families Forgoing Genetic Testing: Findings from the Swiss CASCADE Cohort. J. Pers. Med. 2022, 12, 1740. https://doi.org/10.3390/jpm12101740
Sarki M, Ming C, Aceti M, Fink G, Aissaoui S, Bürki N, Graffeo R, Heinimann K, Caiata Zufferey M, Monnerat C, et al. Relatives from Hereditary Breast and Ovarian Cancer and Lynch Syndrome Families Forgoing Genetic Testing: Findings from the Swiss CASCADE Cohort. Journal of Personalized Medicine. 2022; 12(10):1740. https://doi.org/10.3390/jpm12101740
Chicago/Turabian StyleSarki, Mahesh, Chang Ming, Monica Aceti, Günther Fink, Souria Aissaoui, Nicole Bürki, Rossella Graffeo, Karl Heinimann, Maria Caiata Zufferey, Christian Monnerat, and et al. 2022. "Relatives from Hereditary Breast and Ovarian Cancer and Lynch Syndrome Families Forgoing Genetic Testing: Findings from the Swiss CASCADE Cohort" Journal of Personalized Medicine 12, no. 10: 1740. https://doi.org/10.3390/jpm12101740
APA StyleSarki, M., Ming, C., Aceti, M., Fink, G., Aissaoui, S., Bürki, N., Graffeo, R., Heinimann, K., Caiata Zufferey, M., Monnerat, C., Rabaglio, M., Zürrer-Härdi, U., Chappuis, P. O., Katapodi, M. C., & the CASCADE Consortium. (2022). Relatives from Hereditary Breast and Ovarian Cancer and Lynch Syndrome Families Forgoing Genetic Testing: Findings from the Swiss CASCADE Cohort. Journal of Personalized Medicine, 12(10), 1740. https://doi.org/10.3390/jpm12101740









