ILB® Attenuates Clinical Symptoms and Serum Biomarkers of Oxidative/Nitrosative Stress and Mitochondrial Dysfunction in Patients with Amyotrophic Lateral Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Oversight
2.2. Patients and Controls
2.3. Clinical Assessment of Patients with ALS Using the ALSFRS-R
2.4. Investigational Medicinal Product (IMP), Dosing and Administration
2.5. Serum Sampling
2.6. List of ALS Patient Samples, Sample Processing and Analyses
2.7. Statistics
3. Results
3.1. Raw Data
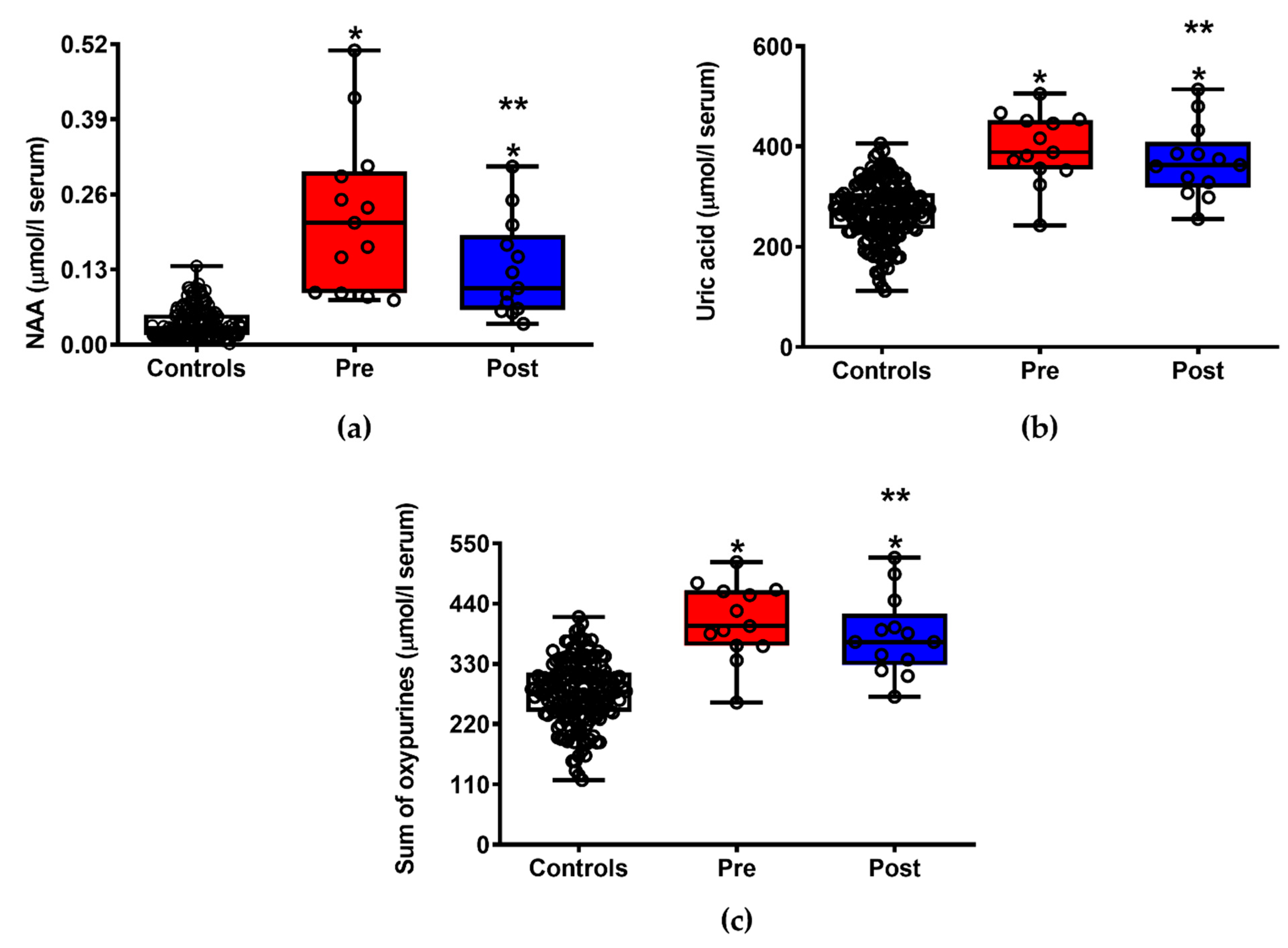
3.2. ILB® Improves Patients’ Clinical Conditions and Decreases Neuronal Damage and Energy Metabolism Impairment
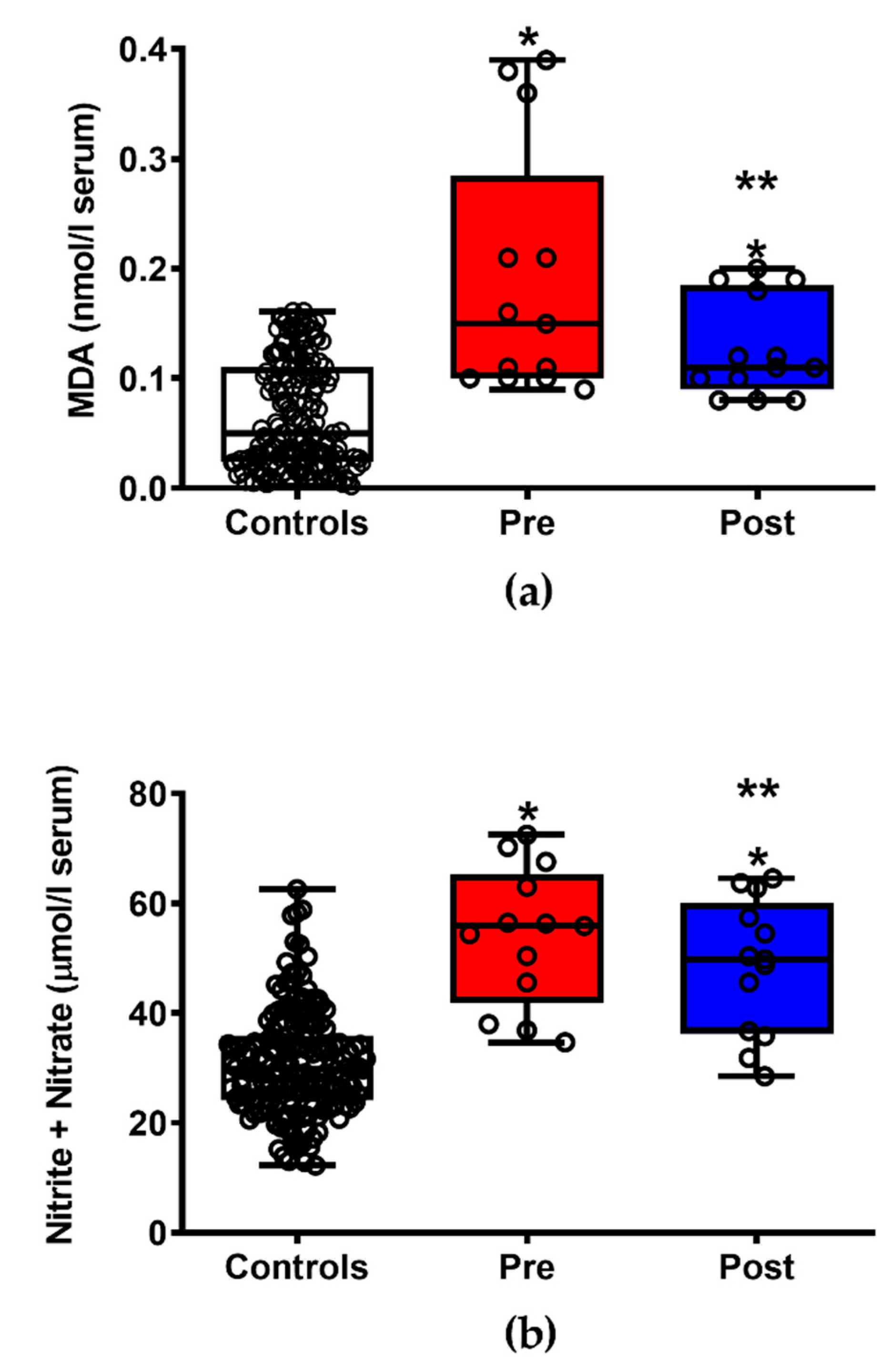
3.3. ILB® Decreases ALS-Related Oxidative/Nitrosative Stress
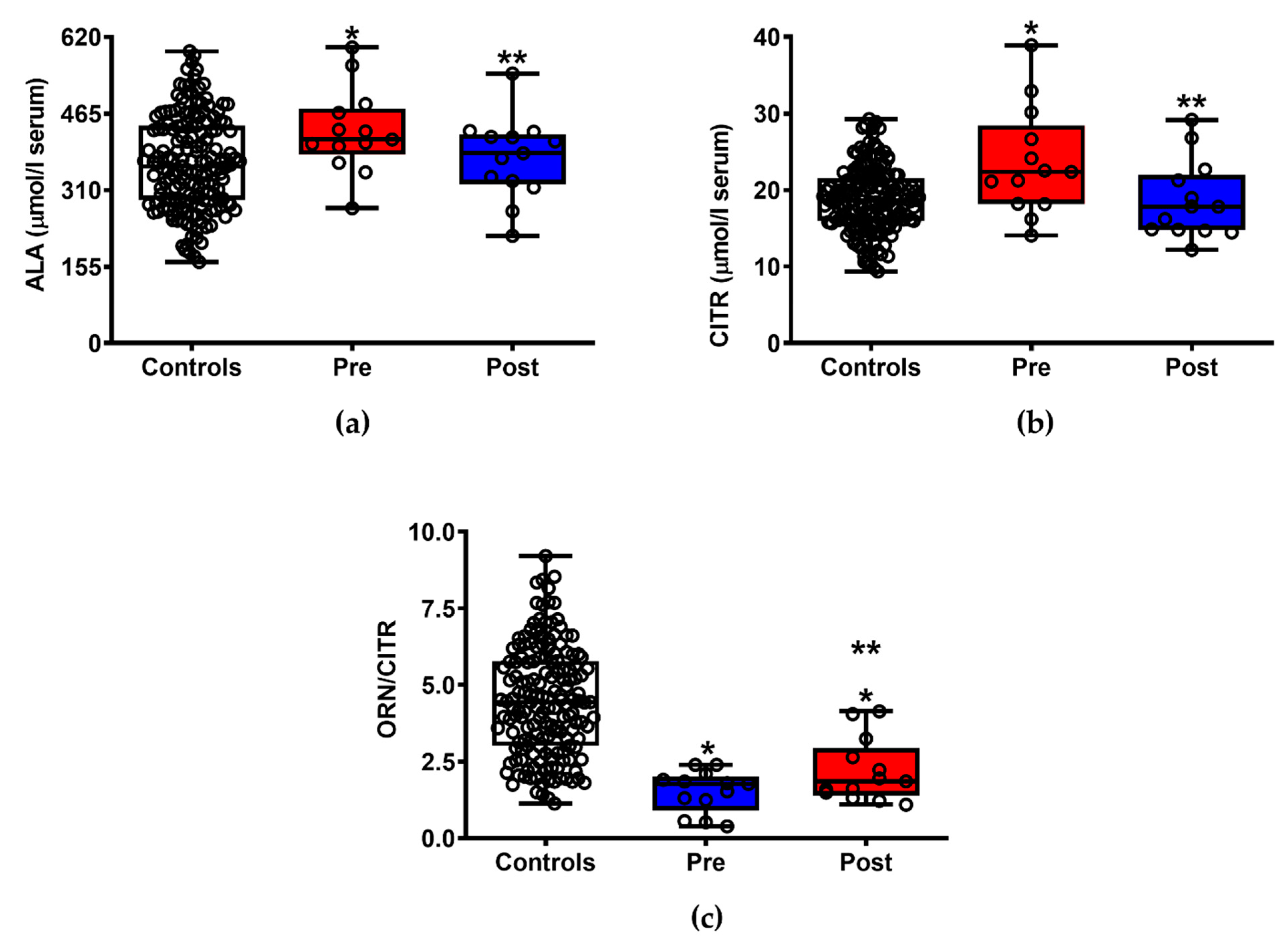
3.4. ILB® Ameliorates ALS-Induced Changes of Serum Amino Acids
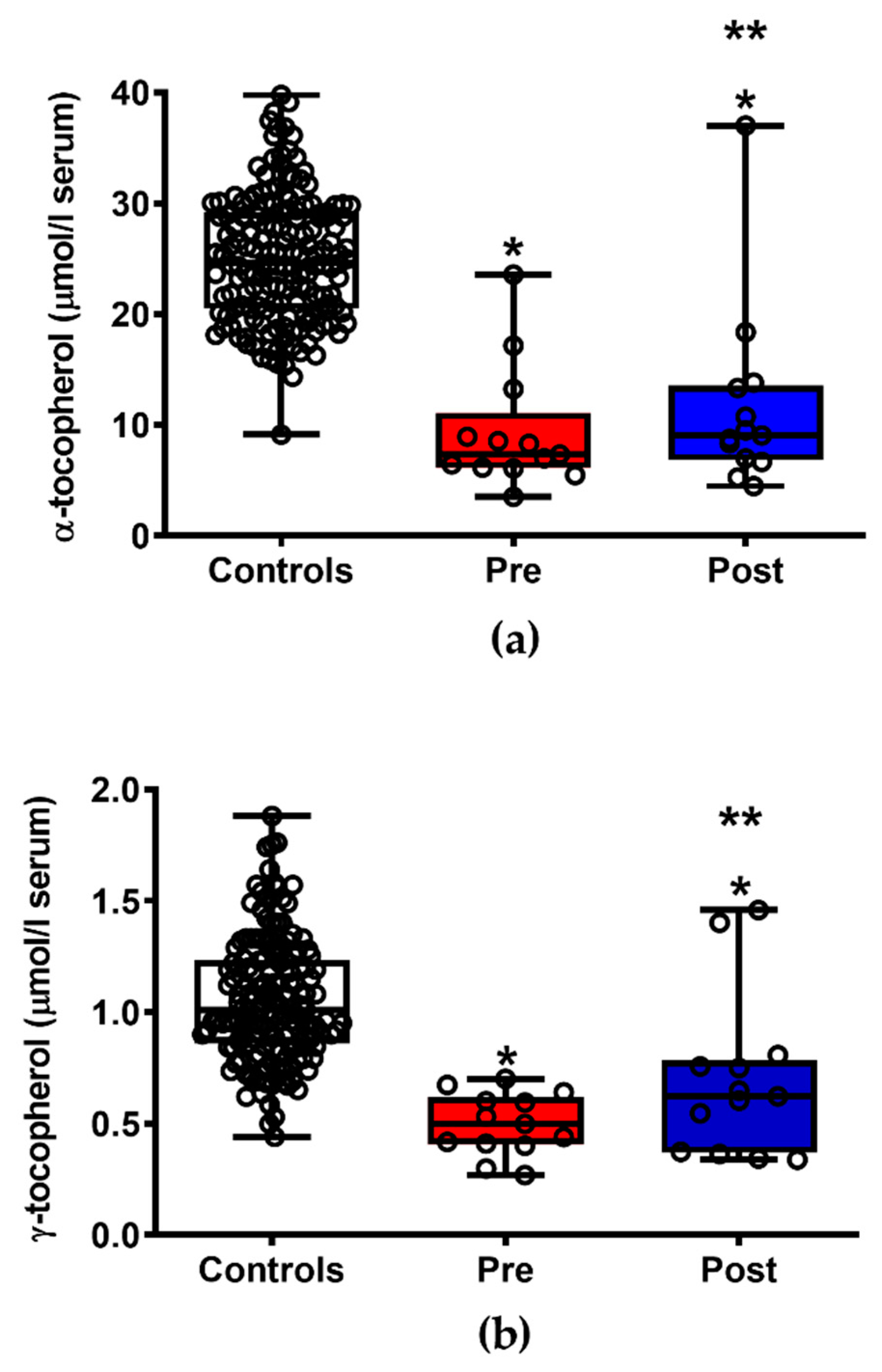
3.5. ILB® Improves the Pattern of Circulating Fat-Soluble Antioxidants
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mitchell, J.D.; Borasio, G.D. Amyotrophic lateral sclerosis. Lancet 2007, 369, 2031–2041. [Google Scholar] [CrossRef]
- Andrews, J.A.; Jackson, C.E.; Heiman-Patterson, T.D.; Bettica, P.; Brooks, B.R.; Pioro, E.P. Real-world evidence of riluzole effectiveness in treating amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Front. Degener. 2020, 21, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Le Gall, L.; Anakor, E.; Connolly, O.; Vijayakumar, U.G.; Duddy, W.J.; Duguez, S. Molecular and cellular mechanisms affected in ALS. J. Pers. Med. 2020, 10, 101. [Google Scholar] [CrossRef]
- Aydemir, D.; Ulusu, N.N. Importance of the serum biochemical parameters as potential biomarkers for rapid diagnosis and evaluating preclinical stage of ALS. Med. Hypotheses 2020, 141, 109736. [Google Scholar] [CrossRef]
- von Zur-Mühlen, B.; Lundgren, T.; Bayman, L.; Berne, C.; Bridges, N.; Eggerman, T.; Foss, A.; Goldstein, J.; Jenssen, T.; Jorns, C.; et al. Open randomized multicenter study to evaluate safety and efficacy of low molecular weight sulphated dextran in islet transplantation. Transplantation 2019, 103, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Lazzarino, G.; Amorini, A.M.; Barnes, N.M.; Bruce, L.; Mordente, A.; Lazzarino, G.; Di Pietro, V.; Tavazzi, B.; Belli, A.; Logan, A. Low molecular weight dextran sulfate (ILB®) restores brain energy metabolism following severe traumatic brain injury in the rat. Antioxidants 2020, 9, 850. [Google Scholar] [CrossRef]
- Logan, A.; Nagy, Z.; Barnes, N.M.; Belli, A.; Di Pietro, V.; Tavazzi, B.; Lazzarino, G.; Lazzarino, G.; Bruce, L.; Ropero, B.M.; et al. A phase II open label clinical study of the safety, tolerability and efficacy of a low molecular weight dextran sulphate for Amyotrophic Lateral Sclerosis. PLoS ONE 2021, submitted. [Google Scholar]
- A Single-Centre, Open Single-Arm Study Where the Safety, Tolerability and Efficacy of Subcutaneously Administered ILB Will Be Evaluated in Patients with Amyotrophic Lateral Sclerosis. Available online: URL https://www.clinicaltrialsregister.eu/ctr-search/trial/2017-005065-47/results (accessed on 11 May 2021).
- Cedarbaum, J.M.; Stambler, N.; Malta, E.; Fuller, C.; Hilt, D.; Thurmond, B.; Nakanishi, A. The ALSFRS-R: A revised ALS functional rating scale that incorporates assessments of respiratory function. BDNF ALS Study Group (Phase III). J. Neurol. Sci. 1999, 169, 13–21. [Google Scholar] [CrossRef]
- Lazzarino, G.; Amorini, A.M.; Fazzina, G.; Vagnozzi, R.; Signoretti, S.; Donzelli, S.; Di Stasio, E.; Giardina, B.; Tavazzi, B. Single-sample preparation for simultaneous cellular redox and energy state determination. Anal. Biochem. 2003, 322, 51–59. [Google Scholar] [CrossRef]
- Tavazzi, B.; Lazzarino, G.; Leone, P.; Amorini, A.M.; Bellia, F.; Janson, C.G.; Di Pietro, V.; Ceccarelli, L.; Donzelli, S.; Francis, J.S.; et al. Simultaneous high performance liquid chromatographic separation of purines, pyrimidines, N-acetylated amino acids, and dicarboxylic acids for the chemical diagnosis of inborn errors of metabolism. Clin. Biochem. 2005, 38, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Lazzarino, G.; Longo, S.; Amorini, A.M.; Di Pietro, V.; D’Urso, S.; Lazzarino, G.; Belli, A.; Tavazzi, B. Single-step preparation of selected biological fluids for the high performance liquid chromatographic analysis of fat-soluble vitamins and antioxidants. J. Chromatogr. A 2017, 1527, 43–52. [Google Scholar] [CrossRef]
- Romitelli, F.; Santini, S.A.; Chierici, E.; Pitocco, D.; Tavazzi, B.; Amorini, A.M.; Lazzarino, G.; Di Stasio, E. Comparison of nitrite/nitrate concentration in human plasma and serum samples measured by the enzymatic batch Griess assay, ion-pairing HPLC and ion-trap GC-MS: The importance of a correct removal of proteins in the Griess assay. J. Chromatogr. B 2007, 851, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Amorini, A.M.; Giorlandino, C.; Longo, S.; D’Urso, S.; Mesoraca, A.; Santoro, M.L.; Picardi, M.; Gullotta, S.; Cignini, P.; Lazzarino, D.; et al. Metabolic profile of amniotic fluid as a biochemical tool to screen for inborn errors of metabolism and fetal anomalies. Mol. Cell. Biochem. 2012, 359, 205–216. [Google Scholar] [CrossRef]
- Artiss, J.D.; Karcher, R.E.; Cavanagh, K.T.; Collins, S.L.; Peterson, V.J.; Varma, S.; Zak, B. A liquid-stable reagent for lactic acid levels. Application to the Hitachi 911 and Beckman CX7. Am. J. Clin. Pathol. 2000, 114, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Sharma, C.; Kim, S.; Nam, Y.; Jung, U.J.; Kim, S.R. Mitochondrial Dysfunction as a Driver of Cognitive Impairment in Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 4850. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.; Laranjinha, J. Nitric oxide and dopamine metabolism converge via mitochondrial dysfunction in the mechanisms of neurodegeneration in Parkinson’s disease. Arch. Biochem. Biophys. 2021, 704, 108877. [Google Scholar] [CrossRef] [PubMed]
- Noori, A.; Mezlini, A.M.; Hyman, B.T.; Serrano-Pozo, A.; Das, S. Systematic review and meta-analysis of human transcriptomics reveals neuroinflammation, deficient energy metabolism, and proteostasis failure across neurodegeneration. Neurobiol. Dis. 2021, 149, 105225. [Google Scholar] [CrossRef]
- McDonald, T.S.; McCombe, P.A.; Woodruff, T.M.; Lee, J.D. The potential interplay between energy metabolism and innate complement activation in amyotrophic lateral sclerosis. FASEB J. 2020, 34, 7225–7233. [Google Scholar] [CrossRef] [PubMed]
- Straub, I.R.; Weraarpachai, W.; Shoubridge, E.A. Multi-OMICS study of a CHCHD10 variant causing ALS demonstrates metabolic rewiring and activation of endoplasmic reticulum and mitochondrial unfolded protein responses. Hum. Mol. Genet. 2021, 30, 687–705. [Google Scholar] [CrossRef] [PubMed]
- Gerou, M.; Hall, B.; Woof, R.; Allsop, J.; Kolb, S.J.; Meyer, K.; Shaw, P.J.; Allen, S.P. Amyotrophic lateral sclerosis alters the metabolic aging profile in patient derived fibroblasts. Neurobiol. Aging 2021, 105, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.P.; Hall, B.; Castelli, L.M.; Francis, L.; Woof, R.; Siskos, A.P.; Kouloura, E.; Gray, E.; Thompson, A.G.; Talbot, K.; et al. Astrocyte adenosine deaminase loss increases motor neuron toxicity in amyotrophic lateral sclerosis. Brain 2019, 142, 586–605. [Google Scholar] [CrossRef] [PubMed]
- Tefera, T.W.; Steyn, F.J.; Ngo, S.T.; Borges, K. CNS glucose metabolism in Amyotrophic Lateral Sclerosis: A therapeutic target? Cell Biosci. 2021, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Yamashita, T.; Nomura, E.; Hishikawa, N.; Ikegami, K.; Osakada, Y.; Matsumoto, N.; Kawahara, Y.; Yunoki, T.; Takahashi, Y.; et al. Improvement of a decreased anti-oxidative activity by edaravone in amyotrophic lateral sclerosis patients. J. Neurol. Sci. 2020, 415, 116906. [Google Scholar] [CrossRef] [PubMed]
- Simone, I.L.; Ruggieri, M.; Tortelli, R.; Ceci, E.; D’Errico, E.; Leo, A.; Zoccolella, S.; Mastrapasqua, M.; Capozzo, R.; Livrea, P.; et al. Serum N-acetylaspartate level in amyotrophic lateral sclerosis. Arch. Neurol. 2011, 68, 1308–1312. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Weerasekera, A.; Peeters, R.; Sima, D.M.; Dresselaers, T.; Sunaert, S.; De Vocht, J.; Claeys, K.; Van Huffel, S.; Van Damme, P.; Himmelreich, U. Motor cortex metabolite alterations in amyotrophic lateral sclerosis assessed in vivo using edited and non-edited magnetic resonance spectroscopy. Brain Res. 2019, 1718, 22–31. [Google Scholar] [CrossRef]
- Reischauer, C.; Gutzeit, A.; Neuwirth, C.; Fuchs, A.; Sartoretti-Schefer, S.; Weber, M.; Czell, D. In-vivo evaluation of neuronal and glial changes in amyotrophic lateral sclerosis with diffusion tensor spectroscopy. Neuroimage Clin. 2018, 20, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Ratai, E.M.; Alshikho, M.J.; Zürcher, N.R.; Loggia, M.L.; Cebulla, C.L.; Cernasov, P.; Reynolds, B.; Fish, J.; Seth, R.; Babu, S.; et al. Integrated imaging of [(11)C]-PBR28 PET, MR diffusion and magnetic resonance spectroscopy (1)H-MRS in amyotrophic lateral sclerosis. Neuroimage Clin. 2018, 20, 357–364. [Google Scholar] [CrossRef]
- Hanstock, C.; Sun, K.; Choi, C.; Eurich, D.; Camicioli, R.; Johnston, W.; Kalra, S. Spectroscopic markers of neurodegeneration in the mesial prefrontal cortex predict survival in ALS. Amyotroph. Lateral Scler. Front. Degener. 2020, 21, 246–251. [Google Scholar] [CrossRef]
- Vicario, N.; Spitale, F.M.; Tibullo, D.; Giallongo, C.; Amorini, A.M.; Scandura, G.; Spoto, G.; Saab, M.W.; D’Aprile, S.; Alberghina, C.; et al. Clobetasol promotes neuromuscular plasticity in mice after motoneuronal loss via sonic hedgehog signaling, immunomodulation and metabolic rebalancing. Cell Death Dis. 2021, 12, 625. [Google Scholar] [CrossRef]
- Vagnozzi, R.; Marmarou, A.; Tavazzi, B.; Signoretti, S.; Di Pierro, D.; del Bolgia, F.; Amorini, A.M.; Fazzina, G.; Sherkat, S.; Lazzarino, G. Changes of cerebral energy metabolism and lipid peroxidation in rats leading to mitochondrial dysfunction after diffuse brain injury. J. Neurotrauma 1999, 16, 903–913. [Google Scholar] [CrossRef]
- Signoretti, S.; Marmarou, A.; Aygok, G.A.; Fatouros, P.P.; Portella, G.; Bullock, R.M. Assessment of mitochondrial impairment in traumatic brain injury using high-resolution proton magnetic resonance spectroscopy. J. Neurosurg. 2008, 108, 42–52. [Google Scholar] [CrossRef]
- Belli, A.; Sen, J.; Petzold, A.; Russo, S.; Kitchen, N.; Smith, M.; Tavazzi, B.; Vagnozzi, R.; Signoretti, S.; Amorini, A.M.; et al. Extracellular N-acetylaspartate depletion in traumatic brain injury. J. Neurochem. 2006, 96, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Vagnozzi, R.; Signoretti, S.; Tavazzi, B.; Floris, R.; Ludovici, A.; Marziali, S.; Tarascio, G.; Amorini, A.M.; Di Pietro, V.; Delfini, R.; et al. Temporal window of metabolic brain vulnerability to concussion: A pilot 1H-magnetic resonance spectroscopic study in concussed athletes—part III. Neurosurgery 2008, 62, 1286–1295. [Google Scholar] [CrossRef] [PubMed]
- Vagnozzi, R.; Signoretti, S.; Cristofori, L.; Alessandrini, F.; Floris, R.; Isgrò, E.; Ria, A.; Marziale, S.; Zoccatelli, G.; Tavazzi, B.; et al. Assessment of metabolic brain damage and recovery following mild traumatic brain injury: A multicentre, proton magnetic resonance spectroscopic study in concussed patients. Brain 2010, 133, 3232–3242. [Google Scholar] [CrossRef] [PubMed]
- Toczek, M.; Zielonka, D.; Zukowska, P.; Marcinkowski, J.T.; Slominska, E.; Isalan, M.; Smolenski, R.T.; Mielcarek, M. An impaired metabolism of nucleotides underpins a novel mechanism of cardiac remodeling leading to Huntington’s disease related cardiomyopathy. Biochim. Biophys. Acta 2016, 1862, 2147–2157. [Google Scholar] [CrossRef] [PubMed]
- Van Wylen, D.G. Effect of ischemic preconditioning on interstitial purine metabolite and lactate accumulation during myocardial ischemia. Circulation 1994, 89, 2283–2289. [Google Scholar] [CrossRef] [PubMed]
- Lazzarino, G.; Raatikainen, P.; Nuutinen, M.; Nissinen, J.; Tavazzi, B.; Di Pierro, D.; Giardina, B.; Peuhkurinen, K. Myocardial release of malondialdehyde and purine compounds during coronary bypass surgery. Circulation 1994, 90, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Cristofori, L.; Tavazzi, B.; Gambin, R.; Vagnozzi, R.; Vivenza, C.; Amorini, A.M.; Di Pierro, D.; Fazzina, G.; Lazzarino, G. Early onset of lipid peroxidation after human traumatic brain injury: A fatal limitation for the free radical scavenger pharmacological therapy? J. Investig. Med. 2001, 49, 450–458. [Google Scholar] [CrossRef]
- Lazzarino, G.; Amorini, A.M.; Petzold, A.; Gasperini, C.; Ruggieri, S.; Quartuccio, M.E.; Lazzarino, G.; Di Stasio, E.; Tavazzi, B. Serum compounds of energy metabolism impairment are related to disability, disease course and neuroimaging in multiple sclerosis. Mol. Neurobiol. 2017, 54, 7520–7533. [Google Scholar] [CrossRef] [PubMed]
- Lazzarino, G.; Amorini, A.M.; Eikelenboom, M.; Killestein, J.; Belli, A.; Di Pietro, V.; Tavazzi, B.; Barkhof, F.; Polman, C.; Uitdehaag, B.; et al. Cerebrospinal fluid ATP metabolites in multiple sclerosis. Mult. Scler. J. 2010, 16, 549–554. [Google Scholar] [CrossRef]
- Tavazzi, B.; Amorini, A.M.; Fazzina, G.; Di Pierro, D.; Tuttobene, M.; Giardina, B.; Lazzarino, G. Oxidative stress induces impairment of human erythrocyte energy metabolism through the oxygen radical-mediated direct activation of AMP-deaminase. J. Biol. Chem. 2001, 276, 48083–48092. [Google Scholar] [CrossRef] [PubMed]
- Bracko, O.; Di Pietro, V.; Lazzarino, G.; Amorini, A.M.; Tavazzi, B.; Artmann, J.; Wong, E.C.; Buxton, R.B.; Weller, M.; Luft, A.R.; et al. 3-Nitropropionic acid-induced ischemia tolerance in the rat brain is mediated by reduced metabolic activity and cerebral blood flow. J. Cereb. Blood Flow Metab. 2014, 34, 1522–1530. [Google Scholar] [CrossRef] [PubMed]
- Vagnozzi, R.; Tavazzi, B.; Signoretti, S.; Amorini, A.M.; Belli, A.; Cimatti, M.; Delfini, R.; Di Pietro, V.; Finocchiaro, A.; Lazzarino, G. Temporal window of metabolic brain vulnerability to concussions: Mitochondrial-related impairment—part I. Neurosurgery 2007, 61, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Sassani, M.; Alix, J.J.; McDermott, C.J.; Baster, K.; Hoggard, N.; Wild, J.M.; Mortiboys, H.J.; Shaw, P.J.; Wilkinson, I.D.; Jenkins, T.M. Magnetic resonance spectroscopy reveals mitochondrial dysfunction in amyotrophic lateral sclerosis. Brain 2020, 143, 3603–3618. [Google Scholar] [CrossRef]
- Ramachandra, C.J.A.; Hernandez-Resendiz, S.; Crespo-Avilan, G.E.; Lin, Y.-H.; Hausenloy, D.J. Mitochondria in acute myocardial infarction and cardioprotection. EBioMedicine 2020, 57, 102884. [Google Scholar] [CrossRef] [PubMed]
- Kaur, I.; Behl, T.; Sehgal, A.; Singh, S.; Sharma, N.; Aleya, L.; Bungau, S. Connecting the dots between mitochondrial dysfunction and Parkinson’s disorder: Focus mitochondria-targeting therapeutic paradigm in mitigating the disease severity. Environ. Sci. Pollut. Res. Int. 2021, 28, 37060–37081. [Google Scholar] [CrossRef]
- Sahel, D.K.; Kaira, M.; Raj, K.; Sharma, S.; Singh, S. Mitochondrial dysfunctioning and neuroinflammation: Recent highlights on the possible mechanisms involved in Traumatic Brain Injury. Neurosci. Lett. 2019, 710, 134347. [Google Scholar] [CrossRef] [PubMed]
- Moosavi, B.; Zhu, X.L.; Yang, W.C.; Yang, G.F. Molecular pathogenesis of tumorigenesis caused by succinate dehydrogenase defect. Eur. J. Cell Biol. 2020, 99, 151057. [Google Scholar] [CrossRef] [PubMed]
- Tavazzi, B.; Vagnozzi, R.; Signoretti, S.; Amorini, A.M.; Belli, A.; Cimatti, M.; Delfini, R.; Di Pietro, V.; Finocchiaro, A.; Lazzarino, G. Temporal window of metabolic brain vulnerability to concussions: Oxidative and nitrosative stresses—part II. Neurosurgery 2007, 61, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Torres, I.; Manzano-Pech, L.; Rubio-Ruíz, M.E.; Soto, M.E.; Guarner-Lans, V. Nitrosative Stress and Its Association with Cardiometabolic Disorders. Molecules 2020, 25, 2555. [Google Scholar] [CrossRef] [PubMed]
- Król, M.; Kepinska, M. Human Nitric Oxide Synthase—Its Functions, Polymorphisms, and Inhibitors in the Context of Inflammation, Diabetes and Cardiovascular Diseases. Int. J. Mol. Sci. 2020, 22, 56. [Google Scholar] [CrossRef] [PubMed]
- Illes, P.; Rubini, P.; Ulrich, H.; Zhao, Y.; Tang, Y. Regulation of Microglial Functions by Purinergic Mechanisms in the Healthy and Diseased CNS. Cells 2020, 9, 1108. [Google Scholar] [CrossRef]
- Debska-Vielhaber, G.; Miller, I.; Peeva, V.; Zuschratter, W.; Walczak, J.; Schreiber, S.; Petri, S.; Machts, J.; Vogt, S.; Szczepanowska, J.; et al. Impairment of mitochondrial oxidative phosphorylation in skin fibroblasts of SALS and FALS patients is rescued by in vitro treatment with ROS scavengers. Exp. Neurol. 2021, 339, 113620. [Google Scholar] [CrossRef] [PubMed]
- Devos, D.; Moreau, C.; Kyheng, M.; Garçon, G.; Rolland, A.S.; Blasco, H.; Gelé, P.; Lenglet, T.T.; Veyrat-Durebex, C.; Corcia, P.; et al. A ferroptosis-based panel of prognostic biomarkers for Amyotrophic Lateral Sclerosis. Sci. Rep. 2019, 9, 2918. [Google Scholar] [CrossRef] [PubMed]
- Golenia, A.; Leśkiewicz, M.; Regulska, M.; Budziszewska, B.; Szczęsny, E.; Jagiełła, J.; Wnuk, M.; Ostrowskam, M.; Lasoń, W.; Basta-Kaim, A.; et al. Catalase activity in blood fractions of patients with sporadic ALS. Pharmacol. Rep. 2014, 66, 704–707. [Google Scholar] [CrossRef] [PubMed]
- Schonhoff, C.M.; Matsuoka, M.; Tummala, H.; Johnson, M.A.; Estevéz, A.G.; Wu, R.; Kamaid, A.; Ricart, K.C.; Hashimoto, Y.; Gaston, B.; et al. S-nitrosothiol depletion in amyotrophic lateral sclerosis. Proc. Natl. Acad Sci. USA 2006, 103, 2404–2409. [Google Scholar] [CrossRef] [PubMed]
- Cheong, I.; Marjańska, M.; Deelchand, D.K.; Eberly, L.E.; Walk, D.; Öz, G. Ultra-High Field Proton MR Spectroscopy in Early-Stage Amyotrophic Lateral Sclerosis. Neurochem. Res. 2017, 42, 1833–1844. [Google Scholar] [CrossRef] [PubMed]
- Weiduschat, N.; Mao, X.; Hupf, J.; Armstrong, N.; Kang, G.; Lange, D.J.; Mitsumoto, H.; Shungu, D.C. Motor cortex glutathione deficit in ALS measured in vivo with the J-editing technique. Neurosci. Lett. 2014, 570, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Kalinina, E.; Novichkova, M. Glutathione in Protein Redox Modulation through S-Glutathionylation and S-Nitrosylation. Molecules 2021, 26, 435. [Google Scholar] [CrossRef]
- Blasco, H.; Garcon, G.; Patin, F.; Veyrat-Durebex, C.; Boyer, J.; Devos, D.; Vourc’h, P.; Andres, C.R.; Corcia, P. Panel of Oxidative Stress and Inflammatory Biomarkers in ALS: A Pilot Study. Can. J. Neurol. Sci. 2017, 44, 90–95. [Google Scholar] [CrossRef]
- Valbuena, G.N.; Rizzardini, M.; Cimini, S.; Siskos, A.P.; Bendotti, C.; Cantoni, L.; Keun, H.C. Metabolomic Analysis Reveals Increased Aerobic Glycolysis and Amino Acid Deficit in a Cellular Model of Amyotrophic Lateral Sclerosis. Mol. Neurobiol. 2016, 53, 2222–2240. [Google Scholar] [CrossRef] [PubMed]
- Palma, A.; de Carvalho, M.; Barata, N.; Evangelista, T.; Chicau, P.; Regalla, M.; Costa, J. Biochemical characterization of plasma in amyotrophic lateral sclerosis: Amino acid and protein composition. Amyotroph. Lateral Scler. 2005, 6, 104–110. [Google Scholar] [CrossRef]
- Bereman, M.S.; Kirkwood, K.I.; Sabaretnam, T.; Furlong, S.; Rowe, D.B.; Guillemin, G.J.; Mellinger, A.L.; Muddiman, D.C. Metabolite Profiling Reveals Predictive Biomarkers and the Absence of beta-Methyl Amino-l-alanine in Plasma from Individuals Diagnosed with Amyotrophic Lateral Sclerosis. J. Proteome Res. 2020, 19, 3276–3285. [Google Scholar] [CrossRef] [PubMed]
| Sample Number | Subject Number and Sampling Time |
|---|---|
| 1 | Subject 101, Day 0 |
| 2 | Subject 101, Day 36 |
| 3 | Subject 102, Day 0 |
| 4 | Subject 10, Day 36 |
| 5 | Subject 103, Day 0 |
| 6 | Subject 103, Day 36 |
| 7 | Subject 104, Day 0 |
| 8 | Subject 104, Day 36 |
| 9 | Subject 105, Day 0 |
| 10 | Subject 105, Day 36 |
| 11 | Subject 106, Day 0 |
| 12 | Subject 106, Day 36 |
| 13 | Subject 107, Day 0 |
| 14 | Subject 107, Day 36 |
| 15 | Subject 108, Day 0 |
| 16 | Subject 108, Day 36 |
| 17 | Subject 109, Day 0 |
| 18 | Subject 109, Day 36 |
| 19 | Subject 110, Day 0 |
| 20 | Subject 110, Day 36 |
| 21 | Subject 111, Day 0 |
| 22 | Subject 111, Day 36 |
| 23 | Subject 112, Day 0 |
| 24 | Subject 112, Day 36 |
| 25 | Subject 113, Day 0 |
| 26 | Subject 113, Day 36 |
| Day | ALSFRS-R ± SD |
|---|---|
| 0 (Pre-treatment) | 36.1 ± 6.7 |
| 29 (Final treatment) | 39.2 ± 6.3 * |
| 36 (Post-treatment) | 40.9 ± 6.9 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazzarino, G.; Mangione, R.; Belli, A.; Di Pietro, V.; Nagy, Z.; Barnes, N.M.; Bruce, L.; Ropero, B.M.; Persson, L.I.; Manca, B.; et al. ILB® Attenuates Clinical Symptoms and Serum Biomarkers of Oxidative/Nitrosative Stress and Mitochondrial Dysfunction in Patients with Amyotrophic Lateral Sclerosis. J. Pers. Med. 2021, 11, 794. https://doi.org/10.3390/jpm11080794
Lazzarino G, Mangione R, Belli A, Di Pietro V, Nagy Z, Barnes NM, Bruce L, Ropero BM, Persson LI, Manca B, et al. ILB® Attenuates Clinical Symptoms and Serum Biomarkers of Oxidative/Nitrosative Stress and Mitochondrial Dysfunction in Patients with Amyotrophic Lateral Sclerosis. Journal of Personalized Medicine. 2021; 11(8):794. https://doi.org/10.3390/jpm11080794
Chicago/Turabian StyleLazzarino, Giacomo, Renata Mangione, Antonio Belli, Valentina Di Pietro, Zsuzsanna Nagy, Nicholas M. Barnes, Lars Bruce, Bernardo M. Ropero, Lennart I. Persson, Benedetta Manca, and et al. 2021. "ILB® Attenuates Clinical Symptoms and Serum Biomarkers of Oxidative/Nitrosative Stress and Mitochondrial Dysfunction in Patients with Amyotrophic Lateral Sclerosis" Journal of Personalized Medicine 11, no. 8: 794. https://doi.org/10.3390/jpm11080794
APA StyleLazzarino, G., Mangione, R., Belli, A., Di Pietro, V., Nagy, Z., Barnes, N. M., Bruce, L., Ropero, B. M., Persson, L. I., Manca, B., Saab, M. W., Amorini, A. M., Tavazzi, B., Lazzarino, G., & Logan, A. (2021). ILB® Attenuates Clinical Symptoms and Serum Biomarkers of Oxidative/Nitrosative Stress and Mitochondrial Dysfunction in Patients with Amyotrophic Lateral Sclerosis. Journal of Personalized Medicine, 11(8), 794. https://doi.org/10.3390/jpm11080794











