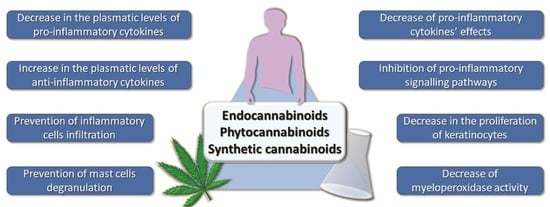
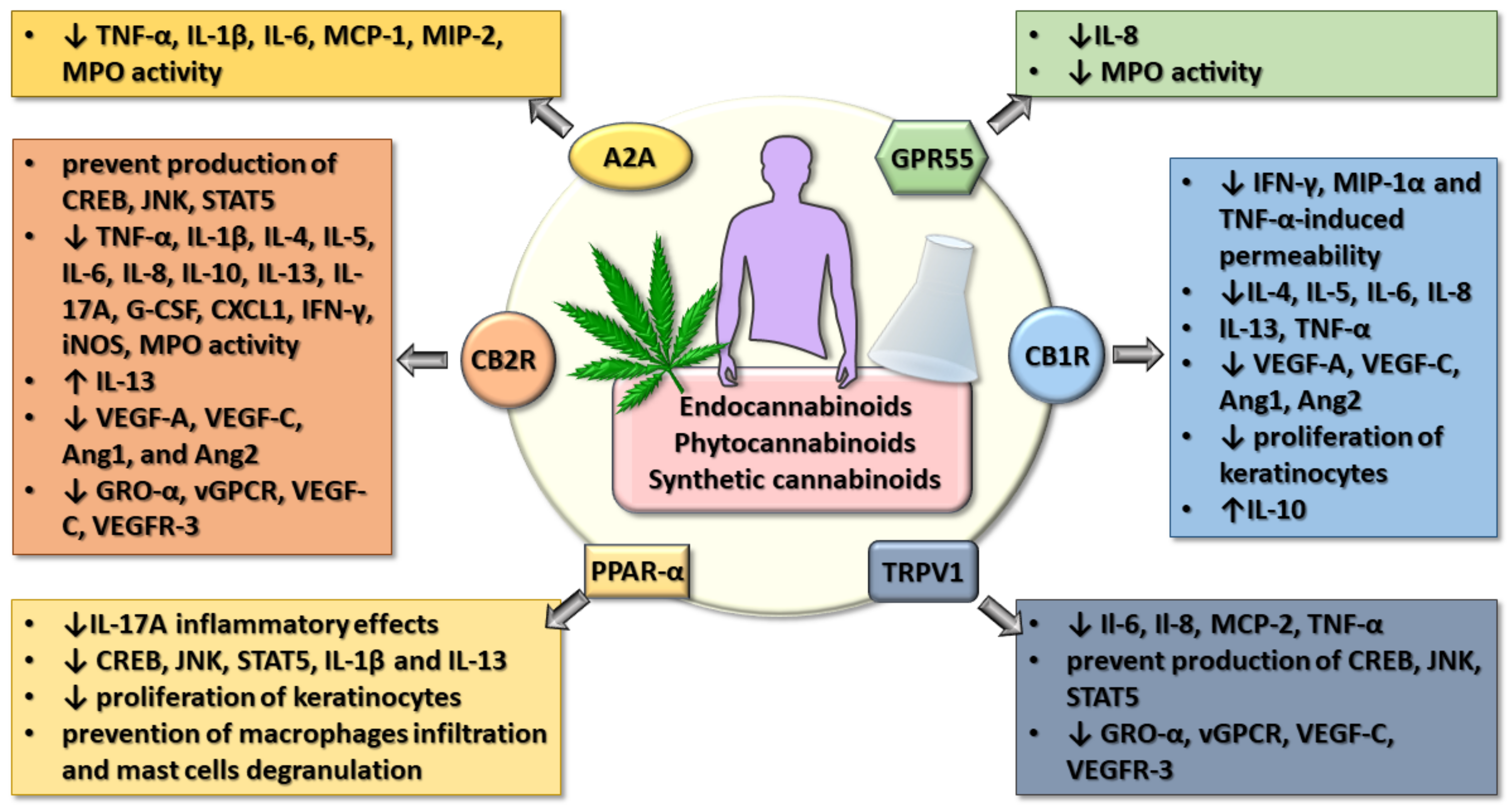
Cannabinoids and Inflammations of the Gut-Lung-Skin Barrier
Abstract
1. Introduction
2. The Gut-Lung-Skin Barrier
3. Cannabinoids and Inflammation
4. Cannabinoids and Gut Inflammations
4.1. In Vitro Studies
4.2. In Vivo Animal Studies
4.3. Human Clinical Trials
5. Cannabinoids and Lung Inflammatory Conditions
5.1. In Vitro Studies
5.2. In Vivo Animal Studies
5.3. Human Clinical Trials
6. Cannabinoids and Inflammatory Skin Disorders
6.1. In Vitro Studies
6.2. In Vivo Animal Studies
6.3. Human Clinical Trials
7. Integrative Vision on the Effects of Cannabinoids in Gut-Lung-Skin Epithelial Inflammation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stanke, F. The Contribution of the Airway Epithelial Cell to Host Defense. Mediat. Inflamm. 2015, 2015, 463016. [Google Scholar] [CrossRef] [PubMed]
- Hornef, M.W.; Normark, S.; Henriques-Normark, B.; Rhen, M. Bacterial evasion of innate defense at epithelial linings. Chem. Immunol. Allergy 2005, 86, 72–98. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.Y.; Chen, Z.J.; Shah, N.P.; El-Nezami, H. Modulation of Intestinal Epithelial Defense Responses by Probiotic Bacteria. Crit. Rev. Food Sci. Nutr. 2016, 56, 2628–2641. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.H.; Zhu, T.R.; Tran, K.A.; Sivamani, R.K.; Shi, V.Y. Epithelial barrier dysfunctions in atopic dermatitis: A skin-gut-lung model linking microbiome alteration and immune dysregulation. Br. J. Dermatol. 2018, 179, 570–581. [Google Scholar] [CrossRef]
- Uluçkan, Ö.; Wagner, E.F. Chronic systemic inflammation originating from epithelial tissues. FEBS J. 2017, 284, 505–516. [Google Scholar] [CrossRef]
- Wang, H.; Liu, J.S.; Peng, S.H.; Deng, X.Y.; Zhu, D.M.; Javidiparsijani, S.; Wang, G.R.; Li, D.Q.; Li, L.X.; Wang, Y.C.; et al. Gut-lung crosstalk in pulmonary involvement with inflammatory bowel diseases. World J. Gastroenterol. 2013, 19, 6794–6804. [Google Scholar] [CrossRef]
- Weaver, C.T.; Hatton, R.D.; Mangan, P.R.; Harrington, L.E. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu. Rev. Immunol. 2007, 25, 821–852. [Google Scholar] [CrossRef]
- Weaver, C.T.; Elson, C.O.; Fouser, L.A.; Kolls, J.K. The Th17 pathway and inflammatory diseases of the intestines, lungs, and skin. Annu. Rev. Pathol. 2013, 8, 477–512. [Google Scholar] [CrossRef]
- Eyerich, K.; Dimartino, V.; Cavani, A. IL-17 and IL-22 in immunity: Driving protection and pathology. Eur. J. Immunol. 2017, 47, 607–614. [Google Scholar] [CrossRef]
- Puel, A.; Cypowyj, S.; Bustamante, J.; Wright, J.F.; Liu, L.; Lim, H.K.; Migaud, M.; Israel, L.; Chrabieh, M.; Audry, M.; et al. Chronic mucocutaneous candidiasis in humans with inborn errors of interleukin-17 immunity. Science 2011, 332, 65–68. [Google Scholar] [CrossRef]
- O’Connor, W., Jr.; Kamanaka, M.; Booth, C.J.; Town, T.; Nakae, S.; Iwakura, Y.; Kolls, J.K.; Flavell, R.A. A protective function for interleukin 17A in T cell-mediated intestinal inflammation. Nat. Immunol. 2009, 10, 603–609. [Google Scholar] [CrossRef]
- Isailovic, N.; Daigo, K.; Mantovani, A.; Selmi, C. Interleukin-17 and innate immunity in infections and chronic inflammation. J. Autoimmun. 2015, 60, 1–11. [Google Scholar] [CrossRef]
- Eyerich, K.; Pennino, D.; Scarponi, C.; Foerster, S.; Nasorri, F.; Behrendt, H.; Ring, J.; Traidl-Hoffmann, C.; Albanesi, C.; Cavani, A. IL-17 in atopic eczema: Linking allergen-specific adaptive and microbial-triggered innate immune response. J. Allergy Clin. Immunol. 2009, 123, 59–66.e54. [Google Scholar] [CrossRef]
- Aujla, S.J.; Kolls, J.K. IL-22: A critical mediator in mucosal host defense. J. Mol. Med. 2009, 87, 451–454. [Google Scholar] [CrossRef]
- Blázquez, A.B.; Mayer, L.; Berin, M.C. Thymic stromal lymphopoietin is required for gastrointestinal allergy but not oral tolerance. Gastroenterology 2010, 139, 1301–1309. [Google Scholar] [CrossRef]
- Luo, J.; Li, Y.; Gong, R. The mechanism of atopic march may be the ‘social’ event of cells and molecules (Review). Int. J. Mol. Med. 2010, 26, 779–785. [Google Scholar] [CrossRef]
- Schett, G.; Elewaut, D.; McInnes, I.B.; Dayer, J.M.; Neurath, M.F. How cytokine networks fuel inflammation: Toward a cytokine-based disease taxonomy. Nat. Med. 2013, 19, 822–824. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, X.; Lu, H.; Li, Y.; Ji, H. Association between Interleukin-8-251A/T polymorphism and gastric cancer susceptibility: A meta-analysis based on 5286 cases and 8000 controls. Int. J. Clin. Exp. Med. 2015, 8, 22393–22402. [Google Scholar]
- Albareda, M.; Ravella, A.; Castelló, M.; Saborit, S.; Peramiquel, L.; Vila, L. Metabolic syndrome and its components in patients with psoriasis. Springerplus 2014, 3, 612. [Google Scholar] [CrossRef]
- Andersen, N.N.; Jess, T. Risk of cardiovascular disease in inflammatory bowel disease. World J. Gastrointest. Pathophysiol. 2014, 5, 359–365. [Google Scholar] [CrossRef]
- Scheau, C.; Mihai, L.G.; Bădărău, I.A.; Caruntu, C. Emerging applications of some important natural compounds in the field of oncology. Farmacia 2020, 68, 8. [Google Scholar] [CrossRef]
- Calina, D.; Buga, A.M.; Mitroi, M.; Buha, A.; Caruntu, C.; Scheau, C.; Bouyahya, A.; El Omari, N.; El Menyiy, N.; Docea, A.O. The Treatment of Cognitive, Behavioural and Motor Impairments from Brain Injury and Neurodegenerative Diseases through Cannabinoid System Modulation-Evidence from In Vivo Studies. J. Clin. Med. 2020, 9, 2395. [Google Scholar] [CrossRef]
- Ilie, M.A.; Caruntu, C.; Tampa, M.; Georgescu, S.R.; Matei, C.; Negrei, C.; Ion, R.M.; Constantin, C.; Neagu, M.; Boda, D. Capsaicin: Physicochemical properties, cutaneous reactions and potential applications in painful and inflammatory conditions (Review). Exp. Ther. Med. 2019, 18, 916–925. [Google Scholar] [CrossRef]
- Salehi, B.; Calina, D.; Docea, A.O.; Koirala, N.; Aryal, S.; Lombardo, D.; Pasqua, L.; Taheri, Y.; Marina Salgado Castillo, C.; Martorell, M.; et al. Curcumin’s Nanomedicine Formulations for Therapeutic Application in Neurological Diseases. J. Clin. Med. 2020, 9, 430. [Google Scholar] [CrossRef]
- Sharma, S.; Naura, A.S. Potential of phytochemicals as immune-regulatory compounds in atopic diseases: A review. Biochem. Pharmacol. 2020, 113790. [Google Scholar] [CrossRef]
- Mintie, C.A.; Singh, C.K.; Ahmad, N. Whole Fruit Phytochemicals Combating Skin Damage and Carcinogenesis. Transl. Oncol. 2019, 13, 146–156. [Google Scholar] [CrossRef]
- Kim, J.E.; Lee, K.W. Molecular Targets of Phytochemicals for Skin Inflammation. Curr. Pharm. Des. 2018, 24, 1533–1550. [Google Scholar] [CrossRef]
- Salehi, B.; Sharifi-Rad, J.; Cappellini, F.; Reiner, Ž.; Zorzan, D.; Imran, M.; Sener, B.; Kilic, M.; El-Shazly, M.; Fahmy, N.M.; et al. The Therapeutic Potential of Anthocyanins: Current Approaches Based on Their Molecular Mechanism of Action. Front. Pharmacol. 2020, 11, 1300. [Google Scholar] [CrossRef]
- Salehi, B.; Quispe, C.; Chamkhi, I.; El Omari, N.; Balahbib, A.; Sharifi-Rad, J.; Bouyahya, A.; Akram, M.; Iqbal, M.; Docea, A.O.; et al. Pharmacological Properties of Chalcones: A Review of Preclinical Including Molecular Mechanisms and Clinical Evidence. Front. Pharmacol. 2020, 11, 592654. [Google Scholar] [CrossRef]
- Makara, G.B. Superficial and deep chemonociception: Differential inhibition by pretreatment with capsaicin. Acta Physiol. Acad. Sci. Hung. 1970, 38, 393–399. [Google Scholar]
- Georgescu, S.R.; Sarbu, M.I.; Matei, C.; Ilie, M.A.; Caruntu, C.; Constantin, C.; Neagu, M.; Tampa, M. Capsaicin: Friend or Foe in Skin Cancer and Other Related Malignancies? Nutrients 2017, 9, 1365. [Google Scholar] [CrossRef] [PubMed]
- Scheau, C.; Badarau, I.A.; Caruntu, C.; Mihai, G.L.; Didilescu, A.C.; Constantin, C.; Neagu, M. Capsaicin: Effects on the Pathogenesis of Hepatocellular Carcinoma. Molecules 2019, 24, 2350. [Google Scholar] [CrossRef] [PubMed]
- Rubin, V. Cannabis and Culture; De Gruyter Mouton: Berlin, Germany, 1975. [Google Scholar]
- Zurier, R.B.; Burstein, S.H. Cannabinoids, inflammation, and fibrosis. FASEB J. 2016, 30, 3682–3689. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. The pharmacology of cannabinoid receptors and their ligands: An overview. Int. J. Obes. 2006, 30, S13–S18. [Google Scholar] [CrossRef]
- Walsh, K.B.; Andersen, H.K. Molecular Pharmacology of Synthetic Cannabinoids: Delineating CB1 Receptor-Mediated Cell Signaling. Int. J. Mol. Sci. 2020, 21, 6115. [Google Scholar] [CrossRef]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef]
- Demuth, D.G.; Molleman, A. Cannabinoid signalling. Life Sci. 2006, 78, 549–563. [Google Scholar] [CrossRef]
- Thapa, D.; Cairns, E.A.; Szczesniak, A.M.; Toguri, J.T.; Caldwell, M.D.; Kelly, M.E.M. The Cannabinoids Δ(8)THC, CBD, and HU-308 Act via Distinct Receptors to Reduce Corneal Pain and Inflammation. Cannabis Cannabinoid Res. 2018, 3, 11–20. [Google Scholar] [CrossRef]
- Britch, S.C.; Goodman, A.G.; Wiley, J.L.; Pondelick, A.M.; Craft, R.M. Antinociceptive and Immune Effects of Delta-9-Tetrahydrocannabinol or Cannabidiol in Male Versus Female Rats with Persistent Inflammatory Pain. J. Pharmacol. Exp. Ther. 2020, 373, 416–428. [Google Scholar] [CrossRef]
- Lunn, C.A.; Fine, J.S.; Rojas-Triana, A.; Jackson, J.V.; Fan, X.; Kung, T.T.; Gonsiorek, W.; Schwarz, M.A.; Lavey, B.; Kozlowski, J.A.; et al. A novel cannabinoid peripheral cannabinoid receptor-selective inverse agonist blocks leukocyte recruitment in vivo. J. Pharmacol. Exp. Ther. 2006, 316, 780–788. [Google Scholar] [CrossRef]
- Henshaw, F.R.; Dewsbury, L.S.; Lim, C.K.; Steiner, G.Z. The Effects of Cannabinoids on Pro- and Anti-Inflammatory Cytokines: A Systematic Review of In Vivo Studies. Cannabis Cannabinoid Res. 2021. [Google Scholar] [CrossRef]
- Costa, B.; Trovato, A.E.; Comelli, F.; Giagnoni, G.; Colleoni, M. The non-psychoactive cannabis constituent cannabidiol is an orally effective therapeutic agent in rat chronic inflammatory and neuropathic pain. Eur. J. Pharmacol. 2007, 556, 75–83. [Google Scholar] [CrossRef]
- Costa, B.; Colleoni, M.; Conti, S.; Parolaro, D.; Franke, C.; Trovato, A.E.; Giagnoni, G. Oral anti-inflammatory activity of cannabidiol, a non-psychoactive constituent of cannabis, in acute carrageenan-induced inflammation in the rat paw. Naunyn Schmiedebergs Arch. Pharmacol. 2004, 369, 294–299. [Google Scholar] [CrossRef]
- Sumariwalla, P.F.; Gallily, R.; Tchilibon, S.; Fride, E.; Mechoulam, R.; Feldmann, M. A novel synthetic, nonpsychoactive cannabinoid acid (HU-320) with antiinflammatory properties in murine collagen-induced arthritis. Arthritis Rheum. 2004, 50, 985–998. [Google Scholar] [CrossRef]
- Buchweitz, J.P.; Karmaus, P.W.; Williams, K.J.; Harkema, J.R.; Kaminski, N.E. Targeted deletion of cannabinoid receptors CB1 and CB2 produced enhanced inflammatory responses to influenza A/PR/8/34 in the absence and presence of Delta9-tetrahydrocannabinol. J. Leukoc. Biol. 2008, 83, 785–796. [Google Scholar] [CrossRef]
- An, D.; Peigneur, S.; Hendrickx, L.A.; Tytgat, J. Targeting Cannabinoid Receptors: Current Status and Prospects of Natural Products. Int. J. Mol. Sci. 2020, 21, 5064. [Google Scholar] [CrossRef]
- Hasenoehrl, C.; Taschler, U.; Storr, M.; Schicho, R. The gastrointestinal tract—A central organ of cannabinoid signaling in health and disease. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2016, 28, 1765–1780. [Google Scholar] [CrossRef]
- Uranga, J.A.; Vera, G.; Abalo, R. Cannabinoid pharmacology and therapy in gut disorders. Biochem. Pharmacol. 2018, 157, 134–147. [Google Scholar] [CrossRef]
- Izzo, A.A.; Sharkey, K.A. Cannabinoids and the gut: New developments and emerging concepts. Pharmacol. Ther. 2010, 126, 21–38. [Google Scholar] [CrossRef]
- Goyal, H.; Singla, U.; Gupta, U.; May, E. Role of cannabis in digestive disorders. Eur. J. Gastroenterol. Hepatol. 2017, 29, 135–143. [Google Scholar] [CrossRef]
- Camilleri, M. Cannabinoids and gastrointestinal motility: Pharmacology, clinical effects, and potential therapeutics in humans. Neurogastroenterol. Motil. 2018, 30, e13370. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Li, Y.J. The vanilloid receptor TRPV1: Role in cardiovascular and gastrointestinal protection. Eur. J. Pharmacol. 2010, 627, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Overton, H.A.; Fyfe, M.C.; Reynet, C. GPR119, a novel G protein-coupled receptor target for the treatment of type 2 diabetes and obesity. Br. J. Pharmacol. 2008, 153 (Suppl. 1), S76–S81. [Google Scholar] [CrossRef] [PubMed]
- Galiazzo, G.; Giancola, F.; Stanzani, A.; Fracassi, F.; Bernardini, C.; Forni, M.; Pietra, M.; Chiocchetti, R. Localization of cannabinoid receptors CB1, CB2, GPR55, and PPARα in the canine gastrointestinal tract. Histochem. Cell Biol. 2018, 150, 187–205. [Google Scholar] [CrossRef]
- Brown, A.J. Novel cannabinoid receptors. Br. J. Pharmacol. 2007, 152, 567–575. [Google Scholar] [CrossRef]
- Harvey, B.S.; Sia, T.C.; Wattchow, D.A.; Smid, S.D. Interleukin 17A evoked mucosal damage is attenuated by cannabidiol and anandamide in a human colonic explant model. Cytokine 2014, 65, 236–244. [Google Scholar] [CrossRef]
- Alhamoruni, A.; Wright, K.L.; Larvin, M.; O’Sullivan, S.E. Cannabinoids mediate opposing effects on inflammation-induced intestinal permeability. Br. J. Pharmacol. 2012, 165, 2598–2610. [Google Scholar] [CrossRef]
- Alhamoruni, A.; Lee, A.C.; Wright, K.L.; Larvin, M.; O’Sullivan, S.E. Pharmacological effects of cannabinoids on the Caco-2 cell culture model of intestinal permeability. J. Pharmacol. Exp. Ther. 2010, 335, 92–102. [Google Scholar] [CrossRef]
- Karwad, M.A.; Couch, D.G.; Theophilidou, E.; Sarmad, S.; Barrett, D.A.; Larvin, M.; Wright, K.L.; Lund, J.N.; O’Sullivan, S.E. The role of CB(1) in intestinal permeability and inflammation. FASEB J. 2017, 31, 3267–3277. [Google Scholar] [CrossRef]
- Couch, D.G.; Tasker, C.; Theophilidou, E.; Lund, J.N.; O’Sullivan, S.E. Cannabidiol and palmitoylethanolamide are anti-inflammatory in the acutely inflamed human colon. Clin. Sci. 2017, 131, 2611–2626. [Google Scholar] [CrossRef]
- Nallathambi, R.; Mazuz, M.; Ion, A.; Selvaraj, G.; Weininger, S.; Fridlender, M.; Nasser, A.; Sagee, O.; Kumari, P.; Nemichenizer, D.; et al. Anti-Inflammatory Activity in Colon Models Is Derived from Δ9-Tetrahydrocannabinolic Acid That Interacts with Additional Compounds in Cannabis Extracts. Cannabis Cannabinoid Res. 2017, 2, 167–182. [Google Scholar] [CrossRef]
- Gigli, S.; Seguella, L.; Pesce, M.; Bruzzese, E.; D’Alessandro, A.; Cuomo, R.; Steardo, L.; Sarnelli, G.; Esposito, G. Cannabidiol restores intestinal barrier dysfunction and inhibits the apoptotic process induced by Clostridium difficile toxin A in Caco-2 cells. United Eur. Gastroenterol. J. 2017, 5, 1108–1115. [Google Scholar] [CrossRef]
- Wright, K.; Rooney, N.; Feeney, M.; Tate, J.; Robertson, D.; Welham, M.; Ward, S. Differential expression of cannabinoid receptors in the human colon: Cannabinoids promote epithelial wound healing. Gastroenterology 2005, 129, 437–453. [Google Scholar] [CrossRef]
- Picciolo, G.; Pallio, G.; Altavilla, D.; Vaccaro, M.; Oteri, G.; Irrera, N.; Squadrito, F. β-Caryophyllene Reduces the Inflammatory Phenotype of Periodontal Cells by Targeting CB2 Receptors. Biomedicines 2020, 8, 164. [Google Scholar] [CrossRef]
- Podolsky, D.K. Inflammatory bowel disease. N. Engl. J. Med. 2002, 347, 417–429. [Google Scholar] [CrossRef]
- Ihenetu, K.; Molleman, A.; Parsons, M.E.; Whelan, C.J. Inhibition of interleukin-8 release in the human colonic epithelial cell line HT-29 by cannabinoids. Eur. J. Pharmacol. 2003, 458, 207–215. [Google Scholar] [CrossRef]
- Ligresti, A.; Bisogno, T.; Matias, I.; De Petrocellis, L.; Cascio, M.G.; Cosenza, V.; D’Argenio, G.; Scaglione, G.; Bifulco, M.; Sorrentini, I.; et al. Possible endocannabinoid control of colorectal cancer growth. Gastroenterology 2003, 125, 677–687. [Google Scholar] [CrossRef]
- Couch, D.G.; Maudslay, H.; Doleman, B.; Lund, J.N.; O’Sullivan, S.E. The Use of Cannabinoids in Colitis: A Systematic Review and Meta-Analysis. Inflamm. Bowel Dis. 2018, 24, 680–697. [Google Scholar] [CrossRef]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; et al. Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochem. Pharmacol. 2013, 85, 1306–1316. [Google Scholar] [CrossRef]
- Borrelli, F.; Aviello, G.; Romano, B.; Orlando, P.; Capasso, R.; Maiello, F.; Guadagno, F.; Petrosino, S.; Capasso, F.; Di Marzo, V.; et al. Cannabidiol, a safe and non-psychotropic ingredient of the marijuana plant Cannabis sativa, is protective in a murine model of colitis. J. Mol. Med. 2009, 87, 1111–1121. [Google Scholar] [CrossRef]
- Pagano, E.; Iannotti, F.A.; Piscitelli, F.; Romano, B.; Lucariello, G.; Venneri, T.; Di Marzo, V.; Izzo, A.A.; Borrelli, F. Efficacy of combined therapy with fish oil and phytocannabinoids in murine intestinal inflammation. Phytother. Res. 2021, 35, 517–529. [Google Scholar] [CrossRef]
- Silvestri, C.; Pagano, E.; Lacroix, S.; Venneri, T.; Cristiano, C.; Calignano, A.; Parisi, O.A.; Izzo, A.A.; Di Marzo, V.; Borrelli, F. Fish Oil, Cannabidiol and the Gut Microbiota: An Investigation in a Murine Model of Colitis. Front. Pharmacol. 2020, 11, 585096. [Google Scholar] [CrossRef]
- Manichanh, C.; Borruel, N.; Casellas, F.; Guarner, F. The gut microbiota in IBD. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 599–608. [Google Scholar] [CrossRef]
- Muccioli, G.G.; Naslain, D.; Bäckhed, F.; Reigstad, C.S.; Lambert, D.M.; Delzenne, N.M.; Cani, P.D. The endocannabinoid system links gut microbiota to adipogenesis. Mol. Syst. Biol. 2010, 6, 392. [Google Scholar] [CrossRef]
- Kienzl, M.; Storr, M.; Schicho, R. Cannabinoids and Opioids in the Treatment of Inflammatory Bowel Diseases. Clin. Transl. Gastroenterol. 2020, 11, e00120. [Google Scholar] [CrossRef]
- Zoppi, S.; Madrigal, J.L.; Pérez-Nievas, B.G.; Marín-Jiménez, I.; Caso, J.R.; Alou, L.; García-Bueno, B.; Colón, A.; Manzanares, J.; Gómez-Lus, M.L.; et al. Endogenous cannabinoid system regulates intestinal barrier function in vivo through cannabinoid type 1 receptor activation. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G565–G571. [Google Scholar] [CrossRef]
- Cao, M.H.; Li, Y.Y.; Xu, J.; Feng, Y.J.; Lin, X.H.; Li, K.; Han, T.; Chen, C.J. Cannabinoid HU210 protects isolated rat stomach against impairment caused by serum of rats with experimental acute pancreatitis. PLoS ONE 2012, 7, e52921. [Google Scholar] [CrossRef]
- Lin, S.; Li, Y.; Shen, L.; Zhang, R.; Yang, L.; Li, M.; Li, K.; Fichna, J. The Anti-Inflammatory Effect and Intestinal Barrier Protection of HU210 Differentially Depend on TLR4 Signaling in Dextran Sulfate Sodium-Induced Murine Colitis. Dig. Dis. Sci. 2017, 62, 372–386. [Google Scholar] [CrossRef]
- Krohn, R.M.; Parsons, S.A.; Fichna, J.; Patel, K.D.; Yates, R.M.; Sharkey, K.A.; Storr, M.A. Abnormal cannabidiol attenuates experimental colitis in mice, promotes wound healing and inhibits neutrophil recruitment. J. Inflamm. 2016, 13, 21. [Google Scholar] [CrossRef]
- Negreanu, L.; Voiosu, T.; State, M.; Mateescu, R.B. Quality of colonoscopy preparation in patients with inflammatory bowel disease: Retrospective analysis of 348 colonoscopies. J. Int. Med. Res. 2020, 48. [Google Scholar] [CrossRef]
- Negreanu, L.; Voiosu, T.; State, M.; Voiosu, A.; Bengus, A.; Mateescu, B.R. Endoscopy in inflammatory bowel disease: From guidelines to real life. Ther. Adv. Gastroenterol. 2019, 12. [Google Scholar] [CrossRef]
- Voiosu, T.; Benguş, A.; Dinu, R.; Voiosu, A.M.; Bălănescu, P.; Băicuş, C.; Diculescu, M.; Voiosu, R.; Mateescu, B. Rapid fecal calprotectin level assessment and the SIBDQ score can accurately detect active mucosal inflammation in IBD patients in clinical remission: A prospective study. J. Gastrointest. Liver Dis. 2014, 23, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Phatak, U.P.; Rojas-Velasquez, D.; Porto, A.; Pashankar, D.S. Prevalence and Patterns of Marijuana Use in Young Adults With Inflammatory Bowel Disease. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Storr, M.; Devlin, S.; Kaplan, G.G.; Panaccione, R.; Andrews, C.N. Cannabis use provides symptom relief in patients with inflammatory bowel disease but is associated with worse disease prognosis in patients with Crohn’s disease. Inflamm. Bowel Dis. 2014, 20, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Merker, A.M.; Riaz, M.; Friedman, S.; Allegretti, J.R.; Korzenik, J. Legalization of Medicinal Marijuana Has Minimal Impact on Use Patterns in Patients with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2018, 24, 2309–2314. [Google Scholar] [CrossRef] [PubMed]
- Couch, D.G.; Cook, H.; Ortori, C.; Barrett, D.; Lund, J.N.; O’Sullivan, S.E. Palmitoylethanolamide and Cannabidiol Prevent Inflammation-induced Hyperpermeability of the Human Gut In Vitro and In Vivo—A Randomized, Placebo-controlled, Double-blind Controlled Trial. Inflamm. Bowel Dis. 2019, 25, 1006–1018. [Google Scholar] [CrossRef] [PubMed]
- Naftali, T.; Bar-Lev Schleider, L.; Dotan, I.; Lansky, E.P.; Sklerovsky Benjaminov, F.; Konikoff, F.M. Cannabis induces a clinical response in patients with Crohn’s disease: A prospective placebo-controlled study. Clin. Gastroenterol. Hepatol. 2013, 11, 1276–1280.e1. [Google Scholar] [CrossRef]
- Naftali, T.; Mechulam, R.; Marii, A.; Gabay, G.; Stein, A.; Bronshtain, M.; Laish, I.; Benjaminov, F.; Konikoff, F.M. Low-Dose Cannabidiol Is Safe but Not Effective in the Treatment for Crohn’s Disease, a Randomized Controlled Trial. Dig. Dis. Sci. 2017, 62, 1615–1620. [Google Scholar] [CrossRef]
- Kafil, T.S.; Nguyen, T.M.; MacDonald, J.K.; Chande, N. Cannabis for the treatment of Crohn’s disease. Cochrane Database Syst. Rev. 2018, 11, CD012853. [Google Scholar] [CrossRef]
- Irving, P.M.; Iqbal, T.; Nwokolo, C.; Subramanian, S.; Bloom, S.; Prasad, N.; Hart, A.; Murray, C.; Lindsay, J.O.; Taylor, A.; et al. A Randomized, Double-blind, Placebo-controlled, Parallel-group, Pilot Study of Cannabidiol-rich Botanical Extract in the Symptomatic Treatment of Ulcerative Colitis. Inflamm. Bowel Dis. 2018, 24, 714–724. [Google Scholar] [CrossRef]
- Naftali, T.; Benjaminov, F.; Lish, I. Cannabis induces clinical and endoscopic improvement in moderately active ulcerative colitis. J. Crohns Colitis 2018, 12, S306. [Google Scholar] [CrossRef]
- Tashkin, D.P.; Roth, M.D. Pulmonary effects of inhaled cannabis smoke. Am. J. Drug Alcohol Abus. 2019, 45, 596–609. [Google Scholar] [CrossRef]
- Yayan, J.; Rasche, K. Damaging Effects of Cannabis Use on the Lungs. Adv. Exp. Med. Biol. 2016, 952, 31–34. [Google Scholar] [CrossRef]
- Fligiel, S.E.; Beals, T.F.; Tashkin, D.P.; Paule, M.G.; Scallet, A.C.; Ali, S.F.; Bailey, J.R.; Slikker, W., Jr. Marijuana exposure and pulmonary alterations in primates. Pharmacol. Biochem. Behav. 1991, 40, 637–642. [Google Scholar] [CrossRef]
- Gong, H., Jr.; Fligiel, S.; Tashkin, D.P.; Barbers, R.G. Tracheobronchial changes in habitual, heavy smokers of marijuana with and without tobacco. Am. Rev. Respir. Dis. 1987, 136, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Burstein, S.; Hunter, S.A.; Latham, V.; Mechoulam, R.; Melchior, D.L.; Renzulli, L.; Tefft, R.E., Jr. Prostaglandins and cannabis XV. Comparison of enantiomeric cannabinoids in stimulating prostaglandin synthesis in fibroblasts. Life Sci. 1986, 39, 1813–1823. [Google Scholar] [CrossRef]
- Roth, M.D.; Whittaker, K.; Salehi, K.; Tashkin, D.P.; Baldwin, G.C. Mechanisms for impaired effector function in alveolar macrophages from marijuana and cocaine smokers. J. Neuroimmunol. 2004, 147, 82–86. [Google Scholar] [CrossRef]
- Turcotte, C.; Blanchet, M.R.; Laviolette, M.; Flamand, N. Impact of Cannabis, Cannabinoids, and Endocannabinoids in the Lungs. Front. Pharmacol. 2016, 7, 317. [Google Scholar] [CrossRef]
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic. Br. J. Pharmacol. 2020, 177, 4967–4970. [Google Scholar] [CrossRef]
- Raj, V.; Park, J.G.; Cho, K.H.; Choi, P.; Kim, T.; Ham, J.; Lee, J. Assessment of antiviral potencies of cannabinoids against SARS-CoV-2 using computational and in vitro approaches. Int. J. Biol. Macromol. 2021, 168, 474–485. [Google Scholar] [CrossRef]
- Costiniuk, C.T.; Jenabian, M.A. Acute inflammation and pathogenesis of SARS-CoV-2 infection: Cannabidiol as a potential anti-inflammatory treatment? Cytokine Growth Factor Rev. 2020, 53, 63–65. [Google Scholar] [CrossRef]
- Brown, J.D. Cannabidiol as prophylaxis for SARS-CoV-2 and COVID-19? Unfounded claims versus potential risks of medications during the pandemic. Res. Soc. Adm. Pharm. 2021, 17, 2053. [Google Scholar] [CrossRef]
- Calignano, A.; Kátona, I.; Désarnaud, F.; Giuffrida, A.; La Rana, G.; Mackie, K.; Freund, T.F.; Piomelli, D. Bidirectional control of airway responsiveness by endogenous cannabinoids. Nature 2000, 408, 96–101. [Google Scholar] [CrossRef]
- Bozkurt, T.E. Endocannabinoid System in the Airways. Molecules 2019, 24, 4626. [Google Scholar] [CrossRef]
- Fantauzzi, M.F.; Aguiar, J.A.; Tremblay, B.J.-M.; Yanagihara, T.; Chandiramohan, A.; Revill, S.; Ryu, M.H.; Carlsten, C.; Ask, K.; Stämpfli, M.; et al. Expression of the endocannabinoid system in the human airway epithelial cells—Impact of sex and chronic respiratory disease status. bioRxiv 2020. [Google Scholar] [CrossRef]
- Sarafian, T.A.; Habib, N.; Oldham, M.; Seeram, N.; Lee, R.P.; Lin, L.; Tashkin, D.P.; Roth, M.D. Inhaled marijuana smoke disrupts mitochondrial energetics in pulmonary epithelial cells in vivo. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 290, L1202–L1209. [Google Scholar] [CrossRef]
- Sarafian, T.A.; Magallanes, J.A.; Shau, H.; Tashkin, D.; Roth, M.D. Oxidative stress produced by marijuana smoke. An adverse effect enhanced by cannabinoids. Am. J. Respir. Cell Mol. Biol. 1999, 20, 1286–1293. [Google Scholar] [CrossRef]
- Srivastava, M.D.; Srivastava, B.I.; Brouhard, B. Delta9 tetrahydrocannabinol and cannabidiol alter cytokine production by human immune cells. Immunopharmacology 1998, 40, 179–185. [Google Scholar] [CrossRef]
- Liu, A.P.; Yuan, Q.H.; Zhang, B.; Yang, L.; He, Q.W.; Chen, K.; Liu, Q.S.; Li, Z.; Zhan, J. Cannabinoid receptor 2 activation alleviates septic lung injury by promoting autophagy via inhibition of inflammatory mediator release. Cell. Signal. 2020, 69, 109556. [Google Scholar] [CrossRef]
- Muthumalage, T.; Rahman, I. Cannabidiol differentially regulates basal and LPS-induced inflammatory responses in macrophages, lung epithelial cells, and fibroblasts. Toxicol. Appl. Pharmacol. 2019, 382, 114713. [Google Scholar] [CrossRef]
- Adcock, I.M.; Caramori, G. Cross-talk between pro-inflammatory transcription factors and glucocorticoids. Immunol. Cell Biol. 2001, 79, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Staiano, R.I.; Loffredo, S.; Borriello, F.; Iannotti, F.A.; Piscitelli, F.; Orlando, P.; Secondo, A.; Granata, F.; Lepore, M.T.; Fiorelli, A.; et al. Human lung-resident macrophages express CB1 and CB2 receptors whose activation inhibits the release of angiogenic and lymphangiogenic factors. J. Leukoc. Biol. 2016, 99, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Helyes, Z.; Kemény, Á.; Csekő, K.; Szőke, É.; Elekes, K.; Mester, M.; Sándor, K.; Perkecz, A.; Kereskai, L.; Márk, L.; et al. Marijuana smoke induces severe pulmonary hyperresponsiveness, inflammation, and emphysema in a predictive mouse model not via CB1 receptor activation. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 313, L267–L277. [Google Scholar] [CrossRef] [PubMed]
- Kleinman, M.T.; Arechavala, R.J.; Herman, D.; Shi, J.; Hasen, I.; Ting, A.; Dai, W.; Carreno, J.; Chavez, J.; Zhao, L.; et al. E-cigarette or Vaping Product Use-Associated Lung Injury Produced in an Animal Model from Electronic Cigarette Vapor Exposure Without Tetrahydrocannabinol or Vitamin E Oil. J. Am. Heart Assoc. 2020, 9, e017368. [Google Scholar] [CrossRef] [PubMed]
- Zawatsky, C.N.; Abdalla, J.; Cinar, R. Synthetic cannabinoids induce acute lung inflammation via cannabinoid receptor 1 activation. ERJ Open Res. 2020, 6. [Google Scholar] [CrossRef]
- Ribeiro, A.; Almeida, V.I.; Costola-de-Souza, C.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Gimenes-Junior, J.A.; Akamine, A.T.; Crippa, J.A.; Tavares-de-Lima, W.; et al. Cannabidiol improves lung function and inflammation in mice submitted to LPS-induced acute lung injury. Immunopharmacol. Immunotoxicol. 2015, 37, 35–41. [Google Scholar] [CrossRef]
- Ribeiro, A.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Mariano-Souza, D.P.; Quinteiro-Filho, W.M.; Akamine, A.T.; Almeida, V.I.; Quevedo, J.; Dal-Pizzol, F.; et al. Cannabidiol, a non-psychotropic plant-derived cannabinoid, decreases inflammation in a murine model of acute lung injury: Role for the adenosine A(2A) receptor. Eur. J. Pharmacol. 2012, 678, 78–85. [Google Scholar] [CrossRef]
- Berdyshev, E.; Boichot, E.; Corbel, M.; Germain, N.; Lagente, V. Effects of cannabinoid receptor ligands on LPS-induced pulmonary inflammation in mice. Life Sci. 1998, 63, PL125–PL129. [Google Scholar] [CrossRef]
- Karmaus, P.W.; Wagner, J.G.; Harkema, J.R.; Kaminski, N.E.; Kaplan, B.L. Cannabidiol (CBD) enhances lipopolysaccharide (LPS)-induced pulmonary inflammation in C57BL/6 mice. J. Immunotoxicol. 2013, 10, 321–328. [Google Scholar] [CrossRef]
- Arruza, L.; Pazos, M.R.; Mohammed, N.; Escribano, N.; Lafuente, H.; Santos, M.; Alvarez-Díaz, F.J.; Hind, W.; Martínez-Orgado, J. Cannabidiol reduces lung injury induced by hypoxic-ischemic brain damage in newborn piglets. Pediatr. Res. 2017, 82, 79–86. [Google Scholar] [CrossRef]
- Tauber, S.; Paulsen, K.; Wolf, S.; Synwoldt, P.; Pahl, A.; Schneider-Stock, R.; Ullrich, O. Regulation of MMP-9 by a WIN-binding site in the monocyte-macrophage system independent from cannabinoid receptors. PLoS ONE 2012, 7, e48272. [Google Scholar] [CrossRef]
- Vuolo, F.; Petronilho, F.; Sonai, B.; Ritter, C.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.; Dal-Pizzol, F. Evaluation of Serum Cytokines Levels and the Role of Cannabidiol Treatment in Animal Model of Asthma. Mediat. Inflamm. 2015, 2015, 538670. [Google Scholar] [CrossRef]
- Vuolo, F.; Abreu, S.C.; Michels, M.; Xisto, D.G.; Blanco, N.G.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.; Reis, C.; Bahl, M.; et al. Cannabidiol reduces airway inflammation and fibrosis in experimental allergic asthma. Eur. J. Pharmacol. 2019, 843, 251–259. [Google Scholar] [CrossRef]
- Frei, R.B.; Luschnig, P.; Parzmair, G.P.; Peinhaupt, M.; Schranz, S.; Fauland, A.; Wheelock, C.E.; Heinemann, A.; Sturm, E.M. Cannabinoid receptor 2 augments eosinophil responsiveness and aggravates allergen-induced pulmonary inflammation in mice. Allergy 2016, 71, 944–956. [Google Scholar] [CrossRef]
- Ferrini, M.E.; Hong, S.; Stierle, A.; Stierle, D.; Stella, N.; Roberts, K.; Jaffar, Z. CB2 receptors regulate natural killer cells that limit allergic airway inflammation in a murine model of asthma. Allergy 2017, 72, 937–947. [Google Scholar] [CrossRef]
- Tahamtan, A.; Tavakoli-Yaraki, M.; Shadab, A.; Rezaei, F.; Marashi, S.M.; Shokri, F.; Mokhatri-Azad, T.; Salimi, V. The Role of Cannabinoid Receptor 1 in the Immunopathology of Respiratory Syncytial Virus. Viral Immunol. 2018, 31, 292–298. [Google Scholar] [CrossRef]
- Tahamtan, A.; Samieipoor, Y.; Nayeri, F.S.; Rahbarimanesh, A.A.; Izadi, A.; Rashidi-Nezhad, A.; Tavakoli-Yaraki, M.; Farahmand, M.; Bont, L.; Shokri, F.; et al. Effects of cannabinoid receptor type 2 in respiratory syncytial virus infection in human subjects and mice. Virulence 2018, 9, 217–230. [Google Scholar] [CrossRef]
- Karmaus, P.W.; Chen, W.; Crawford, R.; Kaplan, B.L.; Kaminski, N.E. Δ9-tetrahydrocannabinol impairs the inflammatory response to influenza infection: Role of antigen-presenting cells and the cannabinoid receptors 1 and 2. Toxicol. Sci. 2013, 131, 419–433. [Google Scholar] [CrossRef]
- Buchweitz, J.P.; Karmaus, P.W.; Harkema, J.R.; Williams, K.J.; Kaminski, N.E. Modulation of airway responses to influenza A/PR/8/34 by Delta9-tetrahydrocannabinol in C57BL/6 mice. J. Pharmacol. Exp. Ther. 2007, 323, 675–683. [Google Scholar] [CrossRef]
- Rao, R.; Nagarkatti, P.S.; Nagarkatti, M. Δ(9) Tetrahydrocannabinol attenuates Staphylococcal enterotoxin B-induced inflammatory lung injury and prevents mortality in mice by modulation of miR-17-92 cluster and induction of T-regulatory cells. Br. J. Pharmacol. 2015, 172, 1792–1806. [Google Scholar] [CrossRef]
- Zeng, J.; Li, X.; Cheng, Y.; Ke, B.; Wang, R. Activation of cannabinoid receptor type 2 reduces lung ischemia reperfusion injury through PI3K/Akt pathway. Int. J. Clin. Exp. Pathol. 2019, 12, 4096–4105. [Google Scholar]
- Çakır, M.; Tekin, S.; Okan, A.; Çakan, P.; Doğanyiğit, Z. The ameliorating effect of cannabinoid type 2 receptor activation on brain, lung, liver and heart damage in cecal ligation and puncture-induced sepsis model in rats. Int. Immunopharmacol. 2020, 78, 105978. [Google Scholar] [CrossRef]
- Tschöp, J.; Kasten, K.R.; Nogueiras, R.; Goetzman, H.S.; Cave, C.M.; England, L.G.; Dattilo, J.; Lentsch, A.B.; Tschöp, M.H.; Caldwell, C.C. The cannabinoid receptor 2 is critical for the host response to sepsis. J. Immunol. 2009, 183, 499–505. [Google Scholar] [CrossRef]
- Liu, M.W.; Su, M.X.; Wang, Y.H.; Wei, W.; Qin, L.F.; Liu, X.; Tian, M.L.; Qian, C.Y. Effect of melilotus extract on lung injury by upregulating the expression of cannabinoid CB2 receptors in septic rats. BMC Complement. Altern. Med. 2014, 14, 94. [Google Scholar] [CrossRef]
- Fujii, M.; Sherchan, P.; Soejima, Y.; Doycheva, D.; Zhao, D.; Zhang, J.H. Cannabinoid Receptor Type 2 Agonist Attenuates Acute Neurogenic Pulmonary Edema by Preventing Neutrophil Migration after Subarachnoid Hemorrhage in Rats. Acta Neurochir. Suppl. 2016, 121, 135–139. [Google Scholar] [CrossRef]
- Kopczyńska, B.; Sulejczak, D.; Wełniak-Kamińska, M.; Gietka, A.; Grieb, P. Anandamide enhances expression of heat shock proteins Hsp70 and Hsp25 in rat lungs. Eur. J. Pharmacol. 2011, 668, 257–263. [Google Scholar] [CrossRef]
- Andrade-Silva, M.; Correa, L.B.; Candéa, A.L.; Cavalher-Machado, S.C.; Barbosa, H.S.; Rosas, E.C.; Henriques, M.G. The cannabinoid 2 receptor agonist β-caryophyllene modulates the inflammatory reaction induced by Mycobacterium bovis BCG by inhibiting neutrophil migration. Inflamm. Res. 2016, 65, 869–879. [Google Scholar] [CrossRef]
- Conuel, E.J.; Chieng, H.C.; Fantauzzi, J.; Pokhrel, K.; Goldman, C.; Smith, T.C.; Tiwari, A.; Chopra, A.; Judson, M.A. Cannabinoid Oil Vaping-Associated Lung Injury and its Radiographic Appearance. Am. J. Med. 2020, 133, 865–867. [Google Scholar] [CrossRef]
- Öcal, N.; Doğan, D.; Çiçek, A.F.; Yücel, O.; Tozkoparan, E. Acute Eosinophilic Pneumonia with Respiratory Failure Induced by Synthetic Cannabinoid Inhalation. Balk. Med. J. 2016, 33, 688–690. [Google Scholar] [CrossRef]
- Alhadi, S.; Tiwari, A.; Vohra, R.; Gerona, R.; Acharya, J.; Bilello, K. High times, low sats: Diffuse pulmonary infiltrates associated with chronic synthetic cannabinoid use. J. Med. Toxicol. 2013, 9, 199–206. [Google Scholar] [CrossRef]
- Alon, M.H.; Saint-Fleur, M.O. Synthetic cannabinoid induced acute respiratory depression: Case series and literature review. Respir. Med. Case Rep. 2017, 22, 137–141. [Google Scholar] [CrossRef]
- Chmiel, J.F.; Flume, P.; Downey, D.G.; Dozor, A.J.; Colombo, C.; Mazurek, H.; Sapiejka, E.; Rachel, M.; Constantine, S.; Conley, B.; et al. Safety and efficacy of lenabasum in a phase 2 randomized, placebo-controlled trial in adults with cystic fibrosis. J. Cyst. Fibros. 2021, 20, 78–85. [Google Scholar] [CrossRef]
- Investigation of Cannabis for Pain and Inflammation in Lung Cancer. Available online: https://ClinicalTrials.gov/show/NCT02675842 (accessed on 3 April 2021).
- Tashkin, D.P.; Shapiro, B.J.; Frank, I.M. Acute pulmonary physiologic effects of smoked marijuana and oral (Delta)9 -tetrahydrocannabinol in healthy young men. N. Engl. J. Med. 1973, 289, 336–341. [Google Scholar] [CrossRef]
- Abdallah, S.J.; Smith, B.M.; Ware, M.A.; Moore, M.; Li, P.Z.; Bourbeau, J.; Jensen, D. Effect of Vaporized Cannabis on Exertional Breathlessness and Exercise Endurance in Advanced Chronic Obstructive Pulmonary Disease. A Randomized Controlled Trial. Ann. Am. Thorac. Soc. 2018, 15, 1146–1158. [Google Scholar] [CrossRef]
- Gong, H., Jr.; Tashkin, D.P.; Calvarese, B. Comparison of bronchial effects of nabilone and terbutaline in healthy and asthmatic subjects. J. Clin. Pharmacol. 1983, 23, 127–133. [Google Scholar] [CrossRef]
- Tashkin, D.P.; Reiss, S.; Shapiro, B.J.; Calvarese, B.; Olsen, J.L.; Lodge, J.W. Bronchial effects of aerosolized delta 9-tetrahydrocannabinol in healthy and asthmatic subjects. Am. Rev. Respir. Dis. 1977, 115, 57–65. [Google Scholar] [CrossRef]
- Abboud, R.T.; Sanders, H.D. Effect of oral administration of delta-tetrahydrocannabinol on airway mechanics in normal and asthmatic subjects. Chest 1976, 70, 480–485. [Google Scholar] [CrossRef]
- Van Dam, N.T.; Earleywine, M. Pulmonary function in cannabis users: Support for a clinical trial of the vaporizer. Int. J. Drug Policy 2010, 21, 511–513. [Google Scholar] [CrossRef]
- Scheau, C.; Badarau, I.A.; Mihai, G.L.; Scheau, A.-E.; Costache, D.O.; Constantin, C.; Calina, D.; Caruntu, C.; Costache, R.S.; Caruntu, A. Cannabinoids in the pathophysiology of skin inflammation. Molecules 2020, 25, 652. [Google Scholar] [CrossRef]
- Sheriff, T.; Lin, M.J.; Dubin, D.; Khorasani, H. The potential role of cannabinoids in dermatology. J. Dermatol. Treat. 2020, 31, 839–845. [Google Scholar] [CrossRef]
- Baswan, S.M.; Klosner, A.E.; Glynn, K.; Rajgopal, A.; Malik, K.; Yim, S.; Stern, N. Therapeutic Potential of Cannabidiol (CBD) for Skin Health and Disorders. Clin. Cosmet. Investig. Dermatol. 2020, 13, 927–942. [Google Scholar] [CrossRef]
- Cintosun, A.; Lara-Corrales, I.; Pope, E. Mechanisms of Cannabinoids and Potential Applicability to Skin Diseases. Clin. Drug Investig. 2020, 40, 293–304. [Google Scholar] [CrossRef]
- Zagórska-Dziok, M.; Bujak, T.; Ziemlewska, A.; Nizioł-Łukaszewska, Z. Positive Effect of Cannabis sativa L. Herb Extracts on Skin Cells and Assessment of Cannabinoid-Based Hydrogels Properties. Molecules 2021, 26, 802. [Google Scholar] [CrossRef]
- Eagleston, L.R.M.; Kalani, N.K.; Patel, R.R.; Flaten, H.K.; Dunnick, C.A.; Dellavalle, R.P. Cannabinoids in dermatology: A scoping review. Dermatol. Online J. 2018, 24, 1. [Google Scholar]
- Nickles, M.A.; Lio, P.A. Cannabinoids in Dermatology: Hope or Hype? Cannabis Cannabinoid Res. 2020, 5, 279–282. [Google Scholar] [CrossRef]
- Bíró, T.; Tóth, B.I.; Haskó, G.; Paus, R.; Pacher, P. The endocannabinoid system of the skin in health and disease: Novel perspectives and therapeutic opportunities. Trends Pharmacol. Sci. 2009, 30, 411–420. [Google Scholar] [CrossRef]
- Dobrosi, N.; Tóth, B.I.; Nagy, G.; Dózsa, A.; Géczy, T.; Nagy, L.; Zouboulis, C.C.; Paus, R.; Kovács, L.; Bíró, T. Endocannabinoids enhance lipid synthesis and apoptosis of human sebocytes via cannabinoid receptor-2-mediated signaling. FASEB J. 2008, 22, 3685–3695. [Google Scholar] [CrossRef]
- Telek, A.; Bíró, T.; Bodó, E.; Tóth, B.I.; Borbíró, I.; Kunos, G.; Paus, R. Inhibition of human hair follicle growth by endo- and exocannabinoids. FASEB J. 2007, 21, 3534–3541. [Google Scholar] [CrossRef]
- Tóth, B.I.; Oláh, A.; Szöllősi, A.G.; Bíró, T. TRP channels in the skin. Br. J. Pharmacol. 2014, 171, 2568–2581. [Google Scholar] [CrossRef]
- Gouin, O.; L’Herondelle, K.; Lebonvallet, N.; Le Gall-Ianotto, C.; Sakka, M.; Buhé, V.; Plée-Gautier, E.; Carré, J.L.; Lefeuvre, L.; Misery, L.; et al. TRPV1 and TRPA1 in cutaneous neurogenic and chronic inflammation: Pro-inflammatory response induced by their activation and their sensitization. Protein Cell 2017, 8, 644–661. [Google Scholar] [CrossRef] [PubMed]
- Hänel, K.H.; Cornelissen, C.; Lüscher, B.; Baron, J.M. Cytokines and the skin barrier. Int. J. Mol. Sci. 2013, 14, 6720–6745. [Google Scholar] [CrossRef]
- Zaalberg, A.; Moradi Tuchayi, S.; Ameri, A.H.; Ngo, K.H.; Cunningham, T.J.; Eliane, J.-P.; Livneh, M.; Horn, T.D.; Rosman, I.S.; Musiek, A.; et al. Chronic Inflammation Promotes Skin Carcinogenesis in Cancer-Prone Discoid Lupus Erythematosus. J. Investig. Dermatol. 2019, 139, 62–70. [Google Scholar] [CrossRef]
- Solomon, I.; Voiculescu, V.M.; Caruntu, C.; Lupu, M.; Popa, A.; Ilie, M.A.; Albulescu, R.; Caruntu, A.; Tanase, C.; Constantin, C.; et al. Neuroendocrine Factors and Head and Neck Squamous Cell Carcinoma: An Affair to Remember. Dis. Markers 2018, 2018, 9787831. [Google Scholar] [CrossRef]
- Ion, A.; Popa, I.M.; Papagheorghe, L.M.; Lisievici, C.; Lupu, M.; Voiculescu, V.; Caruntu, C.; Boda, D. Proteomic Approaches to Biomarker Discovery in Cutaneous T-Cell Lymphoma. Dis. Markers 2016, 2016, 9602472. [Google Scholar] [CrossRef]
- Karsak, M.; Gaffal, E.; Date, R.; Wang-Eckhardt, L.; Rehnelt, J.; Petrosino, S.; Starowicz, K.; Steuder, R.; Schlicker, E.; Cravatt, B.; et al. Attenuation of Allergic Contact Dermatitis Through the Endocannabinoid System. Science 2007, 316, 1494–1497. [Google Scholar] [CrossRef]
- Petrosino, S.; Cristino, L.; Karsak, M.; Gaffal, E.; Ueda, N.; Tüting, T.; Bisogno, T.; De Filippis, D.; D’Amico, A.; Saturnino, C.; et al. Protective role of palmitoylethanolamide in contact allergic dermatitis. Allergy 2010, 65, 698–711. [Google Scholar] [CrossRef]
- Petrosino, S.; Verde, R.; Vaia, M.; Allara, M.; Iuvone, T.; Di Marzo, V. Anti-inflammatory Properties of Cannabidiol, a Nonpsychotropic Cannabinoid, in Experimental Allergic Contact Dermatitis. J. Pharm. Exp. 2018, 365, 652–663. [Google Scholar] [CrossRef]
- Rio, C.D.; Millan, E.; Garcia, V.; Appendino, G.; DeMesa, J.; Munoz, E. The endocannabinoid system of the skin. A potential approach for the treatment of skin disorders. Biochem. Pharmacol. 2018, 157, 122–133. [Google Scholar] [CrossRef]
- Wilkinson, J.D.; Williamson, E.M. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J. Dermatol. Sci. 2007, 45, 87–92. [Google Scholar] [CrossRef]
- Ramot, Y.; Sugawara, K.; Zakany, N.; Toth, B.I.; Biro, T.; Paus, R. A novel control of human keratin expression: Cannabinoid receptor 1-mediated signaling down-regulates the expression of keratins K6 and K16 in human keratinocytes in vitro and in situ. PeerJ 2013, 1, e40. [Google Scholar] [CrossRef]
- Robinson, E.S.; Alves, P.; Bashir, M.M.; Zeidi, M.; Feng, R.; Werth, V.P. Cannabinoid Reduces Inflammatory Cytokines, Tumor Necrosis Factor-alpha, and Type I Interferons in Dermatomyositis In Vitro. J. Investig. Dermatol. 2017, 137, 2445–2447. [Google Scholar] [CrossRef]
- Oláh, A.; Tóth, B.I.; Borbíró, I.; Sugawara, K.; Szöllõsi, A.G.; Czifra, G.; Pál, B.; Ambrus, L.; Kloepper, J.; Camera, E.; et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J. Clin. Investig. 2014, 124, 3713–3724. [Google Scholar] [CrossRef]
- Maor, Y.; Yu, J.; Kuzontkoski, P.M.; Dezube, B.J.; Zhang, X.; Groopman, J.E. Cannabidiol inhibits growth and induces programmed cell death in kaposi sarcoma-associated herpesvirus-infected endothelium. Genes Cancer 2012, 3, 512–520. [Google Scholar] [CrossRef]
- Bechara, C.; Chai, H.; Lin, P.H.; Yao, Q.; Chen, C. Growth related oncogene-alpha (GRO-alpha): Roles in atherosclerosis, angiogenesis and other inflammatory conditions. Med. Sci. Monit. 2007, 13, Ra87–Ra90. [Google Scholar]
- Del Rio, C.; Navarrete, C.; Collado, J.A.; Bellido, M.L.; Gomez-Canas, M.; Pazos, M.R.; Fernandez-Ruiz, J.; Pollastro, F.; Appendino, G.; Calzado, M.A.; et al. The cannabinoid quinol VCE-004.8 alleviates bleomycin-induced scleroderma and exerts potent antifibrotic effects through peroxisome proliferator-activated receptor-gamma and CB2 pathways. Sci. Rep. 2016, 6, 21703. [Google Scholar] [CrossRef]
- Balistreri, E.; Garcia-Gonzalez, E.; Selvi, E.; Akhmetshina, A.; Palumbo, K.; Lorenzini, S.; Maggio, R.; Lucattelli, M.; Galeazzi, M.; Distler, J.W. The cannabinoid WIN55, 212–2 abrogates dermal fibrosis in scleroderma bleomycin model. Ann. Rheum. Dis. 2011, 70, 695–699. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, B.; Park, B.M.; Jeon, J.E.; Lee, S.H.; Mann, S.; Ahn, S.K.; Hong, S.P.; Jeong, S.K. Topical cannabinoid receptor 1 agonist attenuates the cutaneous inflammatory responses in oxazolone-induced atopic dermatitis model. Int. J. Dermatol. 2015, 54, e401–e408. [Google Scholar] [CrossRef]
- Formukong, E.A.; Evans, A.T.; Evans, F.J. Analgesic and antiinflammatory activity of constituents of Cannabis sativa L. Inflammation 1988, 12, 361–371. [Google Scholar] [CrossRef]
- Armstrong, J.L.; Hill, D.S.; McKee, C.S.; Hernandez-Tiedra, S.; Lorente, M.; Lopez-Valero, I.; Eleni Anagnostou, M.; Babatunde, F.; Corazzari, M.; Redfern, C.P.F.; et al. Exploiting cannabinoid-induced cytotoxic autophagy to drive melanoma cell death. J. Investig. Dermatol. 2015, 135, 1629–1637. [Google Scholar] [CrossRef]
- Calenic, B.; Greabu, M.; Caruntu, C.; Tanase, C.; Battino, M. Oral keratinocyte stem/progenitor cells: Specific markers, molecular signaling pathways and potential uses. Periodontology 2015, 69, 68–82. [Google Scholar] [CrossRef]
- Lupu, M.; Caruntu, C.; Ghita, M.A.; Voiculescu, V.; Voiculescu, S.; Rosca, A.E.; Caruntu, A.; Moraru, L.; Popa, I.M.; Calenic, B.; et al. Gene Expression and Proteome Analysis as Sources of Biomarkers in Basal Cell Carcinoma. Dis. Markers 2016, 2016, 9831237. [Google Scholar] [CrossRef] [PubMed]
- Voiculescu, V.; Calenic, B.; Ghita, M.; Lupu, M.; Caruntu, A.; Moraru, L.; Voiculescu, S.; Ion, A.; Greabu, M.; Ishkitiev, N.; et al. From Normal Skin to Squamous Cell Carcinoma: A Quest for Novel Biomarkers. Dis. Markers 2016, 2016, 4517492. [Google Scholar] [CrossRef]
- Tampa, M.; Caruntu, C.; Mitran, M.; Mitran, C.; Sarbu, I.; Rusu, L.C.; Matei, C.; Constantin, C.; Neagu, M.; Georgescu, S.R. Markers of Oral Lichen Planus Malignant Transformation. Dis. Markers 2018, 2018, 1959506. [Google Scholar] [CrossRef] [PubMed]
- Scheau, C.; Badarau, I.A.; Costache, R.; Caruntu, C.; Mihai, G.L.; Didilescu, A.C.; Constantin, C.; Neagu, M. The Role of Matrix Metalloproteinases in the Epithelial-Mesenchymal Transition of Hepatocellular Carcinoma. Anal. Cell. Pathol. 2019, 2019, 9423907. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Bode, A.M.; Zhao, Q.; Cho, Y.Y.; Zhu, F.; Ma, W.Y.; Dong, Z. The cannabinoid receptors are required for ultraviolet-induced inflammation and skin cancer development. Cancer Res. 2008, 68, 3992–3998. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Akhtar, N. The safety and efficacy of 3% Cannabis seeds extract cream for reduction of human cheek skin sebum and erythema content. Pak. J. Pharm. Sci. 2015, 28, 1389–1395. [Google Scholar] [PubMed]
- Evaluation of BTX 1503 in Patients with Moderate to Severe Acne Vulgaris. Available online: https://clinicaltrials.gov/ct2/show/NCT03573518 (accessed on 10 January 2020).
- Spiera, R.; Hummers, L.; Chung, L.; Frech, T.; Domsic, R.; Furst, D.; Gordon, J.; Mayes, M.; Simms, R.; Constantine, S.; et al. OP0126 A phase 2 study of safety and efficacy of anabasum (JBT-101) in systemic sclerosis. Ann. Rheum. Dis. 2017, 76, 105. [Google Scholar] [CrossRef]
- Burstein, S.H. Ajulemic acid: Potential treatment for chronic inflammation. Pharmacol. Res. Perspect. 2018, 6, e00394. [Google Scholar] [CrossRef]
- Chen, K.; Zeidi, M.; Reddy, N.; White, B.; Werth, V. Fri0307 lenabasum, a cannabinoid type 2 receptor agonist, reduces cd4 cell populations and downregulates type 1 and 2 interferon activities in lesional dermatomyositis skin. Ann. Rheum. Dis. 2019, 78, 835. [Google Scholar] [CrossRef]
- Werth, V.; Oddis, C.V.; Lundberg, I.E.; Fiorentino, D.; Cornwall, C.; Dgetluck, N.; Constantine, S.; White, B. Sat0303 design of phase 3 study of lenabasum for the treatment of dermatomyositis. Ann. Rheum. Dis. 2019, 78, 1228. [Google Scholar] [CrossRef]
- Dariš, B.; Tancer Verboten, M.; Knez, Ž.; Ferk, P. Cannabinoids in cancer treatment: Therapeutic potential and legislation. Bosn. J. Basic Med. Sci. Udruz. Basicnih Med. Znan. 2019, 19, 14–23. [Google Scholar] [CrossRef]
- Jin, J.; Sklar, G.E.; Min Sen Oh, V.; Chuen Li, S. Factors affecting therapeutic compliance: A review from the patient’s perspective. Ther. Clin. Risk Manag. 2008, 4, 269–286. [Google Scholar] [CrossRef]
| Cannabinoid | Impacted Molecules | Receptors/Pathway | Experimental Model | Study |
|---|---|---|---|---|
| AEA and CBD | IL-17A inflammatory effects (↓) | Mostly CB2, possibly PPAR- γ-mediated effects | Confluent Caco-2 cell monolayers | Harvey et al. [57] |
| THC and CBD | IFN-γ and TNF-α-induced permeability (↓) | CB1-mediated effects | Confluent Caco-2 cell monolayers | Alhamoruni et al. [58] |
| AEA and 2-AG | IFN-γ and TNF-α-induced permeability (↑) | CB1-mediated effects | Confluent Caco-2 cell monolayers | Alhamoruni et al. [58] |
| AEA | IL-6, IL-8 (↓) | CB1-mediated effects, possibly TRPV1 modulation | Caco-2 cell monolayers | Karwad et al. [60] |
| CBD and PEA | prevents production of CREB, JNK, STAT5 | CB2, TRPV1 (CBD) PPAR-α (PEA) | Caco-2 cells | Couch et al. [61] |
| CBD and THCA | IL-8 (↓) | partially GPR55-mediated (THCA) | HCT116, HT29, and Caco-2 colon cells | Nallathambi et al. [62] |
| BPC | TNF-α, IL-1β, IL-6, IL-17A (↓) IL-13 (↑) | CB2-mediated STAT-3 downregulation | Human gingival fibroblasts and mucosa epithelial cells | Picciolo et al. [65] |
| CBD | IL-6, G-CSF, CXCL1 (↓) IL-8, GM-CSF, CXCL2 (↑) | Unspecified, possibly CB2-mediated | Normal bronchial cells treated with TNF-α | Muthumalage et al. [111] |
| CBD | MCP-1 (↓) | NF-κB inhibition | BEAS-2B, U937, and HFL-1 cells | Muthumalage et al. [111] |
| ACEA and JWH-133 | VEGF-A, VEGF-C, Ang1, and Ang2 (↓) | CB1 (ACEA) CB2 (JWH-133) | Human lung macrophages | Staiano et al. [113] |
| AJA | TNF-α, IFN-α and β (↓) | Unspecified, possibly CB2-mediated | Peripheral blood mononuclear cells from dermatomyositis patients | Robinson et al. [173] |
| PEA | MCP-2 (↓) | “Entourage” effect on TRPV1 ligands (AEA and OEA) | HaCaT keratinocytes treated with polyinosinic:polycytidylic acid | Petrosino et al. [168] |
| CBD | MCP-2, IL-6, IL-8, TNF-α (↓) | CB2-mediated as well as through TRPV1 activation/desensitization | HaCaT keratinocytes treated with polyinosinic:polycytidylic acid | Petrosino et al. [169] |
| THC, CBN, CBD, and CBG | Keratinocytes (inhibits proliferation) | Predominantly mediated by PPAR-γ | HPV-16 E6/E7 transformed human skin keratinocytes | Wilkinson et al. [171] |
| ACEA | keratinocytes (inhibits proliferation) | CB1-mediated signaling | Isolated human skin samples of psoriasis lesions | Ramot et al. [172] |
| CBD | GRO-α, vGPCR, VEGF-C, VEGFR-3 (↓) | Unspecified, possibly mediated by multiple receptors (CB1, CB2, TRPVs, and/or GPRs) | Kaposi’s sarcoma-associated herpesvirus-infected primary human dermal endothelial cells | Maor et al. [175] |
| CBD | TNF-α, IL-1β, IL-6 (↓) | A2a adenosine receptor-cAMP-TRIB3-NF-κB pathway | SZ95 human sebocytes cultures | Oláh et al. [174] |
| Cannabinoid | Impacted Molecules | Receptors/Pathway | Experimental Model | Study |
|---|---|---|---|---|
| CBG | IL-1β, IL-10, IFN-γ, iNOS expression, MPO activity (↓) | CB2 and possibly TRPV4-mediated | Murine colitis induced by DNBS | Borrelli et al. [70] |
| CBD | IL-1β, IL-10, iNOS expression (↓) | Unspecified, possibly CB2-mediated | Murine colitis induced by DNBS | Borrelli et al. [71] |
| CBD and CBG combined with fish oil | IL-1β, MPO activity (↓) | Possibly by regulating endocannabinoids and their derivates | Murine colitis induced by DNBS | Pagano et al. [72] |
| CBD combined with fish oil | IL-1β, IL-6, MPO activity (↓) IL-10 (↑) | Unspecified | DSS model of murine colitis | Silvestri et al. [73] |
| Abn-CBD | MPO activity (↓) | Non-CB1/2, possibly GPR18 and GPR55 | TNBS-induced colitis in CD1 mice | Krohn et al. [80] |
| HU210 | IL-1β, IL-6, IL-17, TNF-α, MPO activity (↓) | TLR4/MAPK signaling pathway | DSS model of murine colitis | Lin et al. [79] |
| HU210 | IL-6, chemokine KC (↓) | CB1/2 receptor agonism | Gastric mucosa inflammation secondary to acute pancreatitis in rats | Cao et al. [78] |
| CBD | IL-6, TNF-α, MCP-1, MIP-2, MPO activity (↓) | Non-CB1/2, possibly through the adenosine A2A receptor | Lipopolysaccharide-induced acute lung injury in mice | Ribeiro et al. [117,118] |
| WIN 55,212-2, PEA and THC | TNF-α (↓) | Partially CB2-mediated | Lipopolysaccharide-induced acute lung injury in mice | Beryshev et al. [119] |
| CBD | IL-5, IL-23, G-CSF, TNF-α (↑) | Increased activation of NFAT and Ca2+ signaling | Lipopolysaccharide-induced acute lung injury in mice | Karmaus et al. [120] |
| CBD | IL-1 and total protein content (↓) | 5-HT1A receptor | Lung inflammation induced by brain ischemia in newborn piglets | Arruza et al. [121] |
| WIN55,212-2 | MMP-9 (↓) | ERK1/2 signaling pathway | Lung inflammation in mice exposed to cigarette smoke | Tauber et al. [122] |
| CBD | IL-4, IL-5, IL-13, IL-6, and TNF-α (↓) | CB1/2-mediated | Ovalbumin-induced asthma in mice | Vuolo et al. [123,124] |
| JWH-133 | CD11b surface expression/adhesion, ROS production (↑) | CB2-mediated | Ovalbumin-induced asthma in mice | Frei et al. [125] |
| JWH-133 combined with JZL184 | IFN-γ, MIP-1α (↓) IL-10 (↑) | CB1 (JZL184) CB2 (JWH-133) | Lung inflammation in RSV infection in mice | Tahamtan et al. [127,128] |
| THC | IFN-γ (↓) | PI3K/Akt pathway signaling inhibition | Murine model of lung injury caused by SEB | Rao et al. [131] |
| JWH-133 | IL-6, TNF-α, MPO activity (↓) SOD activity (↑) | CB2-mediated activation of PI3K/Akt pathway signaling | Lung ischemia-reperfusion injury model in mice | Zeng et al. [132] |
| JWH-133 | IL-1β, IL-6, TNF-α, caspase-3 (↓) IL-10 (↑) | NF-κB signaling inhibition | Mice with CLP-induced sepsis | Çakır et al. [133] |
| GP1a | IL-6, chemokine KC, MIP-2 (↓) | CB2-mediated | Mice with CLP-induced sepsis | Tschöp et al. [134] |
| M. suaveolens | IL-1β, IL-6, TNF-α, VEGF (↓) IL-4, IL-10 (↑) | NF-κB signaling inhibition | Mice with CLP-induced sepsis | Liu et al. [135] |
| JWH-133 | MPO activity (↓) | CB2-mediated | Murine model of NPE after subarachnoid hemorrhage | Fujii et al. [136] |
| BPC and GP1a | IL-12, chemokine KC, leukotriene B4, NO (↓) | CB2-mediated | Mycobacterium bovis-induced pulmonary inflammation | Andrade-Silva et al. [138] |
| Possible unspecified agonist * | TNF-α, NF-κB and MAP kinases * (↓) | CB1 and CB2-mediated NF-κB and MAP/ERK signaling | Murine UV-induced skin carcinogenesis | Zheng et al. [187] |
| VCE-004.8 | IL-1β and IL-13. Prevention of macrophages infiltration and mast cells degranulation. Indirectly decreased TGF-β | PPAR-γ and CB2-mediated SMAD-signaling transcriptional activity modulation | Mice with bleomycin-induced scleroderma | del Río et al. [177] |
| WIN55,212-2 | TGF-β, CTGF, and PDGF (↓) | Non-CB1, non-CB2 mediated downregulation of PDGF/TGFβ signaling pathways | Mice with bleomycin-induced scleroderma | Balistreri et al. [178] |
| α-OOS | Mast cells degranulation and IL-4 (↓) | “Entourage” effect on CB1 with possible PPAR-γ and GPR55 involvement | Oxazolone induced atopic dermatitis in Balb/c and hairless mice | Kim et al. [179] |
| CBD | Arachidonate (↑) and prostaglandins (↓) | Increased PLA2 activity and inhibition of cyclooxygenase and lipoxygenase | Tetradecanoylphorbol acetate-induced erythema of mouse skin | Formukong et al. [180] |
| CBD | ROS production and caspase activation (↓) | Undetermined anti-inflammatory pathway *. TRIB3-Akt/mTOR signaling pathway | Mice bearing BRAF wild-type melanoma xenografts | Armstrong et al. [181] |
| Cannabinoid | Effects | Inflammatory Condition | Clinical Trial Stage | Study |
|---|---|---|---|---|
| PEA and CBD | Decreased intestinal permeability | Aspirin-induced intestinal inflammation | Phase 1 | Couch et al. [87] |
| THC | Decreased CDAI and CRP Increased quality of life | Crohn’s disease | Phase 1 | Naftali et al. [88] |
| CBD | No beneficial effects | Crohn’s disease | Phase 2 | Naftali et al. [89] |
| CBD | Minor improvements in rectal bleeding and endoscopic scores. Increased quality of life | Ulcerative colitis | Phase 2 | Irving et al. [91] |
| THC | Decreased DAI and endoscopic score | Ulcerative colitis | Phase 1 | Naftali et al. [92] |
| Lenabasum | Decreased IgG, IL-8, exacerbations, and lymphocyte infiltration | Cystic fibrosis | Phase 2 | Chmiel et al. [143] |
| Cannabis (high CBD/low THC) | Results pending | Pain and Inflammation in Lung Cancer | Phase 1 | Martinez et al. [144] |
| Cannabis (high CBD/low THC) | No clinical positive or negative effects | Chronic obstructive pulmonary disease | Phase 2 | Abdallah et al. [146] |
| Nabilone | No significant bronchodilation | Asthma | Phase 1 | Gong et al. [147] |
| THC | Antagonistic effects on bronchodilation. Irritating effect on airways | Asthma | Phase 1 | Tashkin et al. [148] |
| THC | Mild bronchodilation effect. Significant psychoactive effects | Asthma | Phase 1 | Abboud et al. [149] |
| Cannabis seeds mixture | Decreased erythema | Acne | Phase 1 | Ali et al. [188] |
| BTX 1503 | Results pending | Acne | Phase 2 | Botanix Pharmaceuticals [189] |
| AJA | Decreased expression of key genes related to inflammation | Cutaneous systemic sclerosis | Phase 2 | Spiera et al. [190] |
| AJA | Decreased IL-31, IFN-β and γ, and T-helper cell inflammation | Dermatomyositis | Phase 2 | Chen et al. [192] |
| AJA | Initiated study | Cutaneous systemic sclerosis | Phase 3 | Burstein et al. [191] |
| AJA | Developed study protocol | Dermatomyositis | Phase 3 | Werth et al. [193] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scheau, C.; Caruntu, C.; Badarau, I.A.; Scheau, A.-E.; Docea, A.O.; Calina, D.; Caruntu, A. Cannabinoids and Inflammations of the Gut-Lung-Skin Barrier. J. Pers. Med. 2021, 11, 494. https://doi.org/10.3390/jpm11060494
Scheau C, Caruntu C, Badarau IA, Scheau A-E, Docea AO, Calina D, Caruntu A. Cannabinoids and Inflammations of the Gut-Lung-Skin Barrier. Journal of Personalized Medicine. 2021; 11(6):494. https://doi.org/10.3390/jpm11060494
Chicago/Turabian StyleScheau, Cristian, Constantin Caruntu, Ioana Anca Badarau, Andreea-Elena Scheau, Anca Oana Docea, Daniela Calina, and Ana Caruntu. 2021. "Cannabinoids and Inflammations of the Gut-Lung-Skin Barrier" Journal of Personalized Medicine 11, no. 6: 494. https://doi.org/10.3390/jpm11060494
APA StyleScheau, C., Caruntu, C., Badarau, I. A., Scheau, A.-E., Docea, A. O., Calina, D., & Caruntu, A. (2021). Cannabinoids and Inflammations of the Gut-Lung-Skin Barrier. Journal of Personalized Medicine, 11(6), 494. https://doi.org/10.3390/jpm11060494