Internet Usage Habits and Experienced Levels of Psychopathology: A Pilot Study on Association with Spontaneous Eye Blinking Rate
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Procedures
2.2. Psychological Assessment Included
2.2.1. Problematic Internet Use Questionnaire, 9 Items Version
2.2.2. Barratt Impulsiveness Scale
2.2.3. Beck Depression Inventory II
2.2.4. Beck Anxiety Inventory
2.2.5. Clark–Beck Obsessive–Compulsive Inventory
2.3. sEBR Assessment
2.4. Statistical Evaluation
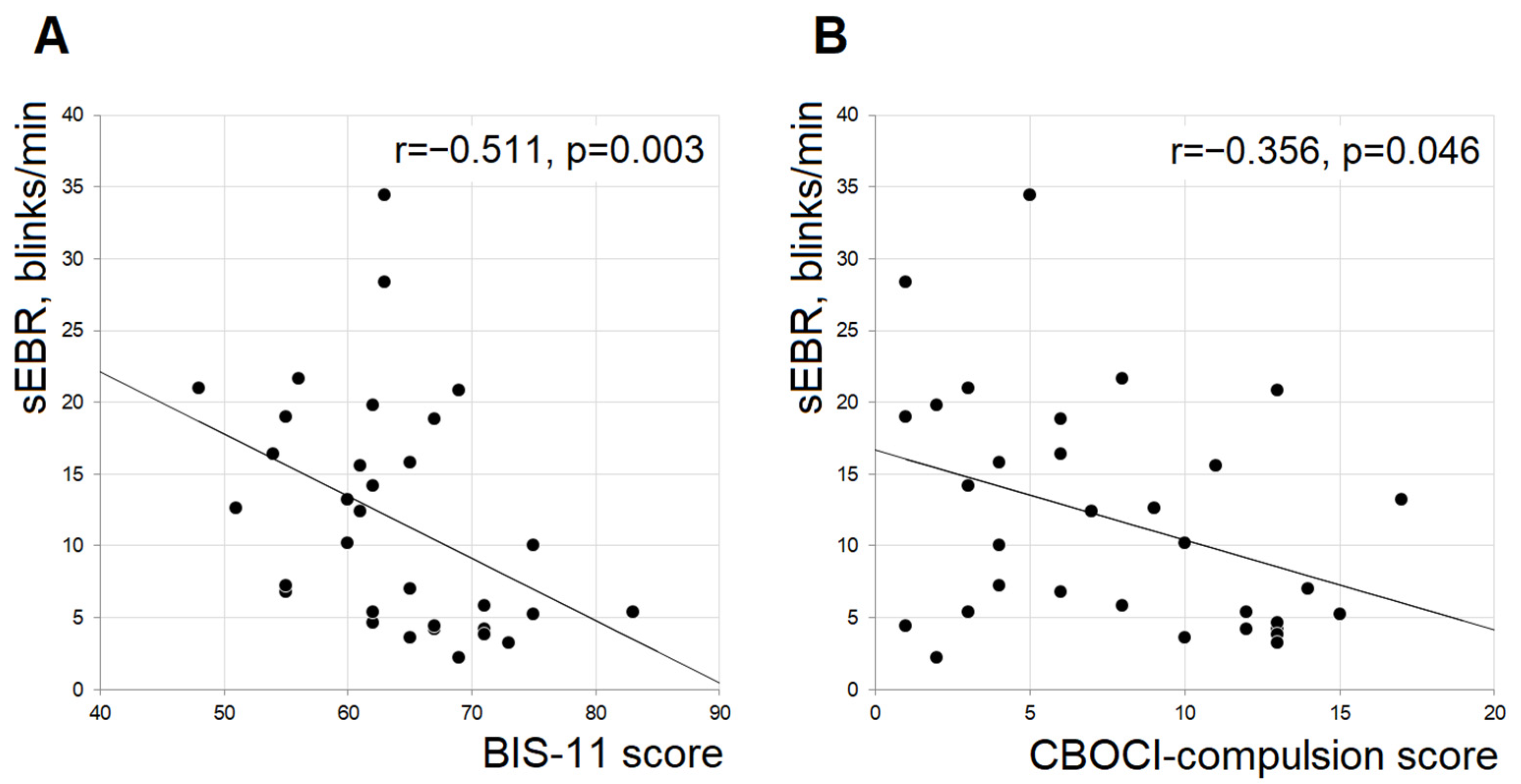
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Durkee, T.; Kaess, M.; Carli, V.; Parzer, P.; Wasserman, C.; Floderus, B.; Apter, A.; Balazs, J.; Barzilay, S.; Bobes, J.; et al. Prevalence of pathological internet use among adolescents in Europe: Demographic and social factors. Addiction 2012, 107, 2210–2222. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Mondal, A. A study on Internet addiction and its relation to psychopathology and self-esteem among college students. Ind. Psychiatry J. 2018, 27, 61. [Google Scholar]
- Maras, D.; Flament, M.F.; Murray, M.; Buchholz, A.; Henderson, K.A.; Obeid, N.; Goldfield, G.S. Screen time is associated with depression and anxiety in Canadian youth. Prev. Med. 2015, 73, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Mottram, A.J.; Fleming, M.J. Extraversion, Impulsivity, and Online Group Membership as Predictors of Problematic Internet Use. CyberPsychology Behav. 2009, 12, 319–321. [Google Scholar] [CrossRef] [PubMed]
- Tiego, J.; Lochner, C.; Ioannidis, K.; Brand, M.; Stein, D.J.; Yücel, M.; Grant, J.E.; Chamberlain, S.R. Problematic use of the Internet is a unidimensional qua-si-trait with impulsive and compulsive subtypes. BMC Psychiatry 2019, 8, 348. [Google Scholar]
- Feng, W.; Ramo, D.E.; Chan, S.R.; Bourgeois, J.A. Internet gaming disorder: Trends in prevalence 1998–2016. Addict. Behav. 2017, 75, 17–24. [Google Scholar] [CrossRef]
- Kuss, D.; Griffiths, M.; Karila, L.; Billieux, J. Internet Addiction: A Systematic Review of Epidemiological Research for the Last Decade. Curr. Pharm. Des. 2014, 20, 4026–4052. [Google Scholar] [CrossRef] [PubMed]
- Andreassen, C.S.; Billieux, J.; Griffiths, M.D.; Kuss, D.J.; Demetrovics, Z.; Mazzoni, E.; Pallesen, S. The relationship between addictive use of social media and video games and symptoms of psychiatric disorders: A large-scale cross-sectional study. Psychol. Addict. Behav. 2016, 30, 252–262. [Google Scholar] [CrossRef]
- Anderson, E.L.; Steen, E.; Stavropoulos, V. Internet use and Problematic Internet Use: A systematic review of longitudinal re-search trends in adolescence and emergent adulthood. Int. J. Adolesc. Youth 2017, 22, 430–454. [Google Scholar] [CrossRef]
- Kardefelt-Winther, D.; Rees, G.; Livingstone, S. Contextualising the link between adolescents’ use of digital technology and their mental health: A multi-country study of time spent online and life satisfaction. J. Child Psychol. Psychiatry 2020, 61, 875–889. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Morales, M. The Brain on Drugs: From Reward to Addiction. Cell 2015, 162, 712–725. [Google Scholar] [CrossRef]
- A Nader, M.; Morgan, D.; Gage, H.D.; Nader, S.H.; Calhoun, T.L.; Buchheimer, N.; Ehrenkaufer, R.; Mach, R.H. PET imaging of dopamine D2 receptors during chronic cocaine self-administration in monkeys. Nat. Neurosci. 2006, 9, 1050–1056. [Google Scholar] [CrossRef]
- Lee, B.; London, E.D.; Poldrack, R.A.; Farahi, J.; Nacca, A.; Monterosso, J.R.; Mumford, J.A.; Bokarius, A.V.; Dahlbom, M.; Mukherjee, J.; et al. Striatal Dopamine D2/D3 Receptor Availability Is Reduced in Methamphetamine Dependence and Is Linked to Impulsivity. J. Neurosci. 2009, 29, 14734–14740. [Google Scholar] [CrossRef]
- Kim, S.H.; Baik, S.-H.; Park, C.S.; Kim, S.J.; Choi, S.W. Reduced striatal dopamine D2 receptors in people with Internet addiction. Neuroreport 2011, 22, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Jia, S.; Hu, S.; Fan, R.; Sun, W.; Sun, T.; Zhang, H. Reduced Striatal Dopamine Transporters in People with Internet Addiction Disorder. J. Biomed. Biotechnol. 2012, 2012, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.R. Eye blink and schizophrenia: Psychosis or tardive dyskinesia? Am. J. Psychiatry 1978, 135, 223–226. [Google Scholar]
- Jongkees, B.J.; Colzato, L.S. Spontaneous eye blink rate as predictor of dopamine-related cognitive function—A review. Neurosci. Biobehav. Rev. 2016, 71, 58–82. [Google Scholar] [CrossRef]
- Blin, O.; Masson, G.; Azulay, J.P.; Fondarai, J.; Serratrice, G. Apomorphine-induced blinking and yawning in healthy volunteers. Br. J. Clin. Pharmacol. 1990, 30, 769–773. [Google Scholar] [CrossRef]
- Elsworth, J.D.; Lawrence, M.S.; Roth, R.H.; Taylor, J.R.; Mailman, R.B.; E Nichols, D.; Lewis, M.H.; E Redmond, D. D1 and D2 dopamine receptors independently regulate spontaneous blink rate in the vervet monkey. J. Pharmacol. Exp. Ther. 1991, 259, 595–600. [Google Scholar]
- Kleven, M.S.; Koek, W. Differential effects of direct and indirect dopamine agonists on eye blink rate in cynomolgus monkeys. J. Pharmacol. Exp. Ther. 1996, 279, 1211–1219. [Google Scholar]
- Kaminer, J.; Powers, A.S.; Horn, K.G.; Hui, C.; Evinger, C. Characterizing the Spontaneous Blink Generator: An Animal Model. J. Neurosci. 2011, 31, 11256–11267. [Google Scholar] [CrossRef]
- Freed, W.J.; E Kleinman, J.; Karson, C.N.; Potkin, S.G.; Murphy, D.L.; Wyatt, R.J. Eye-blink rates and platelet monoamine oxidase activity in chronic schizophrenic patients. Biol. Psychiatry 1980, 15, 329–332. [Google Scholar] [PubMed]
- Mackert, A.; Flechtner, K.M.; Woyth, C.; Frick, K. Increased blink rates in schizophrenics. Influences of neuroleptics and psycho-pathology. Schizophr. Res. 1991, 4, 41–47. [Google Scholar] [CrossRef]
- Colzato, L.S.; Slagter, H.A.; Wildenberg, W.P.V.D.; Hommel, B. Closing one’s eyes to reality: Evidence for a dopaminergic basis of Psychoticism from spontaneous eye blink rates. Pers. Individ. Differ. 2009, 46, 377–380. [Google Scholar] [CrossRef]
- Colzato, L.S.; van den Wildenberg, W.P.M.; Hommel, B. Reduced spontaneous eye blink rates in recreational cocaine users: Evi-dence for dopaminergic hypoactivity. PLoS ONE 2008, 3, e3461. [Google Scholar] [CrossRef]
- Kowal, M.A.; Colzato, L.S.; Hommel, B. Decreased Spontaneous Eye Blink Rates in Chronic Cannabis Users: Evidence for Striatal Cannabinoid-Dopamine Interactions. PLoS ONE 2011, 6, e26662. [Google Scholar] [CrossRef][Green Version]
- Paprocki, R.; Lenskiy, A. What Does Eye-Blink Rate Variability Dynamics Tell Us About Cognitive Performance? Front. Hum. Neurosci. 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Mathar, D.; Wiehler, A.; Chakroun, K.; Goltz, D.; Peters, J. A potential link between gambling addiction severity and central dopamine levels: Evidence from spontaneous eye blink rates. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Kruis, A.; Slagter, H.A.; Bachhuber, D.R.W.; Davidson, R.J.; Lutz, A. Effects of meditation practice on spontaneous eyeblink rate. Psychophysiology 2016, 53, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Burkauskas, J.; Király, O.; Demetrovics, Z.; Podlipskyte, A.; Steibliene, V. Psychometric Properties of the Nine-Item Problematic Internet Use Questionnaire (PIUQ-9) in a Lithuanian Sample of Students. Front. Psychiatry 2020, 11. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Manual for the Beck Depression Inventory-II; Psychological Corporation: San Antonio, TX, USA, 1996. [Google Scholar]
- Beck, A.T.; Epstein, N.; Brown, G.; Steer, R.A. An inventory for measuring clinical anxiety: Psychometric properties. J. Consult Clin. Psychol. 1988, 56, 893–897. [Google Scholar] [CrossRef]
- Clark, D.A.; Anthony, M.M.; Beck, A.T.; Swinson, R.P.; Steer, R.A. Clark–Beck Obsessive–Compulsive Inventory; American Psychological Association (APA): San Antonio, TX, USA, 2002. [Google Scholar]
- Patton, J.H.; Stanford, M.S.; Barratt, E.S. Factor structure of the Barratt impulsiveness scale. J. Clin. Psychol. 1995, 51, 768–774. [Google Scholar] [CrossRef]
- Lange, F.; Wagner, A.; Müller, A.; Eggert, F. Subscales of the Barratt Impulsiveness Scale differentially relate to the Big Five factors of personality. Scand. J. Psychol. 2017, 6, 133–259. [Google Scholar] [CrossRef]
- Janavičiūtė, J.; Šinkariova, L. Psychometric properties of the Lithuanian version of Barratt Impulsiveness Scale-11 (BIS-11) in a nonclinical sample. Cogn. Brain Behav. Interdiscip. J. 2020, 24, 123–138. [Google Scholar] [CrossRef]
- Wang, Y.-P.; Gorenstein, C. Assessment of depression in medical patients: A systematic review of the utility of the Beck Depres-sion Inventory-II. Clinics 2013, 68, 1274–1287. [Google Scholar] [CrossRef]
- Bunevicius, A.; Staniute, M.; Brozaitiene, J.; Bunevicius, R. Diagnostic accuracy of self-rating scales for screening of depression in coronary artery disease patients. J. Psychosom. Res. 2012, 72, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-P.; Gorenstein, C. Psychometric properties of the Beck Depression Inventory-II: A comprehensive review. Rev. Bras. Psiquiatr. 2013, 35, 416–431. [Google Scholar] [CrossRef]
- Doughty, M.J. Consideration of three types of spontaneous eyeblink activity in normal humans: During reading and video display terminal use, in primary gaze, and while in conversation. Optom. Vis. Sci. 2001, 78, 712–725. [Google Scholar] [CrossRef] [PubMed]
- Zaman, M.L.; Doughty, M.J. Some methodological issues in the assessment of the spontaneous eyeblink frequency in man. Ophthalmic Physiol. Opt. 1997, 17, 421–432. [Google Scholar] [CrossRef]
- Kleifges, K.; Bigdely-Shamlo, N.; Kerick, S.E.; Robbins, K.A. BLINKER: Automated Extraction of Ocular Indices from EEG Enabling Large-Scale Analysis. Front. Neurosci. 2017, 11, 12. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, D.K. What is the proper way to apply the multiple comparison test? Korean J. Anesthesiol. 2018, 71, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Cruz, A.A.; Garcia, D.M.; Pinto, C.T.; Cechetti, S.P. Spontaneous Eyeblink Activity. Ocul. Surf. 2011, 9, 29–41. [Google Scholar] [CrossRef]
- Van Slooten, J.C.; Jahfari, S.; Theeuwes, J. Spontaneous eye blink rate predicts individual differences in exploration and exploitation during reinforcement learning. Sci. Rep. 2019, 9, 17436. [Google Scholar] [CrossRef] [PubMed]
- Korponay, C.; Dentico, D.; Kral, T.; Ly, M.; Kruis, A.; Goldman, R.; Lutz, A.; Davison, R.J. Neurobiological correlates of impulsivity in healthy adults: Lower prefrontal gray matter volume and spontaneous eye-blink rate but greater resting-state functional connectivity in basal ganglia-thalamo-cortical circuitry. Neuroimage 2017, 157, 288–296. [Google Scholar] [CrossRef]
- Müller, K.W.; Beutel, M.E.; Wölfling, K. A contribution to the clinical characterization of Internet addiction in a sample of treat-ment seekers: Validity of assessment, severity of psychopathology and type of co-morbidity. Compr. Psychiatry 2014, 55, 770–777. [Google Scholar] [CrossRef]
- Floros, G.; Siomos, K.; Stogiannidou, A.; Giouzepas, I.; Garyfallos, G. Comorbidity of psychiatric disorders with Internet addiction in a clinical sample: The effect of personality, defense style and psychopathology. Addict. Behav. 2014, 39, 1839–1845. [Google Scholar] [CrossRef]
- de Vries, H.T.; Nakamae, T.; Fukui, K.; Denys, D.; Narumoto, J. Problematic internet use and psychiatric co-morbidity in a population of Japanese adult psychiatric patients. BMC Psychiatry 2018, 18, 9. [Google Scholar] [CrossRef]
- Hetzel-Riggin, M.D.; Pritchard, J.R. Predicting Problematic Internet Use in Men and Women: The Contributions of Psychological Distress, Coping Style, and Body Esteem. Cyberpsychology Behav. Soc. Netw. 2011, 14, 519–525. [Google Scholar] [CrossRef]
- Cherian, A.V.; Anand, N.; Jain, P.A.; Prabhu, S.; Thomas, C.; Bhat, A.; Prathyusha, P.V.; Bhat, S.U.; Young, K. Prevalence of excessive internet use and its association with psychological distress among university students in South India. Ind. Psychiatry J. 2018, 27, 131–140. [Google Scholar] [CrossRef]
- Gecaite-Stonciene, J.; Saudargiene, A.; Pranckeviciene, A.; Liaugaudaite, V.; Griskova-Bulanova, I.; Simkute, D.; Naginiene, R.; Dainauskas, L.L.; Ceidaite, G.; Burkauskas, J. Impulsivity Mediates Associations Between Problematic Internet Use, Anxiety, and Depressive Symptoms in Students: A Cross-Sectional COVID-19 Study. Front. Psychiatry 2021, 12, 634464. [Google Scholar] [CrossRef]
- Chou, C.; Condron, L.; Belland, J.C. A Review of the Research on Internet Addiction. Educ. Psychol. Rev. 2005, 17, 363–388. [Google Scholar] [CrossRef]
- Cecilia, M.; Mazza, M.; Cenciarelli, S.; Grassi, M.; Cofini, V. The Relationship between Compulsive Behaviour and Internet Addiction. Styles Commun. 2013, 1, 5. [Google Scholar]
- Kim, K.; Ryu, E.; Chon, M.-Y.; Yeun, E.-J.; Choi, S.-Y.; Seo, J.-S.; Nam, B.-W. Internet addiction in Korean adolescents and its relation to depression and suicidal ideation: A questionnaire survey. Int. J. Nurs. Stud. 2006, 43, 185–192. [Google Scholar] [CrossRef]
- Jeong, H.; Yim, H.W.; Lee, S.Y.; Lee, H.K.; Potenza, M.N.; Kwon, J.H.; Koo, H.J.; Kwen, Y.-S.; Bhang, S.; Choi, J.-S. Discordance between self-report and clinical diagnosis of Internet gaming disorder in adolescents. Sci. Rep. 2018, 8, 10084. [Google Scholar] [CrossRef]
- Ko, C.-H.; Yen, C.-F.; Yen, C.-N.; Yen, J.-Y.; Chen, C.-C.; Chen, S.-H. Screening for Internet Addiction: An Empirical Study on Cut-off Points for the Chen Internet Addiction Scale. Kaohsiung J. Med Sci. 2005, 21, 545–551. [Google Scholar] [CrossRef]
- Spada, M.M. An overview of problematic Internet use. Addict. Behav. 2014, 39, 3–6. [Google Scholar] [CrossRef]
- de Wit, H.; Enggasser, J.L.; Richards, J.B. Acute administration of d-amphetamine decreases impulsivity in healthy volunteers. Neuropsychopharmacology 2002, 27, 813–825. [Google Scholar] [CrossRef]
- Gu, B.-M.; Park, J.-Y.; Kang, D.-H.; Lee, S.J.; Yoo, S.Y.; Jo, H.J.; Choi, C.-H.; Lee, J.-M.; Kwon, J.S. Neural correlates of cognitive inflexibility during task-switching in obsessive-compulsive disorder. Brain 2007, 131, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, S.R.; Fineberg, N.A.; Menzies, L.A.; Blackwell, A.D.; Bullmore, E.T.; Robbins, T.W.; Sahakian, B.J. Impaired cognitive flexibility and motor inhibition in unaffected first-degree relatives of patients with obsessive-compulsive disorder. Am. J. Psychiatry 2007, 164, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Remijnse, P.L.; Nielen, M.M.; van Balkom, A.J.; Cath, D.C.; van Oppen, P.; Uylings, H.B.; Veltman, D.J. Reduced orbitofrontal-striatal activity on a reversal learning task in obsessive-compulsive disorder. Arch. Gen. Psychiatry 2006, 63, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Holland, M.K.; Tarlow, G. Blinking and Mental Load. Psychol. Rep. 1972, 31, 119–127. [Google Scholar] [CrossRef]
- Oh, J.; Jeong, S.-Y.; Jeong, J. The timing and temporal patterns of eye blinking are dynamically modulated by attention. Hum. Mov. Sci. 2012, 31, 1353–1365. [Google Scholar] [CrossRef] [PubMed]
- Seiler, J.L.; Cosme, C.V.; Sherathiya, V.N.; Bianco, J.M.; Lerner, T.N. Dopamine Signaling in the Dorsomedial Striatum Promotes Compulsive Behavior. bioRxiv 2020, bioRxiv: 2020.03.30.016238. [Google Scholar]
- Sescousse, G.; Ligneul, R.; van Holst, R.J.; Janssen, L.K.; de Boer, F.; Janssen, M.; Berry, A.M.; Jagust, W.J.; Cools, R. Spontaneous eye blink rate and dopamine synthesis capacity: Preliminary evi-dence for an absence of positive correlation. Eur. J. Neurosci. 2018, 47, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Dang, L.C.; Samanez-Larkin, G.R.; Castrellon, J.J.; Perkins, S.F.; Cowan, R.L.; Newhouse, P.A.; Zald, D.H. Spontaneous Eye Blink Rate (EBR) Is Uncorrelated with Dopamine D2 Receptor Availability and Unmodulated by Dopamine Agonism in Healthy Adults. Eneuro 2017, 4. [Google Scholar] [CrossRef] [PubMed]
| PIU 1 | Control | Mann–Whitney U | p | |
|---|---|---|---|---|
| F/M | 20/12 | 14/16 | ||
| Age | 25 (7.0) | 24 (5.0) | 544.00 | 0.365 |
| PIUQ-9 | 24 (5.0) | 14 (3.0) | 740.00 | <0.001 |
| BIS-11 | 63 (9.0) | 57 (7.5) | 700.00 | 0.002 |
| BDI-II | 8 (9.8) | 4 (9.0) | 594.00 | 0.108 |
| BAI | 29 (8.8) | 26 (6.0) | 568.50 | 0.130 |
| CBOCI | 22.5 (13.8) | 12.0 (7.0) | 740.00 | <0.001 |
| sEBR | PIUQ-9 | BIS-11 | BDI-II | BAI | CBOCI | ||
|---|---|---|---|---|---|---|---|
| PIUQ-9 | r | 0.009 | 1.000 | 0.361 | 0.252 | 0.240 | 0.437 |
| p | 0.942 | 0.004 | 0.048 | 0.062 | <0.001 | ||
| sEBR | r | 1.000 | 0.009 | −0.284 | −0.155 | −0.040 | −0.038 |
| p | 0.942 | 0.025 | 0.228 | 0.758 | 0.768 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simkute, D.; Nagula, I.; Tarailis, P.; Burkauskas, J.; Griskova-Bulanova, I. Internet Usage Habits and Experienced Levels of Psychopathology: A Pilot Study on Association with Spontaneous Eye Blinking Rate. J. Pers. Med. 2021, 11, 288. https://doi.org/10.3390/jpm11040288
Simkute D, Nagula I, Tarailis P, Burkauskas J, Griskova-Bulanova I. Internet Usage Habits and Experienced Levels of Psychopathology: A Pilot Study on Association with Spontaneous Eye Blinking Rate. Journal of Personalized Medicine. 2021; 11(4):288. https://doi.org/10.3390/jpm11040288
Chicago/Turabian StyleSimkute, Dovile, Igor Nagula, Povilas Tarailis, Julius Burkauskas, and Inga Griskova-Bulanova. 2021. "Internet Usage Habits and Experienced Levels of Psychopathology: A Pilot Study on Association with Spontaneous Eye Blinking Rate" Journal of Personalized Medicine 11, no. 4: 288. https://doi.org/10.3390/jpm11040288
APA StyleSimkute, D., Nagula, I., Tarailis, P., Burkauskas, J., & Griskova-Bulanova, I. (2021). Internet Usage Habits and Experienced Levels of Psychopathology: A Pilot Study on Association with Spontaneous Eye Blinking Rate. Journal of Personalized Medicine, 11(4), 288. https://doi.org/10.3390/jpm11040288








