Early Evaluation of Immunotherapy Response in Lymphoma Patients by 18F-FDG PET/CT: A Literature Overview
Abstract
1. Introduction
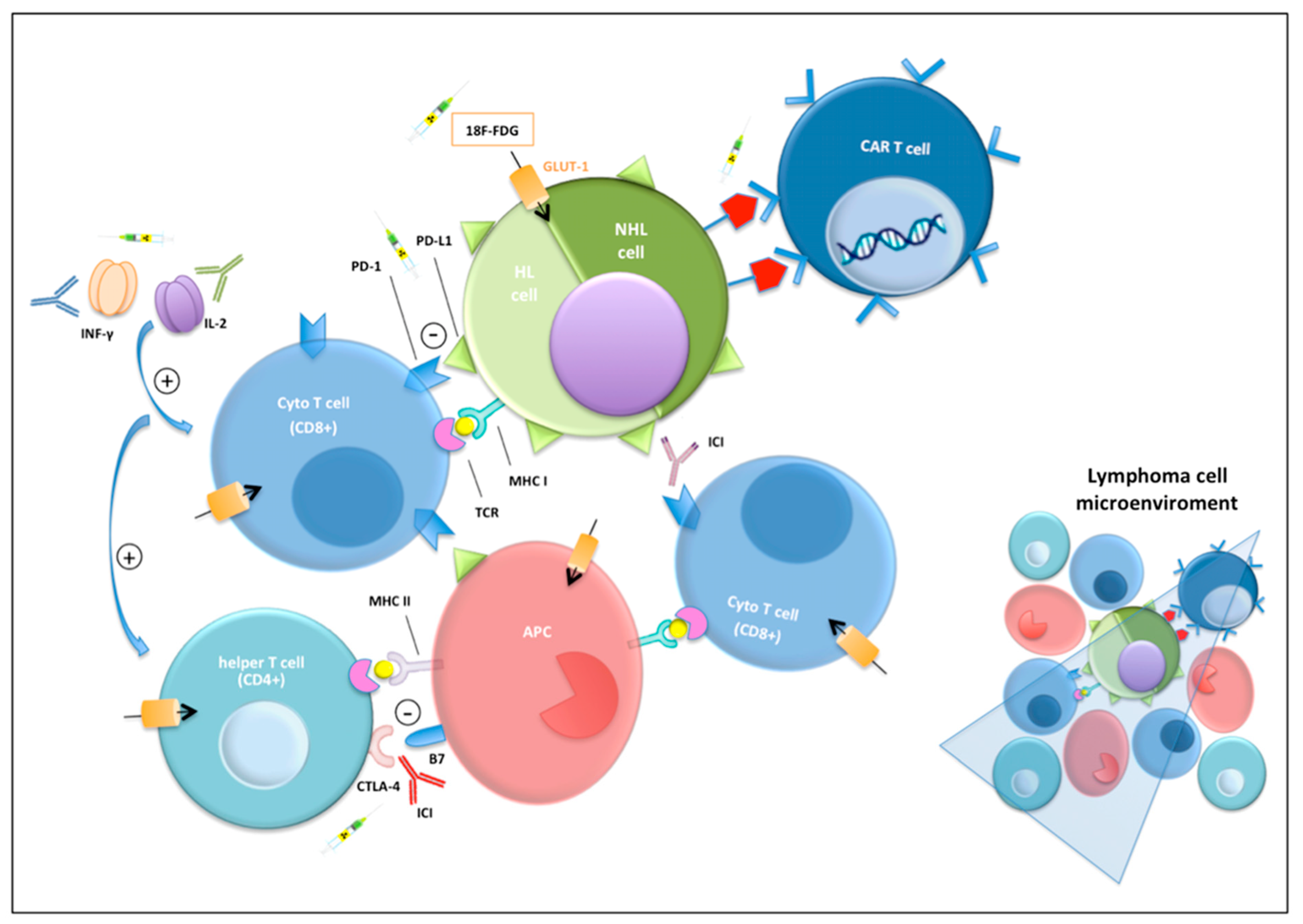
2. The Basis of Immunotherapy
Challenge in Treatment Response Assessment
3. Search Strategy
4. Evidence Based Medicine of Early 18F-FDG PET/CT during Immunotherapy
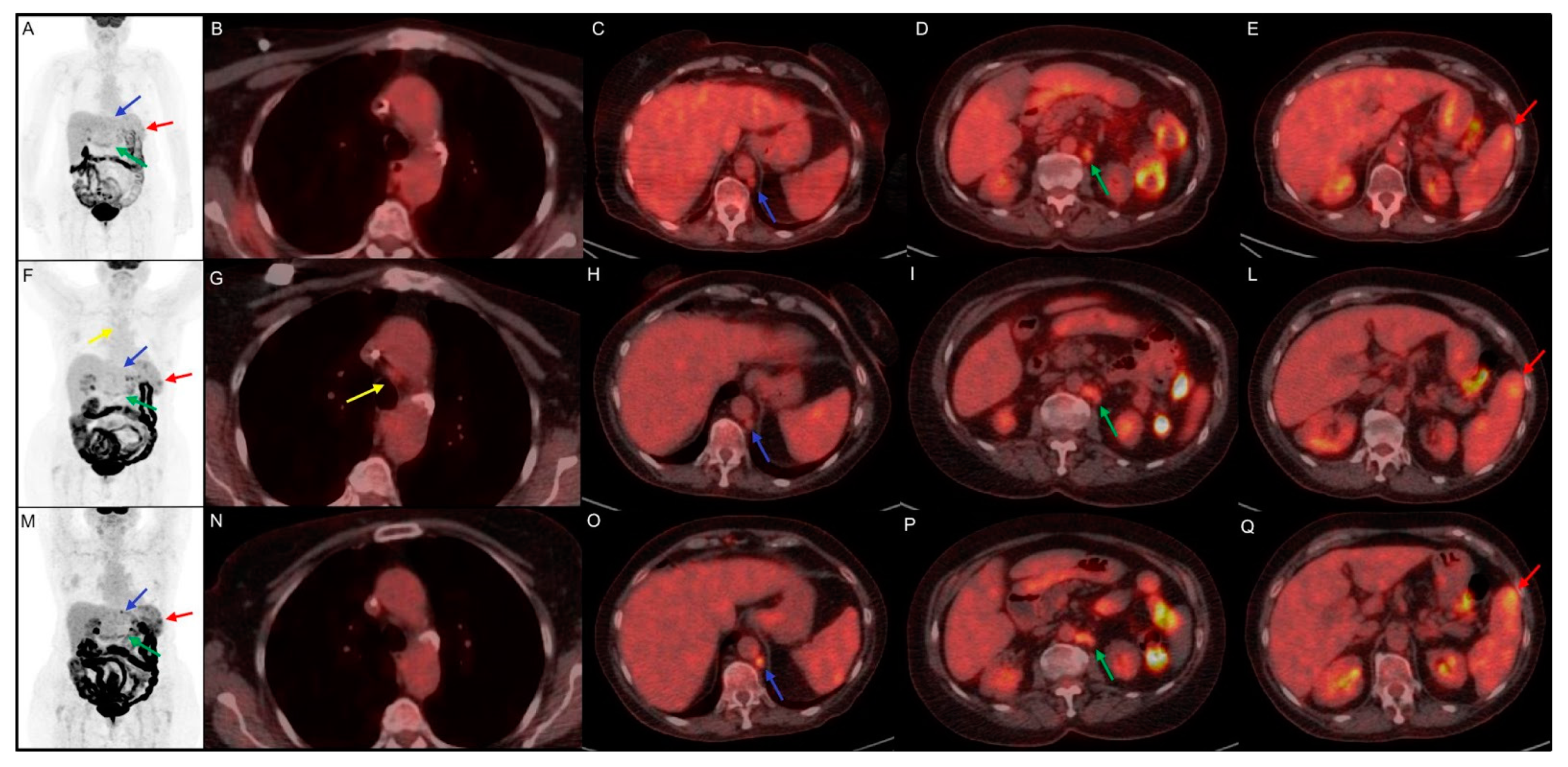
4.1. Hodgkin Lymphoma
4.2. Non Hodgkin Lymphoma
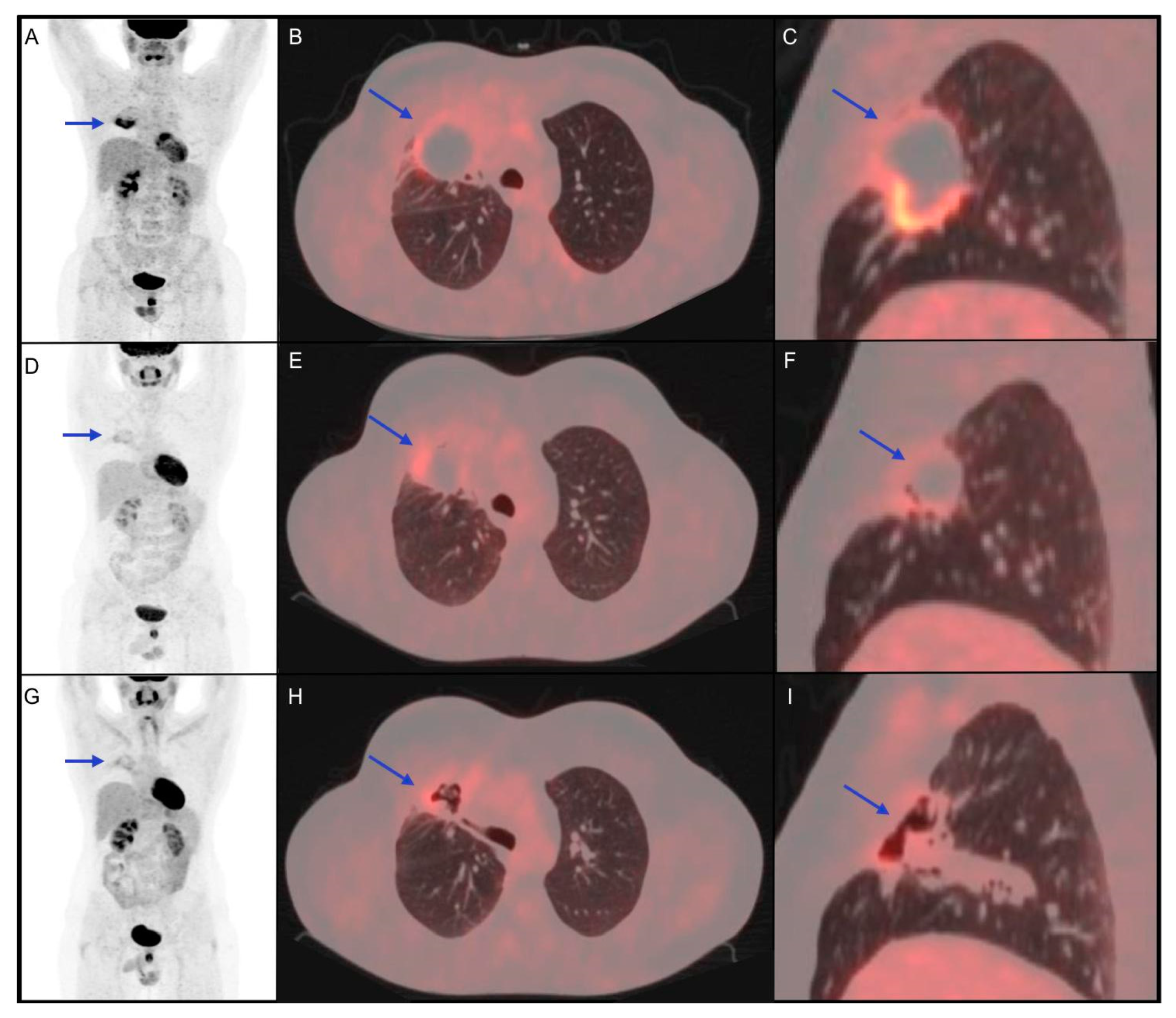
4.3. Role of 18F-FDG PET/CT in IrAEs
5. Looking Forward
5.1. Ongoing Studies
5.2. Immuno-PET
6. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Armand, P.; Shipp, M.A.; Ribrag, V.; Michot, J.M.; Zinzani, P.L.; Kuruvilla, J.; Snyder, E.S.; Ricart, A.D.; Balakumaran, A.; Rose, S.; et al. Programmed death-1 blockade with pembrolizumab in patients with classical hodgkin lymphoma after brentuximab vedotin failure. J. Clin. Oncol. 2016, 34, 3733–3739. [Google Scholar] [CrossRef]
- Ansell, S.M.; Lesokhin, A.M.; Borrello, I.; Halwani, A.; Scott, E.C.; Gutierrez, M.; Schuster, S.J.; Millenson, M.M.; Cattry, D.; Freeman, G.J.; et al. PD-1 Blockade with Nivolumab in Relapsed or Refractory Hodgkin’s Lymphoma. N. Engl. J. Med. 2015, 372, 311–319. [Google Scholar] [CrossRef]
- Castello, A.; Grizzi, F.; Qehajaj, D.; Rahal, D.; Lutman, F.; Lopci, E. 18 F-FDG PET/CT for response assessment in Hodgkin lymphoma undergoing immunotherapy with checkpoint inhibitors. Leuk. Lymphoma 2019, 60, 367–375. [Google Scholar] [CrossRef]
- Armand, P.; Engert, A.; Younes, A.; Fanale, M.; Santoro, A.; Zinzani, P.L.; Timmerman, J.M.; Collins, G.P.; Ramchandren, R.; Cohen, J.B.; et al. Nivolumab for relapsed/refractory classic hodgkin lymphoma after failure of autologous hematopoietic cell transplantation: Extended follow-up of the multicohort single-arm phase II checkmate 205 trial. J. Clin. Oncol. 2018, 36, 1428–1439. [Google Scholar] [CrossRef]
- Chen, R.; Zinzani, P.L.; Fanale, M.A.; Armand, P.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; et al. Phase II study of the efficacy and safety of pembrolizumab for relapsed/refractory classic Hodgkin Lymphoma. J. Clin. Oncol. 2017, 35, 2125–2132. [Google Scholar] [CrossRef]
- Kostakoglu, L.; Gallamini, A. Interim 18F-FDG PET in hodgkin lymphoma: Would PET-adapted clinical trials lead to a paradigm shift? J. Nucl. Med. 2013, 54, 1082–1093. [Google Scholar] [CrossRef] [PubMed]
- Kasamon, Y.L.; De Claro, R.A.; Wang, Y.; Shen, Y.L.; Farrell, A.T.; Pazdur, R. FDA Approval Summary: Nivolumab for the Treatment of Relapsed or Progressive Classical Hodgkin Lymphoma. Oncologist 2017, 22, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Mokrane, F.Z.; Chen, A.; Schwartz, L.H.; Morschhauser, F.; Stamatoullas, A.; De Colella, J.M.S.; Vercellino, L.; Casasnovas, O.; Chauchet, A.; Delmer, A.; et al. Performance of CT compared with 18F-FDG PET in predicting the efficacy of nivolumab in relapsed or Refractory Hodgkin lymphoma. Radiology 2020, 295, 651–661. [Google Scholar] [CrossRef]
- Ansell, S.M.; Minnema, M.C.; Johnson, P.; Timmerman, J.M.; Armand, P.; Shipp, M.A.; Rodig, S.J.; Ligon, A.H.; Roemer, M.G.M.; Reddy, N.; et al. Nivolumab for relapsed/refractory diffuse large B-cell lymphoma in patients ineligible for or having failed autologous transplantation: A single-arm, phase II study. J. Clin. Oncol. 2019, 37, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Egen, J.G.; Kuhns, M.S.; Allison, J.P. CTLA-4: New insights into its biological function and use in tumor immunotherapy. Nat. Immunol. 2002, 3, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, T.; Chikuma, S.; Iwai, Y.; Fagarasan, S.; Honjo, T. A rheostat for immune responses: The unique properties of PD-1 and their advantages for clinical application. Nat. Immunol. 2013, 14, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Decazes, P.; Bohn, P. Immunotherapy by immune checkpoint inhibitors and nuclear medicine imaging: Current and future applications. Cancers 2020, 12, 371. [Google Scholar] [CrossRef] [PubMed]
- Green, M.R.; Monti, S.; Rodig, S.J.; Juszczynski, P.; Currie, T.; O’Donnell, E.; Chapuy, B.; Takeyama, K.; Neuberg, D.; Golub, T.R.; et al. Integrative analysis reveals selective 9p24.1 amplification, increased PD-1 ligand expression, and further induction via JAK2 in nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Blood 2010, 116, 3268–3277. [Google Scholar] [CrossRef] [PubMed]
- Roemer, M.G.M.; Advani, R.H.; Ligon, A.H.; Natkunam, Y.; Redd, R.A.; Homer, H.; Connelly, C.F.; Sun, H.H.; Daadi, S.E.; Freeman, G.J.; et al. PD-L1 and PD-L2 genetic alterations define classical hodgkin lymphoma and predict outcome. J. Clin. Oncol. 2016, 34, 2690–2697. [Google Scholar] [CrossRef] [PubMed]
- Steidl, C.; Shah, S.P.; Woolcock, B.W.; Rui, L.; Kawahara, M.; Farinha, P.; Johnson, N.A.; Zhao, Y.; Telenius, A.; Ben Neriah, S.; et al. MHC class II transactivator CIITA is a recurrent gene fusion partner in lymphoid cancers. Nature 2011, 471, 377–383. [Google Scholar] [CrossRef]
- Green, M.R.; Rodig, S.; Juszczynski, P.; Ouyang, J.; Sinha, P.; O’Donnell, E.; Neuberg, D.; Shipp, M.A. Constitutive AP-1 activity and EBV infection induce PD-l1 in Hodgkin lymphomas and posttransplant lymphoproliferative disorders: Implications for targeted therapy. Clin. Cancer Res. 2012, 18, 1611–1618. [Google Scholar] [CrossRef]
- Zou, W.; Wolchok, J.D.; Chen, L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci. Transl. Med. 2016, 8. [Google Scholar] [CrossRef]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Kwak, J.J.; Tirumani, S.H.; Van den Abbeele, A.D.; Koo, P.J.; Jacene, H.A. Cancer immunotherapy: Imaging assessment of novel treatment response patterns and immune-related adverse events. Radiographics 2015, 35, 424–437. [Google Scholar] [CrossRef]
- Wang, G.X.; Kurra, V.; Gainor, J.F.; Sullivan, R.J.; Flaherty, K.T.; Lee, S.I.; Fintelmann, F.J. Immune checkpoint inhibitor cancer therapy: Spectrum of imaging findings. Radiographics 2017, 37, 2132–2144. [Google Scholar] [CrossRef] [PubMed]
- Küppers, R. The biology of Hodgkin’s lymphoma. Nat. Rev. Cancer 2009, 9, 15–27. [Google Scholar] [CrossRef]
- Jona, A.; Szodoray, P.; Illés, A. Immunologic pathomechanism of Hodgkin’s lymphoma. Exp. Hematol. 2013, 41, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Chemnitz, J.M.; Eggle, D.; Driesen, J.; Classen, S.; Riley, J.L.; Debey-Pascher, S.; Beyer, M.; Popov, A.; Zander, T.; Schultze, J.L. RNA fingerprints provide direct evidence for the inhibitory role of TGFβ and PD-1 on CD4+ T cells in Hodgkin lymphoma. Blood 2007, 110, 3226–3233. [Google Scholar] [CrossRef]
- Dercle, L.; Seban, R.D.; Lazarovici, J.; Schwartz, L.H.; Houot, R.; Ammari, S.; Danu, A.; Edeline, V.; Marabelle, A.; Ribrag, V.; et al. 18 F-FDG PET and CT scans detect new imaging patterns of response and progression in patients with hodgkin lymphoma treated by Anti–Programmed death 1 immune checkpoint inhibitor. J. Nucl. Med. 2018, 59, 15–24. [Google Scholar] [CrossRef]
- Ansell, S.M. Nivolumab in the treatment of Hodgkin lymphoma. Clin. Cancer Res. 2017, 23, 1623–1626. [Google Scholar] [CrossRef]
- Yamamoto, R.; Nishikori, M.; Kitawaki, T.; Sakai, T.; Hishizawa, M.; Tashima, M.; Kondo, T.; Ohmori, K.; Kurata, M.; Hayashi, T.; et al. PD-1 PD-1 ligand interaction contributes to immunosuppressive microenvironment of Hodgkin lymphoma. Blood 2008, 111, 3220–3224. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.J.; Chapuy, B.; Ouyang, J.; Sun, H.H.; Roemer, M.G.M.; Xu, M.L.; Yu, H.; Fletcher, C.D.M.; Freeman, G.J.; Shipp, M.A.; et al. PD-L1 expression is characteristic of a subset of aggressive B-cell lymphomas and virus-associated malignancies. Clin. Cancer Res. 2013, 19, 3462–3473. [Google Scholar] [CrossRef] [PubMed]
- Andorsky, D.J.; Yamada, R.E.; Said, J.; Pinkus, G.S.; Betting, D.J.; Timmerman, J.M. Programmed death ligand 1 is expressed by non-Hodgkin lymphomas and inhibits the activity of tumor-associated T cells. Clin. Cancer Res. 2011, 17, 4232–4244. [Google Scholar] [CrossRef]
- Kershaw, M.H.; Teng, M.W.L.; Smyth, M.J.; Darcy, P.K. Supernatural T cells: Genetic modification of T cells for cancer therapy. Nat. Rev. Immunol. 2005, 5, 928–940. [Google Scholar] [CrossRef] [PubMed]
- Batlevi, C.L.; Matsuki, E.; Brentjens, R.J.; Younes, A. Novel immunotherapies in lymphoid malignancies. Nat. Rev. Clin. Oncol. 2016, 13, 25–40. [Google Scholar] [CrossRef]
- Ferrari, C.; Niccoli Asabella, A.; Merenda, N.; Altini, C.; Fanelli, M.; Muggeo, P.; De Leonardis, F.; Perillo, T.; Santoro, N.; Rubini, G. Pediatric Hodgkin Lymphoma: Predictive value of interim 18F-FDG PET/CT in therapy response assessment. Medicine 2017. [Google Scholar] [CrossRef] [PubMed]
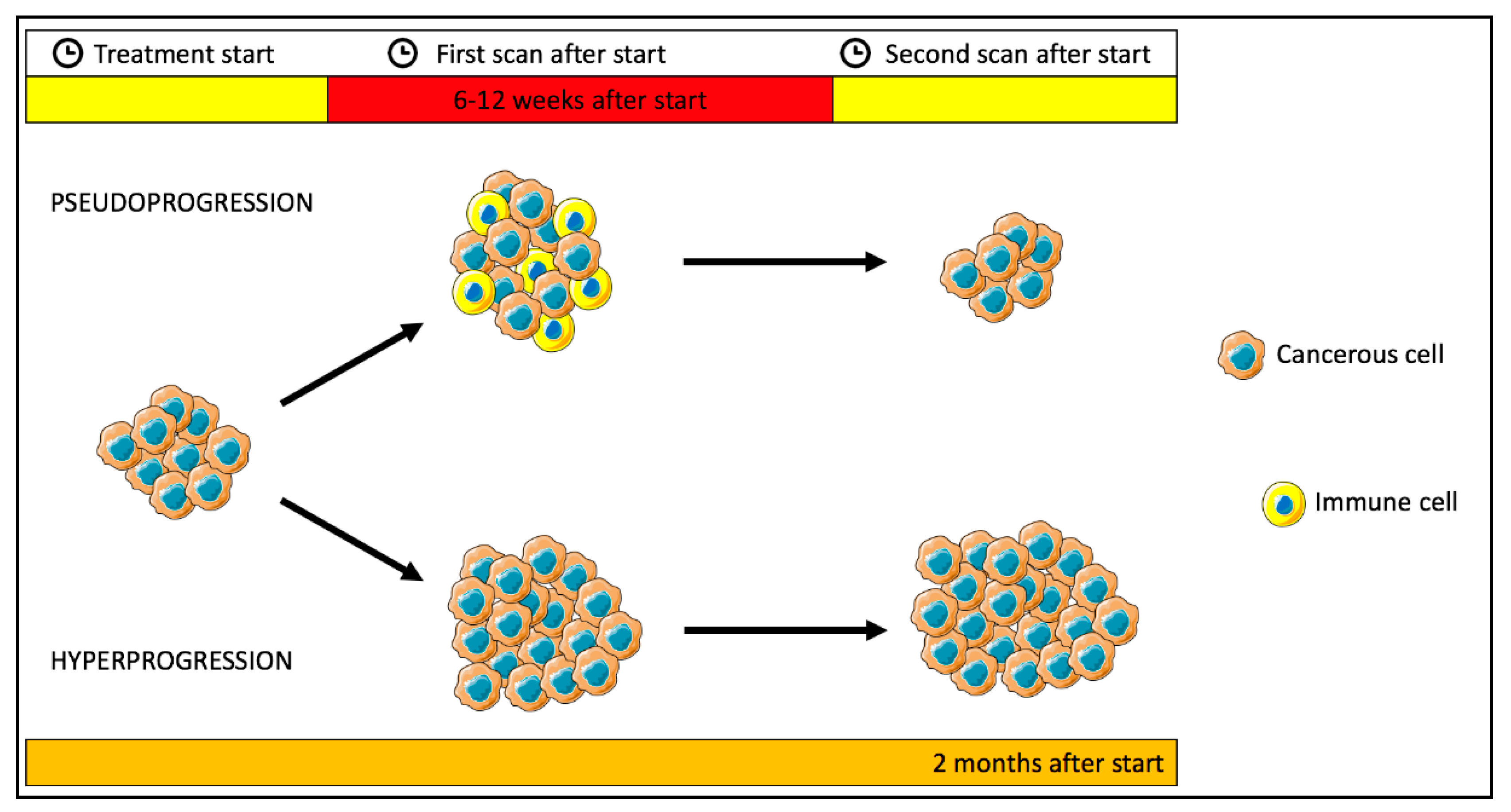
- Kanjanapan, Y.; Day, D.; Wang, L.; Al-Sawaihey, H.; Abbas, E.; Namini, A.; Siu, L.L.; Hansen, A.; Razak, A.A.; Spreafico, A.; et al. Hyperprogressive disease in early-phase immunotherapy trials: Clinical predictors and association with immune-related toxicities. Cancer 2019, 125, 1341–1349. [Google Scholar] [CrossRef]
- Borcoman, E.; Kanjanapan, Y.; Champiat, S.; Kato, S.; Servois, V.; Kurzrock, R.; Goel, S.; Bedard, P.; Le Tourneau, C. Novel patterns of response under immunotherapy. Ann. Oncol. 2019, 30, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Seymour, L.; Bogaerts, J.; Perrone, A.; Ford, R.; Schwartz, L.H.; Mandrekar, S.; Lin, N.U.; Litière, S.; Dancey, J.; Chen, A.; et al. iRECIST: Guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017, 18, e143–e152. [Google Scholar] [CrossRef]
- Baues, C.; Trommer-Nestler, M.; Jablonska, K.; Bröckelmann, P.J.; Schlaak, M.; Von Bergwelt-Baildon, M.; Engert, A.; Semrau, R.; Marnitz, S.; Theurich, S. Short review of potential synergies of immune checkpoint inhibition and radiotherapy with a focus on Hodgkin lymphoma: Radio-immunotherapy opens new doors. Immunotherapy 2017, 9, 423–433. [Google Scholar] [CrossRef]
- Sardaro, A.; Ferrari, C.; Carbonara, R.; Altini, C.; Lavelli, V.; Rubini, G. Synergism Between Immunotherapy and Radiotherapy in Esophageal Cancer: An Overview of Current Knowledge and Future Perspectives. Cancer Biother. Radiopharm. 2020. [Google Scholar] [CrossRef] [PubMed]
- Shahabi, V.; Postow, M.A.; Tuck, D.; Wolchok, J.D. Immune-priming of the tumor microenvironment by radiotherapy: Rationale for combination with immunotherapy to improve anticancer efficacy. Am. J. Clin. Oncol. Cancer Clin. Trials 2015, 38, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Stamell, E.F.; Wolchok, J.D.; Gnjatic, S.; Lee, N.Y.; Brownell, I. The abscopal effect associated with a systemic anti-melanoma immune response. Int. J. Radiat. Oncol. Biol. Phys. 2013, 85, 293–295. [Google Scholar] [CrossRef]
- Postow, M.A.; Callahan, M.K.; Barker, C.A.; Yamada, Y.; Yuan, J.; Kitano, S.; Mu, Z.; Rasalan, T.; Adamow, M.; Ritter, E.; et al. Immunologic Correlates of the Abscopal Effect in a Patient with Melanoma. N. Engl. J. Med. 2012, 366, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Reynders, K.; Illidge, T.; Siva, S.; Chang, J.Y.; De Ruysscher, D. The abscopal effect of local radiotherapy: Using immunotherapy to make a rare event clinically relevant. Cancer Treat. Rev. 2015, 41, 503–510. [Google Scholar] [CrossRef]
- Burnette, B.C.; Liang, H.; Lee, Y.; Chlewicki, L.; Khodarev, N.N.; Weichselbaum, R.R.; Fu, Y.X.; Auh, S.L. The efficacy of radiotherapy relies upon induction of type I interferon-dependent innate and adaptive immunity. Cancer Res. 2011, 71, 2488–2496. [Google Scholar] [CrossRef]
- Tseng, Y.D.; Chen, Y.H.; Catalano, P.J.; Ng, A. Rates and durability of response to salvage radiation therapy among patients with refractory or relapsed aggressive non-Hodgkin lymphoma. Int. J. Radiat. Oncol. Biol. Phys. 2015, 91, 223–231. [Google Scholar] [CrossRef]
- Quéro, L.; Gilardin, L.; Fumagalli, I.; Martin, V.; Guillerm, S.; Bauduceau, O.; Kirova, Y.M.; Hennequin, C.; Brice, P. Anti-PD-1 immunotherapy in combination with sequential involved-site radiotherapy in heavily pretreated refractory Hodgkin lymphoma. Cancer Radiother. 2019, 23, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Nan, X.; Miller, T.; Fisher, R.; Teh, B.; Pandita, S.; Farach, A.M.; Pingali, S.R.; Pandita, R.K.; Butler, E.B.; et al. Complete Local and Abscopal Responses from a Combination of Radiation and Nivolumab in Refractory Hodgkin’s Lymphoma. Radiat. Res. 2018, 190, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, G.; Marech, I.; Asabella, A.N.; Di Palo, A.; Porcelli, M.; Lavelli, V.; Rubini, G.; Ferrari, C.; Gadaleta, C.D. Tyrosine-Kinase inhibitors therapies with mainly anti-angiogenic activity in advanced renal cell carcinoma: Value of PET/CT in response evaluation. Int. J. Mol. Sci. 2017, 18, 1937. [Google Scholar] [CrossRef]
- Cheson, B.D.; Ansell, S.; Schwartz, L.; Gordon, L.I.; Advani, R.; Jacene, H.A.; Hoos, A.; Barrington, S.F.; Armand, P. Refinement of the Lugano Classification lymphoma response criteria in the era of immunomodulatory therapy. Blood 2016, 128, 2489–2496. [Google Scholar] [CrossRef]
- Chen, A.; Mokrane, F.Z.; Schwartz, L.H.; Morschhauser, F.; Stamatoullas, A.; Schiano de Colella, J.M.; Vercellino, L.; Casasnovas, O.; Chauchet, A.; Delmer, A.; et al. Early 18F-FDG PET/CT Response Predicts Survival in Relapsed or Refractory Hodgkin Lymphoma Treated with Nivolumab. J. Nucl. Med. 2020, 61, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Dercle, L.; Mokrane, F.Z.; Schiano de Colella, J.M.; Stamatoullas, A.; Morschhauser, F.; Brice, P.; Ghesquières, H.; Casasnovas, O.; Chen, A.; Manson, G.; et al. Unconventional immune-related phenomena observed using 18F-FDG PET/CT in Hodgkin lymphoma treated with anti PD-1 monoclonal antibodies. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1391–1392. [Google Scholar] [CrossRef]
- Chang, C.H.; Qiu, J.; O’Sullivan, D.; Buck, M.D.; Noguchi, T.; Curtis, J.D.; Chen, Q.; Gindin, M.; Gubin, M.M.; Van Der Windt, G.J.W.; et al. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell 2015, 162, 1229–1241. [Google Scholar] [CrossRef]
- Dercle, L.; Ammari, S.; Seban, R.D.; Schwartz, L.H.; Houot, R.; Labaied, N.; Mokrane, F.Z.; Lazarovici, J.; Danu, A.; Marabelle, A.; et al. Kinetics and nadir of responses to immune checkpoint blockade by anti-PD1 in patients with classical Hodgkin lymphoma. Eur. J. Cancer 2018, 91, 136–144. [Google Scholar] [CrossRef]
- Wang, J.; Hu, Y.; Yang, S.; Wei, G.; Zhao, X.; Wu, W.; Zhang, Y.; Zhang, Y.; Chen, D.; Wu, Z.; et al. Role of Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography in Predicting the Adverse Effects of Chimeric Antigen Receptor T Cell Therapy in Patients with Non-Hodgkin Lymphoma. Biol. Blood Marrow Transplant. 2019, 25, 1092–1098. [Google Scholar] [CrossRef]
- Shah, N.N.; Nagle, S.J.; Torigian, D.A.; Farwell, M.D.; Hwang, W.T.; Frey, N.; Nasta, S.D.; Landsburg, D.; Mato, A.; June, C.H.; et al. Early positron emission tomography/computed tomography as a predictor of response after CTL019 chimeric antigen receptor—T-cell therapy in B-cell non-Hodgkin lymphomas. Cytotherapy 2018, 20, 1415–1418. [Google Scholar] [CrossRef]
- Derlin, T.; Schultze-Florey, C.; Werner, R.A.; Möhn, N.; Skripuletz, T.; David, S.; Beutel, G.; Eder, M.; Ross, T.L.; Bengel, F.M.; et al. 18F-FDG PET/CT of off-target lymphoid organs in CD19-targeting chimeric antigen receptor T-cell therapy for relapsed or refractory diffuse large B-cell lymphoma. Ann. Nucl. Med. 2020, 35. [Google Scholar] [CrossRef]
- Haverkos, B.M.; Abbott, D.; Hamadani, M.; Armand, P.; Flowers, M.E.; Merryman, R.; Kamdar, M.; Kanate, A.S.; Saad, A.; Mehta, A.; et al. PD-1 blockade for relapsed lymphoma post-allogeneic hematopoietic cell transplant: High response rate but frequent GVHD. Blood 2017, 130, 221–228. [Google Scholar] [CrossRef]
- Younes, A.; Santoro, A.; Shipp, M.; Zinzani, P.L.; Timmerman, J.M.; Ansell, S.; Armand, P.; Fanale, M.; Ratanatharathorn, V.; Kuruvilla, J.; et al. Nivolumab for classical Hodgkin’s lymphoma after failure of both autologous stem-cell transplantation and brentuximab vedotin: A multicentre, multicohort, single-arm phase 2 trial. Lancet Oncol. 2016, 17, 1283–1294. [Google Scholar] [CrossRef]
- Chen, R.; Zinzani, P.L.; Lee, H.J.; Armand, P.; Johnson, N.A.; Brice, P.; Radford, J.; Ribrag, V.; Molin, D.; Vassilakopoulos, T.P.; et al. Pembrolizumab in relapsed or refractory Hodgkin lymphoma: 2-year follow-up of KEYNOTE-087. Blood 2019, 134, 1144–1153. [Google Scholar] [CrossRef]
- Isohashi, K.; Tatsumi, M.; Higuchi, I.; Inoue, A.; Nakajo, K.; Ishikawa, J.; Shimosegawa, E.; Kanakura, Y.; Nakamura, H.; Hatazawa, J. 18F-FDG-PET in patients with malignant lymphoma having long-term follow-up: Staging and restaging, and evaluation of treatment response and recurrence. Ann. Nucl. Med. 2008, 22, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Barrington, S.F.; Kirkwood, A.A.; Franceschetto, A.; Fulham, M.J.; Roberts, T.H.; Almquist, H.; Brun, E.; Hjorthaug, K.; Viney, Z.N.; Pike, L.C.; et al. PET-CT for staging and early response: Results from the Response-Adapted Therapy in Advanced Hodgkin Lymphoma study. Blood 2016, 127, 1531–1538. [Google Scholar] [CrossRef]
- Tatsumi, M.; Cohade, C.; Nakamoto, Y.; Fishman, E.K.; Wahl, R.L. Direct comparison of FDG PET and CT findings in patients with lymphoma: Initial experience. Radiology 2005, 237, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Manson, G.; Herbaux, C.; Brice, P.; Bouabdallah, K.; Stamatoullas, A.; Schiano, J.M.; Ghesquieres, H.; Dercle, L.; Houot, R. Prolonged remissions after anti-PD-1 discontinuation in patients with Hodgkin lymphoma. Blood 2018, 131, 2856–2859. [Google Scholar] [CrossRef] [PubMed]
- Rossi, C.; Gilhodes, J.; Maerevoet, M.; Herbaux, C.; Morschhauser, F.; Brice, P.; Garciaz, S.; Borel, C.; Ysebaert, L.; Obéric, L.; et al. Efficacy of chemotherapy or chemo-anti-PD-1 combination after failed anti-PD-1 therapy for relapsed and refractory hodgkin lymphoma: A series from lysa centers. Am. J. Hematol. 2018, 93, 1042–1049. [Google Scholar] [CrossRef]
- Ansell, S.M. Immunotherapy in Hodgkin Lymphoma: The Road Ahead. Trends Immunol. 2019, 40, 380–386. [Google Scholar] [CrossRef]
- Xie, W.; Medeiros, L.J.; Li, S.; Yin, C.C.; Khoury, J.D.; Xu, J. PD-1/PD-L1 Pathway and Its Blockade in Patients with Classic Hodgkin Lymphoma and Non-Hodgkin Large-Cell Lymphomas. Curr. Hematol. Malig. Rep. 2020, 15, 372–381. [Google Scholar] [CrossRef]
- Merryman, R.W.; Armand, P.; Wright, K.T.; Rodig, S.J. Checkpoint blockade in Hodgkin and non-Hodgkin lymphoma. Blood Adv. 2017. [Google Scholar] [CrossRef] [PubMed]
- Imber, B.S.; Sadelain, M.; DeSelm, C.; Batlevi, C.; Brentjens, R.J.; Dahi, P.B.; Giralt, S.; Park, J.H.; Sauter, C.; Scordo, M.; et al. Early experience using salvage radiotherapy for relapsed/refractory non-Hodgkin lymphomas after CD19 chimeric antigen receptor (CAR) T cell therapy. Br. J. Haematol. 2020, 190, 45–51. [Google Scholar] [CrossRef]
- Weist, M.R.; Starr, R.; Aguilar, B.; Chea, J.; Miles, J.K.; Poku, E.; Gerdts, E.; Yang, X.; Priceman, S.J.; Forman, S.J.; et al. PET of adoptively transferred chimeric antigen receptor T Cells with 89Zr-Oxine. J. Nucl. Med. 2018, 59, 1531–1537. [Google Scholar] [CrossRef]
- Mulazzani, M.; Fräßle, S.P.; Von Mücke-Heim, I.; Langer, S.; Zhou, X.; Ishikawa-Ankerhold, H.; Leube, J.; Zhang, W.; Dötsch, S.; Svec, M.; et al. Long-term in vivo microscopy of CAR T cell dynamics during eradication of CNS lymphoma in mice. Proc. Natl. Acad. Sci. USA 2019, 116, 24275–24284. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Zhao, L.; Zhang, Y.; Qin, Y.; Guan, Y.; Zhang, T.; Liu, C.; Zhou, J. Understanding the Mechanisms of Resistance to CAR T-Cell Therapy in Malignancies. Front. Oncol. 2019, 9, 1237. [Google Scholar] [CrossRef] [PubMed]
- Kochenderfer, J.N.; Somerville, R.P.T.; Lu, T.; Shi, V.; Bot, A.; Rossi, J.; Xue, A.; Goff, S.L.; Yang, J.C.; Sherry, R.M.; et al. Lymphoma remissions caused by anti-CD19 chimeric antigen receptor T cells are associated with high serum interleukin-15 levels. J. Clin. Oncol. 2017, 35, 1803–1813. [Google Scholar] [CrossRef]
- Lee, D.W.; Santomasso, B.D.; Locke, F.L.; Ghobadi, A.; Turtle, C.J.; Brudno, J.N.; Maus, M.V.; Park, J.H.; Mead, E.; Pavletic, S.; et al. ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells. Biol. Blood Marrow Transplant. 2019, 25, 625–638. [Google Scholar] [CrossRef]
- Giavridis, T.; Van Der Stegen, S.J.C.; Eyquem, J.; Hamieh, M.; Piersigilli, A.; Sadelain, M. CAR T cell-induced cytokine release syndrome is mediated by macrophages and abated by IL-1 blockade letter. Nat. Med. 2018, 24, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Norelli, M.; Camisa, B.; Barbiera, G.; Falcone, L.; Purevdorj, A.; Genua, M.; Sanvito, F.; Ponzoni, M.; Doglioni, C.; Cristofori, P.; et al. Monocyte-derived IL-1 and IL-6 are differentially required for cytokine-release syndrome and neurotoxicity due to CAR T cells. Nat. Med. 2018, 24, 739–748. [Google Scholar] [CrossRef]
- Wu, X.; Pertovaara, H.; Korkola, P.; Vornanen, M.; Eskola, H.; Kellokumpu-Lehtinen, P.L. Glucose metabolism correlated with cellular proliferation in diffuse large B-cell lymphoma. Leuk. Lymphoma 2012, 53, 400–405. [Google Scholar] [CrossRef]
- Rubin, D.B.; Danish, H.H.; Ali, A.B.; Li, K.; Larose, S.; Monk, A.D.; Cote, D.J.; Spendley, L.; Kim, A.H.; Robertson, M.S.; et al. Neurological toxicities associated with chimeric antigen receptor T-cell therapy. Brain 2019, 142, 1334–1348. [Google Scholar] [CrossRef]
- Makuku, R.; Khalili, N.; Razi, S.; Keshavarz-Fathi, M.; Rezaei, N. Current and Future Perspectives of PD-1/PDL-1 Blockade in Cancer Immunotherapy. J. Immunol. Res. 2021, 2021, 1–15. [Google Scholar] [CrossRef]
- Costa, L.B.; Queiroz, M.A.; Barbosa, F.G.; Nunes, R.F.; Zaniboni, E.C.; Ruiz, M.M.; Jardim, D.; Marin, J.F.G.; Cerri, G.G.; Buchpiguel, C.A. Reassessing patterns of response to immunotherapy with pet: From morphology to metabolism. Radiographics 2021, 41, 120–143. [Google Scholar] [CrossRef] [PubMed]
- Nobashi, T.; Baratto, L.; Reddy, S.A.; Srinivas, S.; Toriihara, A.; Hatami, N.; Yohannan, T.K.; Mittra, E. Predicting Response to Immunotherapy by Evaluating Tumors, Lymphoid Cell-Rich Organs, and Immune-Related Adverse Events Using FDG-PET/CT. Clin. Nucl. Med. 2019, 44, e272–e279. [Google Scholar] [CrossRef]
- Petersen, H.; Holdgaard, P.C.; Madsen, P.H.; Knudsen, L.M.; Gad, D.; Gravergaard, A.E.; Rohde, M.; Godballe, C.; Engelmann, B.E.; Bech, K.; et al. FDG PET/CT in cancer: Comparison of actual use with literature-based recommendations. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Sachpekidis, C.; Kopp-Schneider, A.; Hakim-Meibodi, L.; Dimitrakopoulou-Strauss, A.; Hassel, J.C. 18 F-FDG PET/CT longitudinal studies in patients with advanced metastatic melanoma for response evaluation of combination treatment with vemurafenib and ipilimumab. Melanoma Res. 2019, 29, 178–186. [Google Scholar] [CrossRef]
- Aide, N.; Hicks, R.J.; Le Tourneau, C.; Lheureux, S.; Fanti, S.; Lopci, E. FDG PET/CT for assessing tumour response to immunotherapy: Report on the EANM symposium on immune modulation and recent review of the literature. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 238–250. [Google Scholar] [CrossRef]
- Pektor, S.; Hilscher, L.; Walzer, K.C.; Miederer, I.; Bausbacher, N.; Loquai, C.; Schreckenberger, M.; Sahin, U.; Diken, M.; Miederer, M. In vivo imaging of the immune response upon systemic RNA cancer vaccination by FDG-PET. EJNMMI Res. 2018, 8. [Google Scholar] [CrossRef]
- Maschmeyer, G.; De Greef, J.; Mellinghoff, S.C.; Nosari, A.; Thiebaut-Bertrand, A.; Bergeron, A.; Franquet, T.; Blijlevens, N.M.A.; Maertens, J.A. Infections associated with immunotherapeutic and molecular targeted agents in hematology and oncology. A position paper by the European Conference on Infections in Leukemia (ECIL). Leukemia 2019, 33, 844–862. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C.; François, P.; Faivre-Chauvet, A.; Bourgeois, M.; Guérard, F.; Haddad, F.; Barbet, J.; Chérel, M.; Kraeber-Bodéré, F.; Carlier, T.; et al. Immuno-pet for clinical theranostic approaches. Int. J. Mol. Sci. 2017, 18, 57. [Google Scholar] [CrossRef] [PubMed]
- Knowles, S.M.; Wu, A.M. Advances in immuno-positron emission tomography: Antibodies for molecular imaging in oncology. J. Clin. Oncol. 2012, 30, 3884–3892. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.A.; Drake, C.G.; Mintz, A. Molecular imaging for cancer immunotherapy. Immuno-Oncol. Technol. 2020, 5, 10–21. [Google Scholar] [CrossRef]
- Wei, W.; Rosenkrans, Z.T.; Liu, J.; Huang, G.; Luo, Q.Y.; Cai, W. ImmunoPET: Concept, Design, and Applications. Chem. Rev. 2020, 120, 3787–3851. [Google Scholar] [CrossRef] [PubMed]
- Muylle, K.; Flamen, P.; Vugts, D.J.; Guiot, T.; Ghanem, G.; Meuleman, N.; Bourgeois, P.; Vanderlinden, B.; Van Dongen, G.A.M.S.; Everaert, H.; et al. Tumour targeting and radiation dose of radioimmunotherapy with 90Y-rituximab in CD20+ B-cell lymphoma as predicted by 89Zr-rituximab immuno-PET: Impact of preloading with unlabelled rituximab. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1304–1314. [Google Scholar] [CrossRef]
- Rizvi, S.N.F.; Visser, O.J.; Vosjan, M.J.W.D.; Van Lingen, A.; Hoekstra, O.S.; Zijlstra, J.M.; Huijgens, P.C.; Van Dongen, G.A.M.S.; Lubberink, M. Biodistribution, radiation dosimetry and scouting of 90Y- ibritumomab tiuxetan therapy in patients with relapsed B-cell non-Hodgkin’s lymphoma using 89Zr-ibritumomab tiuxetan and PET. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 512–520. [Google Scholar] [CrossRef]
- Jauw, Y.W.S.; Zijlstra, J.M.; De Jong, D.; Vugts, D.J.; Zweegman, S.; Hoekstra, O.S.; Van Dongen, G.A.M.S.; Huisman, M.C. Performance of 89Zr-labeled-rituximab-PET as an imaging biomarker to assess CD20 targeting: A pilot study in patients with relapsed/refractory diffuse large B cell lymphoma. PLoS ONE 2017, 12, e169828. [Google Scholar] [CrossRef]
- Simonetta, F.; Alam, I.S.; Lohmeyer, J.K.; Sahaf, B.; Good, Z.; Chen, W.; Xiao, Z.; Hirai, T.; Scheller, L.; Engels, P.; et al. Molecular Imaging of Chimeric Antigen Receptor T Cells by ICOS-ImmunoPET. Clin. Cancer Res. 2020. [Google Scholar] [CrossRef]
| LUGANO | LYRIC | |
|---|---|---|
| Complete Response (CR) | PET/CT: DS 1, 2, or 3 with or without a residual mass OR CT: target nodes/nodal masses must regress to ≤1.5 cm in longest diameter | |
| Partial Response (PR) | PET/CT: DS 4 or 5 with reduced uptake compared with baseline and residual mass(es) of any size OR CT: ≥50% decrease in SPD of up to 6 target measurable nodes and extranodal sites | |
| Progressive Disease (PD) | PET/CT: DS 4 or 5 with an increase in intensity of uptake from baseline and/or new FDG-avid foci consistent with lymphoma at interim or EoT assessment. OR CT: an individual node/lesion must be abnormal with: longest diameter >1.5 cm and increase by ≥50% from product of the perpendicular diameters nadir and an increase in longest or short diameter from nadir 0.5 cm for lesions ≤2 cm 1.0 cm for lesions >2 cm | |
| In the setting of splenomegaly, the splenic length must increase by >50% of the extent of its prior increase beyond baseline (eg, a 15-cm spleen must increase to >16 cm). If no prior splenomegaly, must increase by ≥2 cm from baseline | ||
| New or recurrent splenomegaly AND/OR involvement of the bone marrow | ||
| New or clear progression of preexisiting non measured lesions | ||
| Regrowth of previously resolved lesions | ||
| A new node >1.5 cm in any axis or a new extranodal site >1.0 cm in any axis; if <1.0 cm in any axis, its presence must be unequivocal and must be attributable to lymphoma | ||
| Assessable disease of any size unequivocally attributable to lymphoma | ||
| IR(1): ≥50% increase in SPD in first 12 weeks | ||
| IR(2): <50% increase in SPD with | ||
| a. New lesion(s), or | ||
| b. ≥50% increase in PPD of a lesion or set of lesions at any time during treatment | ||
| IR(3): Increase in FDG uptake without a concomitant increase in lesion size meeting criteria for PD | ||
| Author PMID | Year | Study Type | pts | Hystology | Immunotherapy | Imaging Timing | Response Criteria | Main Faindings |
|---|---|---|---|---|---|---|---|---|
| Chen et al., 31,628,220 [47] | 2019 | retrospective | 45 | HL | anti PD-1 (nivolumab) | 3 mo (SD +\−2.3) | Lugano/LYRIC | In R/R HL patients the first early 18F-FDG PET/CT assessment, using either Lugano or LYRIC, predicted OS and allowed early risk stratification. |
| Castello et al., 30,032,683 [3] | 2018 | retrospective | 43 | HL | anti PD-1 (42 nivolumab; 1 pembrolizumab) | 8 weeks and 17 weeks | Lugano/LYRIC | Decrease in glucose metabolism (DSUVmax) and tumor burden (DMTV, DTLG) on early/interim 18F-FDG PET/CT resulted significant in responders to anti PD-1 immunotherapy. |
| Mokrane et al., 32,286,191 [8] | 2020 | retrospective | 45 | HL | anti PD-1 (nivolumab) | 2 mo (range 1.7–3.7) | Lugano/LYRIC | In R/R HL early CT and 18F-FDG PET/CT at a median of 2 months after initiation of anti-PD1 immunotherapy predicted OS. |
| Dercle et al., 28,596,157 [24] | 2018 | retrospective | 16 | HL | anti PD-1 (15 nivolumab; 1 pembrolizumab) | 3 mo | Lugano/LYRIC | HL responders (CR, PR) at 3-month 18F-FDG PET/CT assessment could confirm/convert in a complete response in the successive evaluation |
| Dercle et al., 29,360,605 [50] | 2018 | retrospective | 16 | HL | anti PD-1 (15 nivolumab; 1 pembrolizumab) | 2–3 mo | Lugano/LYRIC | Reduction in tumor volume (DMTV, DTLG), in tumor glucose metabolism (DSUVmax) and increasing in spleen metabolism is associated to anti PD-1 therapy response |
| Wang et al., 30,769,193 [51] | 2019 | retrospective | 19 | NHL (14 DLBCL, 3 FL, 1 MALT, 1 Burkitt, 1 ovarian LL) | CD19 CAR-T | baseline | PERCIST | In NHL patients treated with CAR-T cells therapy, higher baseline disease burden (DMTV; DTLG) on 18F-FDG PET/CT is associated have more severe CRS |
| Shah et al., 30,385,043 [52] | 2018 | prospective | 7 | NHL (3 DLBCL, 4 FL) | CTL019 CAR-T | 1 mo | Lugano | In patients with DLBCL and FL receiving CTL019 CAR-T cells, the early 18F-FDG PET/CT and the total MTV could predict immunotherapy response |
| Derlin et al., 33,174,144 [53] | 2020 | retrospective | 10 | NHL (DLBCL) | CD19 CAR-T | 30 d and 90 d | Lugano | Early metabolic assessment in lymphoma lesions and off-target lymphoid organs (spleen and lymph nodes) could predict medium-term response to CAR-T-cell therapy, as well as identify patients at risk for severe toxicity |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrari, C.; Maggialetti, N.; Masi, T.; Nappi, A.G.; Santo, G.; Niccoli Asabella, A.; Rubini, G. Early Evaluation of Immunotherapy Response in Lymphoma Patients by 18F-FDG PET/CT: A Literature Overview. J. Pers. Med. 2021, 11, 217. https://doi.org/10.3390/jpm11030217
Ferrari C, Maggialetti N, Masi T, Nappi AG, Santo G, Niccoli Asabella A, Rubini G. Early Evaluation of Immunotherapy Response in Lymphoma Patients by 18F-FDG PET/CT: A Literature Overview. Journal of Personalized Medicine. 2021; 11(3):217. https://doi.org/10.3390/jpm11030217
Chicago/Turabian StyleFerrari, Cristina, Nicola Maggialetti, Tamara Masi, Anna Giulia Nappi, Giulia Santo, Artor Niccoli Asabella, and Giuseppe Rubini. 2021. "Early Evaluation of Immunotherapy Response in Lymphoma Patients by 18F-FDG PET/CT: A Literature Overview" Journal of Personalized Medicine 11, no. 3: 217. https://doi.org/10.3390/jpm11030217
APA StyleFerrari, C., Maggialetti, N., Masi, T., Nappi, A. G., Santo, G., Niccoli Asabella, A., & Rubini, G. (2021). Early Evaluation of Immunotherapy Response in Lymphoma Patients by 18F-FDG PET/CT: A Literature Overview. Journal of Personalized Medicine, 11(3), 217. https://doi.org/10.3390/jpm11030217







