Poor-Prognosis Patients Affected by Glioblastoma: Retrospective Study of Hypofractionated Radiotherapy with Simultaneous Integrated Boost and Concurrent/Adjuvant Temozolomide
Abstract
:1. Introduction
2. Materials and Methods
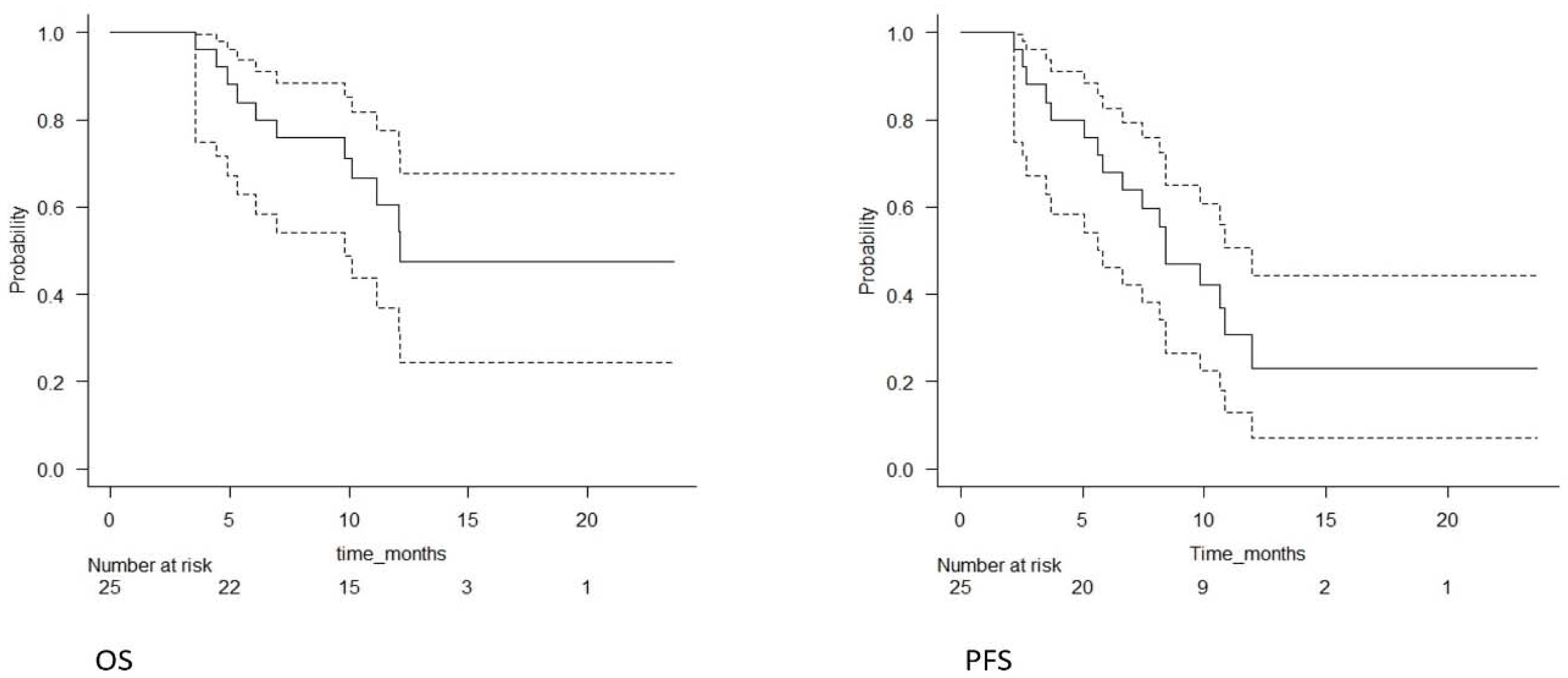
3. Results
Prognostic Factors for OS and PFS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stupp, R.; Mason, W.; Van der Bent, M.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups, National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant Temozolomide for glioblatoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.D.; Strike, T.A. An evaluation of methyl-CCNU, BCNU and radiotherapy in the treatment of malignant glioma. Proc. Am. Assoc. Cancer Res. 1976, 17, 63. [Google Scholar]
- Balducci, M.; Apicella, G.; Manfrida, S.; Mangiola, A.; Fiorentino, A.; Azario, L.; D’Agostino, G.R.; Frascino, V.; Dinapoli, N.; Mantini, G.; et al. Single- Arm Phase II Study of Conformal Radiation Therapy and Temozolomide plus fractionated Stereotactic Conformal Boost in High grade Gliomas. Strahlentherapie und Onkologie 2010, 186, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Balducci, M.; Chiesa, S.; Diletto, B.; D’Agostino, G.R.; Mangiola, A.; Manfrida, S.; Mantini, G.; Albanese, A.; Fiorentino, A.; Frascino, V.; et al. Low-dose fractionated radiotherapy and concomitant chemotherapy in glioblastoma multiforme with poor prognosis: A feasibility study. Neuro Oncol. 2012, 14, 79–86. [Google Scholar] [CrossRef]
- Lee, S.W.; Fraass, B.A.; Marsh, L.H.; Herbort, K.; Gebarski, S.S.; Martel, M.K.; Radany, E.H.; Lichter, A.S.; Sandler, H.M. Patterns of failure following high-dose 3-D conformal radiotherapy for high-grade astrocytomas: A quantitative dosimetric study. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 79–88. [Google Scholar] [CrossRef]
- Mazzola, R.; Corradini, S.; Gregucci, F.; Figlia, V.; Fiorentino, A.; Alongi, F. Role of Radiosurgery/Stereotactic Radiotherapy in Oligometastatic Disease: Brain Oligometastases. Front. Oncol. 2019, 9, 206. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, R.; Corradini, S.; Gregucci, F.; Figlia, V.; Fiorentino, A.; Alongi, F. Volume de-escalation in radiation therapy: State of the art and new perspectives. J. Cancer Res. Clin. Oncol. 2020, 146, 909–924. [Google Scholar]
- McAleese, J.J.; Stenning, S.P.; Ashley, S.; Traish, D.; Hines, F.; Sardell, S.; Guerrero, D.; Brada, M. Hypofractionated radiotherapy for poor prognosis malignant glioma: Matched pair survival analysis with MRC controls. Radiother. Oncol. 2003, 67, 177–182. [Google Scholar] [CrossRef]
- Phillips, C.; Guiney, M.; Smith, J.; Hughes, P.; Narayan, K.; Quong, G. A randomized trial comparing 35 Gy in ten fractions with 60 Gy in 30 fractions of cerebral irradiation for glioblastoma multiforme and older patients with anaplastic astrocytoma. Radiother. Oncol. 2003, 68, 23–26. [Google Scholar] [CrossRef]
- Chang, E.L.; Yi, W.; Allen, P.K.; Levin, V.A.; Sawaya, R.E.; Maor, M.H. Hypofractionated radiotherapy for elderly or younger low performance status glioblastoma patients: Outcome and prognostic factors. Int. J. Radiat. Oncol. Biol. Phys. 2003, 56, 519–528. [Google Scholar] [CrossRef]
- Roa, W.; Brasher, P.M.A.; Bauman, G.; Anthes, M.; Bruera, E.; Chan, A.; Fisher, B.; Fulton, D.; Gulavita, S.; Hao, C.; et al. Abbreviated course of radiation therapy in older patients with glioblastoma multiforme: A prospective randomized clinical trial. J. Clin. Oncol. 2004, 22, 1583–1588. [Google Scholar] [CrossRef] [PubMed]
- Malmström, A.; Grønberg, B.H.; Marosi, C.; Stupp, R.; Frappaz, D.; Schultz, H.; Abacioglu, U.; Tavelin, B.; Lhermitte, B.; Hegi, M.E.; et al. Temozolomide versus standard 6-week radiotherapy versus hypofractionated radiotherapy in patients older than 60 years with glioblastoma: The Nordic randomised, phase 3 trial. Lancet Oncol. 2012, 13, 916–926. [Google Scholar] [CrossRef]
- Scoccianti, S.; Krengli, M.; Marrazzo, L.; Magrini, S.M.; Detti, B.; Fusco, V.; Pirtoli, L.; Doino, D.; Fiorentino, A.; Masini, L.; et al. Hypofractionated radiotherapy with simultaneous integrated boost (SIB) plus temozolomide in good prognosis patients with glioblastoma: A multicenter phase II study by the Brain Study Group of the Italian Association of Radiation Oncology (AIRO). Radiol. Med. 2018, 123, 48–62. [Google Scholar] [CrossRef] [Green Version]
- Wick, W.; Platten, M.; Meisner, C.; Felsberg, J.; Tabatabai, G.; Simon, M.; Nikkhah, G.; Papsdorf, K.; Steinbach, J.P.; Sabel, M.; et al. Temozolomide chemotherapy alone versus radiotherapy alone for malignant astrocytoma in the elderly: The NOA-08 randomised, phase 3 trial. Lancet Oncol. 2012, 13, 707–715. [Google Scholar] [CrossRef] [Green Version]
- Jablonska, P.A.; Diez-Valle, R.; Pérez-Larraya, J.G.; Moreno-Jiménez, M.; Idoate, M.Á.; Arbea, L.; Tejada, S.; Garcia de Eulate, M.R.; Ramos, L.; Arbizu, J.; et al. Hypofractionated radiation therapy and temozolomide in patients with glioblastoma and poor prognostic factors. A prospective, single-institution experience. PLoS ONE 2019, 14, e0217881. [Google Scholar]
- Perry, J.R.; Laperriere, N.; O’Callaghan, C.J.; Brandes, A.A.; Menten, J.; Phillips, C.; Fay, M.; Nishikawa, R.; Cairncross, J.G.; Roa, W.; et al. Trial Investigators. Short-Course Radiation plus Temozolomide in Elderly Patients with Glioblastoma. N. Engl. J. Med. 2017, 376, 1027–1037. [Google Scholar] [CrossRef] [Green Version]
- Navarria, P.; Pessina, F.; Cozzi, L.; Tomatis, S.; Reggiori, G.; Simonelli, M.; Santoro, A.; Clerici, E.; Franzese, C.; Carta, G.; et al. Phase II study of hypofractionated radiation therapy in elderly patients with newly diagnosed glioblastoma with poor prognosis. Tumori 2019, 105, 47–54. [Google Scholar] [CrossRef]
- Roa, W.; Kepka, L.; Kumar, N.; Sinaika, V.; Matiello, J.; Lomidze, D.; Hentati, D.; Guedes de Castro, D.; Dyttus-Cebulok, K.; Drodge, S.; et al. International Atomic Energy Agency Randomized Phase III Study of Radiation Therapy in Elderly and/or Frail Patients With Newly Diagnosed Glioblastoma Multiforme. J. Clin. Oncol. 2015, 33, 4145–4150. [Google Scholar] [CrossRef] [PubMed]
- de Castro, D.G.; Matiello, J.; Roa, W.; Ghosh, S.; Kepka, L.; Kumar, N.; Sinaika, V.; Lomidze, D.; Hentati, D.; Rosenblatt, E.; et al. Survival Outcomes With Short-Course Radiation Therapy in Elderly Patients With Glioblastoma: Data From a Randomized Phase 3 Trial. Int. J. Radiat. Oncol. Biol. Phys. 2017, 98, 931–938. [Google Scholar] [CrossRef]
- Hulshof, M.C.; Schimmel, E.C.; Bosch, D.A.; González, D.G. Hypofractionation in glioblastoma multiforme. Radiother. Oncol. 2000, 54, 143–148. [Google Scholar] [CrossRef]
- Minniti, G.; De Sanctis, V.; Muni, R.; Rasio, D.; Lanzetta, G.; Bozzao, A.; Osti, M.F.; Salvati, M.; Valeriani, M.; Cantore, G.P.; et al. Hypofractionated radiotherapy followed by adjuvant chemotherapy with temozolomide in elderly patients with glioblastoma. J. Neurooncol. 2009, 91, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; James, N.; Guerrero, D.; Ashley, S.; Gregor, A.; Brada, M. Hypofractionated radiotherapy as palliative treatment in poor prognosis patients with high grade glioma. Radiother. Oncol. 1994, 33, 113–116. [Google Scholar] [CrossRef]
- Minniti, G.; Scaringi, C.; Lanzetta, G.; Terrenato, I.; Esposito, V.; Arcella, A.; Pace, A.; Giangaspero, F.; Bozzao, A.; Enrici, R.M. Standard (60 Gy) or short-course (40 Gy) irradiation plus concomitant and adjuvant temozolomide for elderly patients with glioblastoma: A propensity-matched analysis. Int. J. Radiat. Oncol. Biol. Phys. 2015, 91, 109–115. [Google Scholar] [CrossRef]
- Pedretti, S.; Masini, L.; Turco, E.; Triggiani, L.; Krengli, M.; Meduri, B.; Pirtoli, L.; Borghetti, P.; Pegurri, L.; Riva, N.; et al. Hypofractionated radiation therapy versus chemotherapy with temozolomide in patients affected by RPA class V and VI glioblastoma: A randomized phase II trial. J. Neurooncol. 2019, 143, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Chen, L.; Lv, S.; Li, Q.; Chen, G.; Luo, W.; Zhou, P.; Li, G. Efficacy of moderately hypofractionated simultaneous integrated boost intensity-modulated radiotherapy combined with temozolomide for the postoperative treatment of glioblastoma multiforme: A single-institution experience. Radiat Oncol. 2019, 14, 104. [Google Scholar] [CrossRef]
- Omuro, A.; Beal, K.; Gutin, P.; Karimi, S.; Correa, D.D.; Kaley, T.J.; DeAngelis, L.M.; Chan, T.A.; Gavrilovic, I.T.; Nolan, C.; et al. Phase II study of bevacizumab, temozolomide, and hypofractionated stereotactic radiotherapy for newly diagnosed glioblastoma. Clin. Cancer Res. 2014, 20, 5023–5031. [Google Scholar] [CrossRef] [Green Version]
- Bauman, S.; Gaspar, L.E.; Fisher, B.F.; Halperin, E.C.; Macdonald, D.R.; Cairncross, J.G. A prospective study of short-course radiotherapy in poor prognosis glioblastoma multiforme. Int. J. Radiat. Oncol. Biol. Phys. 1994, 29, 835–839. [Google Scholar] [CrossRef]
- Curran, W.J., Jr.; Scott, C.B.; Horton, J.; Nelson, J.S.; Weinstein, A.S.; Fischbach, A.J.; Chang, C.H.; Rotman, M.; Asbell, S.O.; Krisch, R.E.; et al. Recursive partitioning analysis of prognostic factors in three Radiation Therapy Oncology Group malignant glioma trials. J. Natl. Cancer Inst. 1993, 85, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; Van Glabbeke, M.; van Oosterom, A.T.; Christian, M.C.; et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl. Cancer Inst. 2000, 92, 205–216. [Google Scholar] [CrossRef] [Green Version]
- Fokas, E.; Wacker, U.; Gross, M.W.; Henzel, M.; Encheva, E.; Engenhart-Cabillic, R. Hypofractionated stereotactic reirradiation of recurrent glioblastomas: A beneficial treatment option after high-dose radiotherapy? Strahlenther Onkol. 2009, 185, 235–240. [Google Scholar] [CrossRef]
- Navarria, P.; Minniti, G.; Clerici, E.; Tomatis, S.; Pinzi, V.; Ciammella, P.; Galaverni, M.; Amelio, D.; Scartoni, D.; Scoccianti, S.; et al. Re-irradiation for recurrent glioma: Outcome evaluation, toxicity and prognostic factors assessment. A multicenter study of the Radiation Oncology Italian Association (AIRO). J. Neurooncol. 2019, 142, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, G.; De Salvo, G.L.; Brandes, A.A.; Eoli, M.; Rudà, R.; Faedi, M.; Lolli, I.; Pace, A.; Daniele, B.; Pasqualetti, F.; et al. Regorafenib compared with lomustine in patients with relapsed glioblastoma (REGOMA): A multicentre, open-label, randomised, controlled, phase 2 trial. Lancet Oncol. 2019, 20, 110–119. [Google Scholar] [CrossRef]
- Fiorentino, A.; Balducci, M.; De Bonis, P.; Chiesa, S.; De Filippo, L.; Mangiola, A.; De Rose, F.; Autorino, R.; Rinaldi, C.; Fersino, S.; et al. Can elderly patients with newly diagnosed glioblastoma be enrolled in radiochemotherapy trials? Am. J. Clin. Oncol. 2015, 38, 23–27. [Google Scholar] [CrossRef]
- Balducci, M.; Fiorentino, A.; De Bonis, P.; Chiesa, S.; Manfrida, S.; D’Agostino, G.R.; Mantini, G.; Frascino, V.; Mattiucci, G.C.; De Bari, B.; et al. Impact of age and co-morbidities in patients with newly diagnosed glioblastoma: A pooled data analysis of three prospective mono-institutional phase II studies. Med. Oncol. 2012, 29, 3478–3483. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, A.; Caivano, R.; Chiumento, C.; Cozzolino, M.; Clemente, S.; Pedicini, P.; Fusco, V. Comorbidity assessment and adjuvant radiochemotherapy in elderly affected by glioblastoma. Med. Oncol. 2012, 29, 3467–3471. [Google Scholar] [CrossRef] [PubMed]
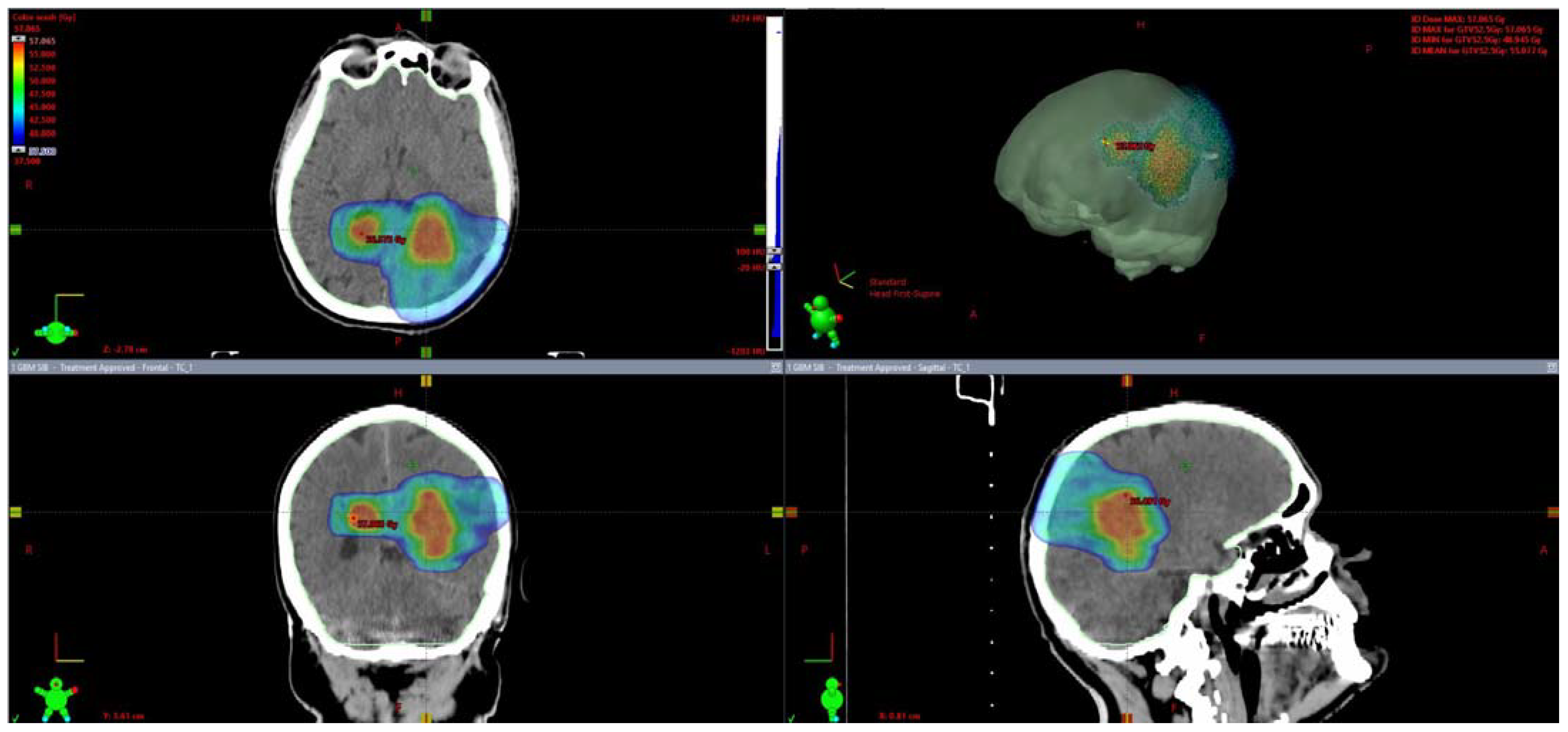
| Number of Patients | 25 | ||
| Sex | |||
| Male | 18 | (72%) | |
| Female | 7 | (28%) | |
| Age | |||
| Median (range) in years | 65 | (37–82) | |
| <60 years | 5 | (20%) | |
| 60–65 years | 11 | (44%) | |
| >65 years | 9 | (36%) | |
| KPS Score | |||
| Median (range), in % | 60 | (50–90) | |
| <60% | 5 | (20%) | |
| 60–70% | 16 | (64%) | |
| >70% | 4 | (16%) | |
| RPA Class | |||
| Median (range) | V | (IV–VI) | |
| IV | 7 | (28%) | |
| V | 10 | (40%) | |
| VI | 8 | (32%) | |
| CCI | |||
| Median (range) | 8 | (5–14) | |
| <7 | 6 | (24%) | |
| 7–9 | 18 | (72%) | |
| >9 | 2 | (4%) | |
| Mass Effect | |||
| Yes | 15 | (60%) | |
| No | 10 | (40%) | |
| Surgery | |||
| Complete | 3 | (12%) | |
| Incomplete | 12 | (48%) | |
| Unresectable (biopsy) | 10 | (40%) | |
| Multifocal Tumor | |||
| Yes | 7 | (28%) | |
| No | 18 | (72%) | |
| Poor Molecular Factors | |||
| wild-type IDH | 25 | (100%) | |
| Unmethylated MGMT | 19 | (76%) | |
| Gross Tumor Volume | |||
| Median (range), in cc | 50 | (31–135) | |
| ≤50 cc | 16 | (64%) | |
| >50 cc | 9 | (36%) | |
| Planning Target Volume | |||
| Median (range), in cc | 220 | (117–358) | |
| ≤200 cc | 12 | (48%) | |
| >200 cc | 13 | (52%) |
| Variable | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | p | HR | 95%CI | p | |
| Sex | 1.23 | 0.3–2.3 | 0.7 | - | - | - |
| Age (≥65 years) | 1.622 | 0.48–5.38 | 0.04 | 2.88 | 0.72–11.43 | 0.1 |
| KPS (≤60%) | 0.42 | 0.05–3.32 | 0.4 | - | - | - |
| RPA (≥ V) | 1.78 | 0.37–8.38 | 0.04 | 0.19 | 0.01–2.78 | 0.2 |
| CCI (≥8) | 1.201 | 0.34–4.17 | 0.7 | - | - | - |
| Mass effect (yes) | 1.51 | 0.39–5.81 | 0.5 | - | - | - |
| Multifocal tumor (yes) | 1.92 | 0.56–6.61 | 0.02 | 2.95 | 0.51–16.98 | 0.2 |
| Surgery (yes) | 0.82 | 0.24–2.73 | 0.7 | - | - | - |
| Resection (incomplete) | 2.29 | 0.04–6.43 | 0.01 | 0.96 | 0.16–5.78 | 0.9 |
| MGMT methylation (absent) | 0.25 | 0.03–2.02 | 0.04 | 0.61 | 0.02–13.66 | 0.05 |
| GTV (>50 cc) | 5.208 | 1.37–19.71 | 0.01 | 4.83 | 0.83–27.88 | 0.01 |
| PTV (>200 cc) | 0.29 | 0.08–1.06 | 0.06 | 2.14 | 0.01–9.87 | 0.02 |
| Variable | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| Sex | 0.81 | 0.27–2.39 | 0.7 | - | - | - |
| Age (≥65 years) | 0.77 | 0.28–2.14 | 0.6 | - | - | - |
| KPS (≤60%) | 0.89 | 0.25–3.17 | 0.8 | - | - | - |
| RPA (≥ V) | 0.83 | 0.28–2.48 | 0.7 | - | - | - |
| CCI (≥8) | 0.95 | 0.35–2.54 | 0.9 | - | - | - |
| Mass effect (yes) | 1.47 | 0.05–4.23 | 0.04 | 1.32 | 0.29–5.97 | 0.7 |
| Multifocal tumor (yes) | 1.38 | 0.04–6.72 | 0.04 | 1.76 | 0.31–8.12 | 0.05 |
| Surgery (yes) | 1.76 | 0.02–3.04 | 0.05 | 1.32 | o.18–3.27 | 0.7 |
| Resection (incomplete) | 2.96 | 0.3–22.42 | 0.02 | 2.91 | 0.31–27.07 | 0.01 |
| MGMT methylation (absent) | 1.99 | 0.04–4.06 | 0.06 | 0.79 | 0.11–6.25 | 0.03 |
| GTV (>50 cc) | 0.82 | 0.31–2.24 | 0.7 | - | - | - |
| PTV (>200 cc) | 1.95 | 0.04–3.74 | 0.06 | 1.99 | 0.26–5.37 | 0.05 |
| Author/Publication Year | Study Years | Study Type | Patient Selection | Comparison | No. of Patients | RT Schedule | Median PFS | Median OS | Toxicities |
|---|---|---|---|---|---|---|---|---|---|
| Phillips et al. 2003 [9] | 1990–1996 | Randomized Phase III | Age > 45 y ECOG 0–3 | Hypo-RT Standard RT | 32 36 | 35 Gy/10 fx (WB) 60 Gy/30 fx | NS | 8.7 months 10.3 months | None |
| Roa et al. 2004 [11] | 1996–2001 | Randomized Phase III | Age ≥ 60 y KPS ≥ 50 | Hypo-RT Standard RT | 48 47 | 40 Gy/15 fx 60 Gy/30 fx | NS | 5.6 months 5.1 months | NS |
| Malmström et al. 2012 [12] | 2000–2009 | Randomized Phase III | Age ≥ 60 y ECOG 0–2 | TMZ Hypo-RT Standard RT | 93 98 100 | NA 34 Gy/10 fx 60 Gy/30 fx | NS | 8.3 months 7.5 months 6 months | No G > 3 acute toxicity |
| Roa et al. 2015 [18] | 2010–2013 | Randomized Phase III | Age ≥ 65 y KPS 50–70 | Hypo-RT Hypo-RT | 48 50 | 25 Gy/5 fx 40 Gy/15 fx | 4.2 months 4.2 months | 7.9 months 6.4 months | No G > 3 acute toxicity |
| Guedes de Castro et al. 2017 [19] | NS | Randomized Phase III | Age ≥ 65 y KPS 50–70 | Hypo-RT Hypo-RT | 26 35 | 25 Gy/5 fx 40 Gy/15 fx | 4.3 months 3.2 months | 6.8 months 6.2 months | No G > 3 acute toxicity |
| Perry et al. 2017 [16] | 2007–2013 | Randomized Phase III | Age ≥ 65 y ECOG 0–2 | Hypo-RT + TMZ Hypo-RT | 281 281 | 40 Gy/15 fx | 5.3 months 3.9 months | 9.3 months 7.6 months | No G > 3 acute toxicity |
| Pedretti et al. 2019 [24] | 2010–2015 | Randomized Phase II | RPA Class 5 or 6 | Hypo-RT alone TMZ alone | 14 17 | 30 Gy/6 fx over 2 weeks | 3.8 months | 6.3 months | No G > 3 acute toxicity |
| Bauman et al. 1994 [27] | 1990–1992 | Prospective | Age ≥ 65 y KPS ≤ 50 | Hypo-RT | 29 | 30 Gy/10 fx (WB) | NS | 6 months | NS |
| Thomas et al. 1994 [29] | 1991–1993 | Prospective | KPS ≤ 50 or Age 55–70 y KPS 50–70 or Age ≥ 70 y | Hypo-RT | 38 | 30 Gy/6 fx over 2 weeks | NS | 6 months | None |
| Hulshof et al. 2000 [19] | 1988–1998 | Prospective | Age ≥ 65 y MRC ≥ 2 | Hypo-RT Hypo-RT Standard RT | 48 41 66 | 28 Gy/4 fx 40 Gy/8 fx 66 Gy/33 fx | NS | 6.6 months 5.6 months 7 months | Mild; No difference between groups |
| Minniti et al. 2009 [21] | 2002–2006 | Prospective | Age ≥ 70 y KPS ≥ 60 | Hypo-RT + adj TMZ | 43 | 30 Gy/6 fx over 2 weeks | 6.3 months | 9.3 months | 8 patients presented neurological deterioration (Grade 2/3 confusion and/or somnolence). 12 patients had Grade 3/4 hematological toxic effects |
| Omuro et al. 2014 [26] | NS | Prospective | Age ≥ 18 years (median 55 y) KPS ≥ 70 (median 90) Partial resection or biopsy (75%) | Hypo-RT + TMZ + BEV | 40 | 30 Gy/6 fx over 2 weeks | 10 months | 19 months | None |
| Navarria et al. 2019 [17] | 2013–2016 | Prospective | Age ≥ 70 y KPS ≤ 60 | Hypo-RT | 30 | 52.5 Gy/15 fx | 5 months | 8 months | No severe acute or late neurologic toxicity was recorded |
| McAleese et al. 2003 [8] | 1991–1999 | Retrospective | KPS ≤ 50 or Age 50–70 y KPS 50–90 or Age ≥ 70 y | Hypo-RT | 92 | 30 Gy/6 fx over 2 weeks | NS | 5 months | NS |
| Chang et al. 2003 [10] | 1988–2001 | Retrospective | RPA Class ≥ 4 | Hypo-RT | 59 | 50 Gy/20 fx | 3.9 months | 7 months | 3 patients showed radio-necrosis |
| Minniti el al. 2015 [23] | 2004–2013 | Retrospective | Age ≥ 65 y KPS ≥ 60 | Hypo-RT + TMZ Standard RT + TMZ | 116 127 | 40 Gy/15 fx 59.4–60 Gy/30–33 fx | 6.7 months 5.6 months | 12.5 months 12 months | 28 patients receiving standard RT and 11 subjected to short-course RT had acute worsening of neurologic status. 20 patients receiving standard RT and 3 patients receiving short-course RT had late neurologic deterioration (G2–3 cognitive disability) G3–4 thrombocytopenia and lymphocytopenia were seen in 24 patients and 51 patients. G3 neutropenia developed in 14 patients, and 10 patients displayed G3 anemia |
| Jablonska et al. 2019 [15] | 2010–2017 | Retrospective | RPA Class ≥ 4 | Hypo-RT with SIB + TMZ | 17 | 50–45–40 Gy/15 fx | 7 months | 7 months | No acute G3–5 toxicities were observed. Radio-necrosis occurred in 1 patient. |
| Present study | 2019–2021 | Retrospective | Poor prognosis RPA Class ≥ 4 | Hypo-RT with SIB + TMZ | 25 | 52.5–40 Gy/15 fx | 8.4 months | 13 months | No acute or late neurological side effects of grade ≥ 2 were reported. No cases of radio-necrosis. Grade 3–4 hematologic toxicity occurred in 3 cases. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gregucci, F.; Surgo, A.; Bonaparte, I.; Laera, L.; Ciliberti, M.P.; Carbonara, R.; Gentile, M.A.; Giraldi, D.; Calbi, R.; Caliandro, M.; et al. Poor-Prognosis Patients Affected by Glioblastoma: Retrospective Study of Hypofractionated Radiotherapy with Simultaneous Integrated Boost and Concurrent/Adjuvant Temozolomide. J. Pers. Med. 2021, 11, 1145. https://doi.org/10.3390/jpm11111145
Gregucci F, Surgo A, Bonaparte I, Laera L, Ciliberti MP, Carbonara R, Gentile MA, Giraldi D, Calbi R, Caliandro M, et al. Poor-Prognosis Patients Affected by Glioblastoma: Retrospective Study of Hypofractionated Radiotherapy with Simultaneous Integrated Boost and Concurrent/Adjuvant Temozolomide. Journal of Personalized Medicine. 2021; 11(11):1145. https://doi.org/10.3390/jpm11111145
Chicago/Turabian StyleGregucci, Fabiana, Alessia Surgo, Ilaria Bonaparte, Letizia Laera, Maria Paola Ciliberti, Roberta Carbonara, Maria Annunziata Gentile, David Giraldi, Roberto Calbi, Morena Caliandro, and et al. 2021. "Poor-Prognosis Patients Affected by Glioblastoma: Retrospective Study of Hypofractionated Radiotherapy with Simultaneous Integrated Boost and Concurrent/Adjuvant Temozolomide" Journal of Personalized Medicine 11, no. 11: 1145. https://doi.org/10.3390/jpm11111145
APA StyleGregucci, F., Surgo, A., Bonaparte, I., Laera, L., Ciliberti, M. P., Carbonara, R., Gentile, M. A., Giraldi, D., Calbi, R., Caliandro, M., Sasso, N., D’Oria, S., Somma, C., Martinelli, G., Surico, G., Lombardi, G., & Fiorentino, A. (2021). Poor-Prognosis Patients Affected by Glioblastoma: Retrospective Study of Hypofractionated Radiotherapy with Simultaneous Integrated Boost and Concurrent/Adjuvant Temozolomide. Journal of Personalized Medicine, 11(11), 1145. https://doi.org/10.3390/jpm11111145











