Tract Sealing Techniques for Pneumothorax and Drainage Prevention After CT-Guided Lung Biopsy: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
- Population: Individuals who underwent CLB. Studies reporting CT-guided pulmonary fiducial marker placement were also included, as the procedural technique closely parallels that of biopsy;
- Intervention: Injection of any substance into the lung parenchyma during withdrawal of the coaxial guide, with the purpose of preventing pneumothorax;
- Comparator: Patients in whom no sealing agent was administered following biopsy; patients who received a different sealing agent for pneumothorax prevention;
- Outcomes: pneumothorax and/or pleural drainage insertion.
- Conference abstracts, unpublished manuscripts, reviews, case reports, editorials, and comments;
- Studies including fluoroscopy- or ultrasound-guided biopsies;
- Studies including ablation procedures;
- Studies including the pleural patch technique;
- Studies with no comparator group;
- Duplicate or overlapping patient populations.
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CLB | CT-guided lung biopsy. |
| OR | Odds ratio. |
| CI | Confidence interval. |
| RR | Rapid roll-over. |
Appendix A. Search Strategy
- ScienceDirect (via Elsevier)—multiple keyword combinations were used to run separate searches for each biological agent:
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Blood Patch”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Saline”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Gelfoam”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Hydrogel”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Gelatin”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Plug”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Biosentry”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Collagen”
- −
- (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND “Biopsy” AND (“Lung” OR “Pulmonary”) AND “Glue”
- Scopus (via Elsevier)—(TITLE-ABS-KEY (“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND TITLE-ABS-KEY (“Biopsy” OR “Intervention”) AND TITLE-ABS-KEY (“Lung” OR “Pulmonary”) AND TITLE-ABS-KEY (“Blood Patch” OR “Saline” OR “Gelfoam” OR “Hydrogel” OR “Gelatin” OR “Plug” OR “Sealing” OR “Biosentry” OR “Foam” OR “Polymer” OR “Composite” OR “Composites” OR “Glue” OR “Collagen”) AND (LIMIT-TO (LANGUAGE, “English”)) AND (LIMIT-TO (EXACTKEYWORD, “Observational Study”) OR LIMIT-TO (EXACTKEYWORD, “Randomized Controlled Trial (topic)”) OR LIMIT-TO (EXACTKEYWORD, “Cohort Analysis”) OR LIMIT-TO (EXACTKEYWORD, “Controlled Study”) OR LIMIT-TO (EXACTKEYWORD, “Follow-Up Studies”) OR LIMIT-TO (EXACTKEYWORD, “Prospective Studies”) OR LIMIT-TO (EXACTKEYWORD, “Prospective Study”) OR LIMIT-TO (EXACTKEYWORD, “Randomized Controlled Trial”) OR LIMIT-TO (EXACTKEYWORD, “Retrospective Studies”) OR LIMIT-TO (EXACTKEYWORD, “Retrospective Study”))
- Cochrane Library (via Wiley)—(“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND (“Biopsy” OR “Intervention”) AND (“Lung” OR “Pulmonary”) AND (“Blood Patch” OR “Saline” OR “Gelfoam” OR “Hydrogel” OR “Gelatin” OR “Plug” OR “Sealing” OR “Biosentry” OR “Foam” OR “Polymer” OR “Composite” OR “Composites” OR “Glue” OR “Collagen”)
- Pubmed (via NCBI)—(“Pneumothorax” OR “Complication” OR “Complications” OR “Drainage” OR “Tube”) AND (“Biopsy” OR “Intervention”) AND (“Lung” OR “Pulmonary”) AND (“Blood Patch” OR “Saline” OR “Gelfoam” OR “Hydrogel” OR “Gelatin” OR “Plug” OR “Sealing” OR “Biosentry” OR “Foam” OR “Polymer” OR “Composite” OR “Composites” OR “Glue” OR “Collagen”).
Appendix B
| Author | Year | Conesecutive—Only to Cohort Studies | Study Design | Sealant | Country | Rapid Roll-Over | Number of Patients | Male/Female (n) | Age (y)—Mean | Emphysema | Lesion Size (mm)—Mean | Guide and Biopsy Needle Gauge | Diagnosis of Pneumothorax (h) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sealant | Control | Sealant | Control | Sealant | Control | Sealant | Control | Sealant | Control | Sealant | Control | ||||||||
| Babu [24] | 2020 | Y | cohort | saline (1–3 mL) | Singapore | no | no | 100 | 100 | - | - | 64.8 | 68.3 | 16 | 18 | 39 | 37 | 16/18G or 19/20G | 0 h, 4 h |
| Li [17] | 2015 | - | RCT | saline (1–3 mL) | China | no | no | 161 | 161 | 107/54 | 111/50 | 57.3 | 58.2 | 41 | 37 | 32 | 33 | 19/20G | 0 h, 6 h |
| Billich [25] | 2008 | N | cohort | saline (2–4 mL) | Germany | no | no | 70 | 70 | 52/18 | 49/21 | 63.64 | 62.76 | 3 | 1 | 30.1 | 29.4 | 16/18G | 0 h, 2 h |
| Bourgeais [26] | 2024 | N | cohort | saline (3–5 mL) | France | no | no | 93 | 149 | 64/29 | 97/52 | 70 | 69 | 51 | 82 | 30 | 25 | 17/18G or 19/20G | 0 h |
| Zhang [42] | 2025 | Y | cohort | saline (3–5 mL) | China | no | no | 95 | 132 | 52/43 | 81/51 | 65.7 | 67.5 | 2 | 3 | 28.3 | 29.5 | 17/18G | 0 h, 4 h |
| Satomura [37] | 2024 | Y | cohort | saline (1–5 mL) | Japan | yes | no | 173 | 130 | 105/68 | 84/46 | 71.5 | 69.7 | 80 | 61 | 19.2 | 25.7 | 17/18G | 0 h, 2 h |
| Tangobay [49] | 2025 | N | cohort | saline (5–10 mL) | Turkey | no | no | 74 | 52 | 143/42 | 64.55 | - | - | 32 | 17/18G or 19/20G or 22G | 0 h, 4 h | |||
| Khorochkov [34] | 2018 | Y | cohort | saline (3–5 mL) | Canada | yes | no | 180 | 98 | 82/98 | 39/59 | 71 | 68 | - | - | - | - | 19/20G | 2 h |
| Roman [43] | 2023 | Y | cohort | saline (3–5 mL) | Romania | no | no | 87 | 111 | 40/47 | 69/42 | 63.4 | 63.7 | 31 | 31 | 29.2 | 32.7 | 17/18G | 0 h, 2 h |
| Grage [31] | 2017 | Y | cohort | hydrogel plug | USA | no | no | 100 | 100 | 46/54 | 57/43 | 66.92 | 62.92 | 83 | 86 | 19.7 | 23.3 | 19/20G | 0 h, 2 h |
| Zaetta [16] | 2010 | - | RCT | hydrogel plug (BioSeal) | USA | no | no | 170 | 169 | 78/92 | 94/75 | 66.1 | 67.2 | - | - | under 30 | 19G | 0 h, 24 h, 30 day | |
| Aboudodah [21] | 2021 | N | cohort | hydrogel plug (Biosentry) | USA | no | no | 74 | 447 | 36/38 | 234/213 | 67.3 | 65.4 | 1 | 3 | - | - | - | 0 h, 24 h |
| Ahrar [22] | 2017 | N | cohort | hydrogel plug (Biosentry) | USA | no | no | 317 | 317 | 156/161 | 165/152 | 63.3 | 63.4 | 83 | 87 | 19.8 | 21.2 | 19/20G or 19/22G | 0 h, 3 h |
| Naser-Tavakolian [33] | 2024 | N | cohort | hydrogel plug (Biosentry) | USA | no | no | 125 | 147 | 38/87 | 69/78 | 70.05 | 68.95 | 59 | 44 | 19.2 | 26.3 | 17/18G | 0 h, 1 h |
| Renier [36] | 2020 | Y | cohort | gelatin sponge (Gelfoam) | Belgium | yes | yes | 231 | 213 | 129/102 | 110/103 | 65.7 | 65.1 | 46 | 43 | 23.8 | 25.6 | 19/20G | 0 h, 4 h |
| Sum [38] | 2021 | Y | cohort | gelatin sponge (Gelfoam) | Australia | no | no | 170 | 126 | 67/59 | 94/76 | 69 | 71 | 55 | 42 | - | - | 19/20G | 0 h, 4 h |
| Yang [41] | 2024 | Y | cohort | gelatin sponge (Gelfoam) | China | no | no | 504 | 523 | 359/145 | 170/153 | 68.21 | 67.72 | 212 | 219 | 30.1 | 31.51 | 18/20G | 0 h |
| Salama [15] | 2023 | - | RCT | gelatin sponge (Gelfoam) | Egypt | no | no | 70 | 68 | 55/15 | 50/18 | 60.3 | 55.2 | 32 | 27 | - | - | 17/18G | 0 h, 1 h, 3 h |
| Zhao [46] | 2025 | N | cohort | gelatin sponge (Gelfoam) | China | no | no | 274 | 274 | 137/137 | 136/138 | 62.19 | 62.26 | 56 | 59 | 44.14 | 44.82 | 17/18G | 0 h |
| Zhu [48] | 2025 | N | cohort | gelatin sponge (Gelfoam) | China | no | no | 169 | 169 | 107/62 | 112/57 | 66.9 | 67.3 | 62 | 58 | 32 | 32 | 17/18G | 0 h |
| Grange [44] | 2023 | N | cohort | gelatin sponge (Gelitaspon) | France | no | no | 419 | 419 | 63/42 | 65/35 | 66.2 | 66.7 | 191 | 174 | 25.6 | 26.6 | 18G or 19G or 20G | 0 h, 4 h |
| Wang [45] | 2023 | Y | cohort | gelatin sponge (Jiangxi Xiangen Medical Technology Devel- opment Co.) + haemocoagulase | China | no | no | 626 | 681 | 331/295 | 389/292 | 63.28 | 63 | - | - | 33.67 | 34.9 | 17/18G | 0 h, 3 h |
| Feinggumloon [29] | 2024 | N | cohort | gelatin sponge (Spongostan) | Thailand | no | no | 217 | 217 | 97/120 | 97/120 | 64.9 | 64.4 | 12 | 11 | 32.9 | 28.9 | 16G or 17G | 0 h, 4 h |
| Baadh [23] | 2016 | Y | cohort | gelatin sponge (Surgifoam) | USA | no | no | 125 | 124 | 49/76 | 66/58 | 72.4 | 79.4 | - | - | 29 | 30 | 17/18G or 19/20G | 0 h, 1 h |
| Tran [39] | 2014 | N | cohort | gelatin sponge (Surgifoam) | USA | no | no | 145 | 607 | 81/64 | 304/303 | 68.26 | 63,51 | 19 | 66 | 30.74 | 28.86 | 19/21G or19/20G | 0 h, 4 h, 6 h |
| Jain [32] | 2022 | Y | cohort | ABP clotted | USA | no | no | 309 | 227 | 146/163 | 107/120 | 69 | 68 | - | - | - | - | 19/20G or 17/20G | 0 h |
| Lang [13] | 2000 | - | RCT | ABP clotted | USA | no | no | 50 | 50 | 63/37 | 54 | 48 | 21 | 17 | 20 | 24 | 19G | 0 h, 4 h | |
| Malone [14] | 2013 | - | RCT | ABP clotted | USA | no | no | 123 | 119 | 52/71 | 60/59 | 65 | 66 | 34 | 37 | 2.2 | 2.3 | 17/18G or 19/20G | 1 h, 3 h |
| Clayton [27] | 2016 | Y | cohort | ABP nonclotted | USA | no | no | 245 | 189 | 125/64 | 147/98 | 67 | 66 | 115 | 93 | 23 | 19/20G or 19/22G | 0 h, 2 h | |
| Duignan [28] | 2023 | N | cohort | ABP nonclotted | Ireland | yes | yes | 259 | 393 | 109/150 | 184/209 | 68.4 | 68.1 | 48 | 21 | 24 | 28 | 19/20G | 1 h, 4 h |
| Graffy [30] | 2017 | Y | cohort | ABP nonclotted | USA | yes | yes | 482 | 352 | 278/204 | 206/146 | 66.2 | 63.4 | 200 | 146 | - | - | 19/20G or 19/21G | 1 h |
| Perl [35] | 2019 | Y | cohort | ABP nonclotted | Germany | no | no | 419 | 449 | - | - | - | - | - | - | - | - | 13G or 15G or 17G or 19G | 2 h |
| Tangobay [49] | 2025 | N | cohort | ABP nonclotted | Turkey | no | no | 59 | 52 | 143/42 | 64.55 | - | - | 32 | 17/18G or 19/20G or 22G | 0 h, 4 h | |||
| Turgut [40] | 2020 | Y | cohort | ABP nonclotted | Turkey | no | no | 91 | 171 | 80/11 | 153/18 | 62.6 | 59.3 | 8 | 12 | 32 | 30 | 19/20G | 0 h, 6 h |
| Petsas [18] | 1995 | - | RCT | fibrin glue (Tissucol 0.5) | Greece | yes | yes | 26 | 32 | 41/17 | - | - | - | - | - | - | 19G/22G | 0 h, 1 h, 4 h, 24 h | |
| Zhou [47] | 2022 | N | cohort | haemocoagulase | China | no | no | 50 | 50 | 44/56 | 60 | 64 | - | - | 27 | 28 | 17/18G | 0 h | |
| Studies comparing two different sealing agents | Sealant no.1 | Sealant no.2 | Sealant no.1 | Sealant no.2 | Sealant no.1 | Sealant no.2 | Sealant no.1 | Sealant no.2 | Sealant no.1 | Sealant no.2 | |||||||||
| Maybody [19] | 2019 | - | RCT | ABP (no.1) vs. hydrogel - Biosentry (no.2) | USA | no | no | 179 | 173 | - | - | - | - | - | - | - | - | - | 2 h |
| Dheur [20] | 2024 | - | RCT | saline (no.1) vs. gelatin sponge (no.2) | France | no | no | 134 | 132 | 78/56 | 72/60 | 65.7 | 67.2 | 76 | 67 | 18 | 18 | 19/20G | 0 h, 4 h |
Appendix C. Risk of Bias
References
- Huo, Y.R.; Chan, M.V.; Habib, A.-R.; Lui, I.; Ridley, L. Pneumothorax rates in CT-Guided lung biopsies: A comprehensive systematic review and meta-analysis of risk factors. Br. J. Radiol. 2020, 93, 20190866. [Google Scholar] [CrossRef]
- Heerink, W.J.; de Bock, G.H.; de Jonge, G.J.; Groen, H.J.; Vliegenthart, R.; Oudkerk, M. Complication rates of CT-guided transthoracic lung biopsy: Meta-analysis. Eur. Radiol. 2017, 27, 138–148. [Google Scholar] [CrossRef]
- Değirmenci, M. Morbidity, mortality, and surgical treatment of secondary spontaneous pneumothorax. Ulus. Travma Acil Cerrahi Derg. 2023, 29, 909–919. [Google Scholar] [CrossRef]
- Huo, Y.R.; Chan, M.V.; Habib, A.-R.; Lui, I.; Ridley, L. Post-Biopsy Manoeuvres to Reduce Pneumothorax Incidence in CT-Guided Transthoracic Lung Biopsies: A Systematic Review and Meta-analysis. Cardiovasc. Interv. Radiol. 2019, 42, 1062–1072. [Google Scholar] [CrossRef]
- Wagner, J.M.; Hinshaw, J.L.; Lubner, M.G.; Robbins, J.B.; Kim, D.H.; Pickhardt, P.J.; Lee, F.T., Jr. CT-guided lung biopsies: Pleural blood patching reduces the rate of chest tube placement for postbiopsy pneumothorax. AJR Am. J. Roentgenol. 2011, 197, 783–788. [Google Scholar] [CrossRef]
- Li, T.; Zhang, Q.; Li, W.; Liu, Y. Autologous blood patch intraparenchymal injection reduces the incidence of pneumothorax and the need for chest tube placement following CT-guided lung biopsy: A systematic review and meta-analysis. Eur. J. Med. Res. 2024, 29, 108. [Google Scholar] [CrossRef]
- Singh, P.; Nayak, M.K.; Singhal, T.; Parida, G.K.; Agrawal, K.; Sahoo, B.; Deep, N. Efficacy of needle tract embolization using gelfoam in reducing incidence of pneumothorax in CT-guided transthoracic lung biopsies: A systematic review and meta-analysis. Br. J. Radiol. 2025, 98, 194–200. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Page, M.; Higgins, J.; Sterne, J. Chapter 13: Assessing risk of bias due to missing evidence in a meta-analysis. In Cochrane Handbook for Systematic Reviews of Interventions Version; John Wiley & Sons: Hoboken, NJ, USA, 2024; p. 6. [Google Scholar]
- McMaster University; Evidence Prime. GRADEpro GDT: GRADEpro Guideline Development Tool; McMaster University: Hamilton, ON, Canada; Evidence Prime: Kraków, Poland, 2022. [Google Scholar]
- Lang, E.K.; Ghavami, R.; Schreiner, V.C.; Archibald, S.; Ramirez, J. Autologous blood clot seal to prevent pneumothorax at CT-guided lung biopsy. Radiology 2000, 216, 93–96. [Google Scholar] [CrossRef]
- Malone, L.J.; Stanfill, R.M.; Wang, H.; Fahey, K.M.; Bertino, R.E. Effect of intraparenchymal blood patch on rates of pneumothorax and pneumothorax requiring chest tube placement after percutaneous lung biopsy. AJR Am. J. Roentgenol. 2013, 200, 1238–1243. [Google Scholar] [CrossRef]
- Salama, N.M.; Tabashy, R.H.; Mahmoud, I.H.; Rahman, A.E.R.M.A.E.; Mohamed, D.N.E.; Kassas, H.E. Does Gelfoam slurry embolization post-pulmonary biopsy reduce risk of pneumothorax? A prospective randomized control study. Egypt. J. Radiol. Nucl. Med. 2023, 54, 4. [Google Scholar] [CrossRef]
- Zaetta, J.M.; Licht, M.O.; Fisher, J.S.; Avelar, R.L.; Bio-Seal Study, G. A lung biopsy tract plug for reduction of postbiopsy pneumothorax and other complications: Results of a prospective, multicenter, randomized, controlled clinical study. J. Vasc. Interv. Radiol. 2010, 21, 1235–1243.e3. [Google Scholar] [CrossRef]
- Li, Y.; Du, Y.; Luo, T.Y.; Yang, H.F.; Yu, J.H.; Xu, X.X.; Zheng, H.J.; Li, B. Usefulness of normal saline for sealing the needle track after CT-guided lung biopsy. Clin. Radiol. 2015, 70, 1192–1197. [Google Scholar] [CrossRef]
- Petsas, T.; Siamblis, D.; Giannakenas, C.; Tepetes, K.; Dougenis, D.; Spiropoulos, K.; Fezoulidis, I.; Dimopoulos, I. Fibrin glue for sealing the needle track in fine-needle percutaneous lung biopsy using a coaxial system: Part II—Clinical study. Cardiovasc. Interv. Radiol. 1995, 18, 378–382. [Google Scholar] [CrossRef]
- Maybody, M.; Muallem, N.; Brown, K.T.; Moskowitz, C.S.; Hsu, M.; Zenobi, C.L.; Jihad, M.; Getrajdman, G.I.; Sofocleous, C.T.; Erinjeri, J.P.; et al. Autologous Blood Patch Injection versus Hydrogel Plug in CT-guided Lung Biopsy: A Prospective Randomized Trial. Radiology 2019, 290, 547–554. [Google Scholar] [CrossRef]
- Dheur, S.; Gerard, L.; Lamborelle, P.; Valkenborgh, C.; Grandjean, F.; Gillard, R.; Dardenne, N.; Hustinx, R.; Cousin, F. Track Sealing in CT-Guided Lung Biopsy Using Gelatin Sponge Slurry versus Saline in Reducing Postbiopsy Pneumothorax: A Prospective Randomized Study. J. Vasc. Interv. Radiol. 2024, 35, 1687–1694. [Google Scholar] [CrossRef]
- Abouodah, H.; Werner, G.; Fahrbach, T.M.; Fox, C.; Mazurek, J.; Lott, J.; Walter, C.N.; Clark, L.; Custer, B. Post-Biopsy Pneumothorax Incidence in Patients Treated with Biosentry Plug Device. Kans. J. Med. 2021, 14, 153–155. [Google Scholar] [CrossRef]
- Ahrar, J.U.; Gupta, S.; Ensor, J.E.; Mahvash, A.; Sabir, S.H.; Steele, J.R.; McRae, S.E.; Avritscher, R.; Huang, S.Y.; Odisio, B.C.; et al. Efficacy of a Self-expanding Tract Sealant Device in the Reduction of Pneumothorax and Chest Tube Placement Rates After Percutaneous Lung Biopsy: A Matched Controlled Study Using Propensity Score Analysis. Cardiovasc. Interv. Radiol. 2017, 40, 270–276. [Google Scholar] [CrossRef]
- Baadh, A.S.; Hoffmann, J.C.; Fadl, A.; Danda, D.; Bhat, V.R.; Georgiou, N.; Hon, M. Utilization of the track embolization technique to improve the safety of percutaneous lung biopsy and/or fiducial marker placement. Clin. Imaging 2016, 40, 1023–1028. [Google Scholar] [CrossRef]
- Babu, S.B.; Srinivasan, S.; Chung, R.; Chawla, A.; Tan, H.K.; Lohan, R. Tract sealing with normal saline after percutaneous transthoracic lung biopsies. J. Med. Imaging Radiat. Oncol. 2020, 64, 211–214. [Google Scholar] [CrossRef]
- Billich, C.; Muche, R.; Brenner, G.; Schmidt, S.A.; Kruger, S.; Brambs, H.J.; Pauls, S. CT-guided lung biopsy: Incidence of pneumothorax after instillation of NaCl into the biopsy track. Eur. Radiol. 2008, 18, 1146–1152. [Google Scholar] [CrossRef]
- Bourgeais, G.; Frampas, E.; Liberge, R.; Nicolas, A.; Defrance, C.; Blanc, F.X.; Coudol, S.; Morla, O. Pneumothorax Incidence with Normal Saline Instillation for Sealing the Needle Track After Computed Tomography-Guided Percutaneous Lung Biopsy. Cardiovasc. Interv. Radiol. 2024, 47, 604–612. [Google Scholar] [CrossRef]
- Clayton, J.D.; Elicker, B.M.; Ordovas, K.G.; Kohi, M.P.; Nguyen, J.; Naeger, D.M. Nonclotted Blood Patch Technique Reduces Pneumothorax and Chest Tube Placement Rates After Percutaneous Lung Biopsies. J. Thorac. Imaging 2016, 31, 243–246. [Google Scholar] [CrossRef]
- Duignan, J.A.; Ryan, D.T.; O’Riordan, B.; O’Brien, A.; Healy, G.M.; O’Brien, C.; Butler, M.; Keane, M.P.; McCarthy, C.; Murphy, D.J.; et al. Combined autologous blood patch-immediate patient rollover does not reduce the pneumothorax or chest drain rate following CT-guided lung biopsy compared to immediate patient rollover alone. Eur. J. Radiol. 2023, 160, 110691. [Google Scholar] [CrossRef]
- Feinggumloon, S.; Radchauppanone, P.; Panpikoon, T.; Buangam, C.; Pichitpichatkul, K.; Treesit, T. Reduced Incidence of Pneumothorax and Chest Tube Placement following Transthoracic CT-Guided Lung Biopsy with Gelatin Sponge Torpedo Track Embolization: A Propensity Score-Matched Study. J. Clin. Med. 2024, 13, 4666. [Google Scholar] [CrossRef]
- Graffy, P.; Loomis, S.B.; Pickhardt, P.J.; Lubner, M.G.; Kitchin, D.R.; Lee, F.T., Jr.; Hinshaw, J.L. Pulmonary Intraparenchymal Blood Patching Decreases the Rate of Pneumothorax-Related Complications following Percutaneous CT-Guided Needle Biopsy. J. Vasc. Interv. Radiol. 2017, 28, 608–613.e1. [Google Scholar] [CrossRef]
- Grage, R.A.; Naveed, M.A.; Keogh, S.; Wang, D. Efficacy of a Dehydrated Hydrogel Plug to Reduce Complications Associated With Computed Tomography-guided Percutaneous Transthoracic Needle Biopsy. J. Thorac. Imaging 2017, 32, 57–62. [Google Scholar] [CrossRef]
- Jain, N.; Zurla, Z.; Crowley, S.; Gupta, N.; Bajaj, S.; Khanna, K. Tract injection of autologous blood (intraparenchymal blood patching) in percutaneous transthoracic CT-guided lung biopsy and the incidence of pneumothorax: A retrospective analysis. Precis. Cancer Med. 2022, 5, 34. [Google Scholar] [CrossRef]
- Naser-Tavakolian, K.; Clifton, M.; Mahmoud, A.; Gupta, A. Is the BioSentry Tract Sealant System Effective in Reducing the Pneumothorax Rate After CT-Guided Transthoracic Lung Biopsy With an 18G Biopsy Needle? Cureus 2024, 16, e71078. [Google Scholar] [CrossRef]
- Khorochkov, E.; Garvin, G.J.; Potoczny, S.; Kozak, R.I. Injection of Saline Into the Biopsy Tract and Rapid Patient Rollover Decreases Pneumothorax Size Following Computed Tomography-Guided Transthoracic Needle Biopsy. Can. Assoc. Radiol. J. 2018, 69, 489–492. [Google Scholar] [CrossRef]
- Perl, R.M.; Risse, E.; Hetzel, J.; Bosmuller, H.; Kloth, C.; Fritz, J.; Horger, M. The effect of intraparenchymal blood patching on the rate of pneumothorax in patients undergoing percutaneous CT-guided core biopsy of the lung. Eur. J. Radiol. 2019, 116, 14–20. [Google Scholar] [CrossRef]
- Renier, H.; Gerard, L.; Lamborelle, P.; Cousin, F. Efficacy of the tract embolization technique with gelatin sponge slurry to reduce pneumothorax and chest tube placement after percutaneous CT-guided lung biopsy. Cardiovasc. Interv. Radiol. 2020, 43, 597–603. [Google Scholar] [CrossRef]
- Satomura, H.; Higashihara, H.; Kimura, Y.; Nakamura, M.; Tanaka, K.; Ono, Y.; Kuriu, A.; Tomiyama, N. Normal saline injection and rapid rollover; preventive effect on incidence of pneumothorax after CT-guided lung biopsy: A retrospective cohort study. BMC Pulm. Med. 2024, 24, 505. [Google Scholar] [CrossRef]
- Sum, R.; Lau, T.; Paul, E.; Lau, K. Gelfoam slurry tract occlusion after computed tomography-guided percutaneous lung biopsy: Does it prevent major pneumothorax? J. Med. Imaging Radiat. Oncol. 2021, 65, 678–685. [Google Scholar] [CrossRef]
- Tran, A.A.; Brown, S.B.; Rosenberg, J.; Hovsepian, D.M. Tract embolization with gelatin sponge slurry for prevention of pneumothorax after percutaneous computed tomography-guided lung biopsy. Cardiovasc. Interv. Radiol. 2014, 37, 1546–1553. [Google Scholar] [CrossRef]
- Turgut, B.; Duran, F.M.; Bakdik, S.; Arslan, S.; Tekin, A.F.; Esme, H. Effectiveness of autologous blood injection in reducing the rate of pneumothorax after percutaneous lung core needle biopsy. Diagn. Interv. Radiol. 2020, 26, 470–475. [Google Scholar] [CrossRef]
- Yang, X.; Cheng, H.T.; Huang, Y.; Guo, Y.; Yuan, H.; Chen, Y.Q.; Li, H.L. Safety and efficacy of tract embolization using gelatin sponge particles in reducing pneumothorax after CT-guided percutaneous lung biopsy in patients with emphysema. BMC Pulm. Med. 2024, 24, 329. [Google Scholar] [CrossRef]
- Zhang, X.; An, J.; Wu, J.; Jing, X.; Lu, H.; Tian, Y. Effect of Saline Sealing After CT-Guided Lung Biopsy on Pneumothorax and Hemoptysis. J. Comput. Assist. Tomogr. 2025, 49, 640–645. [Google Scholar] [CrossRef]
- Roman, A.; Brozba, A.; Necula, A.; Muntean, D.D.; Kubelac, P.; Fekete, Z.; Tomuleasa, C.; Csutak, C.; Feier, D.; Pintican, R.; et al. Usefulness of Saline Sealing in Preventing Pneumothorax after CT-Guided Biopsies of the Lung. Diagnostics 2023, 13, 3546. [Google Scholar] [CrossRef]
- Grange, R.; Di Bisceglie, M.; Habert, P.; Resseguier, N.; Sarkissian, R.; Ferre, M.; Dassa, M.; Grange, S.; Izaaryene, J.; Piana, G. Evaluation of preventive tract embolization with standardized gelatin sponge slurry on chest tube placement rate after CT-guided lung biopsy: A propensity score analysis. Insights Imaging 2023, 14, 212. [Google Scholar] [CrossRef]
- Wang, D.X.; Wang, Y.G.; Ding, G.X.; Li, B.; Liu, R.N.; Ai, Z.W.; Wang, Y. The effectiveness of the puncture channel plugging for reduction of complications after CT-guided percutaneous transthoracic needle biopsy. Sci. Rep. 2023, 13, 12318. [Google Scholar] [CrossRef]
- Zhao, X.H.; Zhu, S.B.; Li, X.; Yang, C.M.; Liu, J.X.; Liu, F.; Xing, W.G.; Li, Y. CT-Guided Lung Biopsy with Needle Embolization Using Gelatin Sponge Slurry to Prevent Pneumothorax and Pulmonary Hemorrhage: A Propensity Score Matching Controlled Study. Acad. Radiol. 2025, 32, 5533–5544. [Google Scholar] [CrossRef]
- Zhou, S.Q.; Luo, F.; Gu, M.; Lu, X.J.; Xu, Y.; Wu, R.N.; Xiong, J.; Ran, X. Biopsy-tract haemocoagulase injection reduces major complications after CT-guided percutaneous transthoracic lung biopsy. Clin. Radiol. 2022, 77, e673–e679. [Google Scholar] [CrossRef]
- Zhu, Z.Y.; Yun, Y.Q.; Li, H.; Qin, Z.Q.; Qian, Z.; Zhu, Y.; Li, S.H.; Xie, B.; Yuan, M. Subpleural injection of gelatin sponge particles to reduce pneumothorax incidence in CT-guided lung biopsies: A retrospective single-center case-control study. BMC Cancer 2025, 25, 529. [Google Scholar] [CrossRef]
- Tangobay, E.; Birgi, E.; Ergun, O.; Öztürk, S.; Uyanık, S.A.; Keskin, B.; Eraslan, Ö.; Durmaz, H.; Hekimoğlu, B. Efficacy of Tract Embolization on Reducing Pneumothorax in CT-Guided Transthoracic Lung Biopsies: Comparison of Saline and Autologous Blood Patch. Acad. Radiol. 2025, 32, 3701–3710. [Google Scholar] [CrossRef]


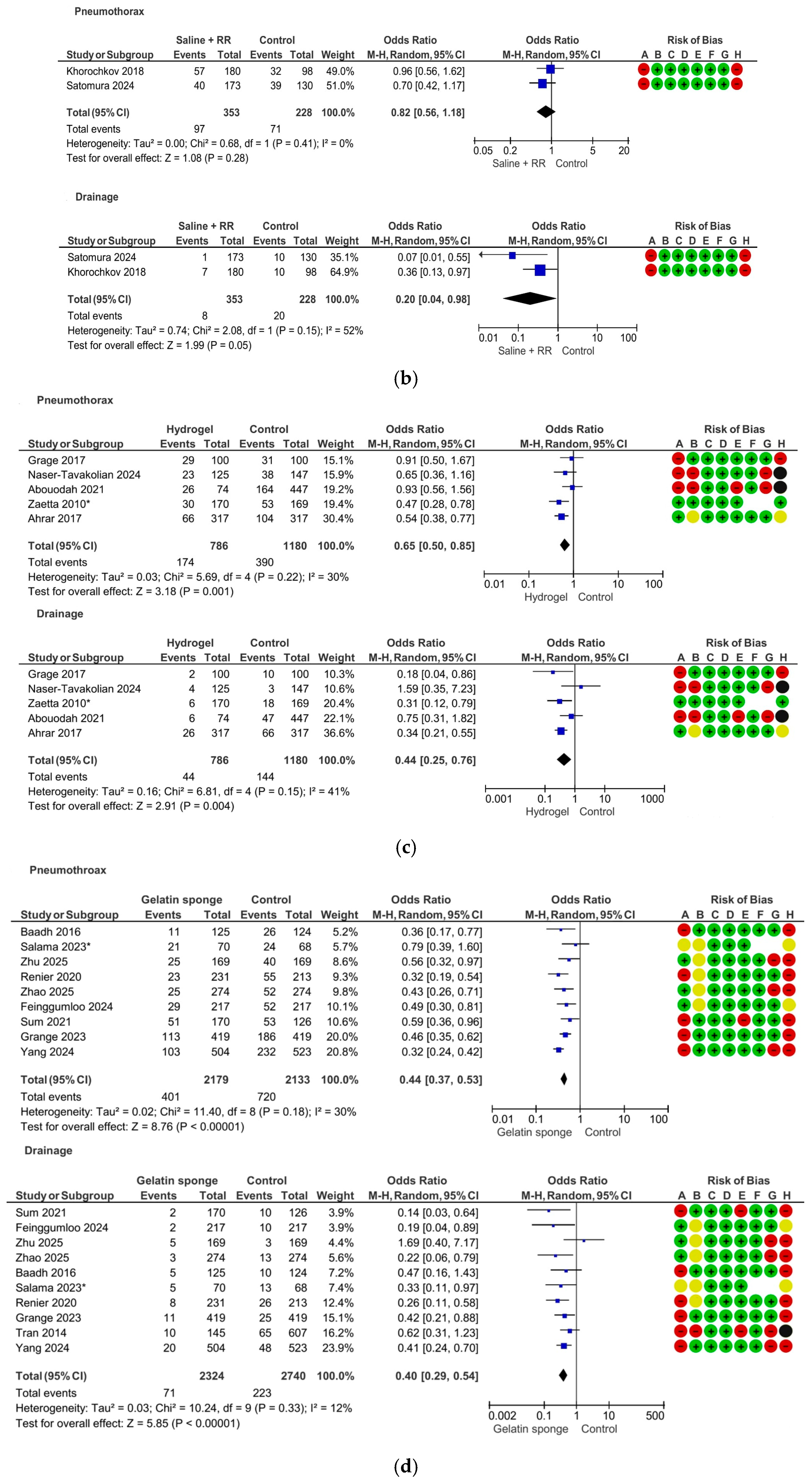


| Analysis | Certainty Assessment | No. of Patients | Effect | Certainty | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | Sealant | Control | Relative (95% CI) | Absolute (95% CI) | ||
| Saline: PTX | 7 | 6 cohort 1 RCT | serious | not serious | not serious | not serious | strong association | 112/680 (16.5%) | 273/775 (35.2%) | OR 0.35 (0.25 to 0.48) | 192 fewer per 1000 (from 233 fewer to 145 fewer) | ⨁⨁⨁⨁ High |
| Saline: Drainage | 7 | 6 cohort 1 RCT | serious | not serious | not serious | serious | strong association | 11/680 (1.6%) | 59/775 (7.6%) | OR 0.22 (0.11 to 0.43) | 58 fewer per 1000 (from 67 fewer to 42 fewer) | ⨁⨁⨁◯ Moderate |
| Saline+RR: PTX | 2 | 2 cohort | serious | not serious | not serious | very serious | none | 97/353 (27.5%) | 71/228 (31.1%) | OR 0.82 (0.56 to 1.18) | 41 fewer per 1000 (from 109 fewer to 37 more) | ⨁◯◯◯ Very low |
| Saline+RR: Drainge | 2 | 2 cohort | serious | not serious | not serious | serious | strong association | 8/353 (2.3%) | 20/228 (8.8%) | OR 0.20 (0.04 to 0.98) | 69 fewer per 1000 (from 84 fewer to 2 fewer) | ⨁⨁⨁◯ Moderate |
| Hydrogel: PTX | 5 | 4 cohort 1 RCT | very serious | not serious | not serious | not serious | none | 174/786 (22.1%) | 390/1180 (33.1%) | OR 0.65 (0.50 to 0.85) | 88 fewer per 1000 (from 133 fewer to 35 fewer) | ⨁⨁◯◯ Low |
| Hydrogel: Drainage | 5 | 4 cohort 1 RCT | very serious | not serious | not serious | serious | strong association | 44/786 (5.6%) | 144/1180 (12.2%) | OR 0.44 (0.25 to 0.76) | 64 fewer per 1000 (from 88 fewer to 26 fewer) | ⨁⨁◯◯ Low |
| Gelatin sponge: PTX | 9 | 8 cohort 1 RCT | serious | not serious | not serious | not serious | none | 401/2179 (18.4%) | 720/2133 (33.8%) | OR 0.44 (0.37 to 0.53) | 154 fewer per 1000 (from 179 fewer to 125 fewer) | ⨁⨁⨁◯ Moderate |
| Gelatin sponge: Drainage | 10 | 9 cohort 1 RCT | serious | not serious | not serious | not serious | strong association | 71/2324 (3.1%) | 223/2740 (8.1%) | OR 0.40 (0.29 to 0.54) | 47 fewer per 1000 (from 56 fewer to 36 fewer) | ⨁⨁⨁⨁ High |
| Blood patch: PTX | 9 | 7 cohort 2 RCT | very serious | not serious | not serious | not serious | none | 401/2037 (19.7%) | 589/2002 (29.4%) | OR 0.50 (0.40 to 0.62) | 122 fewer per 1000 (from 151 fewer to 89 fewer) | ⨁⨁◯◯ Low |
| Blood patch: Drainage | 9 | 7 cohort 2 RCT | very serious | not serious | not serious | not serious | strong association | 78/2037 (3.8%) | 183/2002 (9.1%) | OR 0.40 (0.27 to 0.59) | 53 fewer per 1000 (from 65 fewer to 35 fewer) | ⨁⨁⨁◯ Moderate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Roman, A.; Lobonț-Terec, N.-A.; Pintican, R.; Fetica, B.; Kubelac, P.; Fekete, Z.; Preda, A.C.; Pașca, A.; Schiau, C.; Csutak, C. Tract Sealing Techniques for Pneumothorax and Drainage Prevention After CT-Guided Lung Biopsy: A Systematic Review and Meta-Analysis. Diagnostics 2026, 16, 824. https://doi.org/10.3390/diagnostics16060824
Roman A, Lobonț-Terec N-A, Pintican R, Fetica B, Kubelac P, Fekete Z, Preda AC, Pașca A, Schiau C, Csutak C. Tract Sealing Techniques for Pneumothorax and Drainage Prevention After CT-Guided Lung Biopsy: A Systematic Review and Meta-Analysis. Diagnostics. 2026; 16(6):824. https://doi.org/10.3390/diagnostics16060824
Chicago/Turabian StyleRoman, Andrei, Nicoleta-Anca Lobonț-Terec, Roxana Pintican, Bogdan Fetica, Paul Kubelac, Zsolt Fekete, Alexandra Cristina Preda, Andrei Pașca, Călin Schiau, and Csaba Csutak. 2026. "Tract Sealing Techniques for Pneumothorax and Drainage Prevention After CT-Guided Lung Biopsy: A Systematic Review and Meta-Analysis" Diagnostics 16, no. 6: 824. https://doi.org/10.3390/diagnostics16060824
APA StyleRoman, A., Lobonț-Terec, N.-A., Pintican, R., Fetica, B., Kubelac, P., Fekete, Z., Preda, A. C., Pașca, A., Schiau, C., & Csutak, C. (2026). Tract Sealing Techniques for Pneumothorax and Drainage Prevention After CT-Guided Lung Biopsy: A Systematic Review and Meta-Analysis. Diagnostics, 16(6), 824. https://doi.org/10.3390/diagnostics16060824





