Diagnostic Accuracy and Real-Life Advantages of the MONA.health Artificial Intelligence Software in Screening for Diabetic Retinopathy and Maculopathy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Patients
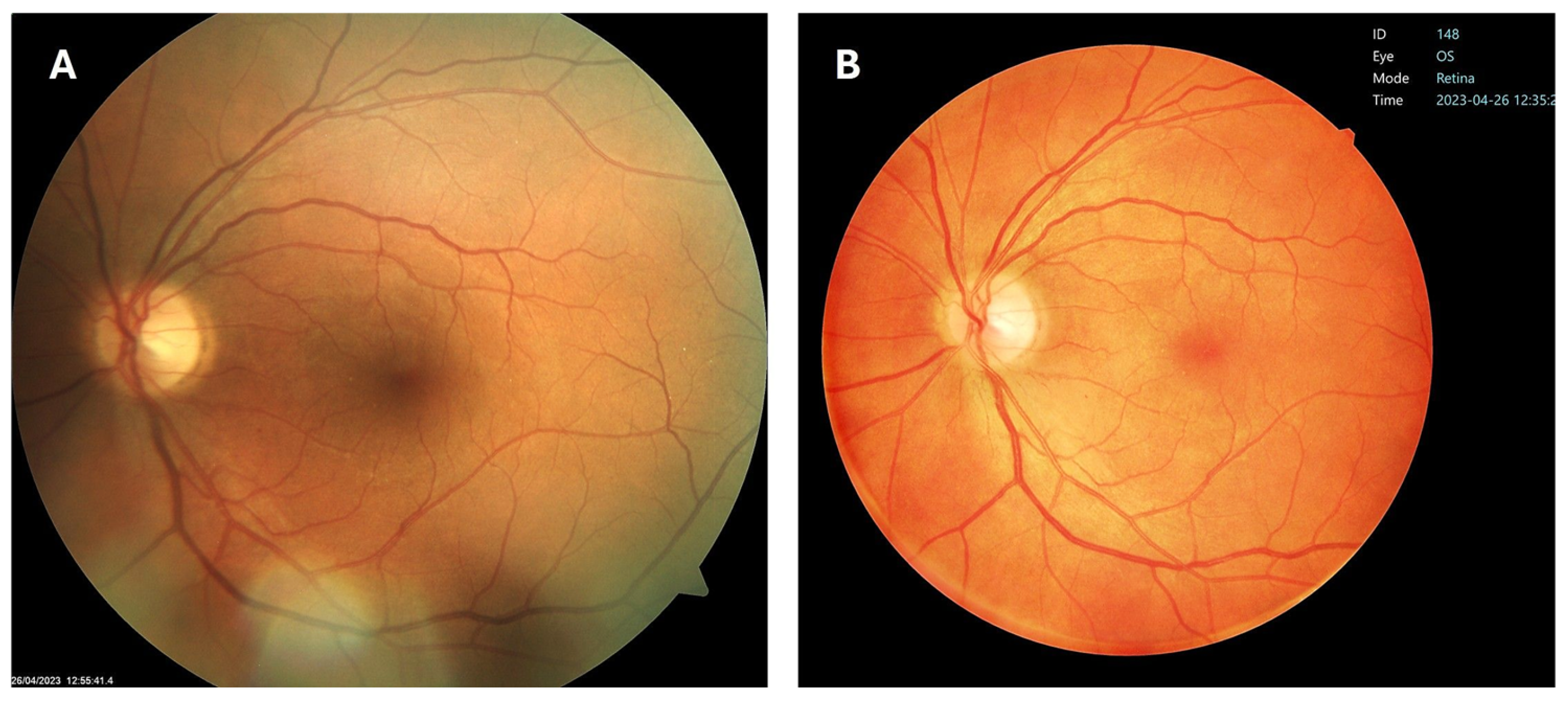
2.3. Fundus Photography and Diabetic Retinopathy Grading
2.4. Statistical Analysis
3. Results
3.1. Demographic Data and Prevalence of Diabetic Retinopathy and Maculopathy
3.2. Diagnostic Accuracy of the MONA.health Artificial Intelligence in the Detection of Referable DR
3.3. Diagnostic Accuracy of the MONA.health Artificial Intelligence in the Detection of DME
3.4. Real-Life Advantages of the MONA.health Artificial Intelligence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ogurtsova, K.; da Rocha Fernandes, J.D.; Huang, Y.; Linnenkamp, U.; Guariguata, L.; Cho, N.H.; Cavan, D.; Shaw, J.E.; Makaroff, L.E. IDF Diabetes Atlas: Global estimates for the prevalence of diabetes for 2015 and 2040. Diabetes Res. Clin. Pract. 2017, 128, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.Y.; Tan, T.-E. The Diabetic Retinopathy “Pandemic” and Evolving Global Strategies: The 2023 Friedenwald Lecture. Investig. Ophthalmol. Vis. Sci. 2023, 64, 47. [Google Scholar] [CrossRef] [PubMed]
- Teo, Z.L.; Tham, Y.C.; Yu, M.; Chee, M.L.; Rim, T.H.; Cheung, N.; Bikbov, M.M.; Wang, Y.X.; Tang, Y.; Lu, Y.; et al. Global prevalence of diabetic retinopathy and projection of burden through 2045: Systematic review and meta-analysis. Ophthalmology 2021, 128, 1580–1591. [Google Scholar] [CrossRef] [PubMed]
- Javitt, J.C.; Aiello, L.P. Cost-effectiveness of detecting and treating diabetic retinopathy. Ann. Intern. Med. 1996, 124, 164–169. [Google Scholar] [CrossRef]
- Rohan, T.E.; Frost, C.D.; Wald, N.J. Prevention of blindness by screening for diabetic retinopathy: A quantitative assessment. BMJ 1989, 299, 1198–1201. [Google Scholar] [CrossRef]
- Wong, T.Y.; Sun, J.; Kawasaki, R.; Ruamviboonsuk, P.; Gupta, N.; Lansingh, V.C.; Maia, M.; Mathenge, W.; Moreker, S.; Muqit, M.M.K.; et al. Guidelines on diabetic eye care: The international council of ophthalmology recommendations for screening, follow-up, referral, and treatment based on resource settings. Ophthalmology 2018, 125, 1608–16622. [Google Scholar] [CrossRef]
- Huang, X.; Wang, H.; She, C.; Feng, J.; Liu, X.; Hu, X.; Chen, L.; Tao, Y. Artificial intelligence promotes the diagnosis and screening of diabetic retinopathy. Front. Endocrinol. 2022, 13, 946915. [Google Scholar] [CrossRef]
- Padhy, S.K.; Takkar, B.; Chawla, R.; Kumar, A. Artificial intelligence in diabetic retinopathy: A natural step to the future. Indian J. Ophthalmol. 2019, 67, 1004–1009. [Google Scholar] [CrossRef]
- Tomić, M.; Vrabec, R.; Hendelja, Đ.; Kolarić, V.; Bulum, T.; Rahelić, D. Diagnostic Accuracy of Hand-Held Fundus Camera and Artificial Intelligence in Diabetic Retinopathy Screening. Biomedicines 2024, 12, 34. [Google Scholar] [CrossRef]
- Lim, J.I.; Regillo, C.D.; Sadda, S.R.; Ipp, E.; Bhaskaranand, M.; Ramachandra, C.; Solanki, K.; for the EyeArt Study Subgroup. Artificial Intelligence Detection of Diabetic Retinopathy. Ophthalmol. Sci. 2022, 3, 100228. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. FDA Permits Marketing of Artificial Intelligence-Based Device to Detect Certain Diabetes-Related Eye Problems; US Food and Drug Administration: Silver Spring, MD, USA, 2018. Available online: https://www.prnewswire.com/news-releases/fda-permits-marketing-of-artificial-intelligence-based-device-to-detect-certain-diabetes-related-eye-problems-300628218.html (accessed on 1 October 2023).
- Bhaskaranand, M.; Ramachandra, C.; Bhat, S.; Cuadros, J.; Nittala, M.G.; Sadda, S.R.; Solanki, K. The value of automated diabetic retinopathy screening with the EyeArt system: A study of more than 100,000 consecutive encounters from people with diabetes. Diabetes Technol. Ther. 2019, 21, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.N.; Dai, L.; Li, S.T.; Kong, H.Y.; Sheng, B.; Wu, Q. Automatic grading system for diabetic retinopathy diagnosis using deep learning artificial intelligence software. Curr. Eye Res. 2020, 45, 1550–1555. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, D.; Tan, Z.; Niu, Y.; Jiang, Y.; Xiong, R.; Peng, G.; He, M. Screening referable diabetic retinopathy using a semi-automated deep learning algorithm assisted approach. Front. Med. 2021, 8, 740987. [Google Scholar] [CrossRef] [PubMed]
- Wu, E.; Wu, K.; Daneshjou, R.; Ouyang, D.; Ho, D.E.; Zou, J. How medical AI devices are evaluated: Limitations and recommendations from an analysis of FDA approvals. Nat. Med. 2021, 27, 582–584. [Google Scholar] [CrossRef]
- Abràmoff, M.D.; Lavin, P.T.; Birch, M.; Shah, N.; Folk, J.C. Pivotal trial of an autonomous AI-based diagnostic system for detection of diabetic retinopathy in primary care offices. NPJ Digit. Med. 2018, 1, 39. [Google Scholar] [CrossRef]
- Rudnicka, A.R.; Shakespeare, R.; Chambers, R.; Bolter, L.; Anderson, J.; Fajtl, J.; Welikala, R.A.; Barman, S.A.; Olvera-Barrios, A.; Webster, L.; et al. Automated retinal image analysis systems to triage for grading of diabetic retinopathy: A large-scale, open-label, national screening programme in England. Lancet Digit. Health 2025, 7, 100914. [Google Scholar] [CrossRef]
- Aldington, S.J.; Kohner, E.M.; Meuer, S.; Klein, R.; Sjølie, A.K. Methodology for retinal photography and assessment of diabetic retinopathy: The EURODIAB IDDM complications study. Diabetologia 1995, 38, 437–4344. [Google Scholar] [CrossRef]
- Wilkinson, C.P.; Ferris, F.L., 3rd; Klein, R.E.; Lee, P.P.; Agardh, C.D.; Davis, M.; Dills, D.; Kampik, A.; Pararajasegaram, R.; Verdaguer, J.T.; et al. Proposed international clinical diabetic retinopathy and diabetic macular edema disease severity scales. Ophthalmology 2003, 110, 1677–1682. [Google Scholar] [CrossRef]
- Peeters, F.; Rommes, S.; Elen, B.; Gerrits, N.; Stalmans, I.; Jacob, J.; De Boever, P. Artificial intelligence software for diabetic eye screening: Diagnostic performance and impact of stratification. J. Clin. Med. 2023, 12, 1408. [Google Scholar] [CrossRef]
- Bouhaimed, M.; Gibbins, R.; Owens, D. Automated Detection of Diabetic Retinopathy: Results of a Screening Study. Diabetes Technol. Ther. 2008, 10, 142–148. [Google Scholar] [CrossRef]
- Lee, A.Y.; Yanagihara, R.T.; Lee, C.S.; Blazes, M.; Jung, H.C.; Chee, Y.E.; Gencarella, M.D.; Gee, H.; Maa, A.Y.; Cockerham, G.C.; et al. Multicenter, Head-to-Head, Real-World Validation Study of Seven Automated Artificial Intelligence Diabetic Retinopathy Screening Systems. Diabetes Care 2021, 44, 1168–1175. [Google Scholar] [CrossRef]
- González-Gonzalo, C.; Sánchez-Gutiérrez, V.; Hernández-Martínez, P.; Contreras, I.; Lechanteur, Y.T.; Domanian, A.; van Ginneken, B.; Sánchez, C.I. Evaluation of a deep learning system for the joint automated detection of diabetic retinopathy and age-related macular degeneration. Acta Ophthalmol. 2020, 98, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Abràmoff, M.D.; Lou, Y.; Erginay, A.; Clarida, W.; Amelon, R.; Folk, J.C.; Niemeijer, M. Improved Automated Detection of Diabetic Retinopathy on a Publicly Available Dataset Through Integration of Deep Learning. Investig. Ophthalmol. Vis. Sci. 2016, 57, 5200. [Google Scholar] [CrossRef] [PubMed]
- Wewetzer, L.; Held, L.A.; Steinhäuser, J. Diagnostic performance of deep-learning-based screening methods for diabetic retinopathy in primary care—A meta-analysis. PLoS ONE 2021, 16, e0255034. [Google Scholar] [CrossRef] [PubMed]
- Krause, J.; Gulshan, V.; Rahimy, E.; Karth, P.; Widner, K.; Corrado, G.S.; Peng, L.; Webster, D.R. Grader Variability and the Importance of Reference Standards for Evaluating Machine Learning Models for Diabetic Retinopathy. Ophthalmology 2018, 125, 1264–1272. [Google Scholar] [CrossRef]
- Taylor, R.; Broadbent, D.M.; Greenwood, R.; Hepburn, D.; Owens, D.R.; Simpson, H. Mobile retinal screening in Britain. Diabet. Med. 1998, 15, 344–347. [Google Scholar] [CrossRef]
- Scanlon, P.H.; Aldington, S.J.; Leal, J.; Luengo-Fernandez, R.; Oke, J.; Sivaprasad, S.; Gazis, A.; Stratton, I.M. Development of a cost-effectiveness model for optimisation of the screening interval in diabetic retinopathy screening. Health Technol. Assess. 2015, 19, 1–116. [Google Scholar] [CrossRef]
- Heydon, P.; Egan, C.; Bolter, L.; Chambers, R.; Anderson, J.; Aldington, S.; Stratton, I.M.; Scanlon, P.H.; Webster, L.; Mann, S.; et al. Prospective evaluation of an artificial intelligence-enabled algorithm for automated diabetic retinopathy screening of 30 000 patients. Br. J. Ophthalmol. 2021, 105, 723–728. [Google Scholar] [CrossRef]
- Tufail, A.; Kapetanakis, V.V.; Salas-Vega, S.; Egan, C.; Rudisill, C.; Owen, C.G.; Lee, A.; Louw, V.; Anderson, J.; Liew, G.; et al. An observational study to assess if automated diabetic retinopathy image assessment software can replace one or more steps of manual imaging grading and to determine their cost-effectiveness. Health Technol. Assess. 2016, 20, 1. [Google Scholar] [CrossRef]
- Rajesh, A.E.; Davidson, O.Q.; Lee, C.S.; Lee, A.Y. Artificial Intelligence and Diabetic Retinopathy: AI Framework, Prospective Studies, Head-to-head Validation, and Cost-effectiveness. Diabetes Care 2023, 46, 1728–1739. [Google Scholar] [CrossRef]
- Styles, C.J. Introducing automated diabetic retinopathy systems: It’s not just about sensitivity and specificity. Eye 2019, 33, 1357–1358. [Google Scholar] [CrossRef]
- Kirkwood, B.J.; Coster, D.J.; Essex, R.W. Ophthalmic nurse practitioner led diabetic retinopathy screening. Results of a 3-month trial. Eye 2006, 20, 173–177. [Google Scholar] [CrossRef]
- Chen, C.; Pazo, E.E.; Liu, H.; Liu, J. Nurse-led model in diabetic retinopathy screening: An exploratory scoping review of nurses’ roles and contributions. Clin. Ophthalmol. 2025, 19, 4335–4347. [Google Scholar] [CrossRef]
- Roux, M.; Rice, J.; Steffen, J. Feasibility of automated artificial intelligence screening for diabetic retinopathy in a resource-limited setting. J. Coll. Med. S. Afr. 2025, 3, a269. [Google Scholar] [CrossRef]



| Total (n = 296) | T1DM (n = 56) | T2DM (n = 240) | Z | p-Value | |
|---|---|---|---|---|---|
| Age (yr) | 63 (19–81) | 39 (19–60) | 66 (31–81) | −10.708 | <0.001 |
| Gender (m/f) (%) | 54/46 | 53.6/46.4 | 54.2/45.8 | 0.006 | 0.936 |
| Diabetes duration (yr) | 9 (1–39) | 14.5 (1–34) | 8 (1–39) | 3.013 | 0.003 |
| MONA DR vs. | VISUCAM NM/FA Zeiss Fundus Camera | |
|---|---|---|
| Estimate | 95% CI | |
| Sensitivity | 100% | 94.40–100% |
| Specificity | 99.81% | 98.95–100% |
| PPV | 99.33% | 95.46–99.91% |
| NPV | 100% | 99.30–100% |
| LR+ | 528.00 | 74.51–3741.49 |
| LR− | 0.00 | |
| Kappa ± SE | 0.99 ± 0.01 | 0.97–1.00 |
| DOR | Infinity | NaN-Infinity |
| DE | 99.85% | 99.10–100% |
| MONA DME vs. | VISUCAM NM/FA Zeiss Fundus Camera | |
|---|---|---|
| Estimate | 95% CI | |
| Sensitivity | 100% | 75.29–100% |
| Specificity | 98.95% | 97.74–99.62% |
| PPV | 85.93% | 73.37–93.12% |
| NPV | 100% | 99.35–100% |
| LR+ | 95.67 | 43.16–212.05 |
| LR− | 0.00 | |
| Kappa ± SE | 0.81 ± 0.08 | 0.66–0.96 |
| DOR | Infinity | NaN-Infinity |
| DE | 99.02% | 97.84–99.65% |
| MONA AI Screening Process | Standard Camera and Human Grading | Z | p-Value | |
|---|---|---|---|---|
| Required time (min) | 5 (3–8) | 13 (10–19) | 18.489 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tomić, M.; Vrabec, R.; Babić, T.; Kljajić, K.; Bulum, T. Diagnostic Accuracy and Real-Life Advantages of the MONA.health Artificial Intelligence Software in Screening for Diabetic Retinopathy and Maculopathy. Diagnostics 2026, 16, 730. https://doi.org/10.3390/diagnostics16050730
Tomić M, Vrabec R, Babić T, Kljajić K, Bulum T. Diagnostic Accuracy and Real-Life Advantages of the MONA.health Artificial Intelligence Software in Screening for Diabetic Retinopathy and Maculopathy. Diagnostics. 2026; 16(5):730. https://doi.org/10.3390/diagnostics16050730
Chicago/Turabian StyleTomić, Martina, Romano Vrabec, Toma Babić, Kristina Kljajić, and Tomislav Bulum. 2026. "Diagnostic Accuracy and Real-Life Advantages of the MONA.health Artificial Intelligence Software in Screening for Diabetic Retinopathy and Maculopathy" Diagnostics 16, no. 5: 730. https://doi.org/10.3390/diagnostics16050730
APA StyleTomić, M., Vrabec, R., Babić, T., Kljajić, K., & Bulum, T. (2026). Diagnostic Accuracy and Real-Life Advantages of the MONA.health Artificial Intelligence Software in Screening for Diabetic Retinopathy and Maculopathy. Diagnostics, 16(5), 730. https://doi.org/10.3390/diagnostics16050730








