Free Thyroxine as a Predictor of Mortality in Critically Ill Septic Patients—A Retrospective Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
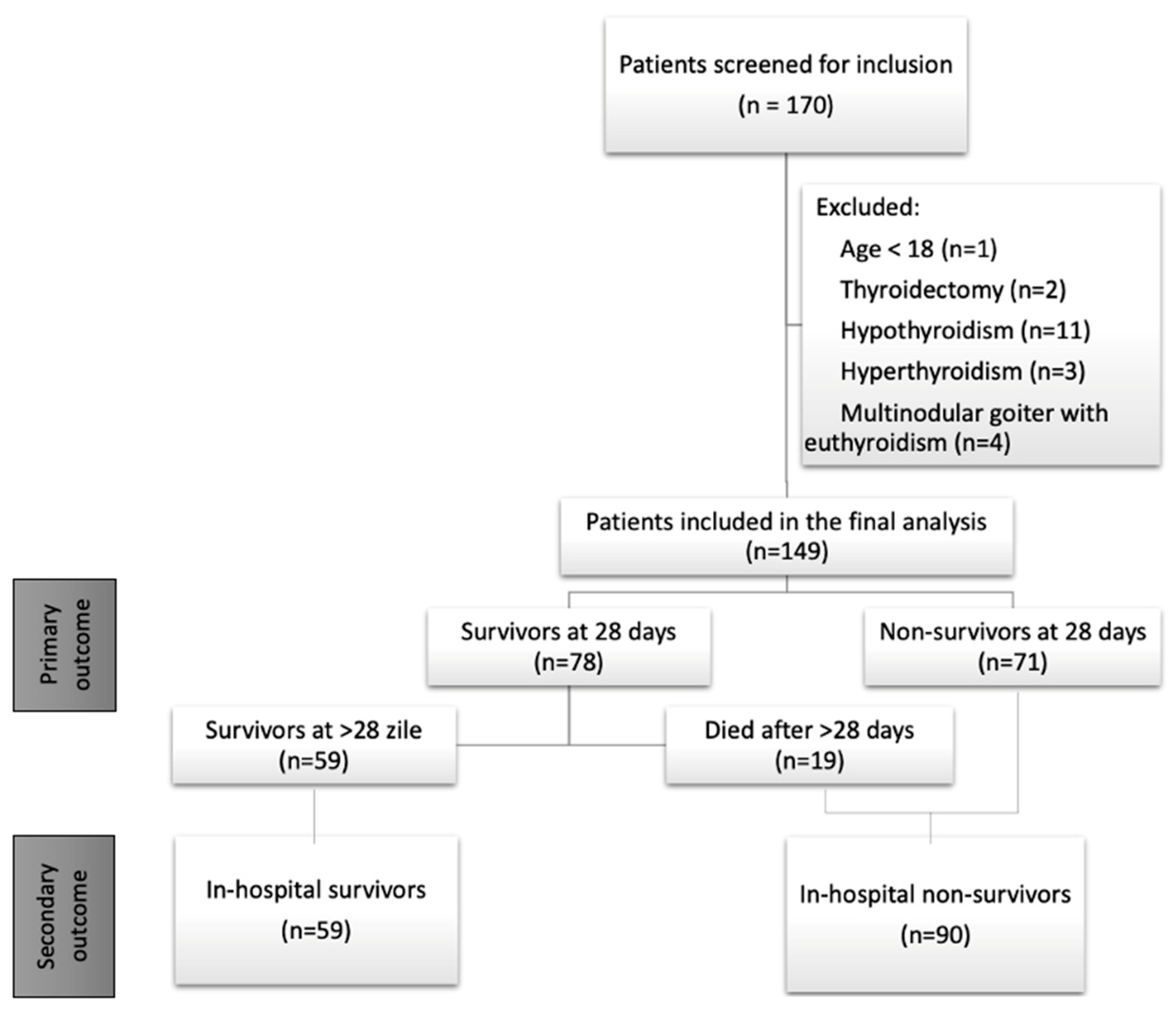
3.1. Study Population
3.2. Analysis of Comorbidities and Clinical Outcome Scores
3.3. Analysis of Laboratory Parameters
3.4. Analysis of Patients Grouped by fT4 Levels
3.5. Univariate Analysis
3.6. Multivariate Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, J.; Wang, Y.; Xu, Z.; Chou, C.; Xiang, J.; Zhang, X.; Hou, X.; Yue, S.; Ye, S.; Chen, F.; et al. Association between Sensitivity to Thyroid Hormone and Prognosis in Septic Patients: A Retrospective Cohort Analysis. Front. Endocrinol. 2025, 16, 1611963. [Google Scholar] [CrossRef] [PubMed]
- Rudd, K.E.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.S.; Kissoon, N.; Finfer, S.; et al. Global, Regional, and National Sepsis Incidence and Mortality, 1990–2017: Analysis for the Global Burden of Disease Study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Mușat, F.; Păduraru, D.N.; Bolocan, A.; Palcău, C.-A.; Bunea, A.-A.; Ion, D.; Andronic, O. Sepsis Burden in a Major Romanian Emergency Center-An 18-Year Retrospective Analysis of Mortality and Risk Factors. Medicina 2025, 61, 864. [Google Scholar] [CrossRef]
- Chechulina, V.; Sheikh, F.; Lóser, M.; Englesakis, M.; Barrett, K. Sepsis Canada Healthcare Costs after Sepsis: A Systematic Review. Crit. Care 2025, 29, 381. [Google Scholar] [CrossRef]
- Bauer, M.; Gerlach, H.; Vogelmann, T.; Preissing, F.; Stiefel, J.; Adam, D. Mortality in Sepsis and Septic Shock in Europe, North America and Australia between 2009 and 2019— Results from a Systematic Review and Meta-Analysis. Crit. Care 2020, 24, 239. [Google Scholar] [CrossRef]
- Liu, Y.-C.; Jiang, T.-Y.; Chen, Z.-S.; Qi, A.-L.; Gao, Y.-L.; Li, S.-X.; Yu, M.-M.; Chai, Y.-F.; Shou, S.-T. Thyroid Hormone Disorders: A Predictor of Mortality in Patients with Septic Shock Defined by Sepsis-3? Intern. Emerg. Med. 2021, 16, 967–973. [Google Scholar] [CrossRef]
- Dai, J.-J.; Du, D.-F.; Ma, G.; Jiang, M.-J. Association between Serum-Free Thyroxine Level and All-Cause Mortality in Critically Ill Patients: A Retrospective Study from MIMIC-IV. Front. Endocrinol. 2023, 14, 1164369. [Google Scholar] [CrossRef]
- Lee, S.; Farwell, A.P. Euthyroid Sick Syndrome. Compr. Physiol. 2016, 6, 1071–1080. [Google Scholar] [CrossRef]
- Luo, B.; Yu, Z.; Li, Y. Thyroid Hormone Disorders and Sepsis. Biomed. Mater. Eng. 2017, 28, S237–S241. [Google Scholar] [CrossRef]
- Kim, J.G.; Shin, H.; Kim, W.; Lim, T.H.; Jang, B.; Cho, Y.; Choi, K.-S.; Ahn, C.; Lee, J.; Na, M.K. The Value of Decreased Thyroid Hormone for Predicting Mortality in Adult Septic Patients: A Systematic Review and Meta-Analysis. Sci. Rep. 2018, 8, 14137. [Google Scholar] [CrossRef] [PubMed]
- Gutch, M.; Kumar, S.; Gupta, K.K. Prognostic Value of Thyroid Profile in Critical Care Condition. Indian J. Endocrinol. Metab. 2018, 22, 387–391. [Google Scholar] [CrossRef] [PubMed]
- da Silveira, C.D.; de Vasconcelos, F.P.; Moura, E.B.; da Silveira, B.T.; Amorim, F.F.; Shintaku, L.S.; de Santana, R.B.; Argotte, P.L.; da Silva, S.F.; de Oliveira Maia, M.; et al. Thyroid Function, Reverse Triiodothyronine, and Mortality in Critically Ill Clinical Patients. Indian J. Crit. Care Med. 2021, 25, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, L.; Shi, J.; Han, W.; Zhu, C.; Zhang, T.; Ma, X.; Liang, Y. Long-Term Prognostic Value of Thyroid Hormone Levels in Chronic Critical Illness Patients. Ann. Med. 2025, 57, 2479583. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, F.; Hong, G.; Lu, Z. Thyroid Hormone Levels as a Predictor Marker Predict the Prognosis of Patients with Sepsis. Am. J. Emerg. Med. 2021, 45, 42–47. [Google Scholar] [CrossRef]
- Siampa, V.N.; Abadi, S.; Aman, A.M.; Bakri, S.; Halim, R.; Zainuddin, A.A. Association between Severity of Sepsis and Thyroid Function Profile. Acta Biomed. 2023, 94, e2023239. [Google Scholar] [CrossRef]
- Foks, M.; Dudek, A.; Polok, K.; Nowak-Kózka, I.; Fronczek, J.; Szczeklik, W. Thyroid Hormones as Potential Prognostic Factors in Sepsis. Anaesthesiol. Intensive Ther. 2019, 51, 205–209. [Google Scholar] [CrossRef]
- Sun, C.; Bao, L.; Guo, L.; Wei, J.; Song, Y.; Shen, H.; Qin, H. Prognostic Significance of Thyroid Hormone T3 in Patients with Septic Shock: A Retrospective Cohort Study. PeerJ 2023, 11, e15335. [Google Scholar] [CrossRef]
- Widmer, A.; Schuetz, P. Endocrine Dysfunction during Sepsis—Are Changes in Hormone Levels a Physiological Adaptation or a Therapeutic Target? J. Lab. Precis. Med. 2018, 3, 61. [Google Scholar] [CrossRef]
- Kovacevic, M.; Nesek-Adam, V.; Klokic, S.; Mujaric, E. Low T3 vs Low T3T4 Euthyroid Sick Syndrome in Septic Shock Patients: A Prospective Observational Cohort Study. World J. Crit. Care Med. 2024, 13, 96132. [Google Scholar] [CrossRef]
- Zhang, L.; Tan, R.; Pan, T.; Qu, H. Impact of Thyroid Hormones on Predicting the Occurrence of Persistent Inflammation, Immunosuppression, and Catabolism Syndrome in Patients with Sepsis. Front. Endocrinol. 2024, 15, 1417846. [Google Scholar] [CrossRef] [PubMed]
- Wajner, S.M.; Maia, A.L. New Insights toward the Acute Non-Thyroidal Illness Syndrome. Front. Endocrinol. 2012, 3, 8. [Google Scholar] [CrossRef]
- Kovacevic, M.; Adam, V.N.; Causevic, S. Triiodothyronine Hormone Supplementation Therapy in Septic Shock Patients with Euthyroid Sick Syndrome: Two Pilot, Placebo-Controlled, Randomized Trials. Anaesth. Crit. Care Pain Med. 2024, 43, 101336. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lu, Y.; Guo, S.-B.; Wang, J.-Y.; Yang, J. Low Serum Thyroid-Stimulating Hormone Levels May Be an Early Predictor of Sepsis. BMJ Support. Palliat. Care 2022. Epub ahead of print. [Google Scholar] [CrossRef]
- Gray, A.P.; Chung, E.; Hsu, R.L.; Araki, D.T.; Hayoon, A.G.; Weaver, N.D.; Swetschinski, L.R.; Wool, E.E.; Han, C.; Mestrovic, T.; et al. Global, Regional, and National Sepsis Incidence and Mortality, 1990–2021: A Systematic Analysis for the Global Burden of Disease Study 2021. Lancet Glob. Health 2025, 13, E2013–E2026. [Google Scholar] [CrossRef]
- Namgung, M.; Ahn, C.; Park, Y.; Kwak, I.-Y.; Lee, J.; Won, M. Mortality among Adult Patients with Sepsis and Septic Shock in Korea: A Systematic Review and Meta-Analysis. Clin. Exp. Emerg. Med. 2023, 10, 157–171. [Google Scholar] [CrossRef]
- Rao, M.-J.; Zhang, Y.; Peng, W.-Z.; Pan, P.-H. Association of Thyroid Hormones and Thyroid-Stimulating Hormone with Mortality in Adults Admitted to the Intensive Care Unit: A Systematic Review and Meta-Analysis. J. Chin. Med. Assoc. 2022, 85, 443–452. [Google Scholar] [CrossRef]
- Ning, N.; Li, J.; Sun, W.; Ma, C.; Li, J.; Sheng, H.; Chen, Y.; Zhao, B.; Zhang, J.; Zhu, J.; et al. Different Subtypes of Nonthyroidal Illness Syndrome on the Prognosis of Septic Patients: A Two-Centered Retrospective Cohort Study. Front. Endocrinol. 2023, 14, 1227530. [Google Scholar] [CrossRef]
- Xu, Y.; Xu, K.; Guo, J.; Fang, M.; Wang, Z. Association between Dynamic Fluctuations in Triiodothyronine Levels and Prognosis among Critically Ill Patients within Comprehensive Intensive Care Units. Front. Endocrinol. 2023, 14, 1282547. [Google Scholar] [CrossRef]
- Krug, N.; Bercker, S.; Busch, T.; Friese, S.; Jahn, N.; Voelker, M.T. Non-Thyroidal Illness Syndrome (NTIS) Is No Independent Predictor for Mortality in ICU Patients. BMC Anesth. 2023, 23, 103. [Google Scholar] [CrossRef]
- Stoian, M.; Azamfirei, L.; Bandila, S.R.; Stoian, A.; Babă, D.-F.; Bănescu, C. Circulating microRNAs and Plasma Gelsolin as Biomarkers of Sepsis: Molecular Insights and Prospects for Precision Medicine. Biomolecules 2025, 15, 1621. [Google Scholar] [CrossRef]

| 28-Day Mortality | In-Hospital Mortality | ||||||
|---|---|---|---|---|---|---|---|
| All Patients (n = 149) | Survivors (n = 78) | Non-Survivors (n = 71) | p | Survivors (n = 59) | Non-Survivors (n = 90) | p | |
| Sex, n (%) | 0.247 | 0.216 | |||||
| M | 64 (42.95) | 37 (47.44) | 27 (38.03) | 29 (49.15) | 35 (38.89) | ||
| F | 85 (57.05) | 41 (52.56) | 44 (61.97) | 30 (50.85) | 55 (61.11) | ||
| Age (years), median (IQR) | 73 (65; 80) | 70.5 (61; 76.25) | 75 (69; 82) | 0.002 | 69 (57; 76) | 74 (69; 82) | 0.002 |
| Primary diagnostic, n (%) | 0.012 | 0.004 | |||||
| Sepsis | 37 (24.83) | 26 (33.33) | 11 (15.49) | 22 (37.29) | 15 (16.67) | ||
| Septic shock | 112 (75.17) | 52 (66.67) | 60 (84.51) | 37 (62.71) | 75 (83.33) | ||
| Location of primary infection, n (%) | 0.977 | 0.422 | |||||
| One source | 103 (69.13) | 54 (69.23) | 49 (69.01) | 43 (72.88) | 60 (66.67) | ||
| Multiple sources | 46 (30.87) | 24 (30.77) | 22 (30.99) | 16 (27.12) | 30 (33.33) | ||
| Sepsis source 1, n (%) | N/A | N/A | |||||
| Pulmonary | 62 (41.61) | 33 (42.31) | 29 (40.85) | 22 (37.29) | 40 (44.44) | ||
| Digestive | 59 (39.6) | 34 (43.59) | 25 (35.21) | 23 (38.98) | 36 (40) | ||
| Urinary | 39 (26.17) | 19 (24.36) | 20 (28.17) | 14 (23.73) | 25 (27.78) | ||
| Cutaneous | 27 (18.12) | 15 (19.23) | 12 (16.9) | 13 (22.03) | 14 (15.56) | ||
| Endocarditis | 4 (2.68) | 1 (1.28) | 3 (4.23) | 1 (1.69) | 3 (3.33) | ||
| Mediastinitis | 1 (0.67) | 0 (0) | 1 (1.41) | 0 (0) | 1 (1.11) | ||
| Septic arthritis | 1 (0.67) | 0 (0) | 1 (1.41) | 0 (0) | 1 (1.11) | ||
| Unknown | 15 (10.07) | 6 (7.69) | 9 (12.68) | 5 (8.47) | 10 (11.11) | ||
| Type of pathology, n (%) | 0.011 | 0.147 | |||||
| Medical | 89 (59.73) | 39 (50) | 50 (70.42) | 31 (52.54) | 58 (64.44) | ||
| Surgical | 60 (40.27) | 39 (50) | 21 (29.58) | 28 (47.46) | 32 (35.56) | ||
| Hospital admission type, n (%) | 0.622 | 0.999 | |||||
| Emergency admission | 145 (97.32) | 75 (96.15) | 70 (98.59) | 58 (98.31) | 87 (96.67) | ||
| Planned admission | 4 (2.68) | 3 (3.85) | 1 (1.41) | 1 (1.69) | 3 (3.33) | ||
| 28-Day Mortality | In-Hospital Mortality | ||||||
|---|---|---|---|---|---|---|---|
| All Patients (n = 149) | Survivors (n = 78) | Non-Survivors (n = 71) | p | Survivors (n = 59) | Non-Survivors (n = 90) | p | |
| HTN, n (%) | 92 (61.74) | 47 (60.26) | 45 (63.38) | 0.695 | 33 (55.93) | 59 (65.56) | 0.237 |
| CHF, n (%) | 84 (56.38) | 38 (48.72) | 46 (64.79) | 0.048 | 25 (42.37) | 59 (65.56) | 0.005 |
| AF, n (%) | 56 (37.58) | 26 (33.33) | 30 (42.25) | 0.262 | 18 (30.51) | 38 (42.22) | 0.149 |
| DM, n (%) | 51 (34.23) | 29 (37.18) | 22 (30.99) | 0.426 | 20 (33.9) | 31 (34.44) | 0.945 |
| CKD, n (%) | 35 (23.49) | 13 (16.67) | 22 (30.99) | 0.039 | 10 (16.95) | 25 (27.78) | 0.127 |
| COPD, n (%) | 31 (20.81) | 11 (14.1) | 20 (28.17) | 0.035 | 10 (16.95) | 21 (23.33) | 0.348 |
| Active or past cancer, n (%) | 26 (17.45) | 15 (19.23) | 11 (15.49) | 0.548 | 8 (13.56) | 18 (20) | 0.311 |
| History of stroking, n (%) | 24 (16.11) | 12 (15.38) | 12 (16.9) | 0.801 | 6 (10.17) | 18 (20) | 0.11 |
| Autoimmune disease, n (%) | 11 (7.38) | 4 (5.13) | 7 (9.86) | 0.27 | 2 (3.39) | 9 (10) | 0.201 |
| APACHE II, median (IQR) | 27 (20.75; 35) | 22 (15; 32) | 32 (26; 38.5) | <0.001 | 21 (14.5; 32) | 31 (23.25; 38) | <0.001 |
| SOFA, mean ± SD | 11.18 ± 4.59 | 9.53 ± 4.73 | 12.97 ± 3.7 | <0.001 | 9.17 ± 4.55 | 12.48 ± 4.14 | <0.001 |
| ICU LOS (days), median (IQR) | 9 (5; 22) | 11 (6; 29) | 7 (4; 15.5) | 0.006 | 7 (5; 12.5) | 11 (5; 22.75) | 0.209 |
| Total hospital LOS (days), median (IQR) | 22 (10; 35) | 30.5 (17.25; 44.75) | 13 (7; 25) | <0.001 | 23 (14; 34.5) | 20 (8.25; 36) | 0.208 |
| 28 Day-Mortality | In-Hospital Mortality | ||||||
|---|---|---|---|---|---|---|---|
| All Patients (n = 149) | Survivors (n = 78) | Non-Survivors (n = 71) | p | Survivors (n = 59) | Non-Survivors (n = 90) | p | |
| CRP (mg/dL), median (IQR) | 16.24 (7.26; 26.94) | 17.21 (8.23; 32.03) | 15 (6.72; 22.91) | 0.234 | 17.19 (7.27; 24.56) | 15.15 (7.27; 24.56) | 0.349 |
| PCT (ng/mL), median (IQR) | 2.56 (0.78; 13.59) | 1.59 (0.6; 14.25) | 3.01 (1.03; 12.3) | 0.241 | 1.57 (0.56; 13.57) | 3.01 (0.98; 14.16) | 0.124 |
| WBC (×109/L), median (IQR) | 13.76 (9.46; 19.08) | 12.61 (8.76; 18.16) | 14.91 (10.68; 20.33) | 0.221 | 12.01 (8.12; 17.35) | 14.93 (10.81; 20.06) | 0.018 |
| Hemoglobin (g/dL), median (IQR) | 10.6 (8.93; 12.05) | 11.4 (9.2; 12.48) | 9.75 (8.5; 11.4) | 0.002 | 11.4 (9.4; 12.55) | 9.8 (8.6; 11.6) | 0.003 |
| Platelet count (×109/L), median (IQR) | 232 (172.75; 308.75) | 242.5 (181.75; 288.5) | 215 (167; 309) | 0.408 | 232 (177; 277.5) | 238 (171.5; 313.5) | 0.919 |
| Serum creatinine (mg/dL), median (IQR) | 1.59 (0.94; 3.18) | 1.48 (0.8; 2.77) | 1.96 (1.15; 3.49) | 0.015 | 1.16 (0.73; 2) | 2.09 (1.18; 3.89) | <0.001 |
| TSH (μUI/mL), median (IQR) | 2.01 (0.94; 3.36) | 2.02 (0.98; 3.14) | 2.01 (0.91; 3.56) | 0.748 | 2 (1.02; 3.24) | 2.05 (0.9; 3.44) | 0.656 |
| fT4 (ng/dL), mean ± SD | 1.08 ± 0.35 | 1.15 ± 0.34 | 1 ± 0.35 | 0.01 | 1.16 ± 0.31 | 1.02 ± 0.37 | 0.014 |
| fT4 Level | p | |||
|---|---|---|---|---|
| Hypothyroid (n = 50) | Euthyroid (n = 92) | Hyperthyroid (n = 7) | ||
| APACHE II, median (IQR) | 26 (21; 34) | 29 (20; 35) | 20 (14.5; 27.5) | 0.325 |
| SOFA, mean ± SD | 10.71 ± 4.13 | 11.71 ± 4.73 | 7.57 ± 4.28 | 0.07 |
| ICU LOS (days), median (IQR) | 12 (5; 22.75) | 8 (5; 17) | 7 (5.5; 20) | 0.59 |
| Total hospital LOS (days), median (IQR) | 22 (10.5; 34.75) | 22.5 (9; 35.25) | 19 (15.5; 28.5) | 0.947 |
| 28-day mortality, n (%) | 31 (62) | 37 (40.22) | 3 (42.86) | 0.044 |
| In-hospital mortality, n (%) | 39 (78) | 46 (50) | 5 (71.43) | 0.004 |
| Hypothyroid Patients | p | ||
|---|---|---|---|
| Concordant TSH (n = 43) | Discordant TSH (n = 7) | ||
| APACHE II, median (IQR) | 27.5 (21.25; 34.75) | 26 (18.5; 28.5) | 0.55 |
| SOFA, mean ± SD | 10.48 ± 4.03 | 12.14 ± 4.78 | 0.328 |
| ICU LOS (days), median (IQR) | 12 (5; 22.5) | 20 (9; 25.5) | 0.237 |
| Total hospital LOS (days), median (IQR) | 22 (12; 34) | 20 (10; 39) | 0.978 |
| 28-day mortality, n (%) | 26 (60.47) | 5 (71.43) | 0.579 |
| In-hospital mortality, n (%) | 33 (76.74) | 6 (85.71) | 0.595 |
| 28 Day-Mortality | In-Hospital Mortality | |||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p | AUC | OR (95% CI) | p | AUC | |
| Age (years) | 1.05 (1.02–1.08) | 0.003 | 0.65 | 1.05 (1.02–1.08) | 0.002 | 0.655 |
| Primary diagnostic (septic shock vs. sepsis) | 2.73 (1.23–6.05) | 0.014 | 0.308 | 2.97 (1.38–6.39) | 0.005 | 0.342 |
| CHF | 1.94 (1.01–3.74) | 0.049 | 0.423 | 2.59 (1.32–5.08) | 0.006 | 0.477 |
| APACHE II | 1.08 (1.04–1.11) | <0.001 | 0.732 | 1.06 (1.03–1.10) | <0.001 | 0.697 |
| SOFA | 1.21 (1.11–1.32) | <0.001 | 0.727 | 1.20 (1.10–1.31) | <0.001 | 0.715 |
| Hemoglobin (g/dL) | 0.82 (0.71–0.95) | 0.007 | 0.653 | 0.83 (0.71–0.96) | 0.011 | 0.645 |
| Serum creatinine (mg/dL) | 1.19 (0.99–1.44) | 0.066 | 0.617 | 1.37 (1.09–1.72) | 0.006 | 0.674 |
| fT4 (ng/dL) | 0.28 (0.10–0.76) | 0.012 | 0.615 | 0.3 (0.11–0.80) | 0.017 | 0.632 |
| 28-Day Mortality | In-Hospital Mortality | |||||||
|---|---|---|---|---|---|---|---|---|
| SE | OR (95% CI) | p | AUC (95% CI) | SE | OR (95% CI) | p | AUC (95% CI) | |
| fT4 (ng/dL) | 0.63 | 0.2 (0.06–0.69) | 0.011 | 0.805 (0.733–0.876) | 0.62 | 0.23 (0.07–0.78) | 0.018 | |
| Hb (g/dL) | 0.08 | 0.82 (0.70–0.96) | 0.014 | 0.08 | 0.83 (0.71–0.98) | 0.029 | 0.799 (0.725–0.873) | |
| Age (years) | 0.02 | 1.06 (1.02–1.10) | 0.005 | 0.02 | 1.05 (1.01–1.08) | 0.008 | ||
| SOFA | 0.05 | 1.22 (1.10–1.35) | <0.001 | 0.05 | 1.2 (1.09–1.32) | <0.001 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Negruț, M.F.; Pastor, V.; Bolcaș, R.; Antal, O.; Szabo, R.; Petrișor, C. Free Thyroxine as a Predictor of Mortality in Critically Ill Septic Patients—A Retrospective Study. Diagnostics 2026, 16, 680. https://doi.org/10.3390/diagnostics16050680
Negruț MF, Pastor V, Bolcaș R, Antal O, Szabo R, Petrișor C. Free Thyroxine as a Predictor of Mortality in Critically Ill Septic Patients—A Retrospective Study. Diagnostics. 2026; 16(5):680. https://doi.org/10.3390/diagnostics16050680
Chicago/Turabian StyleNegruț, Matei Florin, Vlad Pastor, Robert Bolcaș, Oana Antal, Robert Szabo, and Cristina Petrișor. 2026. "Free Thyroxine as a Predictor of Mortality in Critically Ill Septic Patients—A Retrospective Study" Diagnostics 16, no. 5: 680. https://doi.org/10.3390/diagnostics16050680
APA StyleNegruț, M. F., Pastor, V., Bolcaș, R., Antal, O., Szabo, R., & Petrișor, C. (2026). Free Thyroxine as a Predictor of Mortality in Critically Ill Septic Patients—A Retrospective Study. Diagnostics, 16(5), 680. https://doi.org/10.3390/diagnostics16050680







