Using Multimodal Imaging to Correlate Link Between Intracranial Aneurysms and Acute Ischemic Stroke
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Imaging and Stroke Evaluation
2.3. Treatment
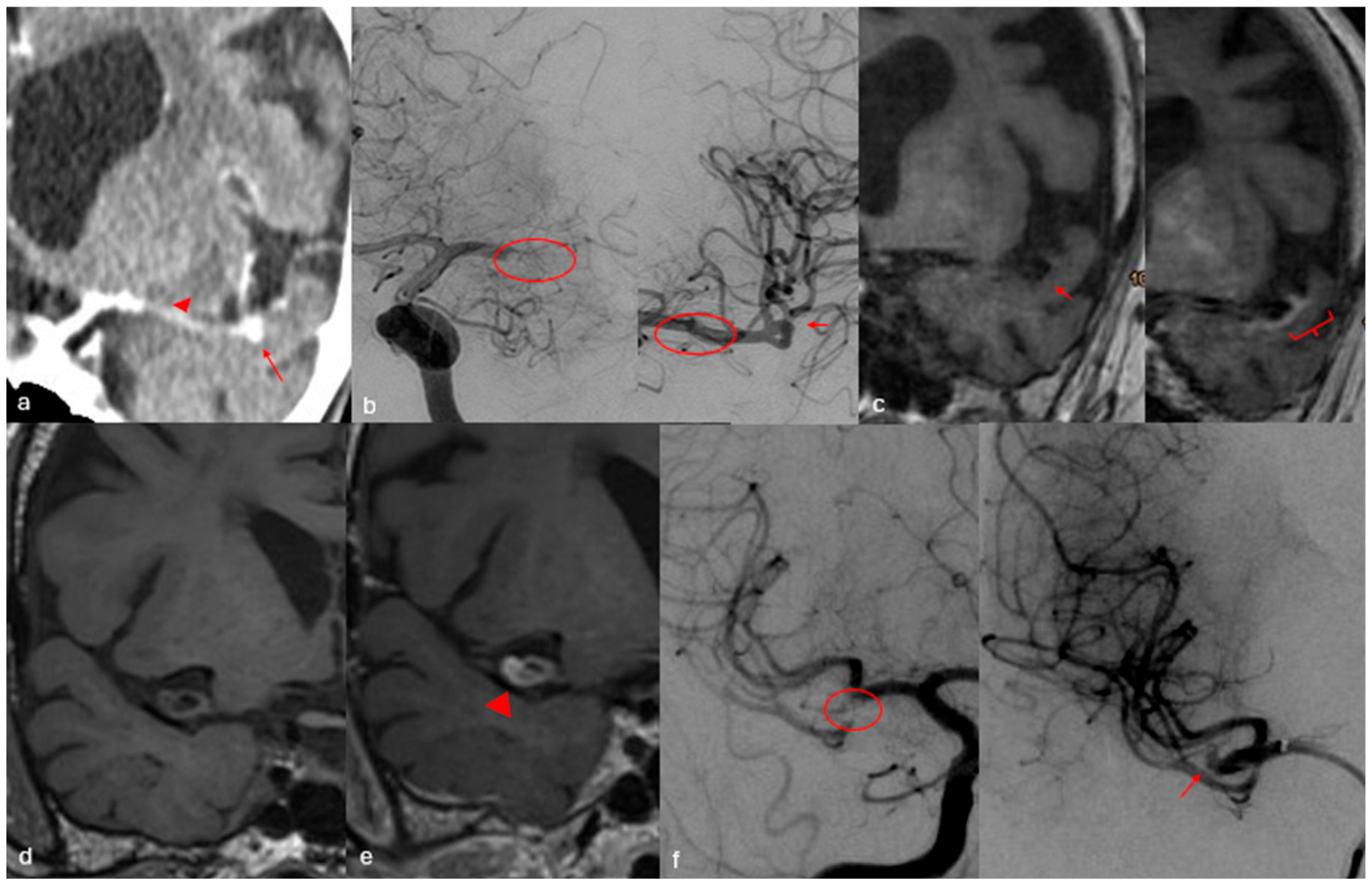
3. Results
3.1. Patients with LVO
3.2. Patients Without LVO
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qureshi, A.I.; Mohammad, Y.; Yahia, A.M.; Luft, A.R.; Sharma, M.; Tamargo, R.J.; Frankel, M.R. Ischemic events associated with unruptured intracranial aneurysms: Multicenter clinical study and review of the literature. Neurosurgery 2000, 46, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Rinkel, G.J.E.; Djibuti, M.; Algra, A.; van Gijn, J. Prevalence and risk of rupture of intracranial aneurysms: A systematic review. Stroke 1998, 29, 251–256. [Google Scholar] [CrossRef]
- Oh, Y.S.; Lee, S.J.; Shon, Y.M.; Yang, D.W.; Kim, B.S.; Cho, A.H. Incidental unruptured intracranial aneurysms in patients with acute ischemic stroke. Cerebrovasc. Dis. 2008, 26, 650–653. [Google Scholar] [CrossRef]
- Calviere, L.; Viguier, A.; Da Silva, N.A., Jr.; Cognard, C.; Larrue, V. Unruptured intracranial aneurysm as a cause of cerebral ischemia. Clin. Neurol. Neurosurg. 2011, 113, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Zibold, F.; Kleine, J.F.; Zimmer, C.; Poppert, H.; Boeckh-Behrens, T. Aneurysms in the target vessels of stroke patients subjected to mechanical thrombectomy: Prevalence and impact on treatment. J. Neurointerv. Surg. 2016, 8, 1016–1020. [Google Scholar] [CrossRef]
- Mokin, M.; Darkhabani, Z.; Binning, M.J.; Levy, E.I.; Siddiqui, A.H. Small unruptured partially thrombosed aneurysms and stroke: Report of three cases and review of the literature. J. Neurointerv. Surg. 2012, 4, e6. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.; Davidson, R.I.; Marcus, E.M. Transient focal cerebral ischemia as a presenting manifestation of unruptured cerebral aneurysms. Ann. Neurol. 1980, 8, 367–372. [Google Scholar] [CrossRef]
- Schaller, B.; Lyrer, P. Focal neurological deficits following spontaneous thrombosis of unruptured giant aneurysms. Eur. Neurol. 2002, 47, 175–182. [Google Scholar] [CrossRef]
- McLaughlin, N.; Bojanowski, M.W. Unruptured cerebral aneurysms presenting with ischemic events. Can. J. Neurol. Sci. 2008, 35, 588–592. [Google Scholar] [CrossRef]
- Sweid, A.; Tjoumakaris, S.; Herial, N.; Gooch, M.R.; Rosenwasser, R.H.; Jabbour, P. Case report of a partially thrombosed ACoA aneurysm presenting with bilateral foot drop. Clin. Neurol. Neurosurg. 2019, 185, 105490. [Google Scholar] [CrossRef]
- Zaidat, O.O.; Derwenskus, J.; Suarez, J.I.; Selman, W.R. Partially thrombosed aneurysm and stroke. Neurology 2004, 63, 1285. [Google Scholar] [CrossRef]
- Fukuoka, S.; Suematsu, K.; Nakamura, J.; Matsuzaki, T.; Satoh, S.; Hashimoto, I. Transient ischemic attacks caused by unruptured intracranial aneurysm. Surg. Neurol. 1982, 17, 464–467. [Google Scholar] [CrossRef]
- Hoffman, W.F.; Wilson, C.B.; Townsend, J.J. Recurrent transient ischemic attacks secondary to an embolizing saccular middle cerebral artery aneurysm. Case report. J. Neurosurg. 1979, 51, 103–106. [Google Scholar] [CrossRef]
- Kuroda, H.; Mochizuki, T.; Shimizu, S.; Kumabe, T. Rupture of Thrombosed Cerebral Aneurysm During Antithrombotic Therapy for Ischemic Stroke: Case Report and Literature Review. World Neurosurg. 2019, 126, 468–471. [Google Scholar] [CrossRef] [PubMed]
- Binning, M.; Duhon, B.; Couldwell, W.T. Partially thrombosed lateral lenticulostriate aneurysm presenting with embolic stroke: Case illustration. J. Neurosurg. Pediatr. 2010, 5, 190. [Google Scholar] [CrossRef]
- Eller, T.W. MRI demonstration of clot in a small unruptured aneurysm causing stroke. Case report. J. Neurosurg. 1986, 65, 411–412. [Google Scholar] [CrossRef] [PubMed]
- Antunes, J.L.; Correll, J.W. Cerebral emboli from intracranial aneurysms. Surg. Neurol. 1976, 6, 7–10. [Google Scholar]
- Kincaid, K.J.; Yu, J.; Echevarria, F.D.; Simpkins, A.N. Giant Vertebrobasilar Fusiform Aneurysm Mass Effect Heralds Rapid in Situ Thrombosis and Ischemic Stroke in the Setting of Ulcerative Colitis. J. Stroke Cerebrovasc. Dis. 2021, 30, 105621. [Google Scholar] [CrossRef]
- Tanaka, T.; Goto, H.; Momozaki, N.; Honda, E. Rapid Change in Shape of Unruptured Intracranial Aneurysm with Acute Perforating Infarction. Case Rep. Neurol. 2022, 14, 400–403. [Google Scholar] [CrossRef]
- Sugita, M.; Sasaki, H.; Kakizawa, T.; Kimura, R.; Fukamachi, A.; Nukui, H. Giant middle cerebral artery aneurysm with parent artery occlusion--case report. Neurol. Med. Chir. 1991, 31, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Yamagami, K.; Hatano, T.; Ando, M.; Chihara, H.; Ogura, T.; Suzuki, K.; Kondo, D.; Kamata, T.; Higashi, E.; Sakai, S.; et al. Symptomatic Cavernous Internal Carotid Artery Aneurysm Complicated by Simultaneous Rapid Growth of the Intra-aneurysmal and Parent Artery Thromboses. NMC Case Rep. J. 2021, 8, 177–182. [Google Scholar] [CrossRef]
- Friedman, J.A.; Piepgras, D.G.; Pichelmann, M.A.; Hansen, K.K.; Brown, R.D., Jr.; Wiebers, D.O. Small cerebral aneurysms presenting with symptoms other than rupture. Neurology 2001, 57, 1212–1216. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, N.; Handa, H. The fate of untreated symptomatic cerebral aneurysms: Analysis of 26 patients with clinical course of more than five years. Surg. Neurol. 1982, 18, 21–26. [Google Scholar] [CrossRef]
- Asari, S.; Ohmoto, T. Natural history and risk factors of unruptured cerebral aneurysms. Clin. Neurol. Neurosurg. 1993, 95, 205–214. [Google Scholar] [CrossRef]
- Juvela, S.; Porras, M.; Heiskanen, O. Natural history of unruptured intracranial aneurysms: A long-term follow-up study. J. Neurosurg. 1993, 79, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.M.; Samson, D.; Diehl, J.; Hinton, R.; Ditmore, Q.M. Unruptured cerebral aneurysms presenting as recurrent transient neurologic deficits. Neurology 1980, 30, 47–51. [Google Scholar] [CrossRef]
- Solomon, R.A.; Fink, M.E.; Pile-Spellman, J. Surgical management of unruptured intracranial aneurysms. J. Neurosurg. 1994, 80, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Sakaki, T.; Kinugawa, K.; Tanigake, T.; Miyamoto, S.; Kyoi, K.; Utsumi, S. Embolism from intracranial aneurysms. J. Neurosurg. 1980, 53, 300–304. [Google Scholar] [CrossRef]
- Vlak, M.H.; Algra, A.; Brandenburg, R.; Rinkel, G.J. Prevalence of unruptured intracranial aneurysms, with emphasis on sex, age, comorbidity, country, and time period: A systematic review and meta-analysis. Lancet Neurol. 2011, 10, 626–636. [Google Scholar] [CrossRef]
- Edwards, N.J.; Kamel, H.; Josephson, S.A. The safety of intravenous thrombolysis for ischemic stroke in patients with pre-existing cerebral aneurysms: A case series and review of the literature. Stroke 2012, 43, 412–416. [Google Scholar] [CrossRef]
- Zhou, T.; Li, T.; Zhu, L.; Wang, Z.; Bai, W.; Xue, J.; Feng, G. Endovascular Thrombectomy for Large-Vessel Occlusion Strokes with Preexisting Intracranial Aneurysms. Cardiovasc. Interv. Radiol. 2018, 41, 1399–1403. [Google Scholar] [CrossRef]
- Marzoughi, S.; Hennawy, M.; Al Hashash, H.; Zhou, L.W. Ischemic Stroke Secondary to Middle Cerebral Artery Branch Occlusion From a Thrombosed Saccular Aneurysm. Stroke 2023, 54, e237–e238. [Google Scholar] [CrossRef]
- Atallah, E.; Valle-Giler, E.; Elarjani, T.; Chalouhi, N.; Tjoumakaris, S.; Rosenwasser, R.H.; Herial, N.; Gooch, M.R.; Zarzour, H.; Jabbour, P. Acute Recanalization of a Partially Thrombosed Large Intracranial Aneurysm. World Neurosurg. 2018, 115, 73–78. [Google Scholar] [CrossRef]
- Cebral, J.; Ollikainen, E.; Chung, B.J.; Mut, F.; Sippola, V.; Jahromi, B.R.; Tulamo, R.; Hernesniemi, J.; Niemela, M.; Robertson, A.; et al. Flow Conditions in the Intracranial Aneurysm Lumen Are Associated with Inflammation and Degenerative Changes of the Aneurysm Wall. AJNR Am. J. Neuroradiol. 2017, 38, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Ha, M.H.; Joo, S.P.; Kim, T.S. Acute Parent Artery Occlusion in Thrombosed Middle Cerebral Artery Aneurysm Presenting with Ischemic Stroke. World Neurosurg. 2020, 136, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Guillon, B.; Daumas-Duport, B.; Delaroche, O.; Warin-Fresse, K.; Sevin, M.; Herisson, F.; Auffray-Calvier, E.; Desal, H. Cerebral ischemia complicating intracranial aneurysm: A warning sign of imminent rupture? AJNR Am. J. Neuroradiol. 2011, 32, 1862–1865. [Google Scholar] [CrossRef] [PubMed]
- Benalia, V.H.; Cortez, G.M.; Monteiro, A.; Siddiqui, A.; Aghaebrahim, A.; Sauvageau, E.; Hanel, R.A. Brain aneurysm rupture during mechanical thrombectomy for large vessel occlusion: Technical case series and complication avoidance strategies. Interv. Neuroradiol. 2022, 31, 567–573. [Google Scholar] [CrossRef]
- Edjlali, M.; Gentric, J.C.; Regent-Rodriguez, C.; Trystram, D.; Hassen, W.B.; Lion, S.; Nataf, F.; Raymond, J.; Wieben, O.; Turski, P.; et al. Does aneurysmal wall enhancement on vessel wall MRI help to distinguish stable from unstable intracranial aneurysms? Stroke 2014, 45, 3704–3706. [Google Scholar] [CrossRef]
- Kulcsar, Z.; Berentei, Z.; Marosfoi, M.; Vajda, J.; Szikora, I. Thromboembolic complication induced stable occlusion of a ruptured basilar tip aneurysm. Case report and review of the literature. Interv. Neuroradiol. 2010, 16, 83–88. [Google Scholar] [CrossRef]
- Lawton, M.T.; Quinones-Hinojosa, A.; Chang, E.F.; Yu, T. Thrombotic intracranial aneurysms: Classification scheme and management strategies in 68 patients. Neurosurgery 2005, 56, 441–454. [Google Scholar] [CrossRef]
- Scerrati, A.; Sabatino, G.; Della Pepa, G.M.; Albanese, A.; Marchese, E.; Puca, A.; Olivi, A.; Sturiale, C.L. Treatment and outcome of thrombosed aneurysms of the middle cerebral artery: Institutional experience and a systematic review. Neurosurg. Rev. 2019, 42, 649–661. [Google Scholar] [CrossRef] [PubMed]


| Pt | Age | Gender | Clinical Presentation | Stroke Age | NIHSS Admission | NIHSS Discharge |
|---|---|---|---|---|---|---|
| 1 | 83 | M | Acute left-sided hemi-syndrome, dysarthria, left-sided neglect in wake-up constellation | Acute | 10 | 0 |
| 2 | 57 | F | Dysarthria, word-finding difficulties, anopsia of the lower right quadrant | Subacute—1 month prior TIA with similar presentation | 3 | 0 |
| 3 | 44 | F | Slight left-sided weakness, dysarthria in wake-up constellation | Acute | 4 | 0 |
| 4 | 80 | F | Right-sided hemisyndrom | Acute—older cerebellar ones | 23 | 2 |
| 5 | 71 | F | Moderate weakness in left leg | Subacute | 2 | 1 |
| 6 | 64 | F | Mouth droop, arm weakness (left), dysarthria | Acute—old from previous M2 occlusion | 3 | 1 |
| 7 | 74 | F | Left-sided hemiparesis, dysarthria, hypoasthesia | Acute—multiple older ones in other territories | 7 | 6 |
| 8 | 80 | F | Left-sided hemiparesis, dysarthria, anisocoria | Acute | 12 | 1 |
| Pt | Location | Thrombus | Size | LVO | VWI | Treatment | Co-Factors | Classification Parameters | Aneurysm-Related Ischemic Event | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | RMCA bifurcation | Near-complete | 7 mm | Yes M1 | + | Coiling | RICA stenosis | Thrombus Enhancement Stroke risk factor | + + + | Probable |
| 2 | LMCA bifurcation | Near-complete | 11 mm | no | + | Clipping | No | Thrombus Enhancement Stroke risk factor | + + − | Probable |
| 3 * | RMCA bifurcation | Unknown—imaging inconclusive | 3 mm | Yes M1 | N/A | Conservative | No | Thrombus Enhancement Stroke risk factor | +/− N/A − | Possible |
| 4 | LMCA bifurcation | No thrombus—CT shows retrograde aneurysm filling | 4.5 mm | Yes M1 | − | Conservative | Atrial fibrillation/older cerebellar infarcts on the left | Thrombus Enhancement Stroke risk factor | − − + | Improbable |
| 5 | RMCA distal M2 | Partial | 10 mm | no | + | Clipping | No | Thrombus Enhancement Stroke risk factor | + + - | Probable |
| 6 * | RMCA bifurcation | Complete | 12 mm | no | N/A | Conservative | Cavernous ICA stenosis/older M2 occlusion | Thrombus Enhancement Stroke risk factor | + N/A + | Possible |
| 7 | RMCA bifurcation | No | 7 mm | no | + | Conservative | Atrial fibrillation/multiple older ischemic lesions in other vascular territories | Thrombus Enhancement Stroke risk factor | − + + | Improbable |
| 8 | RMCA bifurcation | No | 4.5 mm | Yes ICA | + | Conservative | Atrial fibrillation | Thrombus Enhancement Stroke risk factor | − + + | Improbable |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Anagnostakou, V.; Thurner, P.; Madjidyar, J.; Krepuska, M.; Kyselyova, A.; Schubert, T.; Kulcsar, Z. Using Multimodal Imaging to Correlate Link Between Intracranial Aneurysms and Acute Ischemic Stroke. Diagnostics 2026, 16, 1511. https://doi.org/10.3390/diagnostics16101511
Anagnostakou V, Thurner P, Madjidyar J, Krepuska M, Kyselyova A, Schubert T, Kulcsar Z. Using Multimodal Imaging to Correlate Link Between Intracranial Aneurysms and Acute Ischemic Stroke. Diagnostics. 2026; 16(10):1511. https://doi.org/10.3390/diagnostics16101511
Chicago/Turabian StyleAnagnostakou, Vania, Patrick Thurner, Jawid Madjidyar, Miklos Krepuska, Anna Kyselyova, Tilman Schubert, and Zsolt Kulcsar. 2026. "Using Multimodal Imaging to Correlate Link Between Intracranial Aneurysms and Acute Ischemic Stroke" Diagnostics 16, no. 10: 1511. https://doi.org/10.3390/diagnostics16101511
APA StyleAnagnostakou, V., Thurner, P., Madjidyar, J., Krepuska, M., Kyselyova, A., Schubert, T., & Kulcsar, Z. (2026). Using Multimodal Imaging to Correlate Link Between Intracranial Aneurysms and Acute Ischemic Stroke. Diagnostics, 16(10), 1511. https://doi.org/10.3390/diagnostics16101511






