Diagnostic Performance and Prognostic Value of Serum Amyloid A in Patients with Bloodstream Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Assay Methods
2.3. Statistical Analysis
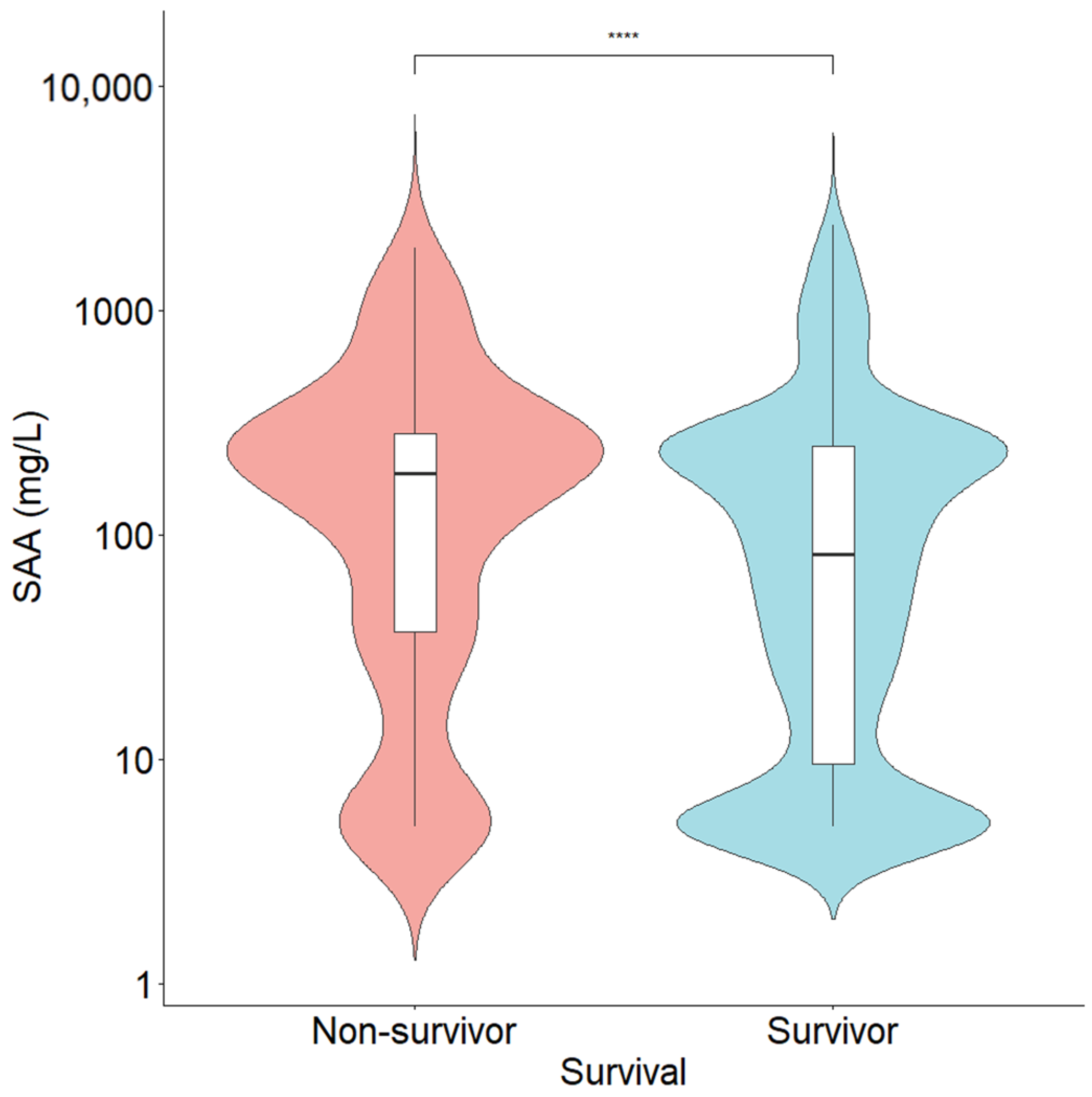
3. Results
3.1. Study Population
3.2. Association Between BC Results and SAA Levels
3.3. Correlation Between SAA and Other Laboratory Markers
3.4. Diagnostic Performance
3.5. Prognostic Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Uhlar, C.M.; Whitehead, A.S. Serum amyloid A, the major vertebrate acute-phase reactant. Eur. J. Biochem. 1999, 265, 501–523. [Google Scholar] [CrossRef]
- Gabay, C.; Kushner, I. Acute-Phase Proteins and Other Systemic Responses to Inflammation. N. Engl. J. Med. 1999, 340, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.J.; Kushner, I. Serum C-reactive protein levels in disease. Ann. N. Y. Acad. Sci. 1982, 389, 406–418. [Google Scholar] [CrossRef]
- Sorić Hosman, I.; Kos, I.; Lamot, L. Serum Amyloid A in Inflammatory Rheumatic Diseases: A Compendious Review of a Renowned Biomarker. Front. Immunol. 2020, 11, 631299. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Kang, S.Y.; Lee, W.I.; Lee, M.Y. Evaluation of serum amyloid A as a biomarker for sepsis diagnosis compared with C-reactive protein, procalcitonin, and presepsin. Lab. Med. 2026, 57, lmaf054. [Google Scholar] [CrossRef] [PubMed]
- Eklund, K.K.; Niemi, K.; Kovanen, P.T. Immune functions of serum amyloid A. Crit. Rev. Immunol. 2012, 32, 335–348. [Google Scholar] [CrossRef]
- Schuetz, P.; Albrich, W.; Mueller, B. Procalcitonin for diagnosis of infection and guide to antibiotic decisions: Past, present and future. BMC Med. 2011, 9, 107. [Google Scholar] [CrossRef]
- Glowicz, J.B.; Landon, E.; Sickbert-Bennett, E.E.; Aiello, A.E.; Dekay, K.; Hoffmann, K.K.; Maragakis, L.; Olmsted, R.N.; Polgreen, P.M.; Trexler, P.A.; et al. SHEA/IDSA/APIC Practice Recommendation: Strategies to prevent healthcare-associated infections through hand hygiene: 2022 Update. Infect. Control Hosp. Epidemiol. 2023, 44, 355–376. [Google Scholar] [CrossRef]
- Kyriazopoulou, E.; Leventogiannis, K.; Tavoulareas, G.; Mainas, E.; Toutouzas, K.; Mathas, C.; Prekates, A.; Sakka, V.; Panagopoulos, P.; Syrigos, K.; et al. Presepsin as a diagnostic and prognostic biomarker of severe bacterial infections and COVID-19. Sci. Rep. 2023, 13, 3814. [Google Scholar] [CrossRef]
- Velissaris, D.; Zareifopoulos, N.; Karamouzos, V.; Karanikolas, E.; Pierrakos, C.; Koniari, I.; Karanikolas, M. Presepsin as a Diagnostic and Prognostic Biomarker in Sepsis. Cureus 2021, 13, e15019. [Google Scholar] [CrossRef]
- Arcenas, R.C. Specific proteins. In Henry’s Clinical Diagnosis and Management by Laboratory Methods, 24th ed.; McPherson, R.A., Pincus, M.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 280–289. [Google Scholar]
- Huang, N.; Chen, J.; Wei, Y.; Liu, Y.; Yuan, K.; Chen, J.; He, M.; Liu, N. Multi-marker approach using C-reactive protein, procalcitonin, neutrophil CD64 index for the prognosis of sepsis in intensive care unit: A retrospective cohort study. BMC Infect. Dis. 2022, 22, 662. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, X.; Shi, P. Recent advances in biomarkers for detection and diagnosis of sepsis and organ dysfunction: A comprehensive review. Eur. J. Med. Res. 2025, 30, 1081. [Google Scholar] [CrossRef] [PubMed]
- Nowak, J.; Ssanyu, J.N.; Namiiro, F.; Mountford, N.; Parducci, A.; Domijan, K.; Daly, M.; O’Brien, D.; Barden, E.; Walshe, K.; et al. Diagnosis of neonatal and adult sepsis using a Serum Amyloid A lateral flow test. PLoS ONE 2025, 20, e0314702. [Google Scholar] [CrossRef] [PubMed]
- Arnon, S.; Litmanovitz, I.; Regev, R.; Lis, M.; Shainkin-Kestenbaum, R.; Dolfin, T. Serum amyloid A protein in the early detection of late-onset bacterial sepsis in preterm infants. J. Perinat. Med. 2002, 30, 329–332. [Google Scholar] [CrossRef]
- Arnon, S.; Litmanovitz, I.; Regev, R.H.; Bauer, S.; Shainkin-Kestenbaum, R.; Dolfin, T. Serum amyloid A: An early and accurate marker of neonatal early-onset sepsis. J. Perinatol. 2007, 27, 297–302. [Google Scholar] [CrossRef]
- Abbas, M.H.; Choudhry, M.N.; Hamza, N.; Ali, B.; Amin, A.A.; Ammori, B.J. Admission levels of serum amyloid a and procalcitonin are more predictive of the diagnosis of acute appendicitis compared with C-reactive protein. Surg. Laparosc. Endosc. Percutan. Tech. 2014, 24, 488–494. [Google Scholar] [CrossRef]
- Stute, M.; Kreysing, M.; Zorn, M.; Michl, P.; Gauss, A. Serum Amyloid A as a Potential Biomarker in Inflammatory Bowel Diseases, Especially in Patients with Low C-Reactive Protein. Int. J. Mol. Sci. 2024, 25, 1177. [Google Scholar] [CrossRef]
- Jung, S.Y.; Park, M.C.; Park, Y.B.; Lee, S.K. Serum amyloid a as a useful indicator of disease activity in patients with ankylosing spondylitis. Yonsei Med. J. 2007, 48, 218–224. [Google Scholar] [CrossRef]
- Chen, R.; Ren, S.; Tang, H.; Wu, Q. Diagnostic value of Serum Amyloid A (SAA) in HIV-associated pulmonary infections and its correlation with inflammatory markers. BMC Infect. Dis. 2025, 25, 1096. [Google Scholar] [CrossRef]
- Xu, S.; Song, Z.; Han, F.; Zhang, C. Effect of appropriate empirical antimicrobial therapy on mortality of patients with Gram-negative bloodstream infections: A retrospective cohort study. BMC Infect. Dis. 2023, 23, 344. [Google Scholar] [CrossRef]
- Lamy, B.; Dargère, S.; Arendrup, M.C.; Parienti, J.-J.; Tattevin, P. How to Optimize the Use of Blood Cultures for the Diagnosis of Bloodstream Infections? A State-of-the Art. Front. Microbiol. 2016, 7, 697. [Google Scholar] [CrossRef]
- VanEpps, J.S. Reducing exposure to broad-spectrum antibiotics for bloodstream infection. J. Lab. Precis. Med. 2018, 3, 100. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Principles and Procedures for Blood Cultures, 2nd ed.; CLSI Guideline M47-ED2; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2022. [Google Scholar]
- Shah, C.; Hari-Dass, R.; Raynes, J.G. Serum amyloid A is an innate immune opsonin for Gram-negative bacteria. Blood 2006, 108, 1751–1757. [Google Scholar] [CrossRef]
- Selinger, M.J.; McAdam, K.P.; Kaplan, M.M.; Sipe, J.D.; Vogel, S.N.; Rosenstreich, D.L. Monokine-induced synthesis of serum amyloid A protein by hepatocytes. Nature 1980, 285, 498–500. [Google Scholar] [CrossRef] [PubMed]
- Benson, M.D.; Kleiner, E. Synthesis and secretion of serum amyloid protein A (SAA) by hepatocytes in mice treated with casein. J. Immunol. 1980, 124, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Liu, Y.; Zou, Y.; Ye, R.D. Recent Advances in Studies of Serum Amyloid A: Implications in Inflammation, Immunity and Tumor Metastasis. Int. J. Mol. Sci. 2025, 26, 987. [Google Scholar] [CrossRef]
- Hagihara, K.; Nishikawa, T.; Sugamata, Y.; Song, J.; Isobe, T.; Taga, T.; Yoshizaki, K. Essential role of STAT3 in cytokine-driven NF-kappaB-mediated serum amyloid A gene expression. Genes Cells 2005, 10, 1051–1063. [Google Scholar] [CrossRef]
- Bae, J.; Choi, M.H.; Park, Y.; Kim, J.-H. Clinical Utility of the Serum Amyloid A Test for the Diagnosis of Bacterial Infections in Patients Visiting the Emergency Room. Lab. Med. Qual. Assur. 2023, 45, 156–165. [Google Scholar]
- Mayer, J.M.; Raraty, M.; Slavin, J.; Kemppainen, E.; Fitzpatrick, J.; Hietaranta, A.; Puolakkainen, P.; Beger, H.G.; Neoptolemos, J.P. Serum amyloid A is a better early predictor of severity than C-reactive protein in acute pancreatitis. Br. J. Surg. 2002, 89, 163–171. [Google Scholar]
- Sack, G.H., Jr. Serum amyloid A—A review. Mol. Med. 2018, 24, 46. [Google Scholar]
- Cunnane, G.; Grehan, S.; Geoghegan, S.; McCormack, C.; Shields, D.; Whitehead, A.S.; Bresnihan, B.; Fitzgerald, O. Serum amyloid A in the assessment of early inflammatory arthritis. J. Rheumatol. 2000, 27, 58–63. [Google Scholar]
- Takata, S.; Wada, H.; Tamura, M.; Koide, T.; Higaki, M.; Mikura, S.I.; Yasutake, T.; Hirao, S.; Nakamura, M.; Honda, K.; et al. Kinetics of c-reactive protein (CRP) and serum amyloid A protein (SAA) in patients with community-acquired pneumonia (CAP), as presented with biologic half-life times. Biomarkers 2011, 16, 530–535. [Google Scholar] [CrossRef]
- Wang, H.; Yao, J.; Huang, B.; Xu, B.; Wang, D. Meta-analysis of serum amyloid A for the diagnosis of neonatal sepsis: A comprehensive evaluation of diagnostic accuracy and clinical utility. Medicine 2025, 104, e44197. [Google Scholar] [CrossRef]
- Arbutina, D.D.; Milic, L.; Cuk, V.V.; Juloski, J.T.; Radulovic, R.; Starcevic, A.; Karamarkovic, A.R. Significance of Biomarkers in Early Diagnosis of Abdominal Sepsis. Chirurgia 2022, 117, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Bengnér, J.; Quttineh, M.; Gäddlin, P.-O.; Salomonsson, K.; Faresjö, M. Serum amyloid A—A prime candidate for identification of neonatal sepsis. Clin. Immunol. 2021, 229, 108787. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xiang, X.; Ren, H.; Xu, L.; Zhao, L.; Chen, X.; Long, H.; Wang, Q.; Wu, Q. Serum Amyloid A is a biomarker of severe Coronavirus Disease and poor prognosis. J. Infect. 2020, 80, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Filev, R.; Lyubomirova, M.; Bogov, B.; Kalinov, K.; Hristova, J.; Svinarov, D.; Rostaing, L. IL-6 and SAA—Strong Predictors for the Outcome in COVID-19 CKD Patients. Int. J. Mol. Sci. 2024, 25, 311. [Google Scholar] [CrossRef]
- Liu, S.L.; Wang, S.Y.; Sun, Y.F.; Jia, Q.Y.; Yang, C.L.; Cai, P.J.; Li, J.Y.; Wang, L.; Chen, Y. Expressions of SAA, CRP, and FERR in different severities of COVID-19. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 11386–11394. [Google Scholar]
- Xie, M.; Min, Z.; Jiang, W.; He, Z.; Xia, X. Prognostic value of multivariate logistic regression analysis and amyloid A lactate monitoring in patients with severe pneumonia-associated sepsis. BMC Pulm. Med. 2025, 25, 191. [Google Scholar] [CrossRef]




| Spearman’s rs (95% CI) | p | |
|---|---|---|
| CRP | 0.884 (0.876 to 0.891) | <0.001 |
| PCT | 0.576 (0.552 to 0.599) | <0.001 |
| WBC | 0.231 (0.197 to 0.264) | <0.001 |
| ANC | 0.300 (0.267 to 0.331) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kang, H.; Park, S. Diagnostic Performance and Prognostic Value of Serum Amyloid A in Patients with Bloodstream Infection. Diagnostics 2026, 16, 1510. https://doi.org/10.3390/diagnostics16101510
Kang H, Park S. Diagnostic Performance and Prognostic Value of Serum Amyloid A in Patients with Bloodstream Infection. Diagnostics. 2026; 16(10):1510. https://doi.org/10.3390/diagnostics16101510
Chicago/Turabian StyleKang, Hyein, and Sunggyun Park. 2026. "Diagnostic Performance and Prognostic Value of Serum Amyloid A in Patients with Bloodstream Infection" Diagnostics 16, no. 10: 1510. https://doi.org/10.3390/diagnostics16101510
APA StyleKang, H., & Park, S. (2026). Diagnostic Performance and Prognostic Value of Serum Amyloid A in Patients with Bloodstream Infection. Diagnostics, 16(10), 1510. https://doi.org/10.3390/diagnostics16101510





