Continuous Wearable-Sensor Monitoring After Colorectal Surgery: A Systematic Review of Clinical Outcomes and Predictive Analytics
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol & Registration
2.2. Search Strategy and Selection Criteria
2.3. Data Extraction and Quality Appraisal
2.4. Outcomes and Data Synthesis
2.5. Statistical Considerations
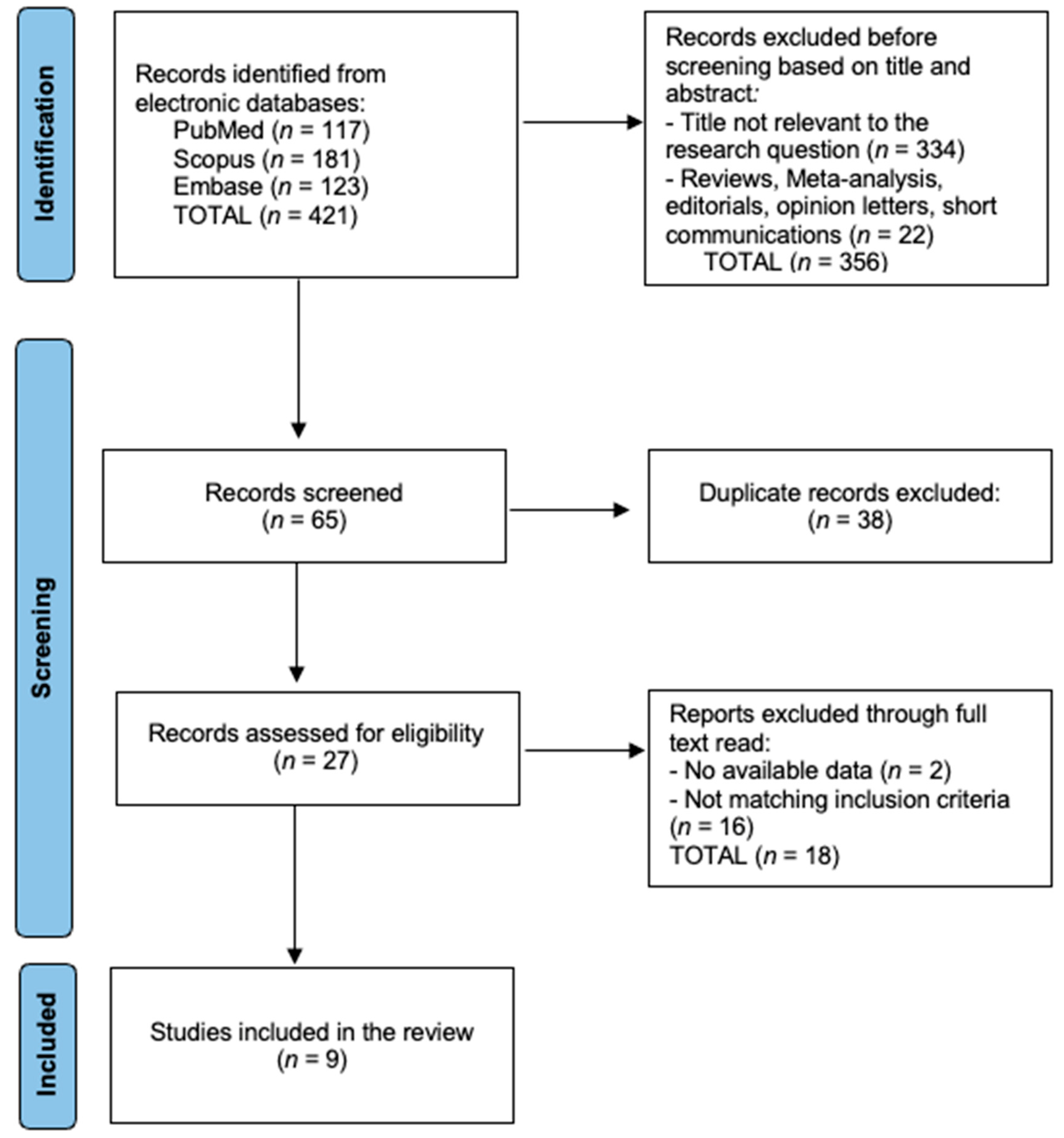
3. Results
4. Discussion
4.1. Summary of Evidence
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dawes, A.J.; Lin, A.Y.; Varghese, C.; Russell, M.M.; Lin, A.Y. Mobile health technology for remote home monitoring after surgery: A meta-analysis. Br. J. Surg. 2021, 108, 1304–1314. [Google Scholar] [CrossRef] [PubMed]
- Talen, A.D.; Leenen, J.P.L.; van der Sluis, G.; Oldenhuis, H.K.E.; Klaase, J.M.; Patijn, G.A. Feasibility of a Comprehensive eCoach to Support Patients Undergoing Colorectal Surgery: Longitudinal Observational Study. JMIR Perioper. Med. 2025, 8, e67425. [Google Scholar] [CrossRef]
- Richards, S.J.G.; Jerram, P.M.; Brett, C.; Falloon, M.; Frizelle, F.A. The association between low pre-operative step count and adverse post-operative outcomes in older patients undergoing colorectal cancer surgery. Perioper. Med. 2020, 9, 20. [Google Scholar] [CrossRef]
- Dornbush, C.; Mishra, A.; Hrabe, J.; Guyton, K.; Axelrod, D.; Blum, J.; Gribovskaja-Rupp, I. Remote monitoring after elective colorectal surgery, a pilot study. Surgery 2025, 179, 108791. [Google Scholar] [CrossRef]
- Leenen, J.P.L.; Ardesch, V.; Patijn, G. Remote Home Monitoring of Continuous Vital Sign Measurements by Wearables in Patients Discharged After Colorectal Surgery: Observational Feasibility Study. JMIR Perioper. Med. 2023, 6, e45113. [Google Scholar] [CrossRef] [PubMed]
- Balvardi, S.; Pecorelli, N.; Castelino, T.; Niculiseanu, P.; Alhashemi, M.; Liberman, A.S.; Charlebois, P.; Stein, B.; Carli, F.; Mayo, N.E.; et al. Impact of Facilitation of Early Mobilization on Postoperative Pulmonary Outcomes After Colorectal Surgery: A Randomized Controlled Trial. Ann. Surg. 2021, 273, 868–875. [Google Scholar] [CrossRef]
- Tang, J.; Liu, X.; Ma, T.; Lv, X.; Jiang, W.; Zhang, J.; Lu, C.; Chen, H.; Li, W.; Li, H.; et al. Application of enhanced recovery after surgery during the perioperative period in infants with Hirschsprung’s disease—A multi-center randomized clinical trial. Clin. Nutr. 2020, 39, 2062–2069. [Google Scholar] [CrossRef]
- Jones, A.S.K.; Kleinstäuber, M.; Akroyd, A.; Mittendorf, A.; Bognuda, P.; Merrie, A.E.H.; Eva, L.; Fernandez, J.; Petrie, K.J. Using animated visualization to improve postoperative mobilization: A randomized controlled trial. Health Psychol. 2019, 38, 748–758. [Google Scholar] [CrossRef]
- Min, J.; An, K.Y.; Park, H.; Cho, W.; Jung, H.J.; Chu, S.H.; Cho, M.; Yang, S.Y.; Jeon, J.Y.; Kim, N.K. Postoperative inpatient exercise facilitates recovery after laparoscopic surgery in colorectal cancer patients: A randomized controlled trial. BMC Gastroenterol. 2023, 23, 127. [Google Scholar] [CrossRef] [PubMed]
- Tazreean, R.; Nelson, G.; Twomey, R. Early mobilization in enhanced recovery after surgery pathways: Current evidence and recent advancements. J. Comp. Eff. Res. 2022, 11, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Ayyoubzadeh, S.M.; Baniasadi, T.; Shirkhoda, M.; Rostam Niakan Kalhori, S.; Mohammadzadeh, N.; Roudini, K.; Ghalehtaki, R.; Memari, F.; Jalaeefar, A. Remote Monitoring of Colorectal Cancer Survivors Using a Smartphone App and Internet of Things-Based Device: Development and Usability Study. JMIR Cancer 2023, 9, e42250. [Google Scholar] [CrossRef]
- Xiao, K.; Yeung, J.C.; Bolger, J.C. The safety and acceptability of using telehealth for follow-up of patients following cancer surgery: A systematic review. Eur. J. Surg. Oncol. 2023, 49, 9–15. [Google Scholar] [CrossRef]
- Bertocchi, E.; Barugola, G.; Masini, G.; Guerriero, M.; Menestrina, N.; Gentile, I.; Meoli, F.; Sanfilippo, L.; Lauria, M.; Freoni, R.; et al. iColon, a patient-focused mobile application for perioperative care in colorectal surgery: Results from 444 patients. J. Telemed. Telecare 2025, 31, 585–591. [Google Scholar] [CrossRef]
- Ștefănescu, V.C.; Ionescu, A.M.; Florea, S.F.; Vasile, M.A.; Bătăilă, V.; Cochior, D. Breaking surgical barriers: ERAS in action in Romania. J. Med. Life 2025, 18, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Ward, W.H.; Meeker, C.R.; Handorf, E.; Hill, M.V.; Einarson, M.; Alpaugh, R.K.; Holden, T.L.; Astsaturov, I.; Denlinger, C.S.; Hall, M.J.; et al. Feasibility of Fitness Tracker Usage to Assess Activity Level and Toxicities in Patients with Colorectal Cancer. JCO Clin. Cancer Inform. 2021, 5, 125–133. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Fiore, J.F., Jr.; Castelino, T.; Pecorelli, N.; Niculiseanu, P.; Balvardi, S.; Hershorn, O.; Liberman, S.; Charlebois, P.; Stein, B.; Carli, F.; et al. Ensuring Early Mobilization Within an Enhanced Recovery Program for Colorectal Surgery: A Randomized Controlled Trial. Ann. Surg. 2017, 266, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Daskivich, T.J.; Houman, J.; Lopez, M.; Luu, M.; Fleshner, P.; Zaghiyan, K.; Cunneen, S.; Burch, M.; Walsh, C.; Paiement, G.; et al. Association of Wearable Activity Monitors with Assessment of Daily Ambulation and Length of Stay Among Patients Undergoing Major Surgery. JAMA Netw. Open 2019, 2, e187673. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hedrick, T.L.; Hassinger, T.E.; Myers, E.; Krebs, E.D.; Chu, D.; Charles, A.N.; Hoang, S.C.; Friel, C.M.; Thiele, R.H. Wearable Technology in the Perioperative Period: Predicting Risk of Postoperative Complications in Patients Undergoing Elective Colorectal Surgery. Dis. Colon. Rectum. 2020, 63, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.; Romain, B.; Pache, B.; Vuagniaux, A.; Guarnero, V.; Hahnloser, D.; Demartines, N.; Hübner, M. Physical Activity and Outcomes in Colorectal Surgery: A Pilot Prospective Cohort Study. Eur. Surg. Res. 2020, 61, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Yin, T.C.; Huang, C.W.; Tsai, H.L.; Su, W.C.; Ma, C.J.; Chang, T.K.; Wang, J.Y. Smartband Use During Enhanced Recovery After Surgery Facilitates Inpatient Recuperation Following Minimally Invasive Colorectal Surgery. Front. Surg. 2021, 7, 608950. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kane, W.J.; Hassinger, T.E.; Myers, E.L.; Chu, D.L.; Charles, A.N.; Hoang, S.C.; Friel, C.M.; Thiele, R.H.; Hedrick, T.L. Wearable technology and the association of perioperative activity level with 30-day readmission among patients undergoing major colorectal surgery. Surg. Endosc. 2022, 36, 1584–1592. [Google Scholar] [CrossRef] [PubMed]
- Wilnerzon Thörn, R.M.; Forsberg, A.; Stepniewski, J.; Hjelmqvist, H.; Magnuson, A.; Ahlstrand, R.; Ljungqvist, O. Immediate mobilization in post-anesthesia care unit does not increase overall postoperative physical activity after elective colorectal surgery: A randomized, double-blinded controlled trial within an enhanced recovery protocol. World J. Surg. 2024, 48, 956–966. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Wang, P.P.; Wang, Z.Y.; Lan, G.R.; Xu, K.W.; Yu, C.H.; Wu, B. Innovative integration of lung ultrasound and wearable monitoring for predicting pulmonary complications in colorectal surgery: A prospective study. World J. Gastrointest. Surg. 2024, 16, 2649–2661. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Inoue, Y.; Kimura, T.; Noro, H.; Yoshikawa, M.; Nomura, M.; Yumiba, T.; Taniguchi, E.; Ohashi, S.; Souda, S.; Matsuda, H. Is laparoscopic colorectal surgery less invasive than classical open surgery? Quantitation of physical activity using an accelerometer to assess postoperative convalescence. Surg. Endosc. 2003, 17, 1269–1273. [Google Scholar] [CrossRef]
- Fuchita, M.; Ridgeway, K.J.; Kimzey, C.; Melanson, E.L.; Fernandez-Bustamante, A. Accelerometer-Measured Inpatient Physical Activity and Associated Outcomes After Major Abdominal Surgery: Systematic Review. Interact. J. Med. Res. 2023, 12, e46629. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Karlsson, P.; Nygren-Bonnier, M.; Henningsohn, L.; Rydwik, E.; Hagströmer, M. The feasibility of using a digital tool to enhance mobilisation following abdominal cancer surgery—A non-randomised controlled trial. Pilot. Feasibility Stud. 2023, 9, 147. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Xu, W.; Wells, C.I.; Seo, S.H.; Sebaratnam, G.; Calder, S.; Gharibans, A.; Bissett, I.P.; O’Grady, G. Feasibility and Accuracy of Wrist-Worn Sensors for Perioperative Monitoring During and After Major Abdominal Surgery: An Observational Study. J. Surg. Res. 2024, 301, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Pooni, A.; Brar, M.S.; Anpalagan, T.; Schmocker, S.; Rashid, S.; Goldstein, R.; Goriawala, A.; Easson, A.; Kennedy, E.D. Home to Stay: A Randomized Controlled Trial Evaluating the Effect of a Postdischarge Mobile App to Reduce 30-Day Readmission Following Elective Colorectal Surgery. Ann. Surg. 2023, 277, e1056–e1062. [Google Scholar] [CrossRef] [PubMed]
- Voigt, K.R.; Wullaert, L.; Höppener, D.J.; Schreinemakers, J.M.J.; Doornebosch, P.G.; Verseveld, M.; Peeters, K.; Verhoef, C.; Husson, O.; Grünhagen, D. Patient-led home-based follow-up after surgery for colorectal cancer: The protocol of the prospective, multicentre FUTURE-primary implementation study. BMJ Open 2023, 13, e074089. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kavallieros, K.; Karakozis, L.; Hayward, R.; Giannas, E.; Selvaggi, L.; Kontovounisios, C. Wearable Devices in Colorectal Surgery: A Scoping Review. Cancers 2024, 16, 2303. [Google Scholar] [CrossRef]
- Liao, Y.T.; Chou, Y.J.; Wu, C.T.; Liu, Y.H.; Liang, J.T.; Lai, F.; Shun, S.C. Wearable mobile health device for monitoring postoperative ambulation among patients with colorectal cancer undergoing minimally invasive surgery: A prospective comparison study. Int. J. Med. Robot. 2024, 20, e2626. [Google Scholar] [CrossRef] [PubMed]
- Hogenbirk, R.N.M.; Wijma, A.G.; van der Plas, W.Y.; Hentzen, J.E.K.R.; de Bock, G.H.; van der Schans, C.P.; Kruijff, S.; Klaase, J.M. Actual physical activity after major abdominal cancer surgery: Far from optimal. Eur. J. Surg. Oncol. 2024, 50, 107949. [Google Scholar] [CrossRef] [PubMed]
- Huber, H.M.; Wei, I.H.; Abbass, M.A.; Karagkounis, G.; Widmar, M.; Smith, J.J.; Nash, G.M.; Weiser, M.R.; Paty, P.B.; Garcia-Aguilar, J.; et al. Impact of Patient-Reported Outcome Monitoring via Recovery Tracker on Post-Discharge Outcomes After Colorectal Surgery: A Comparative Analysis Before and After Implementation. Cancers 2025, 17, 1939. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, Y.I.; Park, I.J.; Ro, J.S.; Lee, J.L.; Kim, C.W.; Yoon, Y.S.; Lim, S.B.; Yu, C.S.; Lee, Y.; Tak, Y.W.; et al. A randomized controlled trial of a digital lifestyle intervention involving postoperative patients with colorectal cancer. npj Digit. Med. 2025, 8, 296. [Google Scholar] [CrossRef]

| Study | Year | Country | Design | n | Wearable/Sensor Metric(s) | Monitoring Window | Primary Endpoint(s) |
|---|---|---|---|---|---|---|---|
| Fiore et al. [17] | 2017 | Canada | RCT | 99 | Pedometer (step count) | POD 0–3 | Out-of-bed time, LOS |
| Daskivich et al. [18] | 2019 | USA | Prospective cohort | 100 | Fitbit Charge HR (steps) | POD 0–2 | Prolonged LOS |
| Hedrick et al. [19] | 2020 | USA | Prospective cohort | 99 | Withings Pulse (pre- & post-op steps) | −30 d & POD 0–3 | Any complication |
| Martin et al. [20] | 2020 | Switzerland | Pilot cohort | 60 | ActiGraph GT9X (steps) | −5 d & POD 0-3 | Clavien-Dindo ≥ II |
| Yin et al. [21] | 2021 | Taiwan | Quasi-experimental | 90 | Xiaomi Mi Band (steps) | POD 1-discharge | LOS, QoR-40 |
| Kane et al. [22] | 2022 | USA | Prospective cohort | 94 | Fitbit Inspire HR (steps) | −30 d & POD 0-discharge | 30-d readmission |
| Wilnerzon Thörn et al. [23] | 2024 | Sweden | RCT | 144 | Axivity AX3 (steps) | PODs 1–3 | Steps PODs 1–3 |
| Lin et al. [24] | 2024 | China | Prospective cohort | 101 | Pedometer + HR | PODs 1–5 | Pulmonary complications |
| Inoue et al. [25] | 2003 | Japan | Comparative cohort | 91 | Uni-axial accelerometer | PODs 0–7 | Time to 90% activity recovery |
| Study | Key Exposure (Cut-Off) | Complications (%) | LOS (Days, Mean ± SD) | 30-d Readmission (%) | Effect Size |
|---|---|---|---|---|---|
| Fiore et al. [17] | Facilitated vs. standard mobilisation | 28.3 vs. 31.4 | 5.2 ± 1.1 vs. 5.4 ± 1.2 | 4.0 vs. 6.2 | ΔPOD1 steps + 843 |
| Daskivich et al. [18] | POD1 < 1000 vs. ≥1000 steps | 34 vs. 22 | OR prolonged LOS 0.63 (95% CI 0.45-0.84) | 8 vs. 6 | Spline plateau > 1000 steps |
| Hedrick et al. [19] | Pre-op inactive (<5000 steps d−1) | 55.9 vs. 27.5 | 6.8 ± 2.0 vs. 5.9 ± 1.7 | 18 vs. 9 | OR any complication 0.39 |
| Martin et al. [20] | Lowest vs. highest peri-op quartile | 46 vs. 20 | 7.1 ± 2.4 vs. 5.8 ± 1.9 | 12 vs. 6 | AUC complication 0.71 |
| Yin et al. [21] | Smart-band ERAS vs. standard | 6.7 vs. 10.0 | 7.8 ± 1.4 vs. 8.9 ± 1.6 | 0 vs. 3.3 | LOS − 1.1 d (p = 0.009) |
| Kane et al. [22] | Return-to-baseline < 28.9% | 38 vs. 23 | 6.1 ± 1.8 vs. 5.6 ± 1.6 | 34 vs. 9 | OR readmission 0.60 per 10% |
| Wilnerzon Thörn et al. [23] | PACU vs. ward mobilisation | 30.5 vs. 29.7 | 5.4 ± 1.3 vs. 5.3 ± 1.2 | 5.6 vs. 4.2 | NS difference in total steps (p = 0.21) |
| Lin et al. [24] | LUS ≥ 6 vs. <6 & ambulation tertiles | 31 vs. 9 | 8.1 ± 2.3 vs. 6.9 ± 1.9 | 21 vs. 9 | OR PPC 5.56 (p < 0.01) |
| Inoue et al. [25] | Laparoscopy vs. open | 18 vs. 31 | 9.0 ± 1.5 vs. 12.2 ± 2.3 | NA | ΔTime-to-90% activity − 3.4 days |
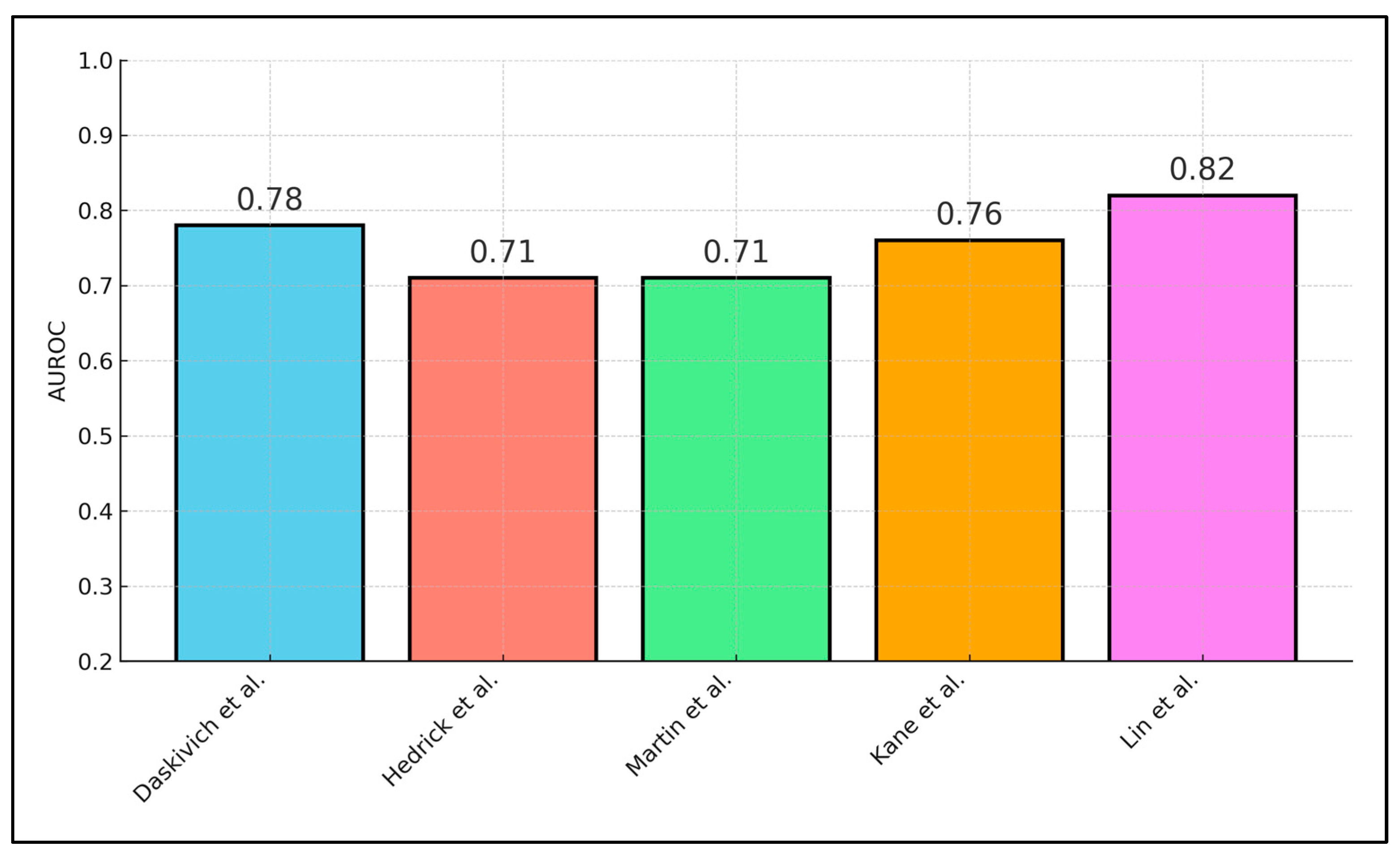
| Study | Sensor Variable(s) | Outcome Predicted | Model | AUROC/Key Statistic |
|---|---|---|---|---|
| Daskivich et al. [18] | POD1 step-count spline | Prolonged LOS | Logistic spline | AUROC 0.78 |
| Hedrick et al. [19] | Pre-op inactivity + NSQIP score | Any complication | Multivariable logistic | AUROC 0.71 (Δ + 0.05 vs. baseline) |
| Martin et al. [20] | Lowest quartile peri-op steps | Clavien ≥ II | ROC analysis | AUROC 0.71 |
| Kane et al. [22] | Return-to-baseline% | 30-d readmission | Optimal-cut-off ROC | AUROC 0.76; Sens 75%, Spec 69% |
| Lin et al. [24] | Distance + HRV + LUS | Pulmonary complications | Mixed logistic | AUROC 0.82 |
| Study | Wear Compliance (%) | Data-Loss Episodes | Device Removal Incidents | Additional Sensors | Notable Implementation Notes |
|---|---|---|---|---|---|
| Fiore et al. [17] | 98 | 2/594 patient-days | None | None | Staff checked pedometers twice daily |
| Daskivich et al. [18] | 95 | Bluetooth drop-outs in 8% records | 3 devices misplaced | None | Fitabase platform auto-uploads data |
| Hedrick et al. [19] | 93 | 5% missing pre-op days | 4 lost chargers | HR, sleep (unused analytically) | Pre-hab counselling improved adherence |
| Martin et al. [20] | 90 | 9% accelerometer gaps > 2 h | 2 water-damage events | None | Raw GT9X files processed in ActiLife |
| Yin et al. [21] | 97 | 3% sync failures | 1 strap allergy | None | Daily nurse-patient step-goal review |
| Kane et al. [22] | 88 | 12% un-synced post-discharge days | 5 device non-returns | HR, sleep | SMS reminders every 48 h to sync data |
| Wilnerzon Thörn et al. [23] | 92 | 7% data gaps | 6 patients removed due to comfort | None | Double-blinded pedometers taped to ankle |
| Lin et al. [24] | 94 | HR data loss 6% | Nil | HRV, skin-temp | Wearable synced with bedside tablet |
| Inoue et al. [25] | 85 | Analogue accelerometer malfunction 10% | NA | None | Pre-Bluetooth era; manual data download |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muntean, C.; Gaborean, V.; Faur, A.M.; Faur, I.F.; Prodan-Bărbulescu, C.; Feier, C.V.I. Continuous Wearable-Sensor Monitoring After Colorectal Surgery: A Systematic Review of Clinical Outcomes and Predictive Analytics. Diagnostics 2025, 15, 2194. https://doi.org/10.3390/diagnostics15172194
Muntean C, Gaborean V, Faur AM, Faur IF, Prodan-Bărbulescu C, Feier CVI. Continuous Wearable-Sensor Monitoring After Colorectal Surgery: A Systematic Review of Clinical Outcomes and Predictive Analytics. Diagnostics. 2025; 15(17):2194. https://doi.org/10.3390/diagnostics15172194
Chicago/Turabian StyleMuntean, Calin, Vasile Gaborean, Alaviana Monique Faur, Ionut Flaviu Faur, Cătălin Prodan-Bărbulescu, and Catalin Vladut Ionut Feier. 2025. "Continuous Wearable-Sensor Monitoring After Colorectal Surgery: A Systematic Review of Clinical Outcomes and Predictive Analytics" Diagnostics 15, no. 17: 2194. https://doi.org/10.3390/diagnostics15172194
APA StyleMuntean, C., Gaborean, V., Faur, A. M., Faur, I. F., Prodan-Bărbulescu, C., & Feier, C. V. I. (2025). Continuous Wearable-Sensor Monitoring After Colorectal Surgery: A Systematic Review of Clinical Outcomes and Predictive Analytics. Diagnostics, 15(17), 2194. https://doi.org/10.3390/diagnostics15172194








