Uterine Artery Pulsatility Index in Singleton Pregnancies Conceived via Assisted Reproductive Technology Versus Spontaneous Conception: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources and Search Strategy
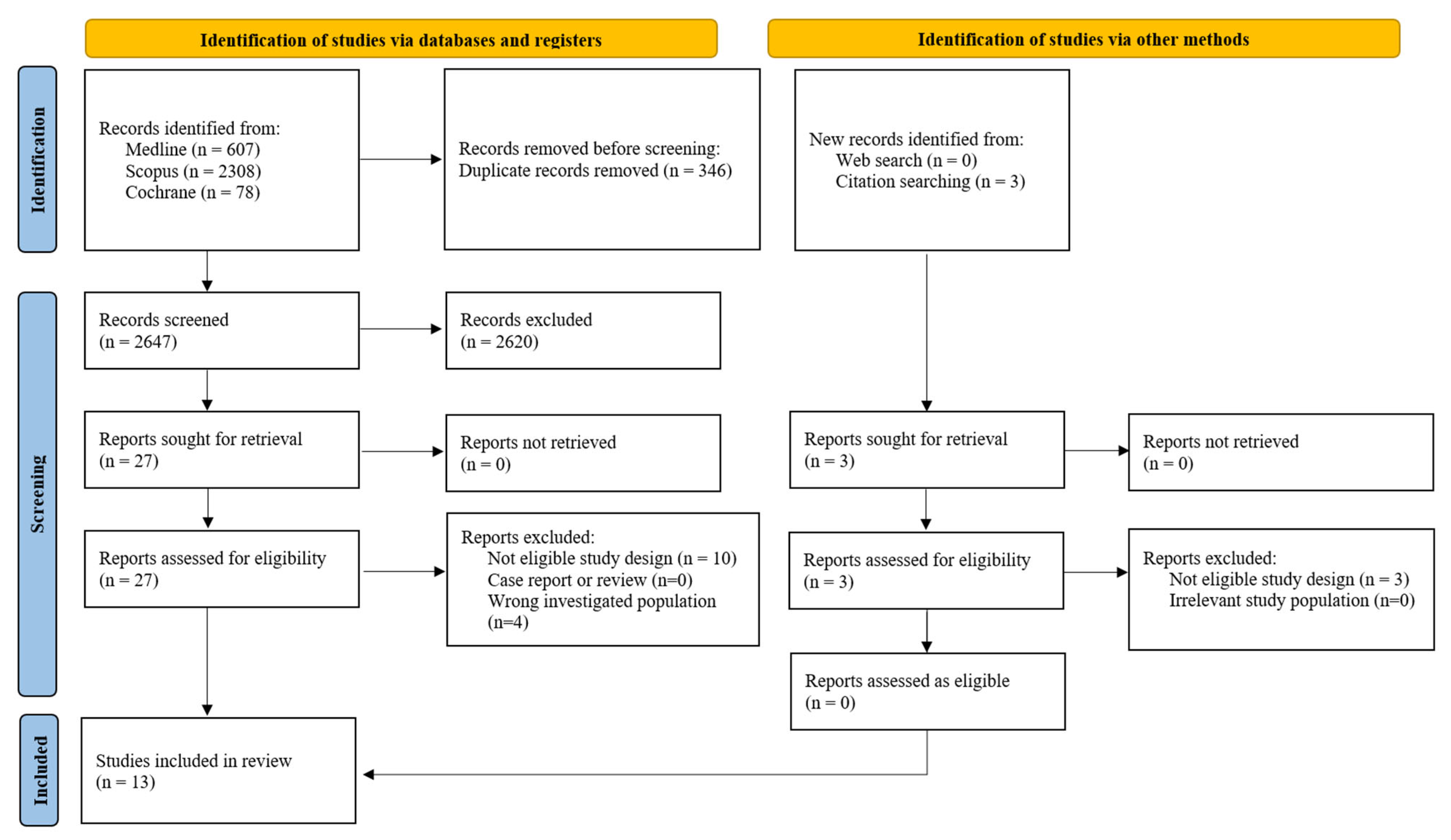
2.3. Study Selection
2.4. Outcomes
2.5. Data Extraction
2.6. Quality and Risk of Bias Assessment
2.7. Data Synthesis and Statistical Analysis
2.8. Confounders and Sensitivity Analysis
2.9. Subgroup Analyses
3. Results
3.1. Quality of the Included Studies
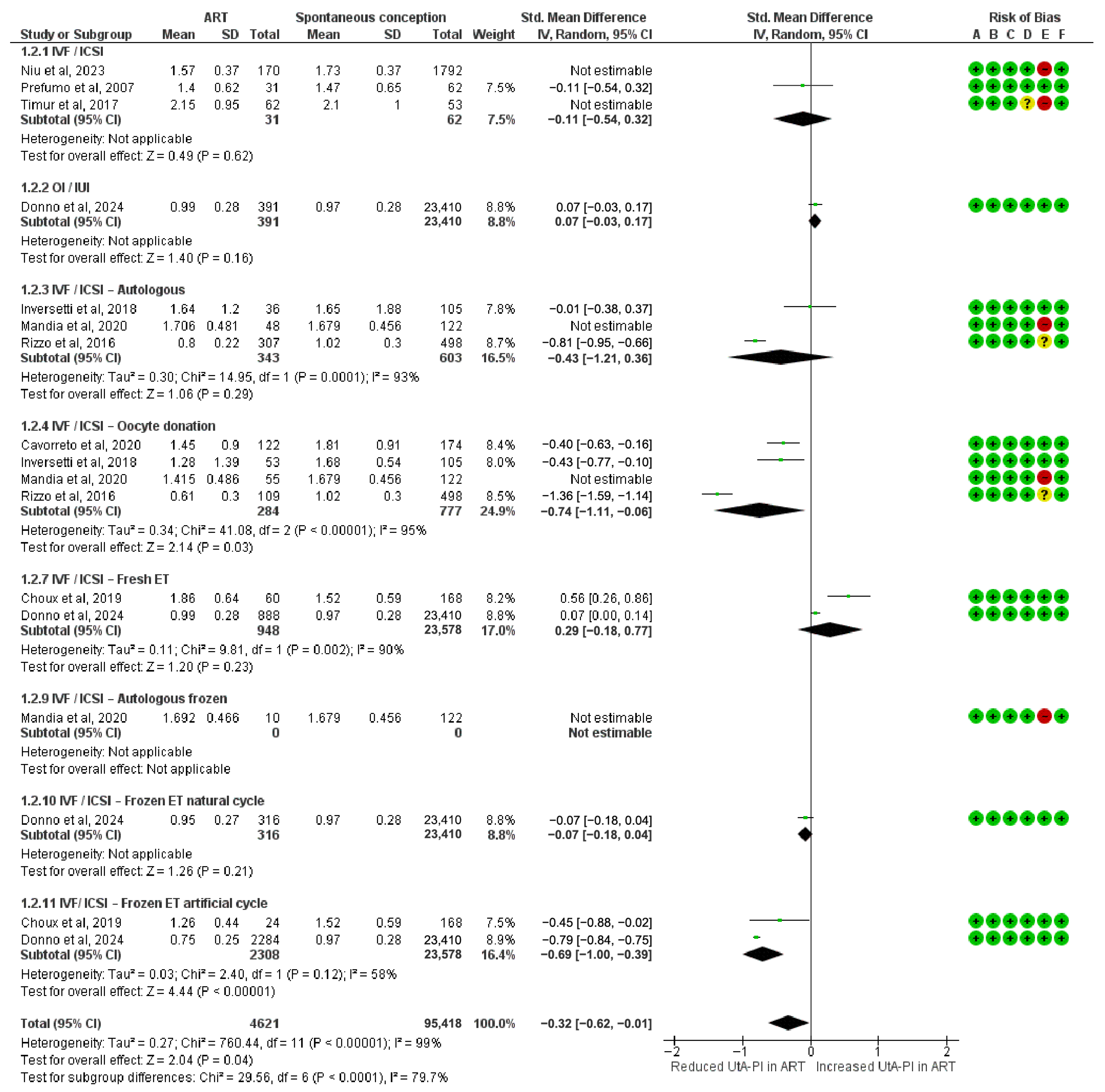
3.2. UtA PI Differences in the First Trimester
3.3. Risk of Bias Sensitivity Analysis
3.4. UtA-PI Differences in Second Trimester
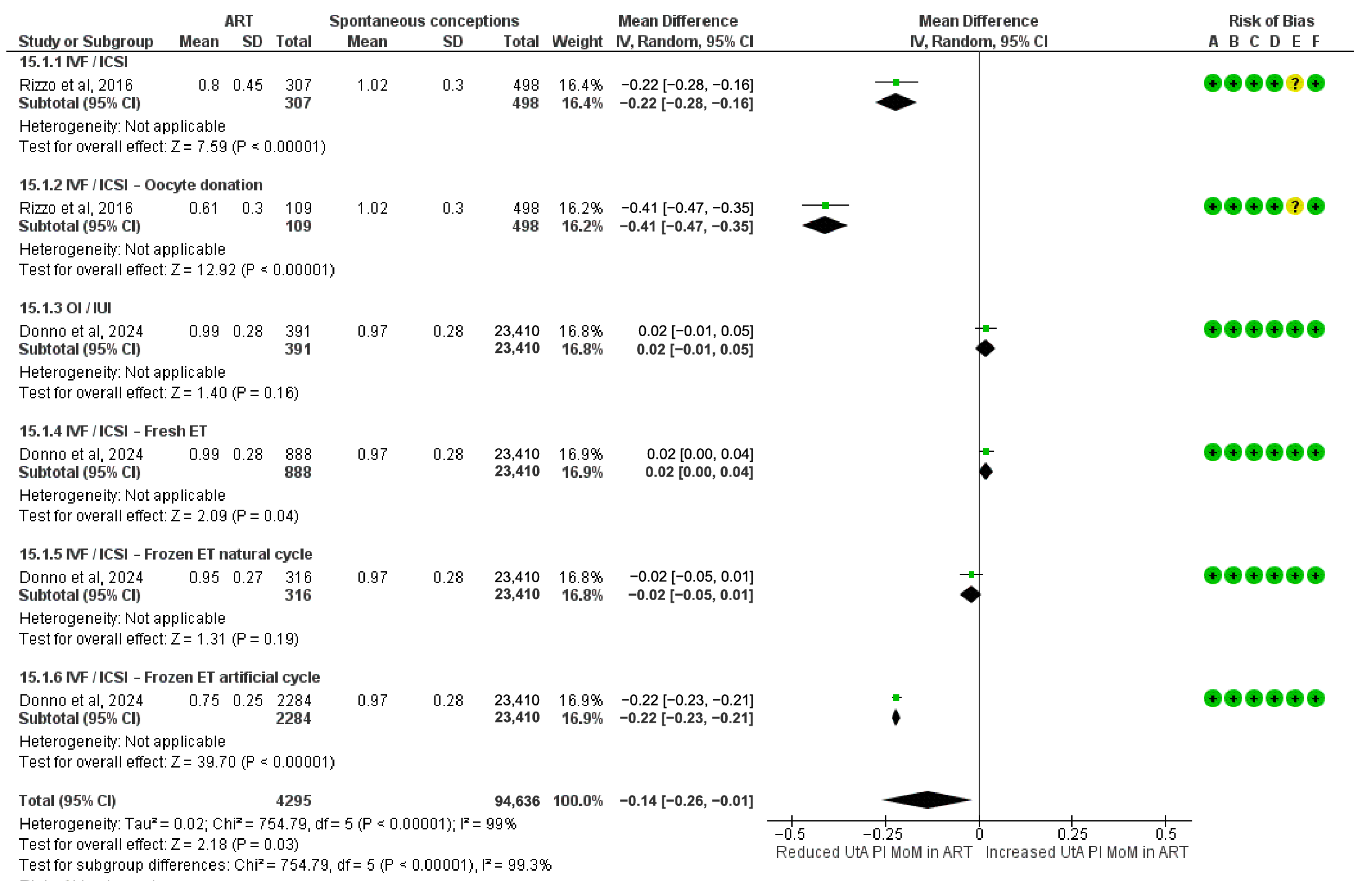
3.5. Meta-Analysis of UtA-PI MoM Values
3.6. Meta-Analysis of the Adjusted Coefficients from Multivariable Analyses
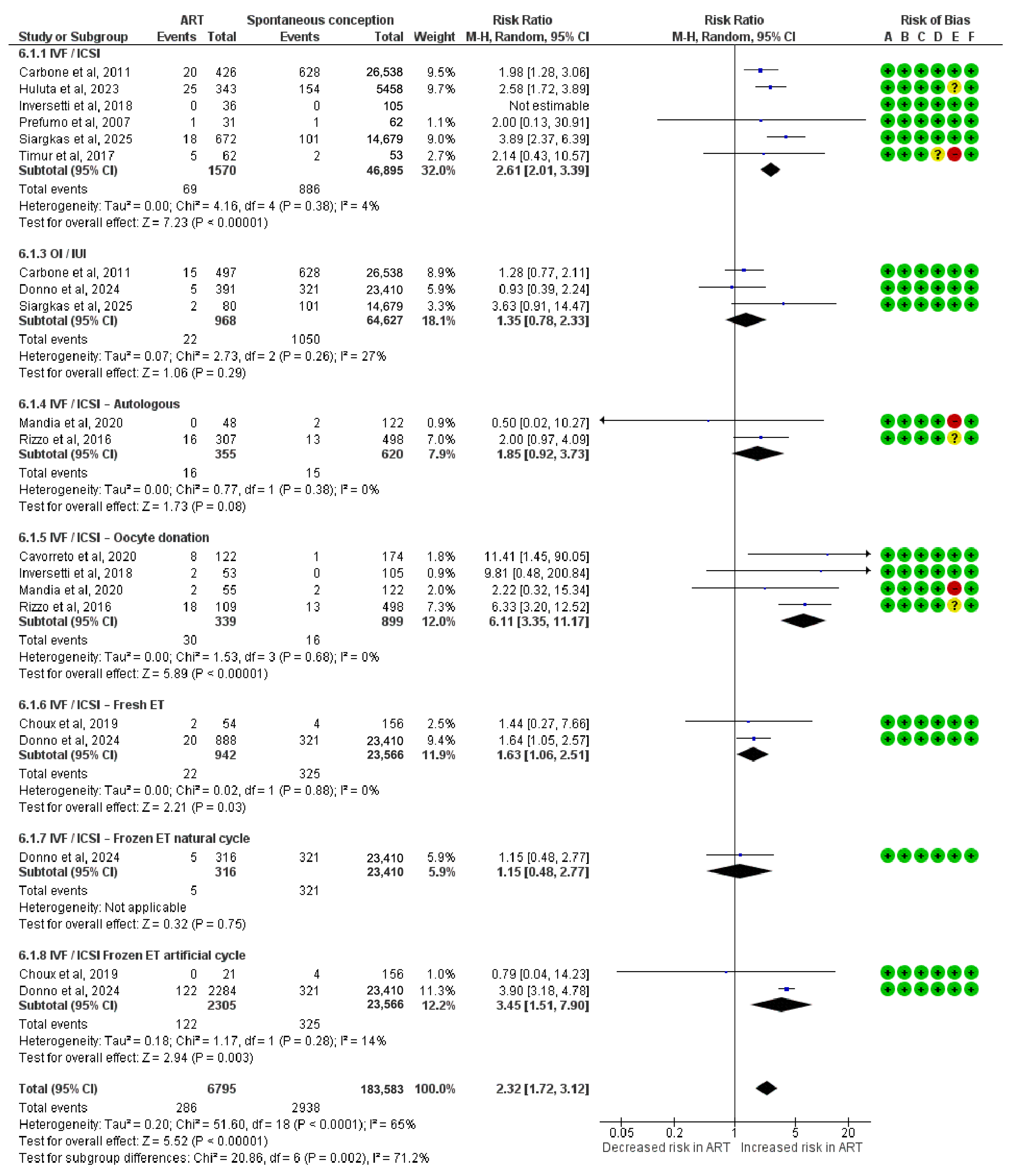
3.7. Risk for PE
3.8. Risk for SGA Neonates
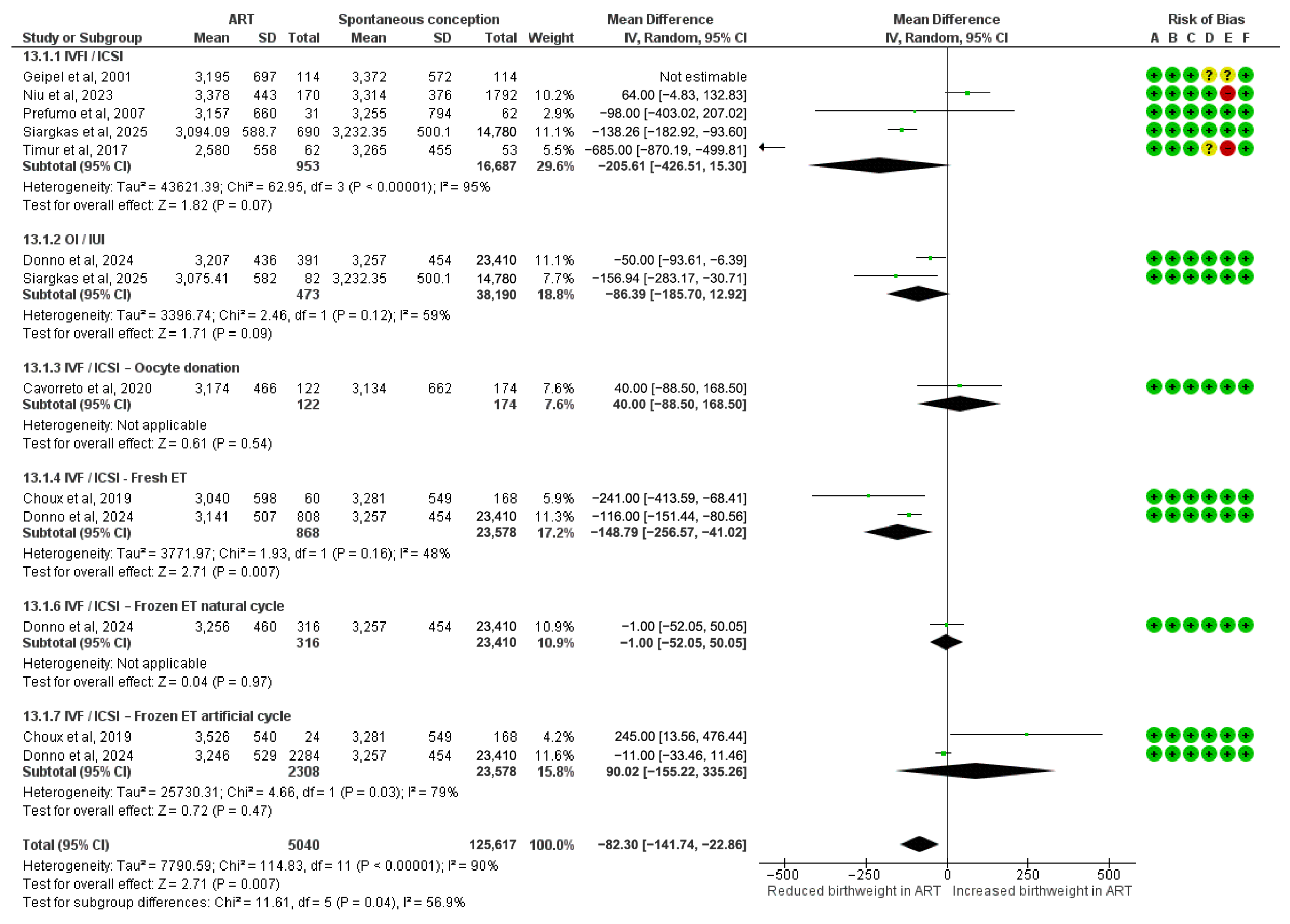
3.9. Mean Difference in Birthweight
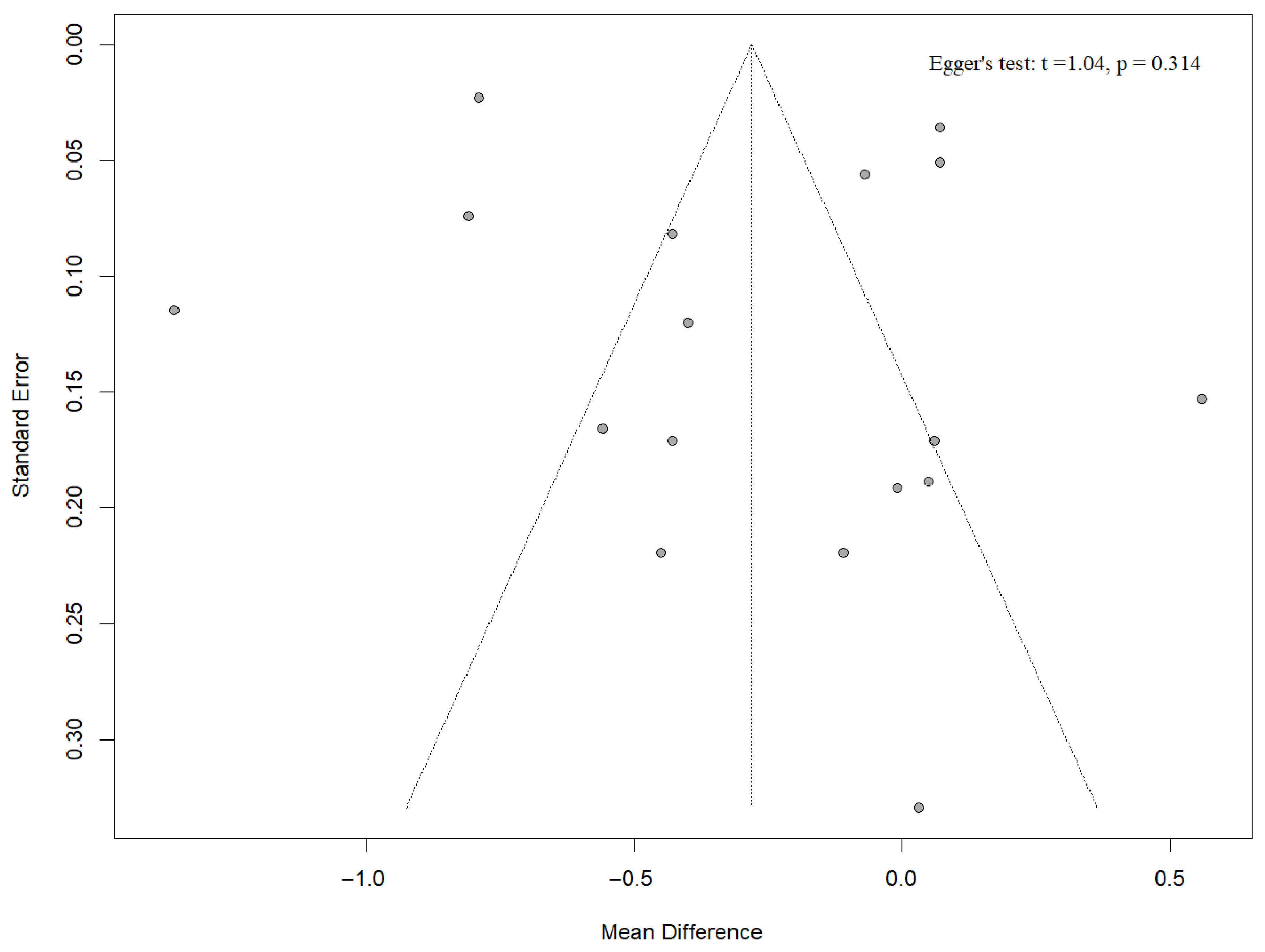
3.10. Publication Bias
4. Discussion
4.1. Primary Findings
4.2. Interpretation
4.3. IVF/ICSI Subgroup
4.4. Oocyte Donation and Autologous Transfer Subgroups
4.5. Artificial and Natural Cycle Subgroups
4.6. Rest of the Subgroups
4.7. Clinical Implications
4.8. Future Directions
4.9. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ART | Assisted Reproductive Technology |
| CI | Confidence Interval |
| Df | Degrees of Freedom |
| ET | Embryo Transfer |
| ICSI | Intracytoplasmic Sperm Injection |
| IUI | Intrauterine Insemination |
| IVF | In Vitro Fertilization |
| MD | Mean Differences |
| MoM | Multiples of the Median |
| OI | Ovulation Induction |
| PE | Preeclampsia |
| PI | Pulsatility Index |
| QUIPS | Quality In Prognosis Studies |
| RR | Risk Ratio |
| SC | Spontaneous Conceptions |
| SD | Standard Deviation |
| SGA | Small-for-gestational-age |
| SMD | Standardized Mean Difference |
| Std | Standardized |
| UtA | Uterine Artery |
| UtA-PI | Uterine Artery Pulsatility Index |
References
- Albaiges, G.; Missfelder-Lobos, H.; Lees, C.; Parra, M.; Nicolaides, K.H. One-stage screening for pregnancy complications by color Doppler assessment of the uterine arteries at 23 weeks’ gestation. Obstet. Gynecol. 2000, 96, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Papageorghiou, A.T.; Yu, C.K.; Bindra, R.; Pandis, G.; Nicolaides, K.H. Multicenter screening for pre-eclampsia and fetal growth restriction by transvaginal uterine artery Doppler at 23 weeks of gestation. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2001, 18, 441–449. [Google Scholar] [CrossRef]
- Papastefanou, I.; Mesaric, V.; Gomes Castello, R.; Nicolaides, K.H.; Charakida, M. At mid-gestation, markers of placental function rather than maternal cardiac function are stronger determinants of birthweight. Am. J. Obstet. Gynecol. 2025, 233, 127.e1–127.e8. [Google Scholar] [CrossRef]
- Papastefanou, I.; Gyokova, E.; Gungil, B.; Syngelaki, A.; Nicolaides, K.H. Prediction of adverse perinatal outcome at midgestation. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2023, 62, 195–201. [Google Scholar] [CrossRef]
- Say, L.; Chou, D.; Gemmill, A.; Tunçalp, Ö.; Moller, A.B.; Daniels, J.; Gülmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: A WHO systematic analysis. Lancet. Glob. Health 2014, 2, e323–e333. [Google Scholar] [CrossRef] [PubMed]
- Papastefanou, I.; Ashoor, G.; Syngelaki, A.; Akolekar, R.; Nicolaides, K.H. Relation of antepartum stillbirth to birthweight and gestational age: Prospective cohort study. BJOG Int. J. Obstet. Gynaecol. 2024, 131, 200–206. [Google Scholar] [CrossRef]
- Brosens, I.; Pijnenborg, R.; Vercruysse, L.; Romero, R. The “Great Obstetrical Syndromes” are associated with disorders of deep placentation. Am. J. Obstet. Gynecol. 2011, 204, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Duley, L. The global impact of pre-eclampsia and eclampsia. Semin. Perinatol. 2009, 33, 130–137. [Google Scholar] [CrossRef]
- Adjahou, S.; Logdanidis, V.; Wright, A.; Syngelaki, A.; Akolekar, R.; Nicolaides, K.H. Assessment of risk for pre-eclampsia at mid-gestation to define subsequent care. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2025, 65. [Google Scholar] [CrossRef]
- Rolnik, D.L.; Wright, D.; Poon, L.C.; O’Gorman, N.; Syngelaki, A.; de Paco Matallana, C.; Akolekar, R.; Cicero, S.; Janga, D.; Singh, M.; et al. Aspirin versus Placebo in Pregnancies at High Risk for Preterm Preeclampsia. N. Engl. J. Med. 2017, 377, 613–622. [Google Scholar] [CrossRef]
- Chen, Y.; Nguyen-Hoang, L.; Dinh, L.T.; Nguyen, D.A.; Pooh, R.K.; Shiozaki, A.; Zheng, M.; Hu, Y.; Ma, R.; Kusuma, A.; et al. Effect of aspirin on small for gestational age neonates in pregnancies at high-risk for preeclampsia: A secondary analysis of a cluster randomised clinical trial. Lancet Reg. Health West. Pac. 2025, 59, 101582. [Google Scholar] [CrossRef]
- Leung, H.H.Y.; Papastefanou, I.; Chen, Y.; Nguyen-Hoang, L.; Nguyen, D.A.; Dinh, L.T.; Pooh, R.K.; Shiozaki, A.; Zheng, M.; Hu, Y.; et al. Aspirin delays preterm birth in pregnancies at high risk for preterm pre-eclampsia: Evidence from randomized clinical trial in Asia. Ultrasound Obs. Gynecol. 2025, 66, 24–32. [Google Scholar] [CrossRef]
- Papastefanou, I.; Chen, Y.; Nguyen-Hoang, L.; Nguyen, D.A.; Dinh, L.T.; Pooh, R.K.; Shiozaki, A.; Zheng, M.; Hu, Y.; Wu, Y.; et al. Impact of Aspirin on Timing of Birth in Pregnancies With Clinical Manifestations of Placental Dysfunction: Evidence From a Multicentre Randomised Clinical Trial. Bjog 2025, 132, 1460–1468. [Google Scholar] [CrossRef]
- Papastefanou, I.; Wright, D.; Syngelaki, A.; Akolekar, R.; Nicolaides, K.H. Personalized stratification of pregnancy care for small for gestational age neonates from biophysical markers at midgestation. Am. J. Obstet. Gynecol. 2023, 229, 57.e1–57.e14. [Google Scholar] [CrossRef] [PubMed]
- Ashoor, G.; Syngelaki, A.; Papastefanou, I.; Nicolaides, K.H.; Akolekar, R. Development and validation of model for prediction of placental dysfunction-related stillbirth from maternal factors, fetal weight and uterine artery Doppler at mid-gestation. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2022, 59, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Papastefanou, I.; Menenez, M.; Szczepkowska, A.; Gungil, B.; Syngelaki, A.; Nicolaides, K.H. Comparison of competing-risks model with angiogenic factors in midgestation screening for preterm growth-related neonatal morbidity. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2024, 63, 613–618. [Google Scholar] [CrossRef]
- Nicolaides, K.H.; Papastefanou, I.; Syngelaki, A.; Ashoor, G.; Akolekar, R. Predictive performance for placental dysfunction related stillbirth of the competing risks model for small-for-gestational-age fetuses. BJOG Int. J. Obstet. Gynaecol. 2022, 129, 1530–1537. [Google Scholar] [CrossRef] [PubMed]
- Papastefanou, I.; Syngelaki, A.; Logdanidis, V.; Akolekar, R.; Nicolaides, K.H. Assessment of risk for small for gestational age at mid-gestation to define subsequent care. Am. J. Obs. Gynecol. 2025. [Google Scholar] [CrossRef]
- Yu, C.K.; Smith, G.C.; Papageorghiou, A.T.; Cacho, A.M.; Nicolaides, K.H. An integrated model for the prediction of preeclampsia using maternal factors and uterine artery Doppler velocimetry in unselected low-risk women. Am. J. Obs. Gynecol. 2005, 193, 429–436. [Google Scholar] [CrossRef]
- Schieve, L.A.; Meikle, S.F.; Ferre, C.; Peterson, H.B.; Jeng, G.; Wilcox, L.S. Low and very low birth weight in infants conceived with use of assisted reproductive technology. New Engl. J. Med. 2002, 346, 731–737. [Google Scholar] [CrossRef]
- Gui, J.; Ling, Z.; Hou, X.; Fan, Y.; Xie, K.; Shen, R. In vitro fertilization is associated with the onset and progression of preeclampsia. Placenta 2020, 89, 50–57. [Google Scholar] [CrossRef]
- Siargkas, A.; Tsakiridis, I.; Kappou, D.; Mamopoulos, A.; Papastefanou, I.; Dagklis, T. The Association Between Uterine Artery Pulsatility Index at Mid-Gestation and the Method of Conception: A Cohort Study. Medicina 2025, 61, 1093. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Aiello, E.; Pietrolucci, M.E.; Arduini, D. Placental volume and uterine artery Doppler evaluation at 11 + 0 to 13 + 6 weeks’ gestation in pregnancies conceived with in-vitro fertilization: Comparison between autologous and donor oocyte recipients. Ultrasound Obs. Gynecol. 2016, 47, 726–731. [Google Scholar] [CrossRef]
- Timur, H.; Çinar, M.; Hançerlioğullari, N.; Alkan, M.; İnal, H.A.; Uygur, D.; Yilmaz, N. Associations between first-trimester uterine artery Doppler velocimetry indices and adverse perinatal outcomes in women conceiving via in vitro fertilization. J. Matern. Fetal Neonatal Med. 2017, 30, 684–688. [Google Scholar] [CrossRef]
- Niu, Z.-H.; Ouyang, Y.-S.; Zhang, Y.-X.; Xu, Z.-H.; Yang, M.; Lu, J.; Wu, X.-N.; Zhang, P.-P.; Dai, Q.; Lv, K.; et al. The multiple reference range of mean uterine artery pulsatility index for natural and in vitro fertilization singletons during 11–14 gestational weeks. Quant. Imaging Med. Surg. 2023, 13, 8587–8598. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. Bmj 2021, 372, n160. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. Jama 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Zhang, J.T.; Lee, R.; Sauer, M.V.; Ananth, C.V. Risks of Placental Abruption and Preterm Delivery in Patients Undergoing Assisted Reproduction. JAMA Netw. Open 2024, 7, e2420970. [Google Scholar] [CrossRef]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, j.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis. 2000. [Google Scholar]
- Hayden, J.A.; van der Windt, D.A.; Cartwright, J.L.; Côté, P.; Bombardier, C. Assessing bias in studies of prognostic factors. Ann. Intern. Med. 2013, 158, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Carbone, I.F.; Cruz, J.J.; Sarquis, R.; Akolekar, R.; Nicolaides, K.H. Assisted conception and placental perfusion assessed by uterine artery Doppler at 11–13 weeks’ gestation. Hum. Reprod. 2011, 26, 1659–1664. [Google Scholar] [CrossRef]
- Cavoretto, P.I.; Farina, A.; Miglio, R.; Zamagni, G.; Girardelli, S.; Vanni, V.S.; Morano, D.; Spinillo, S.; Sartor, F.; Candiani, M. Prospective longitudinal cohort study of uterine arteries Doppler in singleton pregnancies obtained by IVF/ICSI with oocyte donation or natural conception. Hum. Reprod. 2020, 35, 2428–2438. [Google Scholar] [CrossRef] [PubMed]
- Choux, C.; Ginod, P.; Barberet, J.; Rousseau, T.; Bruno, C.; Sagot, P.; Astruc, K.; Fauque, P. Placental volume and other first-trimester outcomes: Are there differences between fresh embryo transfer, frozen-thawed embryo transfer and natural conception? Reprod. Biomed. Online 2019, 38, 538–548. [Google Scholar] [CrossRef]
- Donno, V.; Prats, P.; Rodriguez, I.; Polyzos, N.P. First-trimester uterine artery pulsatility index and preeclampsia risk in pregnancies after artificial frozen embryo transfer: Analysis of over 27,000 pregnancies. Am. J. Obs. Gynecol. 2025, 232, 464.e1–464.e9. [Google Scholar] [CrossRef]
- van Duijn, L.; Rousian, M.; Reijnders, I.F.; Willemsen, S.P.; Baart, E.B.; Laven, J.S.E.; Steegers-Theunissen, R.P.M. The influence of frozen-thawed and fresh embryo transfer on utero-placental (vascular) development: The Rotterdam Periconception cohort. Hum. Reprod. 2021, 36, 2091–2100. [Google Scholar] [CrossRef]
- Huluta, I.; Wright, A.; Cosma, L.M.; Dimopoulou, S.; Nicolaides, K.H.; Charakida, M. Fetal cardiac function at midgestation and conception by in-vitro fertilization. Ultrasound Obs. Gynecol. 2023, 61, 587–592. [Google Scholar] [CrossRef]
- Inversetti, A.; Mandia, L.; Candiani, M.; Cetin, I.; Larcher, A.; Savasi, V.; Papaleo, E.; Cavoretto, P. Uterine artery Doppler pulsatility index at 11-38 weeks in ICSI pregnancies with egg donation. J. Perinat. Med. 2018, 46, 21–27. [Google Scholar] [CrossRef]
- Mandia, L.; Cavoretto, P.; Duca, P.; Candiani, M.; Cetin, I.; Savasi, V. Evaluation of Uterine Artery Doppler and Estrogen Milieu in Oocyte Donation Pregnancies-A Pilot Study. Diagnostics 2020, 10, 254. [Google Scholar] [CrossRef]
- Prefumo, F.; Fratelli, N.; Soares, S.C.; Thilaganathan, B. Uterine artery Doppler velocimetry at 11-14 weeks in singleton pregnancies conceived by assisted reproductive technology. Ultrasound Obs. Gynecol. 2007, 29, 141–145. [Google Scholar] [CrossRef]
- Geipel, A.; Ludwig, M.; Germer, U.; Katalinic, A.; Diedrich, K.; Gembruch, U. Uterine artery Doppler velocimetry and the outcome of pregnancies resulting from ICSI. Hum. Reprod. 2001, 16, 1397–1402. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cnossen, J.S.; Morris, R.K.; ter Riet, G.; Mol, B.W.; van der Post, J.A.; Coomarasamy, A.; Zwinderman, A.H.; Robson, S.C.; Bindels, P.J.; Kleijnen, J.; et al. Use of uterine artery Doppler ultrasonography to predict pre-eclampsia and intrauterine growth restriction: A systematic review and bivariable meta-analysis. Cmaj 2008, 178, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Ramos, D.R.; Araujo Júnior, E.; Petrini, C.G.; Dulgheroff, F.F.; Caldas, T.; Peixoto, A.B. Increased pulsatility index of uterine artery Doppler between 26 and 28 weeks of gestation and adverse perinatal outcomes. J. Matern. Fetal Neonatal Med. 2022, 35, 4810–4817. [Google Scholar] [CrossRef]
- Megan, P.; Megan, H.; Andrew, S.; Lisa, S. The role of placental insufficiency in spontaneous preterm birth: A literature review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2024, 295, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.L.; Proctor, L.; Dodd, J.M.; Toal, M.; Okun, N.; Johnson, J.A.; Windrim, R.; Kingdom, J.C.P. Screening for Placental Insufficiency in High-risk Pregnancies: Is Earlier Better? Placenta 2008, 29, 1034–1040. [Google Scholar] [CrossRef]
- Tan, M.Y.; Syngelaki, A.; Poon, L.C.; Rolnik, D.L.; O’Gorman, N.; Delgado, J.L.; Akolekar, R.; Konstantinidou, L.; Tsavdaridou, M.; Galeva, S.; et al. Screening for pre-eclampsia by maternal factors and biomarkers at 11–13 weeks’ gestation. Ultrasound Obstet. Gynecol. 2018, 52, 186–195. [Google Scholar] [CrossRef]
- Chang, K.; Lubo, Z. Review article: Steroid hormones and uterine vascular adaptation to pregnancy. Reprod. Sci. 2008, 15, 336–348. [Google Scholar] [CrossRef]
- Bonagura, T.W.; Babischkin, J.S.; Aberdeen, G.W.; Pepe, G.J.; Albrecht, E.D. Prematurely elevating estradiol in early baboon pregnancy suppresses uterine artery remodeling and expression of extravillous placental vascular endothelial growth factor and α1β1 and α5β1 integrins. Endocrinology 2012, 153, 2897–2906. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, E.D.; Pepe, G.J. Estrogen regulation of placental angiogenesis and fetal ovarian development during primate pregnancy. Int. J. Dev. Biol. 2010, 54, 397–408. [Google Scholar] [CrossRef]
- Aberdeen, G.W.; Bonagura, T.W.; Harman, C.R.; Pepe, G.J.; Albrecht, E.D. Suppression of trophoblast uterine spiral artery remodeling by estrogen during baboon pregnancy: Impact on uterine and fetal blood flow dynamics. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, H1936–H1944. [Google Scholar] [CrossRef]
- Klatsky, P.C.; Delaney, S.S.; Caughey, A.B.; Tran, N.D.; Schattman, G.L.; Rosenwaks, Z. The role of embryonic origin in preeclampsia: A comparison of autologous in vitro fertilization and ovum donor pregnancies. Obs. Gynecol. 2010, 116, 1387–1392. [Google Scholar] [CrossRef]
- Thilaganathan, B. Association of Higher Maternal Blood Pressure With Lower Infant Birthweight. Hypertension 2016, 67, 499–500. [Google Scholar] [CrossRef] [PubMed]
- Raja, E.-A.; Bhattacharya, S.; Maheshwari, A.; McLernon, D.J. Comparison of perinatal outcomes after frozen or fresh embryo transfer: Separate analyses of singleton, twin, and sibling live births from a linked national in vitro fertilization registry. Fertil. Steril. 2022, 118, 323–334. [Google Scholar] [CrossRef]
- Wennerholm, U.-B.; Henningsen, A.-K.A.; Romundstad, L.B.; Bergh, C.; Pinborg, A.; Skjaerven, R.; Forman, J.; Gissler, M.; Nygren, K.G.; Tiitinen, A. Perinatal outcomes of children born after frozen-thawed embryo transfer: A Nordic cohort study from the CoNARTaS group. Hum. Reprod. 2013, 28, 2545–2553. [Google Scholar] [CrossRef]
- Bhide, A.; Acharya, G.; Baschat, A.; Bilardo, C.M.; Brezinka, C.; Cafici, D.; Ebbing, C.; Hernandez-Andrade, E.; Kalache, K.; Kingdom, J.; et al. ISUOG Practice Guidelines (updated): Use of Doppler velocimetry in obstetrics. Ultrasound Obstet. Gynecol. 2021, 58, 331–339. [Google Scholar] [CrossRef]
- Torricelli, M.; Reis, F.M.; Florio, P.; Severi, F.M.; Bocchi, C.; Picciolini, E.; Guidoni, C.G.; Petraglia, F. Low-molecular-weight heparin improves the performance of uterine artery Doppler velocimetry to predict preeclampsia and small-for-gestational age infant in women with gestational hypertension. Ultrasound Med. Biol. 2006, 32, 1431–1435. [Google Scholar] [CrossRef]
- Adibi, A.; Khadem, M.; Mardanian, F.; Hovsepian, S. Uterine and arcuate arteries blood flow for predicting of ongoing pregnancy in in vitro fertilization. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2015, 20, 879–884. [Google Scholar] [CrossRef]
- Cacciatore, B.; Simberg, N.; Fusaro, P.; Tiitinen, A. Transvaginal Doppler study of uterine artery blood flow in in vitro fertilization-embryo transfer cycles. Fertil. Steril. 1996, 66, 130–134. [Google Scholar] [CrossRef]
- Cacciatore, B.; Simberg, N.; Tiitinen, A.; Ylikorkala, O. Evidence of interplay between plasma endothelin-1 and 17 beta-estradiol in regulation of uterine blood flow and endometrial growth in infertile women. Fertil. Steril. 1997, 67, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.H.; Hsieh, T.T.; Chang, M.Y.; Shiau, C.S.; Hou, H.C.; Hsu, J.J.; Soong, Y.K. Prediction of pregnancy rate of in vitro fertilization and embryo transfer in women aged 40 and over with basal uterine artery pulsatility index. J. Assist. Reprod. Genet. 2000, 17, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Crosby, D.A.; Glover, L.E.; Downey, P.; Mooney, E.E.; McAuliffe, F.M.; O’Farrelly, C.; Brennan, D.J.; Wingfield, M. Mid-luteal uterine artery Doppler indices in the prediction of pregnancy outcome in nulliparous women undergoing assisted reproduction. Hum. Fertil. 2022, 25, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Ergin, R.N.; Yayla, M. Comparison of second trimester uterine artery pulsatility index between IVF and spontaneous twin pregnancies. Clin. Exp. Obs. Gynecol. 2015, 42, 614–616. [Google Scholar] [CrossRef]
- Fan, J.; Zhang, J.; Xu, S.; Liu, H.; Lv, W.; Bi, X.; Liu, Y.; Shi, W.; Zhang, Y.; Wu, X. The predictive value of uterine artery Doppler in the success rate of pregnancy from the first frozen embryo transfer during the implantation window. BMC Pregnancy Childbirth 2023, 23, 825. [Google Scholar] [CrossRef]
- Haapsamo, M.; Martikainen, H.; Räsänen, J. Low-dose aspirin and uterine haemodynamics on the day of embryo transfer in women undergoing IVF/ICSI: A randomized, placebo-controlled, double-blind study. Hum. Reprod. 2009, 24, 861–866. [Google Scholar] [CrossRef]
- Hoozemans, D.A.; Schats, R.; Lambalk, N.B.; Homburg, R.; Hompes, P.G. Serial uterine artery Doppler velocity parameters and human uterine receptivity in IVF/ICSI cycles. Ultrasound Obstet. Gynecol. Off. J. Int. Soc. Ultrasound Obstet. Gynecol. 2008, 31, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Shaikh, A.; Ratnani, R. Ultrasonography and Doppler Study to Predict Uterine Receptivity in Infertile Patients Undergoing Embryo Transfer. J. Obstet. Gynaecol. India 2016, 66, 377–382. [Google Scholar] [CrossRef]
- Li, L.; Du, M.; Wu, S.; Wen, C.; Kong, P.; Zhang, J.; Guan, Y. Analysis of the uterine artery pulsatility index on the day of endometrial transformation and pregnancy outcomes of patients undergoing frozen-thawed embryo transfer. Front. Endocrinol. 2024, 15, 1278504. [Google Scholar] [CrossRef]
- Ng, E.H.; Chan, C.C.; Tang, O.S.; Yeung, W.S.; Ho, P.C. Comparison of endometrial and subendometrial blood flow measured by three-dimensional power Doppler ultrasound between stimulated and natural cycles in the same patients. Hum. Reprod. 2004, 19, 2385–2390. [Google Scholar] [CrossRef] [PubMed]
- Salle, B.; Bied-Damon, V.; Benchaib, M.; Desperes, S.; Gaucherand, P.; Rudigoz, R.C. Preliminary report of an ultrasonography and colour Doppler uterine score to predict uterine receptivity in an in-vitro fertilization programme. Hum. Reprod. 1998, 13, 1669–1673. [Google Scholar] [CrossRef]
- Tayyar, A.; Guerra, L.; Wright, A.; Wright, D.; Nicolaides, K.H. Uterine artery pulsatility index in the three trimesters of pregnancy: Effects of maternal characteristics and medical history. Ultrasound Obs. Gynecol. 2015, 45, 689–697. [Google Scholar] [CrossRef]
- Zaidi, J.; Pittrof, R.; Shaker, A.; Kyei-Mensah, A.; Campbell, S.; Tan, S.L. Assessment of uterine artery blood flow on the day of human chorionic gonadotropin administration by transvaginal color Doppler ultrasound in an in vitro fertilization program. Fertil. Steril. 1996, 65, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Zollner, U.; Specketer, M.-T.; Zollner, K.-P.; Dietl, J. Uterine artery blood flow in the periimplantation period in embryo transfer cycles. Asian Pac. J. Reprod. 2012, 1, 177–182. [Google Scholar] [CrossRef]



| Study, Year | Study Period | Study Type | Country | Inclusion Criteria | Exclusion Criteria | ART Methods | UtA Doppler Measurements | Time of the Measurements | Adjustment |
|---|---|---|---|---|---|---|---|---|---|
| Carbone et al., 2011 [31] | March 2006–October 2009 | Prospective cohort study | UK | Singleton pregnancies, attending for their routine first hospital visit during pregnancy at King’s College Hospital, London, UK and Medway Maritime Hospital, Kent, UK. | Pregnancies conceived by IUI because data on OI drugs were unavailable, fetal aneuploidies or major defects diagnosed either prenatally or in the neonatal period, and pregnancies ending in termination for psychosocial reasons. | OI, IVF/ICSI | MoM UtA-PI | 11+0 to 13+6 weeks of gestation | Fetal crown-rump length, maternal age, maternal weight, maternal height, racial origin, cigarette smoking, method of conception, history of chronic hypertension, history of diabetes mellitus, parity, family history of preeclampsia |
| Cavoretto et al., 2020 [32] | January 2013–December 2018 | Prospective cohort study | Italy | Singleton pregnancies following IVF/ICSI with OD, and matched naturally conceived controls; light smoking (<5 cigarettes/day) and BMI as matching criteria. | Other ARTs (e.g., autologous IVF/ICSI, IUI, gamete/zygote intra-fallopian transfers), heavy smoking (≥5 cigarettes/day), twin pregnancies, fetal aneuploidies, major fetal defects, spontaneous abortions, insulin-dependent diabetes mellitus (type 1 or 2), cardiac, renal or liver diseases, chronic infections, overt autoimmune diseases (e.g., antiphospholipid syndrome, lupus), chronic hypertension (requiring treatment before pregnancy), chromosomal/genetic abnormalities, cancer history, multiple myomectomy with potential uterine artery damage. | IVF/ICSI with OD | Mean UtA-PI | 11+0 to 13+6, 19+0–21+6, 26+0–33+6 gestational weeks | GA, maternal weight, parity |
| Choux et al., 2019 [33] | 1 October 2013–31 January 2015 | Retrospective case–control study | France | Singleton IVF pregnancies (fresh ET or frozen ET), with dates between 1 October 2013 and 31 January 2015, followed at Dijon University Hospital for first-trimester ultrasound and placental acquisition. | Pre-existing maternal diseases (diabetes, chronic hypertension), OD, fetal malformation or abnormal karyotype. | IVF with either fresh ET or frozen ET | Mean UtA-PI, Presence of diastolic notching | 11+0 to 13+6 gestational weeks | GA |
| Donno et al., 2024 [34] | January 2010–May 2023 | Retrospective cohort study | Spain | Singleton pregnancies (natural conception or ART) with full pregnancy follow-up from first-trimester screening to delivery. | - | OI/IUI, IVF and fresh ET or frozen ET in natural cycle or frozen ET in artificial cycle | MoM UtA-PI | 11+0 to 13+6 gestational weeks | No |
| Duijn et al., 2021 [35] | January 2017–March 2018 | Prospective cohort study | The Netherlands | Women 18 years and older, before 10 weeks of gestation with a viable singleton pregnancy, were eligible for participation. | Pregnancies after OD, pregnancies resulting in a miscarriage and drop-outs were excluded. | fresh or frozen ET | Mean UtA-PI, RI | Transvaginal at 7th, 9th, 11+0 to 13+6 gestational weeks; Transabdominal at 22nd week of gestation | GA, parity, periconceptional folic acid supplement use, smoking |
| Huluta et al., 2023 [36] | August 2019–December 2021 | Prospective cohort study | UK | Women with singleton pregnancies attending routine ultrasound at 19+0 to 23+6 weeks, delivering a liveborn fetus without major congenital abnormalities. | Pregnancies with aneuploidy or major fetal abnormality. | IVF/ICSI | MoM UtA-PI | 19+0 to 23+6 gestational weeks | No |
| Inversetti et al., 2018 [37] | 2010–2015 | Prospective cohort study | Italy | Consecutive pregnant women (2010–2015) at two tertiary care centers, with different modes of conception. | Non-singleton pregnancy, aneuploidies, history of preeclampsia, history of hypertension or diabetes, autoimmune disease. | autologous ICSI from fresh transfer and heterologous by ICSI-OD | Mean UtA-PI | First trimester at 11+0 to 13+6 gestational weeks, second trimester at 14+0 to 23+6 gestational weeks | maternal age, BMI, race, parity, smoking status and GA |
| Mandia et al., 2020 [38] | January 2014–January 2016 | Prospective case–control study | Italy | Singleton pregnancies from nulliparous women with different methods of assisted conception or normal pregnancies, attending for routine first-trimester prenatal risk assessment. | Pre-existing maternal diseases (diabetes, chronic hypertension, renal disease, other significant diseases), fetal structural or chromosomal anomalies, vaginal bleeding or threatened miscarriage, multiple pregnancies or pregnancies with vanishing twins above 6 weeks of development (crown rump length > 10 mm), smoking status, data from patients lost to follow-up. | IVF with OD or autologous oocytes from fresh cycles or autologous oocytes from frozen cycles | Mean UtA-PI | 11+0 to 13+6 gestational weeks | No |
| Niu et al., 2023 [25] | December 2017–December 2020 | Prospective cohort study | China | Healthy women with ongoing first-trimester pregnancy, no tobacco, recreational drug use, or excessive alcohol (>50 mL/week), and adequate nutrition; viable singleton with crown rump length 45–84 mm; normal fetal growth (10th–90th percentile) on growth curves; spontaneous or IVF pregnancies. | Participants with pre-existing maternal diseases (e.g., diabetes, immune, cardiovascular, renal, liver, malignancy, gynecological or endocrine disorders, chronic hypertension, or history of preeclampsia/preterm birth), uterine malformations or surgeries (e.g., myomectomy), adverse pregnancy outcomes (fetal growth restriction, preeclampsia, eclampsia), and fetuses with chromosomal abnormalities, major defects, or spontaneous abortions. | IVF/ICSI | Mean UtA-PI, PSV, End-Diastolic Velocity | 11+0–13+6 gestational weeks | No |
| Prefumo et al., 2007 [39] | December 2001–April 2005 | Prospective case–control study | UK | Women with singleton pregnancies attending routine first-trimester Doppler. | Women with a known medical condition (e.g., diabetes mellitus, connective tissue disease, essential hypertension) or a history of recurrent miscarriage. | IVF/ICSI | Mean RI, PI, presence of notching | 11+0–13+6 gestational weeks | No |
| Rizzo et al., 2016 [23] | January 2007–January 2014 | Prospective cohort study | Italy | Singleton pregnancies with successful recordings of UtA Doppler waveforms, no pre-existing maternal diseases (diabetes, chronic hypertension, renal disease), no fetal anomalies, and exhaustive follow-up (delivered at the unit). | - | IVF with autologous and donor oocytes | MoM UtA-PI | 11+0–13+6 gestational weeks | No |
| Siargkas et al., 2025 [22] | February 2015–August 2024 | Retrospective cohort study | Greece | Consecutive singleton pregnancies receiving antenatal care at our institution, confirmed viable from 20+0 weeks until 23+6 weeks of gestation, that had also attended for the first trimester nuchal scan in the same department. | Multiple gestations, singleton pregnancies with known fetal genetic or structural anomalies, termination of pregnancy, miscarriages before 23+6 weeks, and incomplete follow-up data. | IVF/ICSI, OI/IUI | Mean UtA-PI | 20+0–23+6 gestational weeks | GA, maternal weight, height, age, parity, smoking status, history of previous cesarean section, pre-existing diabetes mellitus (type I or II), and pre-existing thyroid disease |
| Timur et al., 2017 [24] | May 2013–June 2015 | Prospective case–control study | Turkey | Healthy nulliparous women with singleton pregnancies following IVF, and matched controls with spontaneous singleton pregnancies. | Women with pre-existing diseases (diabetes, chronic hypertension, renal disease), or fetuses with structural or chromosomal anomalies. | IVF/ICSI | Left and right UtA PI, RI | First trimester | No |
| First Author, Year | Study Type | S1 | S2 | S3 | S4 | C | O1 | O2 | O3 | Total |
|---|---|---|---|---|---|---|---|---|---|---|
| Carbone et al., 2011 [31] | Prospective cohort study | b * | a * | a * | a * | a,b ** | b * | a * | a * | 9 |
| Cavoretto et al., 2020 [32] | Prospective cohort study | b * | a * | a * | a * | a,b ** | b * | a * | a * | 9 |
| Choux et al., 2019 [33] | Retrospective case–control study | a * | a * | a * | a * | a,b ** | a * | a * | a * | 9 |
| Donno et al., 2024 [34] | Retrospective cohort study | b * | a * | a * | a * | a,b ** | b * | a * | a * | 9 |
| Duijn et al., 2021 [35] | Prospective cohort study | a * | a * | a * | a * | a,b ** | a * | a * | a * | 9 |
| Huluta et al., 2023 [36] | Prospective cohort study | b * | a * | a * | a * | a * | b * | a * | a * | 7 |
| Inversetti et al., 2018 [37] | Prospective cohort study | b * | a * | a * | a * | a,b ** | b * | a * | a * | 9 |
| Mandia et al., 2020 [38] | Prospective case–control study | b * | a * | a * | a * | - | b * | a * | a * | 7 |
| Niu et al., 2023 [25] | Prospective cohort study | b * | a * | a * | a * | - | b * | a * | a * | 7 |
| Prefumo et al., 2007 [39] | Prospective case–control study | a * | a * | a * | a * | a,b ** | a * | a * | a * | 9 |
| Rizzo et al., 2016 [23] | Prospective cohort study | b * | a * | a * | a * | a * | b * | a * | a * | 7 |
| Siargkas et al., 2025 [22] | Retrospective cohort study | a * | a * | a * | a * | a,b ** | a * | a * | a * | 9 |
| Timur et al., 2017 [24] | Prospective case–control study | b * | a * | a * | a * | - | b * | a * | a * | 7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siargkas, A.; Tsakiridis, I.; Faka, A.; Kappou, D.; Papastefanou, I.; Dagklis, T. Uterine Artery Pulsatility Index in Singleton Pregnancies Conceived via Assisted Reproductive Technology Versus Spontaneous Conception: A Systematic Review and Meta-Analysis. Diagnostics 2025, 15, 2192. https://doi.org/10.3390/diagnostics15172192
Siargkas A, Tsakiridis I, Faka A, Kappou D, Papastefanou I, Dagklis T. Uterine Artery Pulsatility Index in Singleton Pregnancies Conceived via Assisted Reproductive Technology Versus Spontaneous Conception: A Systematic Review and Meta-Analysis. Diagnostics. 2025; 15(17):2192. https://doi.org/10.3390/diagnostics15172192
Chicago/Turabian StyleSiargkas, Antonios, Ioannis Tsakiridis, Areti Faka, Dimitra Kappou, Ioannis Papastefanou, and Themistoklis Dagklis. 2025. "Uterine Artery Pulsatility Index in Singleton Pregnancies Conceived via Assisted Reproductive Technology Versus Spontaneous Conception: A Systematic Review and Meta-Analysis" Diagnostics 15, no. 17: 2192. https://doi.org/10.3390/diagnostics15172192
APA StyleSiargkas, A., Tsakiridis, I., Faka, A., Kappou, D., Papastefanou, I., & Dagklis, T. (2025). Uterine Artery Pulsatility Index in Singleton Pregnancies Conceived via Assisted Reproductive Technology Versus Spontaneous Conception: A Systematic Review and Meta-Analysis. Diagnostics, 15(17), 2192. https://doi.org/10.3390/diagnostics15172192








