Evaluating the Effect of Bile Acid Levels on Maternal and Perinatal Outcomes in Intrahepatic Cholestasis of Pregnancy: A Retrospective Study
Abstract
1. Introduction
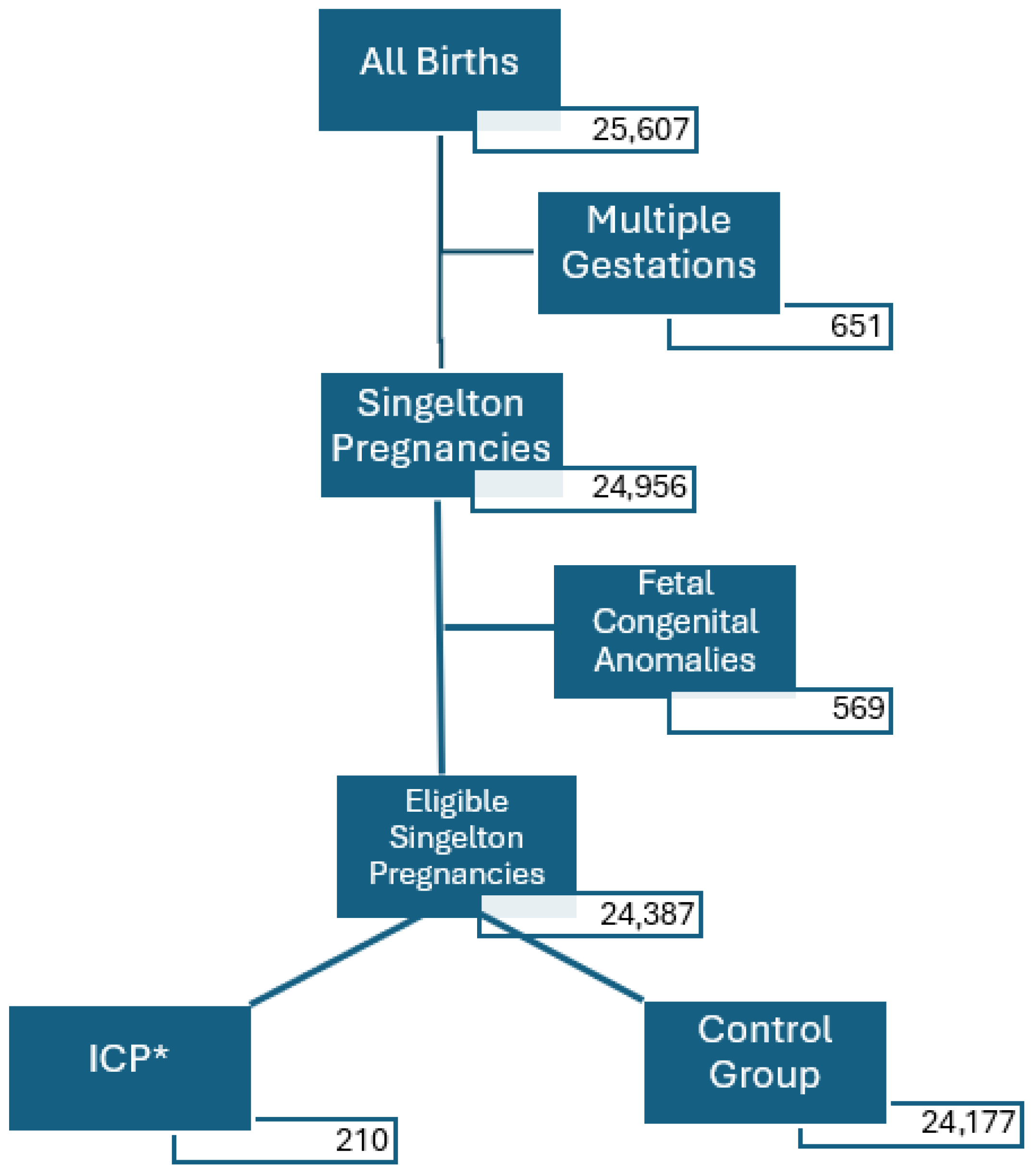
2. Materials and Methods
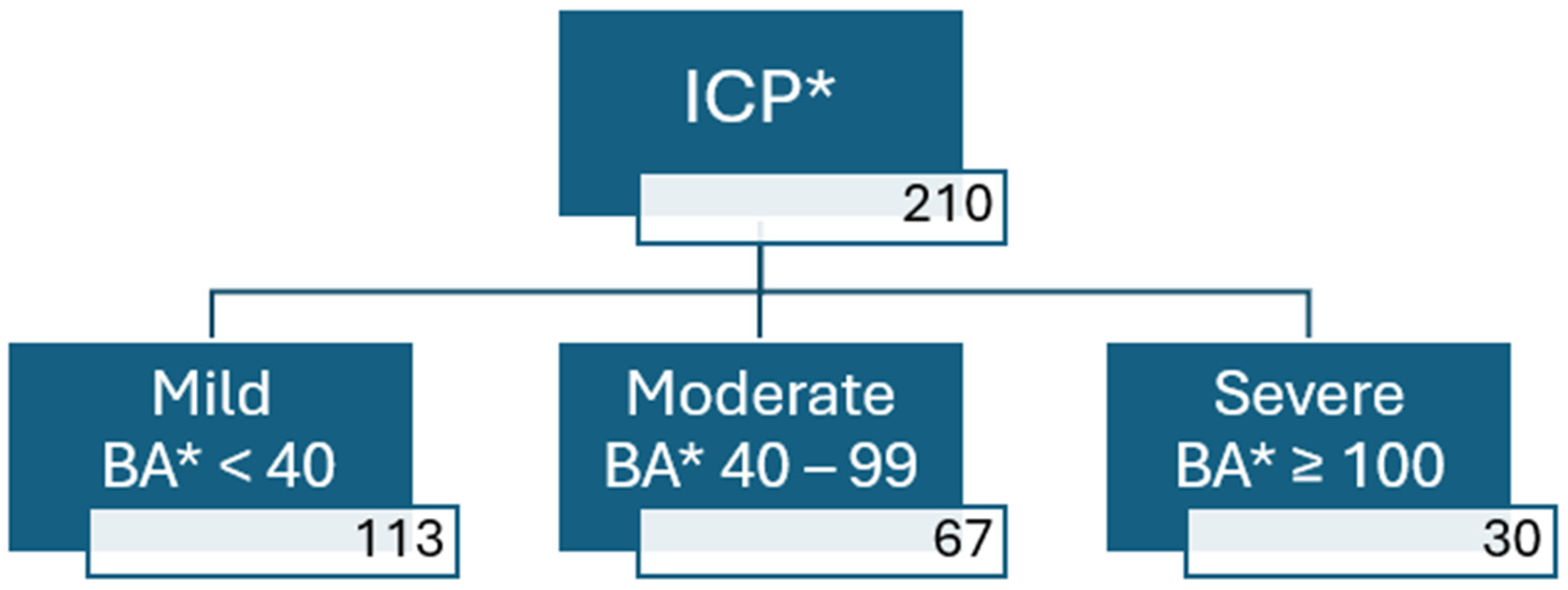
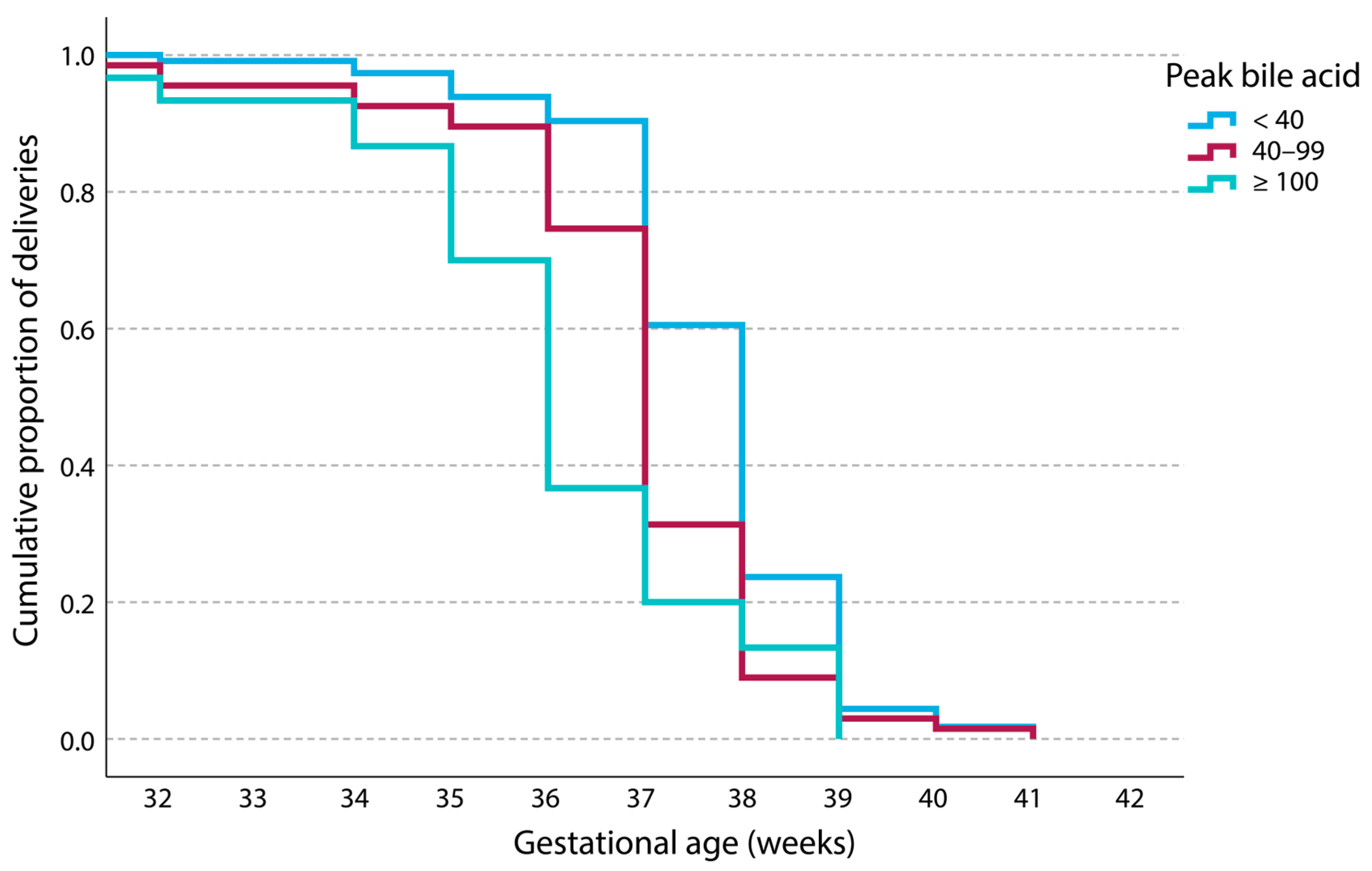
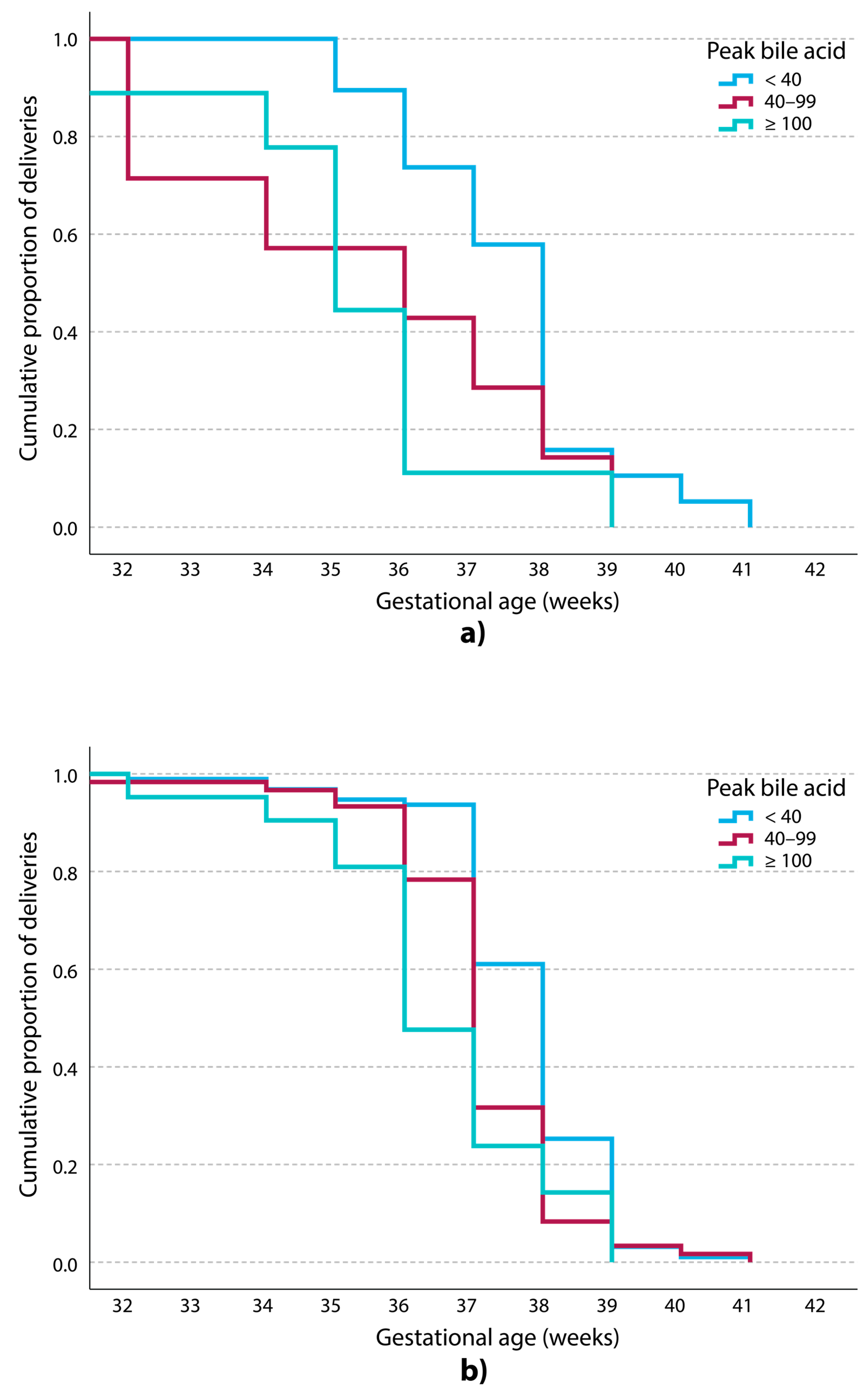
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lata, I. Hepatobiliary diseases during pregnancy and their management: An update. Int. J. Crit. Illn. Inj. Sci. 2013, 3, 175–182. [Google Scholar] [CrossRef]
- Mitchell, A.L.; Ovadia, C.; Syngelaki, A.; Souretis, K.; Martineau, M.; Girling, J.; Vasavan, T.; Fan, H.M.; Seed, P.T.; Chambers, J.; et al. Re-evaluating diagnostic thresholds for intrahepatic cholestasis of pregnancy: Case-control and cohort study. BJOG 2021, 128, 1635–1644. [Google Scholar] [CrossRef] [PubMed]
- Geenes, V.; Williamson, C. Intrahepatic cholestasis of pregnancy. World J. Gastroenterol. 2009, 15, 2049–2066. [Google Scholar] [CrossRef] [PubMed]
- You, P.; Ding, M.; Li, X.; Shao, Y.; Jiang, T.; Jia, Y.; Wang, Y.; Zhang, X. Determining urinary bile acid profiles to predict maternal and neonatal outcomes in patients with intrahepatic cholestasis of pregnancy. Diagnostics 2025, 15, 657. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.M.; Mitchell, A.L.; Williamson, C. ENDOCRINOLOGY IN PREGNANCY: Metabolic impact of bile acids in gestation. Eur. J. Endocrinol. 2021, 184, R69–R83. [Google Scholar] [CrossRef]
- Piechota, J.; Jelski, W. Intrahepatic cholestasis in pregnancy: Review of the literature. J. Clin. Med. 2020, 9, 1361. [Google Scholar] [CrossRef]
- Ovadia, C.; Sajous, J.; Seed, P.T.; Patel, K.; Williamson, N.J.; Attilakos, G.; Azzaroli, F.; Bacq, Y.; Batsry, L.; Broom, K.; et al. Ursodeoxycholic acid in intrahepatic cholestasis of pregnancy: A systematic review and individual participant data meta-analysis. Lancet Gastroenterol. Hepatol. 2021, 6, 547–558. [Google Scholar] [CrossRef]
- Majsterek, M.; Wierzchowska-Opoka, M.; Makosz, I.; Kreczyńska, L.; Kimber-Trojnar, Ż.; Leszczyńska-Gorzelak, B. Bile acids in intrahepatic cholestasis of pregnancy. Diagnostics 2022, 12, 2746. [Google Scholar] [CrossRef]
- Girling, J.; Knight, C.L.; Chappell, L. Intrahepatic cholestasis of pregnancy: Green-top Guideline No. 43. BJOG 2022, 129, e95–e114. [Google Scholar] [CrossRef]
- Nana, M.; Medina, V.; Maxwell, C.; McCormick, C.; Taliani, G.; Beuers, U.; Money, D.; Jacobsson, B.; Kapur, A.; Beyuo, T.; et al. FIGO guideline on liver disease and pregnancy. Int. J. Gynaecol. Obstet. 2025, 170, 28–48. [Google Scholar] [CrossRef]
- Huri, M.; Seravalli, V.; Lippi, C.; Tofani, L.; Galli, A.; Petraglia, F.; Di Tommaso, M. Intrahepatic cholestasis of pregnancy—Time to redefine the reference range of total serum bile acids: A cross-sectional study. BJOG 2022, 129, 1887–1896. [Google Scholar] [CrossRef]
- McIlvride, S.; Dixon, P.H.; Williamson, C. Bile acids and gestation. Mol. Asp. Med. 2017, 56, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Villar, J.; Cavoretto, P.I.; Barros, F.C.; Romero, R.; Papageorghiou, A.T.; Kennedy, S.H. Etiologically based functional taxonomy of the preterm birth syndrome. Clin. Perinatol. 2024, 51, 475–495. [Google Scholar] [CrossRef] [PubMed]
- Ovadia, C.; Seed, P.T.; Sklavounos, A.; Geenes, V.; Di Ilio, C.; Chambers, J.; Kohari, K.; Bacq, Y.; Bozkurt, N.; Brun-Furrer, R.; et al. Association of adverse perinatal outcomes of intrahepatic cholestasis of pregnancy with biochemical markers: Aggregate and individual patient data meta-analyses. Lancet 2019, 393, 899–909. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.H.; Greenberg, M.; Metz, T.D.; Pettker, C.M. Society for Maternal-Fetal Medicine Consult Series #53: Intrahepatic cholestasis of pregnancy. Am. J. Obstet. Gynecol. 2021, 224, B2–B9. [Google Scholar] [CrossRef]
- Zhou, Q.; Yuan, Y.; Wang, Y.; He, Z.; Liang, Y.; Qiu, S.; Chen, Y.; He, Y.; Lv, Z.; Liu, H. ICP severity and adverse outcomes beyond stillbirth: Evidence from 15,826 patients. BMC Pregnancy Childbirth 2024, 24, 476. [Google Scholar] [CrossRef]
- Herrera, C.A.; Manuck, T.A.; Stoddard, G.J.; Varner, M.W.; Esplin, S.; Clark, E.A.; Silver, R.M.; Eller, A.G. Perinatal outcomes associated with intrahepatic cholestasis of pregnancy. J. Matern. Fetal Neonatal Med. 2018, 31, 1913–1920. [Google Scholar] [CrossRef]
- Geenes, V.; Chappell, L.C.; Seed, P.T.; Steer, P.J.; Knight, M.; Williamson, C. Association of severe intrahepatic cholestasis of pregnancy with adverse pregnancy outcomes: A prospective population-based case–control study. Hepatology 2014, 59, 1482–1491. [Google Scholar] [CrossRef]
- Mashburn, S.; Schleckman, E.; Cackovic, P.; Shellhaas, C.; Rood, K.M.; Ma’ayeh, M. Risk factors for severe disease in intrahepatic cholestasis of pregnancy. J. Matern. Fetal Neonatal Med. 2022, 35, 8566–8570. [Google Scholar] [CrossRef]
- Ma, H.; Patti, M.E. Bile acids, obesity, and the metabolic syndrome. Best Pract. Res. Clin. Gastroenterol. 2014, 28, 573–583. [Google Scholar] [CrossRef]
- Granese, R.; Calagna, G.; Alibrandi, A.; Martinelli, C.; Romeo, P.; Filomia, R.; Ferraro, M.I.; Piccione, E.; Ercoli, A.; Saitta, C. Maternal and neonatal outcomes in intrahepatic cholestasis of pregnancy. J. Clin. Med. 2023, 12, 4407. [Google Scholar] [CrossRef]
- Martineau, M.; Raker, C.; Powrie, R.; Williamson, C. Intrahepatic cholestasis of pregnancy is associated with an increased risk of gestational diabetes. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 176, 80–85. [Google Scholar] [CrossRef]
- Gao, X.X.; Ye, M.Y.; Liu, Y.; Li, J.-Y.; Li, L.; Chen, W.; Lu, X.; Nie, G.; Chen, Y.-H. Prevalence and risk factors of intrahepatic cholestasis of pregnancy in a Chinese population. Sci. Rep. 2020, 10, 16307. [Google Scholar] [CrossRef]
- Valdovinos-Bello, V.; García-Romero, C.S.; Cervantes-Peredo, A.; García-Gómez, E.; Martínez-Ibarra, A.; Vázquez-Martínez, E.R.; Valdespino, Y.; Cerbón, M. Body mass index implications in intrahepatic cholestasis of pregnancy and placental histopathological alterations. Ann. Hepatol. 2023, 28, 100879. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xu, L.; Beejadhursing, R.; Li, F. Maternal and neonatal outcomes of intrahepatic cholestasis of pregnancy after in vitro fertilization. BMC Pregnancy Childbirth 2024, 24, 44. [Google Scholar] [CrossRef]
- Dumančić, S.; Mikuš, M.; Palčić, Z.; Habek, D.; Tešanović, M.; Mimica, M.D.; Marušić, J. Severe early-onset intrahepatic cholestasis of pregnancy following ovarian hyperstimulation: Case report and review. Life 2024, 14, 129. [Google Scholar] [CrossRef] [PubMed]
- Bolukbas, F.F.; Bolukbas, C.; Balaban, Y.; Aygun, C.; Ignak, S.; Ergul, E.; Yazicioglu, M.; Ersahin, S. Intrahepatic cholestasis of pregnancy: Spontaneous vs in vitro fertilization. Eur. J. Hepatogastroenterol. 2017, 7, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Alemdaroğlu, S.; Yılmaz Baran, Ş.; Durdağ, G.D.; Yuksel Şimşek, S.; Yetkinel, S.; Alkaş Yağınç, D.; Kalaycı, H.; Şimşek, E. Intrahepatic cholestasis of pregnancy: Are in vitro fertilization pregnancies at risk? J. Matern. Fetal Neonatal Med. 2021, 34, 2548–2553. [Google Scholar] [CrossRef]
- Tsur, A.; Leonard, S.A.; Kan, P.; Datoc, I.A.; Girsen, A.I.; Shaw, G.M.; Stevenson, D.K.; El-Sayed, Y.Y.; Druzin, M.L.; Blumenfeld, Y.J. Vaginal progesterone and intrahepatic cholestasis of pregnancy. Am. J. Perinatol. 2023, 40, 1158–1162. [Google Scholar] [CrossRef]
- Pillarisetty, L.S.; Sharma, A. Pregnancy intrahepatic cholestasis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK551503/ (accessed on 10 July 2025).
- Kamuli, S.M.; Kaiza, I.L.; Kihunrwa, A.; Chibwe, E.; Mabega, N.G. Prevalence of meconium-stained amniotic fluid and fetal outcomes in Tanzania. Eur. J. Prev. Med. 2024, 12, 94–102. [Google Scholar] [CrossRef]
- Berg, B.; Helm, G.; Petersohn, L.; Tryding, N. Cholestasis of pregnancy: Clinical and laboratory studies. Acta Obstet. Gynecol. Scand. 1986, 65, 107–113. [Google Scholar] [CrossRef]
- Sanhal, C.; Dağlar, K.; Kara, Ö.; Kirbas, A.; Uygur, D.; Yücel, A. Seasonal impact on the frequency of intrahepatic cholestasis of pregnancy. Gynecol. Obstet. Reprod. Med. 2016, 22, 5–9. [Google Scholar] [CrossRef]
- Bartolone, S.; Mayrovitz, H.N. Role of baby’s sex on itch severity and bile acid levels in intrahepatic cholestasis of pregnancy. Cureus 2021, 13, e14089. [Google Scholar] [CrossRef]
- Chappell, L.C.; Bell, J.L.; Smith, A.; Linsell, L.; Juszczak, E.; Dixon, P.H.; Chambers, J.; Hunter, R.; Dorling, J.; Williamson, C.; et al. Ursodeoxycholic acid versus placebo in intrahepatic cholestasis of pregnancy (PITCHES trial). Lancet 2019, 394, 849–860. [Google Scholar] [CrossRef]
| Baseline Characteristics of the Groups | ||
|---|---|---|
| Maternal age | Primipara/multipara | GDM * |
| Smoking | Mode of conception: IVF * | Season of birth |
| Pregestational BMI * | DM type 1 * | Sex of newborn |
| Maternal Treatment and Biochemical Markers | ||
| Use of progesterone | Use of cholestyramine | Use of ursodeoxycholic acid |
| Peak pregnancy bile acid levels | ||
| Maternal and Delivery Outcomes | ||
| Gestational age at delivery | Gestational hypertension | Preeclampsia |
| Caesarean section | Vacuum extraction delivery | Intrapartum haemorrhage >500 mL |
| Spontaneous preterm birth | Iatrogenic preterm birth ** | |
| Neonatal Outcome | ||
| Meconium-stained amniotic fluid | Apgar score <7 at 5 min *** | Umbilical artery pH |
| NCIU * admission | Birth weight | |
| ICP Group (N = 210) | Control Group (N = 24,177) | p-Value (%) | |
|---|---|---|---|
| Maternal age (years) mean (st. deviation) | 31.62 (5.903) | 31.10 (5.067) | 0.133 |
| Pregestational BMI, n (%) * | 0.106 | ||
| <18.5, n (%) | 13 (6.2) | 941 (3.9) | |
| 18.5–25, n (%) | 120 (57.1) | 15,279 (63.3) | |
| 25–30, n (%) | 44 (21.0) | 4982 (20.6) | |
| >30, n (%) | 33 (15.7) | 2951 (12.2) | |
| Smoking, n (%) | 21 (10.0) | 2451 (10.1) | 0.947 |
| DM1, n (%) * | 1 (0.5) | 141 (0.6) | 0.839 |
| IVF, n (%) * | 17 (8.1) | 1083 (4.5) | 0.012 |
| GDM, n (%) * | 54 (25.7) | 4836 (20.0) | 0.040 |
| Use of progesterone, n (%) | 20 (9.5) | 2024 (8.4) | 0.549 |
| Primipara, n (%) | 97 (46.2) | 11,326 (46.8) | 0.850 |
| Season of birth—winter, n (%) * | 107 (51.0) | 11,667 (48.3) | 0.436 |
| Foetal sex—male, n (%) | 103 (49.0) | 12,421 (51.4) | 0.794 |
| ICP Group (N = 210) | Control Group (N = 24,177) | p-Value (%) | Adjusted p-Value (%) † | |
|---|---|---|---|---|
| Gestational hypertension, n (%) | 17 (8.1) | 1594 (6.6) | 0.383 | 0.641 |
| Preeclampsia, n (%) | 5 (2.4) | 527 (2.2) | 0.842 | 0.996 |
| Gestational age at delivery (weeks) mean, (st. deviation) | 37.21 (1.656) | 38.73 (2.245) | <0.001 | <0.001 |
| Spontaneous preterm birth, n (%) | 17 (8.1) | 1209 (5.0) | 0.041 | 0.055 |
| Iatrogenic preterm birth, n (%) ** | 30 (14.3) | 661 (2.7) | <0.001 | <0.001 |
| Caesarean section, n (%) | 61 (29.0) | 5038 (20.8) | 0.004 | 0.012 |
| Elective caesarean section, n (%) | 32 (15.2) | 2495 (10.3) | 0.020 | 0.043 |
| Emergency caesarean section, n (%) | 22 (10.5) | 1751 (7.2) | 0.072 | 0.100 |
| Vacuum extraction delivery, n (%) | 8 (3.8) | 586 (2.4) | 0.195 | 0.193 |
| Intrapartum haemorrhage > 500 mL, n (%) | 15 (7.1) | 1607 (6.6) | 0.774 | 0.908 |
| Meconium-stained amniotic fluid, n (%) | 43 (20.5) | 4115 (17.0) | 0.185 | 0.183 |
| Apgar 5 < 7, n (%) * | 4 (1.9) | 231 (1.0) | 0.161 | 0.215 |
| pH a. umbilicalis (n) mediana (st. deviation) | 7.25 (0.077) | 7.26 (0.082) | 0.794 | 0.761 |
| Stillbirth, n (%) | 0 (0) | 89 (0.4) | 0.378 | 0.995 |
| NCIU admission, n (%) * | 20 (9.5) | 1460 (6.0) | 0.035 | 0.051 |
| Birth weight (g) mean, (st. deviation) | 3132.62 (507.642) | 3340.57 (608.622) | <0.001 | <0.001 |
| Peak Bile Acid Level (µmol/L) | <40 (N = 113) | 40–99 (N = 67) | ≥100 (N = 30) | p-Value (%) |
|---|---|---|---|---|
| Maternal age (years) mean, (st. deviation) | 31.75 (5.935) | 31.34 (5.917) | 31.77 (5.929) | 0.896 |
| Pregestational BMI, n (%) * | 0.632 | |||
| <18.5, n (%) | 6 (5.3) | 4 (6.0) | 3 (10.0) | |
| 18.5–25, n (%) | 64 (56.6) | 42 (62.7) | 14 (46.7) | |
| 25–30, n (%) | 25 (22.1) | 10 (14.9) | 9 (30.0) | |
| >30, n (%) | 18 (15.9) | 11 (16.4) | 4 (13.3) | |
| Smoking, n (%) | 12 (10.6) | 8 (11.9) | 1 (3.3) | 0.405 |
| DM 1, n (%) * | 0 (0) | 1 (1.5) | 0 (0) | 0.342 |
| GDM, n (%) * | 29 (25.7) | 19 (28.4) | 6 (20.0) | 0.684 |
| IVF, n (%) * | 5 (4.4) | 7 (10.4) | 5 (16.7) | 0.064 |
| Use of progesterone, n (%) | 9 (8.0) | 7 (10.4) | 4 (13.3) | 0.641 |
| Primipara, n (%) | 50 (44.2) | 29 (43.3) | 18 (60.0) | 0.259 |
| Season of birth—winter, n (%) * | 63 (55.8) | 29 (43.3) | 15 (50.0) | 0.269 |
| Foetal sex—male, n (%) | 57 (50.4) | 30 (44.8) | 16 (53.3) | 0.671 |
| Peak Bile Acid Level (µmol/L) | <40 (N = 113) | 40–99 (N = 67) | ≥100 (N = 30) | p-Value (%) | Adjusted p-Value (%) † |
|---|---|---|---|---|---|
| Gestational hypertension, n (%) | 9 (8.0) | 5 (7.5) | 3 (10.0) | 0.912 | 0.072 |
| Preeclampsia, n (%) | 3 (2.7) | 2 (3.0) | 0 (0) | 0.646 | 0.131 |
| Gestational age at delivery (weeks) mean, (st. deviation) | 37.71 (1.321) | 36.90 (1.698) | 36.07 (1.982) | <0.001 | <0.001 |
| Spontaneous preterm birth, n (%) | 5 (4.4) | 4 (6.0) | 8 (26.7) | <0.001 | 0.002 |
| Iatrogenic preterm birth, n (%) ** | 6 (5.3) | 13 (19.4) | 11 (36.7) | <0.001 | <0.001 |
| Caesarean section, n (%) | 37 (32.7) | 16 (23.9) | 8 (26.7) | 0.428 | 0.146 |
| Elective caesarean section, n (%) | 22 (19.5) | 7 (10.4) | 3 (10.0) | 0.183 | 0.217 |
| Emergency caesarean section, n (%) | 12 (10.6) | 7 (10.4) | 3 (10.0) | 0.995 | 0.208 |
| Vacuum extraction delivery, n (%) | 4 (3.5) | 3 (4.5) | 1 (3.3) | 0.941 | 0.701 |
| Intrapartum haemorrhage > 500 mL, n (%) | 6 (5.3) | 6 (9.0) | 3 (10.0) | 0.529 | 0.795 |
| Meconium-stained amniotic fluid, n (%) | 16 (14.2) | 14 (20.9) | 13 (43.3) | 0.002 | 0.001 |
| Apgar 5 < 7, n (%) * | 0 (0) | 2 (3.0) | 2 (6.7) | 0.043 | 0.115 |
| pH a. umbilicalis (n) mean (st. deviation) | 7.26 (0.070) | 7.25 (0.076) | 7.22 (0.096) | 0.016 | 0.215 |
| Stillbirth, n (%) | 0 (0) | 0 (0) | 0 (0) | ||
| NCIU admission, n (%) * | 6 (5.3) | 7 (10.4) | 7 (23.3) | 0.011 | 0.006 |
| Birth weight (g) mean (st. deviation) | 3256.24 (505.857) | 3059.33 (456.004) | 2830.67 (478.993) | <0.001 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gregorc, P.; Verdenik, I.; Pečlin, P. Evaluating the Effect of Bile Acid Levels on Maternal and Perinatal Outcomes in Intrahepatic Cholestasis of Pregnancy: A Retrospective Study. Diagnostics 2025, 15, 2185. https://doi.org/10.3390/diagnostics15172185
Gregorc P, Verdenik I, Pečlin P. Evaluating the Effect of Bile Acid Levels on Maternal and Perinatal Outcomes in Intrahepatic Cholestasis of Pregnancy: A Retrospective Study. Diagnostics. 2025; 15(17):2185. https://doi.org/10.3390/diagnostics15172185
Chicago/Turabian StyleGregorc, Petra, Ivan Verdenik, and Polona Pečlin. 2025. "Evaluating the Effect of Bile Acid Levels on Maternal and Perinatal Outcomes in Intrahepatic Cholestasis of Pregnancy: A Retrospective Study" Diagnostics 15, no. 17: 2185. https://doi.org/10.3390/diagnostics15172185
APA StyleGregorc, P., Verdenik, I., & Pečlin, P. (2025). Evaluating the Effect of Bile Acid Levels on Maternal and Perinatal Outcomes in Intrahepatic Cholestasis of Pregnancy: A Retrospective Study. Diagnostics, 15(17), 2185. https://doi.org/10.3390/diagnostics15172185






