CT- and MRI-Based 3D Reconstruction of Knee Joint to Assess Cartilage and Bone
Abstract
1. Introduction
1.1. New Cartilage Assessment Methods/Gold Standard
1.2. Use of 3D Modeling Tools
1.3. Machine Learning and Artificial Intelligence
2. Materials and Methods
2.1. Participants
2.1.1. Recruitment
2.1.2. Scanning Process
CT Scanner
MRI
2.2. Data Processing and Analysis
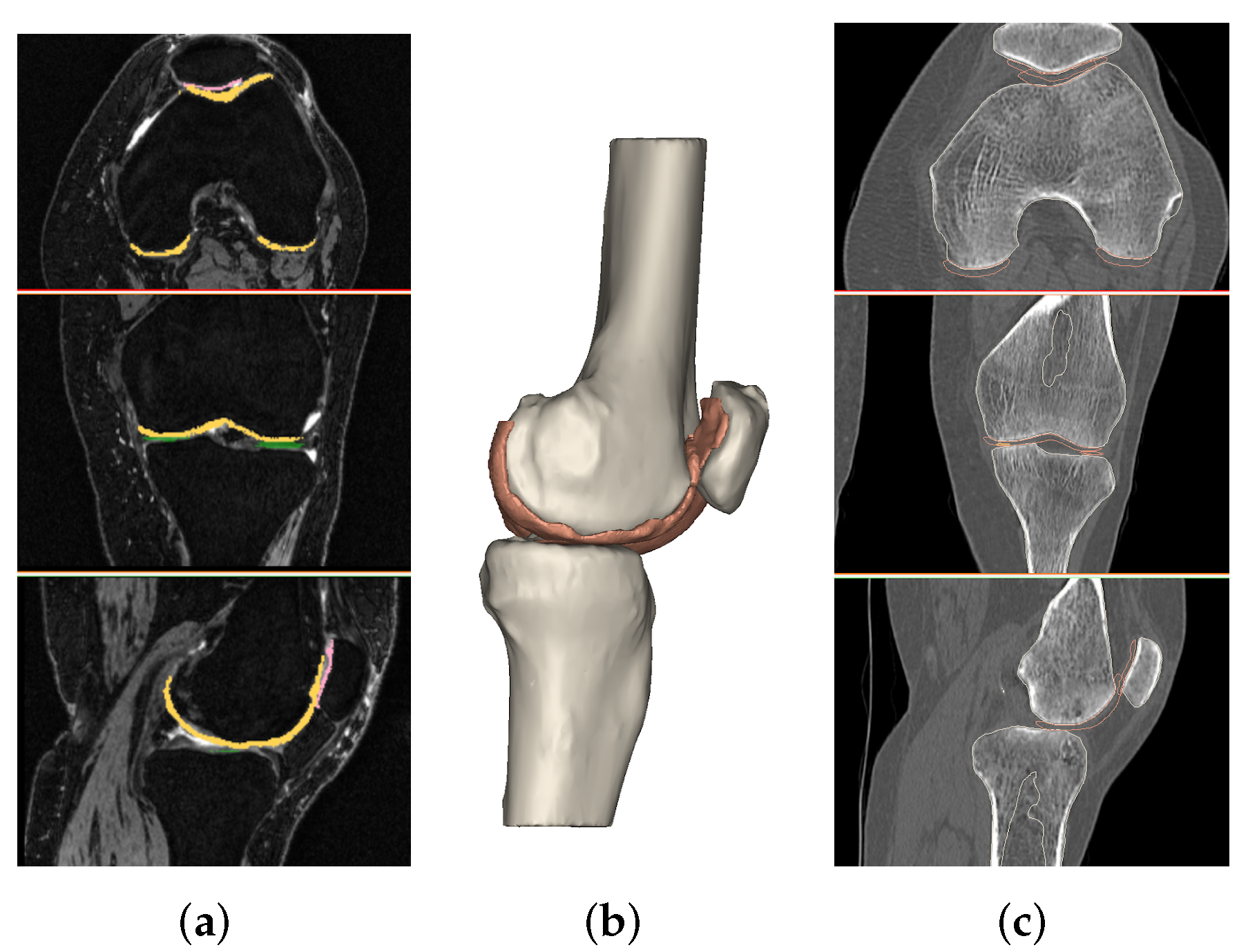
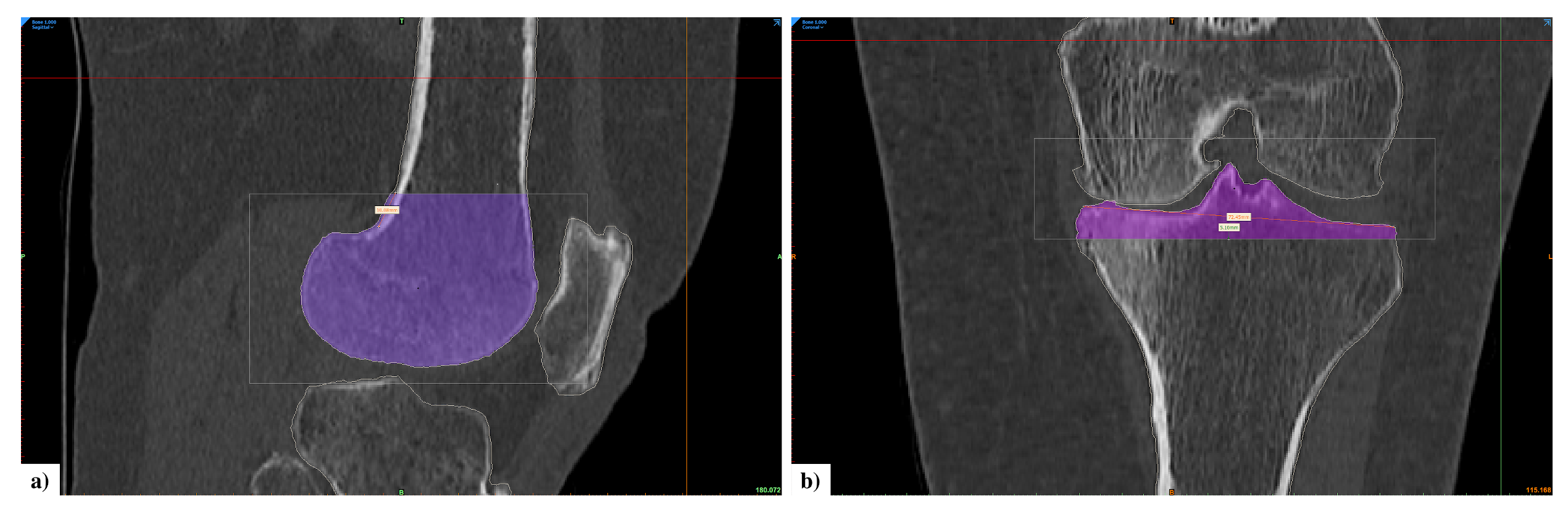
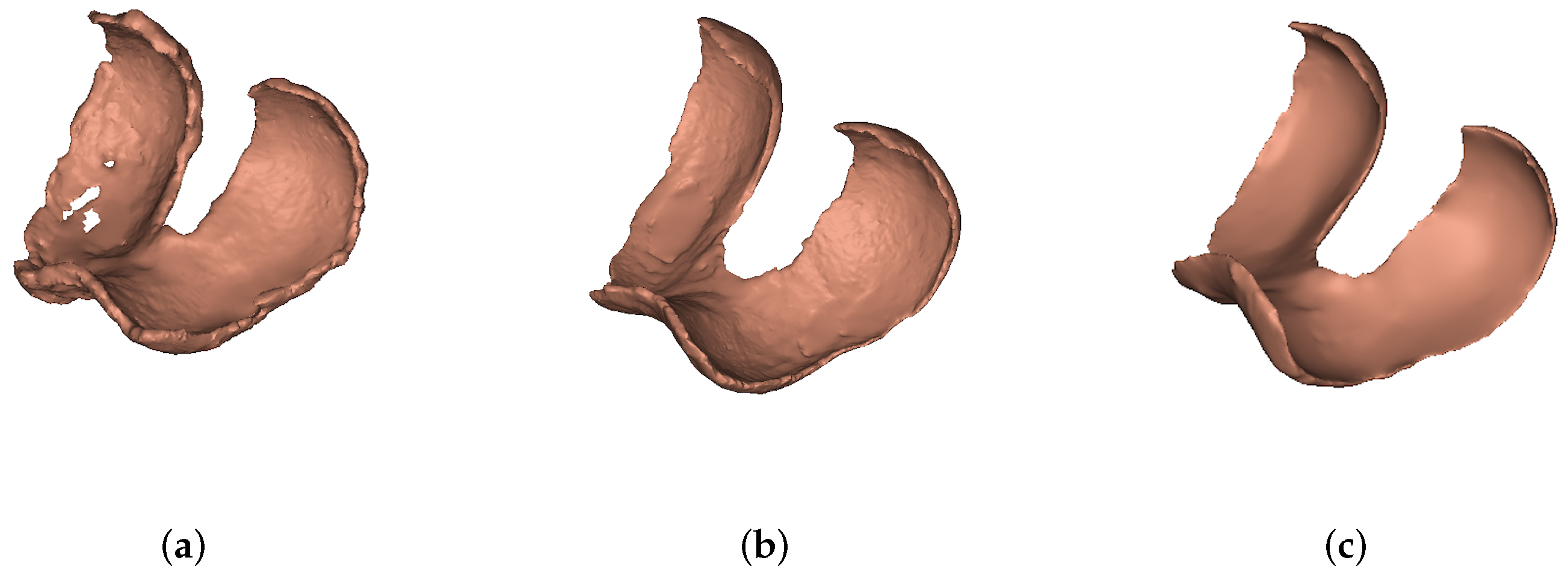
2.2.1. Segmentation
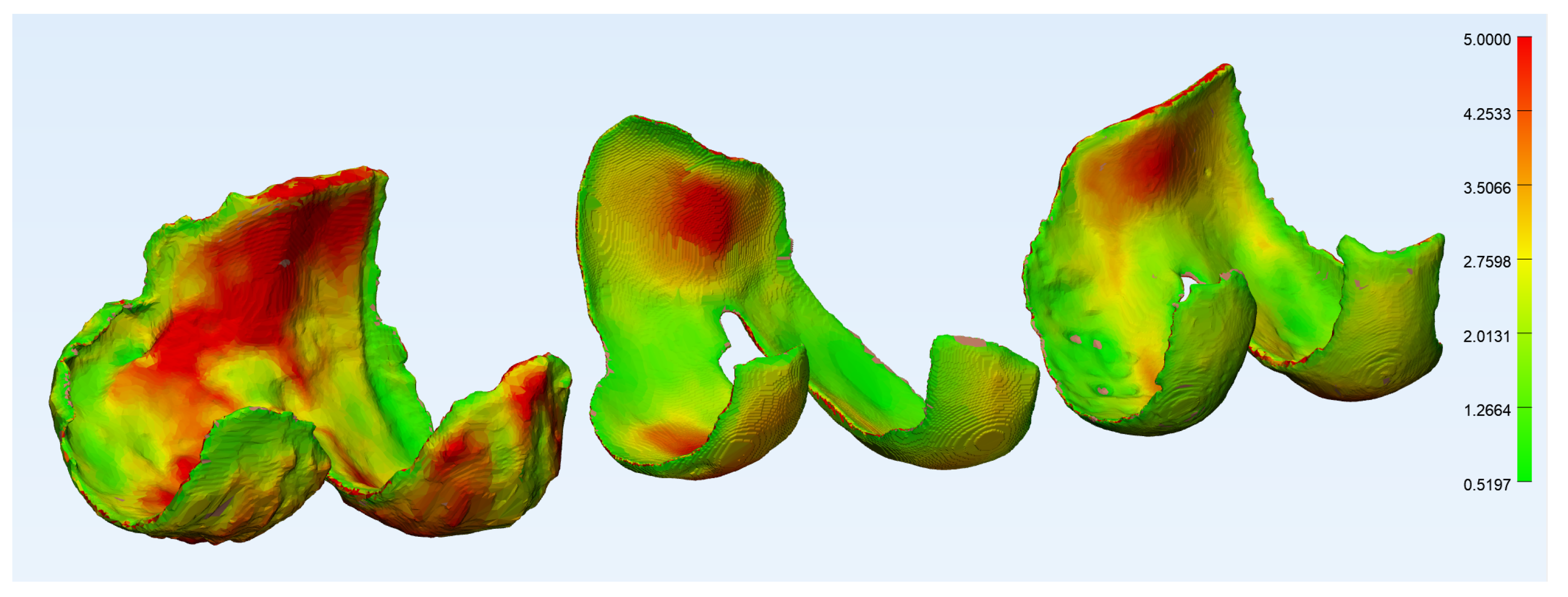
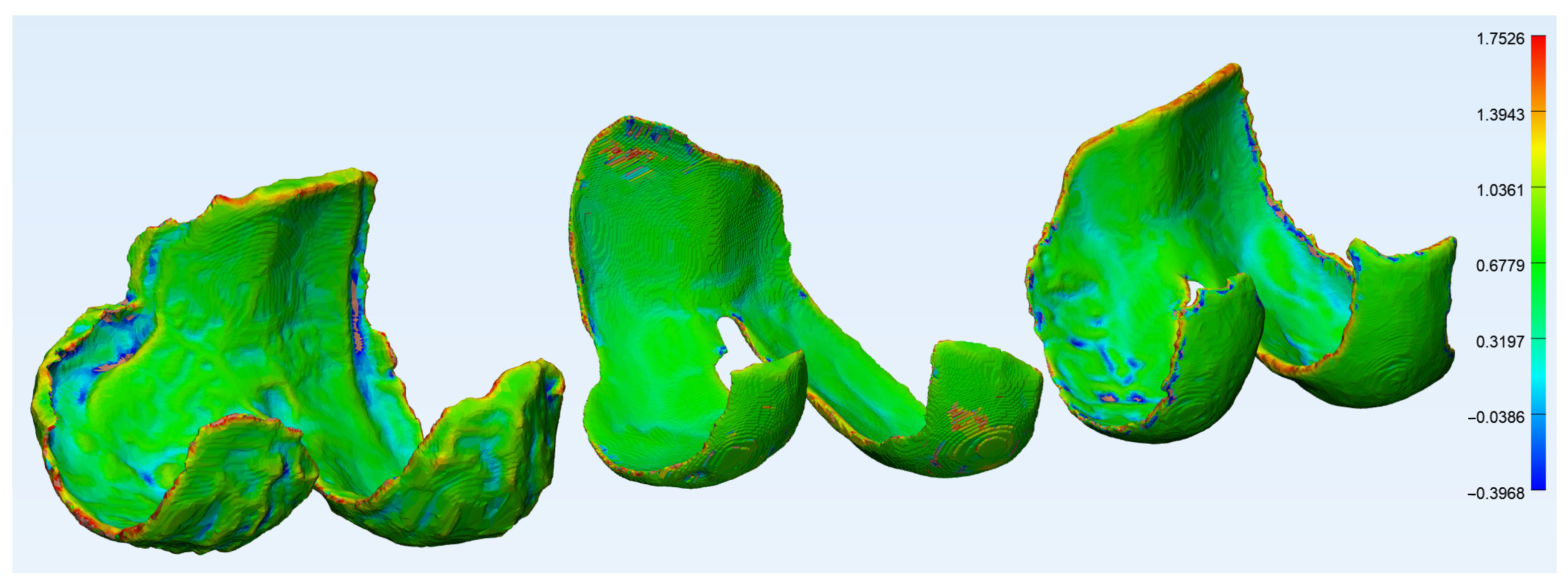
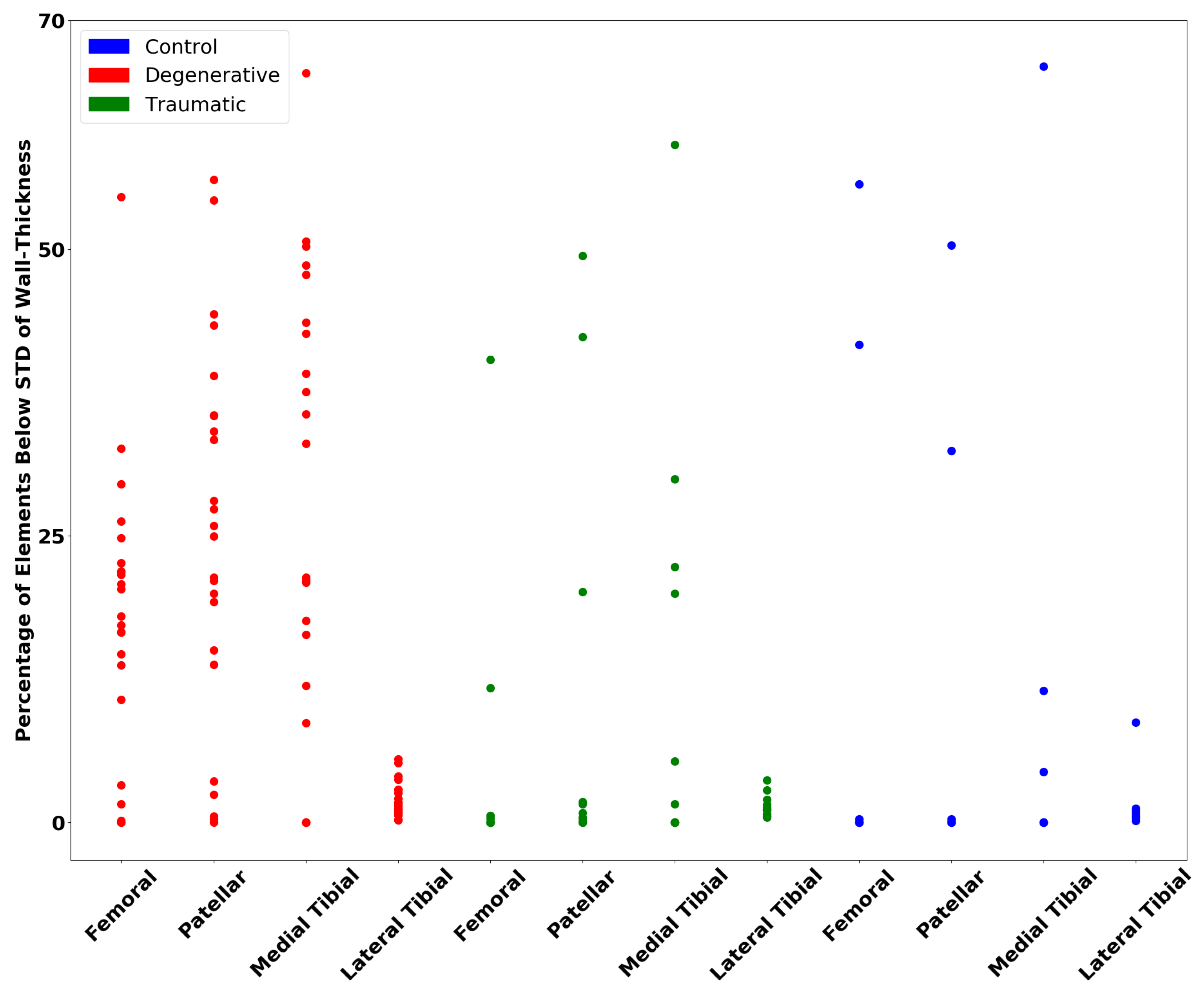
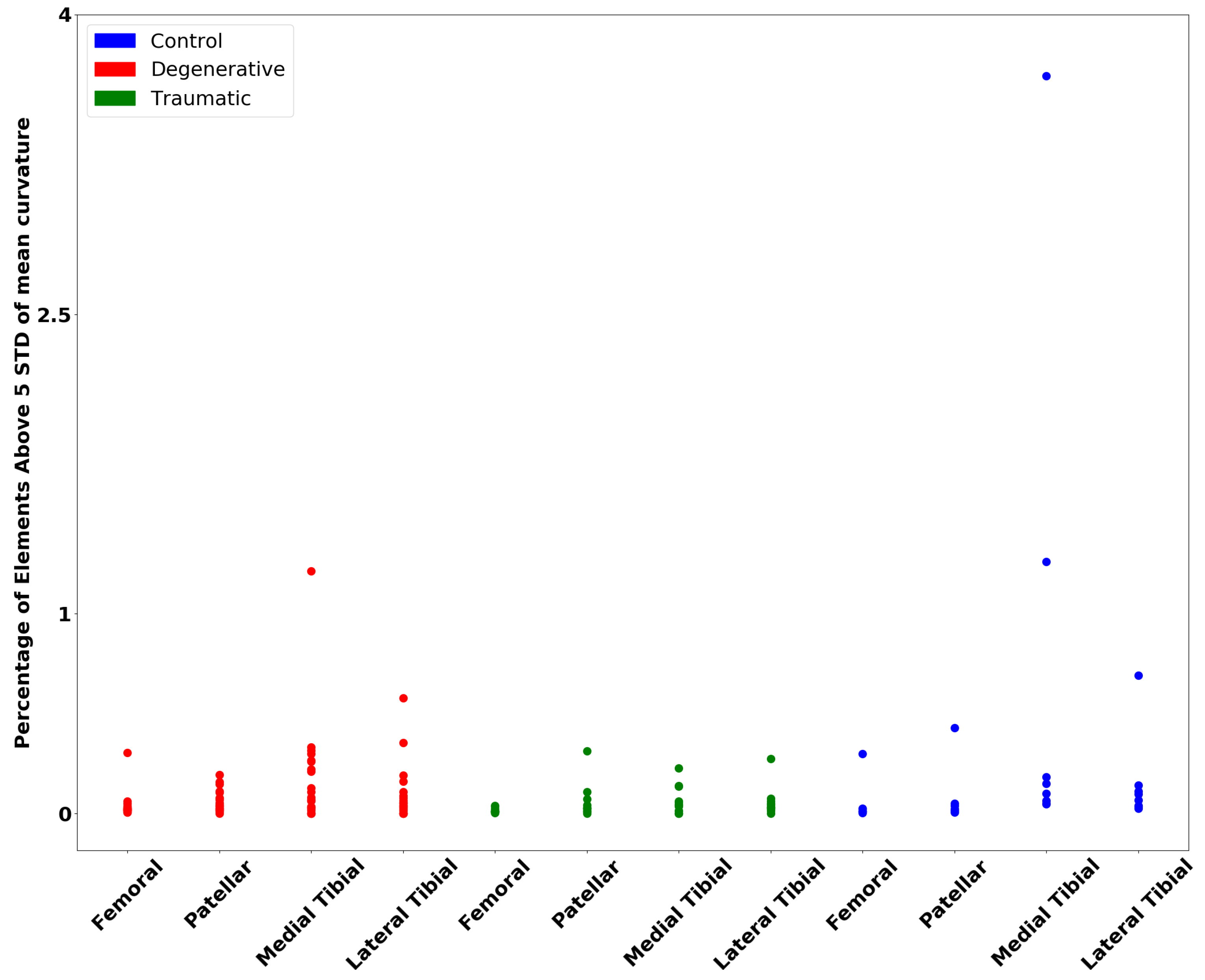
2.2.2. Wall Thickness and Curvature Analysis
2.3. Statistical Analysis
2.4. Machine Learning Classification
- Bone (#8): All the features extracted from the knee bones.
- Cartilage (#16): All the features extracted from the knee cartilages.
- Bone and Cartilage (B-C) (#24): All the features extracted from the knee bones and cartilages.
- Wall Thickness and Curvature (WT-C) (#27): All the features explained in Section 2.2.2. Initially, there were 48 of these features, but 13 of them were not considered because their standard deviation was too high compared to the average values, and these data could affect the classification process. The other 8 are the curvature standard deviation weight and the wall standard deviation weight, which are the same for every subject.
- Total Features (TOT) (#51): B-C and WT-C features together.
3. Results
3.1. 3D Measurements
3.2. Wall Thickness and Curvature Analyses
3.3. Machine Learning
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| B-C | Bone and Cartilage |
| C | Control Group |
| CT | Computed Tomography |
| D | Degenerative Group |
| DL | Deep Learning |
| DT | Decision Tree |
| GB | Gradient Boosting |
| HU | Hounsfield Unit |
| ML | Machine Learning |
| MRI | Magnetic Resonance Imaging |
| OA | Osteoarthritis |
| OARSI | Osteoarthritis Research Society International |
| RF | Random Forest |
| STL | Standard Tessellation Language |
| T | Traumatic Group |
| TOT | Total Features |
| WT-C | Wall Thickness and Curvature |
References
- Zhang, Y.; Jordan, J.M. Epidemiology of Osteoarthritis. Clin. Geriatr. Med. 2010, 26, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Przkora, R.; Sibille, K.; Victor, S.; Meroney, M.; Leeuwenburgh, C.; Gardner, A.; Vasilopoulos, T.; Parvataneni, H.K. Assessing the feasibility of using the short physical performance battery to measure function in the immediate postoperative period after total knee replacement. Eur. J. Transl. Myol. 2021, 31. [Google Scholar] [CrossRef] [PubMed]
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the Global Burden of Disease 2010 study. Ann. Rheumatol. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Murphy, L.; Schwartz, T.A.; Helmick, C.G.; Renner, J.B.; Tudor, G.; Koch, G.; Dragomir, A.; Kalsbeek, W.D.; Luta, G.; Jordan, J.M. Lifetime risk of symptomatic knee osteoarthritis. Arthritis Rheumatol. 2008, 59, 1207–1213. [Google Scholar] [CrossRef]
- Neogi, T. The epidemiology and impact of pain in osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1145–1153. [Google Scholar] [CrossRef]
- Marcucci, L.; Reggiani, C. Increase of resting muscle stiffness, a less considered component of age-related skeletal muscle impairment. Eur. J. Transl. Myol. 2020, 30, 8982. [Google Scholar] [CrossRef]
- Roemer, F.W.; Demehri, S.; Omoumi, P.; Link, T.M.; Kijowski, R.; Saarakkala, S.; Crema, M.D.; Guermazi, A. State of the Art: Imaging of Osteoarthritis—Revisited 2020. Radiology 2020, 296, 5–21. [Google Scholar] [CrossRef]
- Crema, M.D.; Roemer, F.W.; Marra, M.D.; Burstein, D.; Gold, G.E.; Eckstein, F.; Baum, T.; Mosher, T.J.; Carrino, J.A.; Guermazi, A. Articular Cartilage in the Knee: Current MR Imaging Techniques and Applications in Clinical Practice and Research. Radiographics 2011, 31, 37–61. [Google Scholar] [CrossRef]
- Schmitz, R.J.; Wang, H.M.; Polprasert, D.R.; Kraft, R.A.; Pietrosimone, B.G. Evaluation of knee cartilage thickness: A comparison between ultrasound and magnetic resonance imaging methods. Knee 2017, 24, 217–223. [Google Scholar] [CrossRef]
- Hani, A.F.M.; Kumar, D.; Malik, A.S.; Razak, R.; Kiflie, A. Fusion of multinuclear magnetic resonance images of knee for the assessment of articular cartilage. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; IEEE: Osaka, Japan, 2013; pp. 6466–6469. [Google Scholar] [CrossRef]
- Link, T.M.; Neumann, J.; Li, X. Prestructural cartilage assessment using MRI: Prestructural Cartilage Assessment Using MRI. J. Magn. Reson. Imaging 2017, 45, 949–965. [Google Scholar] [CrossRef]
- Pradsgaard, D.Ø.; Fiirgaard, B.; Spannow, A.H.; Heuck, C.; Herlin, T. Cartilage Thickness of the Knee Joint in Juvenile Idiopathic Arthritis: Comparative Assessment by Ultrasonography and Magnetic Resonance Imaging. J. Rheumatol. 2015, 42, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Campbell, A.; Quatman, C.; Schmitt, L.; Knopp, M.; Flanigan, D. Is Magnetic Resonance Imaging Assessment of the Size of Articular Cartilage Defects Accurate? J. Knee Surg. 2013, 27, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Pétursson, T.; Magnússon, B.; Helgason, B.; Magnúsdóttir, G.; Halldórsson, G.; Tribel, J.; Jónsson jr, H.; Gargiulo, P. Bone and muscle assessment in patients undergoing total hip arthroplasty using HU based analysis. Eur. J. Transl. Myol. 2012, 22, 147–152. [Google Scholar] [CrossRef][Green Version]
- Esposito, L.; Bifulco, P.; Gargiulo, P.; Gíslason, M.K.; Cesarelli, M.; Iuppariello, L.; Jónsson, H.; Cutolo, A.; Fraldi, M. Towards a patient-specific estimation of intra-operative femoral fracture risk. Comput. Methods Biomech. Biomed. Eng. 2018, 21, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, P.; Gislason, M.K.; Edmunds, K.J.; Pitocchi, J.; Carraro, U.; Esposito, L.; Fraldi, M.; Bifulco, P.; Cesarelli, M.; Jónsson, H. CT-Based Bone and Muscle Assessment in Normal and Pathological Conditions. In Encyclopedia of Biomedical Engineering; Narayan, R., Ed.; Elsevier: Oxford, UK, 2019; pp. 119–134. [Google Scholar] [CrossRef]
- Johnston, J.; Masri, B.; Wilson, D. Computed tomography topographic mapping of subchondral density (CT-TOMASD) in osteoarthritic and normal knees: Methodological development and preliminary findings. Osteoarthr. Cartil. 2009, 17, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Kellgren, J.H.; Lawrence, J.S. Radiological assessment of Osteo-Arthrosis. Ann. Rheumatol. Dis. 1957, 16, 494. [Google Scholar] [CrossRef] [PubMed]
- Ahlbäck, S. Osteoarthrosis of the knee. A radiographic investigation. Acta Radiol. Diagn. 1968, 277, 7–72. [Google Scholar]
- Galli, M.; De Santis, V.; Tafuro, L. Reliability of the Ahlbäck classification of knee osteoarthritis. Osteoarthrit. Cartil. 2003, 11, 580–584. [Google Scholar] [CrossRef]
- Wright, R.W.; The MARS Group. Osteoarthritis Classification Scales: Interobserver Reliability and Arthroscopic Correlation. J. Bone Jt. Surg. 2014, 96, 1145–1151. [Google Scholar] [CrossRef]
- Keenan, O.J.F.; Holland, G.; Maempel, J.F.; Keating, J.F.; Scott, C.E.H. Correlations between radiological classification systems and confirmed cartilage loss in severe knee osteoarthritis. Bone Jt. J. 2020, 102, 9. [Google Scholar] [CrossRef]
- Eckstein, F.; Le Graverand, M.P.H.; Charles, H.C.; Hunter, D.J.; Kraus, V.B.; Sunyer, T.; Nemirovskyi, O.; Wyman, B.T.; Buck, R.; for the A9001140, investigators. Clinical, radiographic, molecular and MRI-based predictors of cartilage loss in knee osteoarthritis. Ann. Rheumatol. Dis. 2011, 70, 1223. [Google Scholar] [CrossRef] [PubMed]
- MacKay, J.W.; Kaggie, J.D.; Treece, G.M.; McDonnell, S.M.; Khan, W.; Roberts, A.R.; Janiczek, R.L.; Graves, M.J.; Turmezei, T.D.; McCaskie, A.W.; et al. Three-dimensional surface-based analysis of cartilage MRI data in knee osteoarthritis: Validation and initial clinical application. J. Magn. Reson. 2020, 52, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Papernick, S.; Dima, R.; Gillies, D.; Appleton, C.; Fenster, A. Reliability and concurrent validity of three-dimensional ultrasound for quantifying knee cartilage volume. Osteoarthr. Cartil. 2020, 2, 100127. [Google Scholar] [CrossRef]
- Kijowski, R.; Roemer, F.; Englund, M.; Tiderius, C.; Swärd, P.; Frobell, R. Imaging following acute knee trauma. Osteoarthr. Cartil. 2014, 22, 1429–1443. [Google Scholar] [CrossRef] [PubMed]
- Roemer, F.W.; Frobell, R.; Lohmander, L.S.; Niu, J.; Guermazi, A. Anterior Cruciate Ligament OsteoArthritis Score (ACLOAS): Longitudinal MRI-based whole joint assessment of anterior cruciate ligament injury. Osteoarthr. Cartil. 2014, 22, 668–682. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Almajalid, R.; Shan, J.; Zhang, M. A novel method to predict knee osteoarthritis progression on MRI using machine learning methods. IEEE Trans. Nanobiosci. 2018, 17, 228–236. [Google Scholar] [CrossRef]
- Tiulpin, A.; Klein, S.; Bierma-Zeinstra, S.M.; Thevenot, J.; Rahtu, E.; van Meurs, J.; Oei, E.H.; Saarakkala, S. Multimodal machine learning-based knee osteoarthritis progression prediction from plain radiographs and clinical data. Sci. Rep. 2019, 9, 20038. [Google Scholar] [CrossRef]
- Liu, F.; Zhou, Z.; Samsonov, A.; Blankenbaker, D.; Larison, W.; Kanarek, A.; Lian, K.; Kambhampati, S.; Kijowski, R. Deep learning approach for evaluating knee MR images: Achieving high diagnostic performance for cartilage lesion detection. Radiology 2018, 289, 160–169. [Google Scholar] [CrossRef]
- Bien, N.; Rajpurkar, P.; Ball, R.L.; Irvin, J.; Park, A.; Jones, E.; Bereket, M.; Patel, B.N.; Yeom, K.W.; Shpanskaya, K.; et al. Deep-learning-assisted diagnosis for knee magnetic resonance imaging: Development and retrospective validation of MRNet. PLoS Med. 2018, 15, e1002699. [Google Scholar] [CrossRef]
- Tiulpin, A.; Thevenot, J.; Rahtu, E.; Lehenkari, P.; Saarakkala, S. Automatic knee osteoarthritis diagnosis from plain radiographs: A deep learning-based approach. Sci. Rep. 2018, 8, 1727. [Google Scholar] [CrossRef]
- Tolpadi, A.A.; Lee, J.J.; Pedoia, V.; Majumdar, S. Deep learning predicts total knee replacement from magnetic resonance images. Sci. Rep. 2020, 10, 1–12. [Google Scholar]
- Kwon, S.B.; Han, H.S.; Lee, M.C.; Kim, H.C.; Ku, Y. Machine learning-based automatic classification of knee osteoarthritis severity using gait data and radiographic images. IEEE Access 2020, 8, 120597–120603. [Google Scholar] [CrossRef]
- Ramos, Y.F.; Bos, S.D.; Lakenberg, N.; Böhringer, S.; den Hollander, W.J.; Kloppenburg, M.; Slagboom, P.E.; Meulenbelt, I. Genes expressed in blood link osteoarthritis with apoptotic pathways. Ann. Rheumatol. Dis. 2014, 73, 1844–1853. [Google Scholar] [CrossRef]
- Li, J.; Lan, C.N.; Kong, Y.; Feng, S.S.; Huang, T. Identification and analysis of blood gene expression signature for osteoarthritis with advanced feature selection methods. Front. Genet. 2018, 9, 246. [Google Scholar] [CrossRef] [PubMed]
- DenOtter, T.D.; Schubert, J. Hounsfield Unit; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- QRM. Micro-CT HA Phantom for the Calibration of CT-Values. Available online: https://www.qrm.de/en/products/micro-ct-ha-phantom/ (accessed on 23 February 2021).
- Berthold, M.R.; Cebron, N.; Dill, F.; Gabriel, T.R.; Kötter, T.; Meinl, T.; Ohl, P.; Thiel, K.; Wiswedel, B. KNIME-the Konstanz information miner: Version 2.0 and beyond. SIGKDD Explor. 2009, 11, 26–31. [Google Scholar] [CrossRef]
- Recenti, M.; Ricciardi, C.; Edmunds, K.J.; Gislason, M.K.; Sigurdsson, S.; Carraro, U.; Gargiulo, P. Healthy Aging Within an Image: Using Muscle Radiodensitometry and Lifestyle Factors to Predict Diabetes and Hypertension. IEEE J. Biomed. Health Inform. 2020, 25, 2103–2112. [Google Scholar] [CrossRef] [PubMed]
- Recenti, M.; Ricciardi, C.; Aubonnet, R.; Picone, I.; Jacob, D.; Svansson, H.Á.; Agnarsdóttir, S.; Karlsson, G.H.; Baeringsdóttir, V.; Petersen, H.; et al. Toward Predicting Motion Sickness Using Virtual Reality and a Moving Platform Assessing Brain, Muscles, and Heart Signals. Front. Bioeng. Biotechnol. 2021, 9, 132. [Google Scholar] [CrossRef]
- Ho, T.K. Random decision forests. In Proceedings of the 3rd International Conference on Document Analysis and Recognition, Montreal, QC, Canada, 14–16 August 1995; IEEE: Piscataway, NJ, USA, 1995; Volume 1, pp. 278–282. [Google Scholar]
- Friedman, J.H. Greedy function approximation: A gradient boosting machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Ricciardi, C.; Jónsson, H.; Jacob, D.; Improta, G.; Recenti, M.; Gíslason, M.K.; Cesarelli, G.; Esposito, L.; Minutolo, V.; Bifulco, P.; et al. Improving prosthetic selection and predicting BMD from biometric measurements in patients receiving total hip arthroplasty. Diagnostics 2020, 10, 815. [Google Scholar] [CrossRef]
- Recenti, M.; Ricciardi, C.; Gìslason, M.; Edmunds, K.; Carraro, U.; Gargiulo, P. Machine learning algorithms predict body mass index using nonlinear trimodal regression analysis from computed tomography scans. In Mediterranean Conference on Medical and Biological Engineering and Computing; Springer: Berlin/Heidelberg, Germany, 2019; pp. 839–846. [Google Scholar]
- Saeb, S.; Lonini, L.; Jayaraman, A.; Mohr, D.C.; Kording, K.P. The need to approximate the use-case in clinical machine learning. Gigascience 2017, 6, gix019. [Google Scholar] [CrossRef]
- Buckwalter, J.A.; Mankin, H.J. Articular cartilage: Part II. J. Bone Jt. Surg. 1997, 79, 612. [Google Scholar] [CrossRef]
- Teichtahl, A.J.; Wang, Y.; Wluka, A.E.; Strauss, B.J.; Proietto, J.; Dixon, J.B.; Jones, G.; Cicuttini, F.M. Associations between systemic bone mineral density and early knee cartilage changes in middle-aged adults without clinical knee disease: A prospective cohort study. Arthritis Res. Ther. 2017, 19, 98. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hannan, M.; Chaisson, C.; McAlindon, T.; Evans, S.; Aliabadi, P.; Levy, D.; Felson, D. Bone mineral density and risk of incident and progressive radiographic knee osteoarthritis in women: The Framingham Study. J. Rheumatol. 2000, 27, 1032–1037. [Google Scholar] [PubMed]
- Nevitt, M.C.; Zhang, Y.; Javaid, M.K.; Neogi, T.; Curtis, J.R.; Niu, J.; McCulloch, C.E.; Segal, N.A.; Felson, D.T. High systemic bone mineral density increases the risk of incident knee OA and joint space narrowing, but not radiographic progression of existing knee OA: The MOST study. Ann. Rheumatol. Dis. 2010, 69, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.S.; Shin, H.D.; Sim, J.A.; Na, Y.G.; Choi, W.J.; Shin, D.D.; Baik, J.M. Relationship of Bone Mineral Density and Knee Osteoarthritis (Kellgren-Lawrence Grade): Fifth Korea National Health and Nutrition Examination Survey. Clin. Orthop. Surg. 2021, 13, 60. [Google Scholar] [CrossRef]
- Vollnberg, B.; Koehlitz, T.; Jung, T.; Scheffler, S.; Hoburg, A.; Khandker, D.; Hamm, B.; Wiener, E.; Diederichs, G. Prevalence of cartilage lesions and early osteoarthritis in patients with patellar dislocation. Eur. Radiol. 2012, 22, 2347–2356. [Google Scholar] [CrossRef]
- Matzat, S.J.; Kogan, F.; Fong, G.W.; Gold, G.E. Imaging strategies for assessing cartilage composition in osteoarthritis. Curr. Rheumatol. Rep. 2014, 16, 462. [Google Scholar] [CrossRef]
- Nickien, M.; Thambyah, A.; Broom, N.D. How a decreased fibrillar interconnectivity influences stiffness and swelling properties during early cartilage degeneration. J. Mech. Behav. Biomed. Mater. 2017, 75, 390–398. [Google Scholar] [CrossRef]
- Watson, P.J.; Carpenter, T.A.; Hall, L.D.; Tyler, J.A. Cartilage swelling and loss in a spontaneous model of osteoarthritis visualized by magnetic resonance imaging. Osteoarthr. Cartil. 1996, 4, 197–207. [Google Scholar] [CrossRef]
- Vincent, T.L.; Wann, A.K. Mechanoadaptation: Articular cartilage through thick and thin. J. Physiol. 2019, 597, 1271–1281. [Google Scholar] [CrossRef]
- Folkesson, J.; Dam, E.B.; Olsen, O.F.; Karsdal, M.A.; Pettersen, P.C.; Christiansen, C. Automatic quantification of local and global articular cartilage surface curvature: Biomarkers for osteoarthritis? Magn. Reson. Med. Sci. 2008, 59, 1340–1346. [Google Scholar] [CrossRef] [PubMed]
- Chawla, N.V.; Bowyer, K.W.; Hall, L.O.; Kegelmeyer, W.P. SMOTE: Synthetic minority over-sampling technique. J. Artif. Intell. Res. 2002, 16, 321–357. [Google Scholar] [CrossRef]
| D | T | C | |
|---|---|---|---|
| Bone Mineral Density (g/cm3) | |||
| Femur | 1.25 (0.03) | 1.29 (0.03) | 1.27 (0.03) |
| 1.25 | 1.30 | 1.27 | |
| Tibia | 1.28 (0.02) | 1.32 (0.03) | 1.30 (0.03) |
| 1.29 | 1.32 | 1.30 | |
| Patella | 1.34 (0.05) | 1.41 (0.04) | 1.41 (0.05) |
| 1.35 | 1.42 | 1.40 | |
| Patella Volume (mm3) | 20,368.41 (4486.86) | 17,747.93 (4389.93) | 19,375.79 (5009.31) |
| 19,315.82 | 16,575.77 | 20,508.23 | |
| Patella Surface (mm2) | 4867.88 (1614.10) | 5648.82 (3180.33) | 4105.83 (725.54) |
| 4402.45 | 4516.32 | 4234.59 | |
| Radiodensity (HU) | |||
| Femur Cartilage | 88.55 (5.74) | 88.64 (12.34) | 94.06 (7.45) |
| 89.51 | 86.56 | 97.17 | |
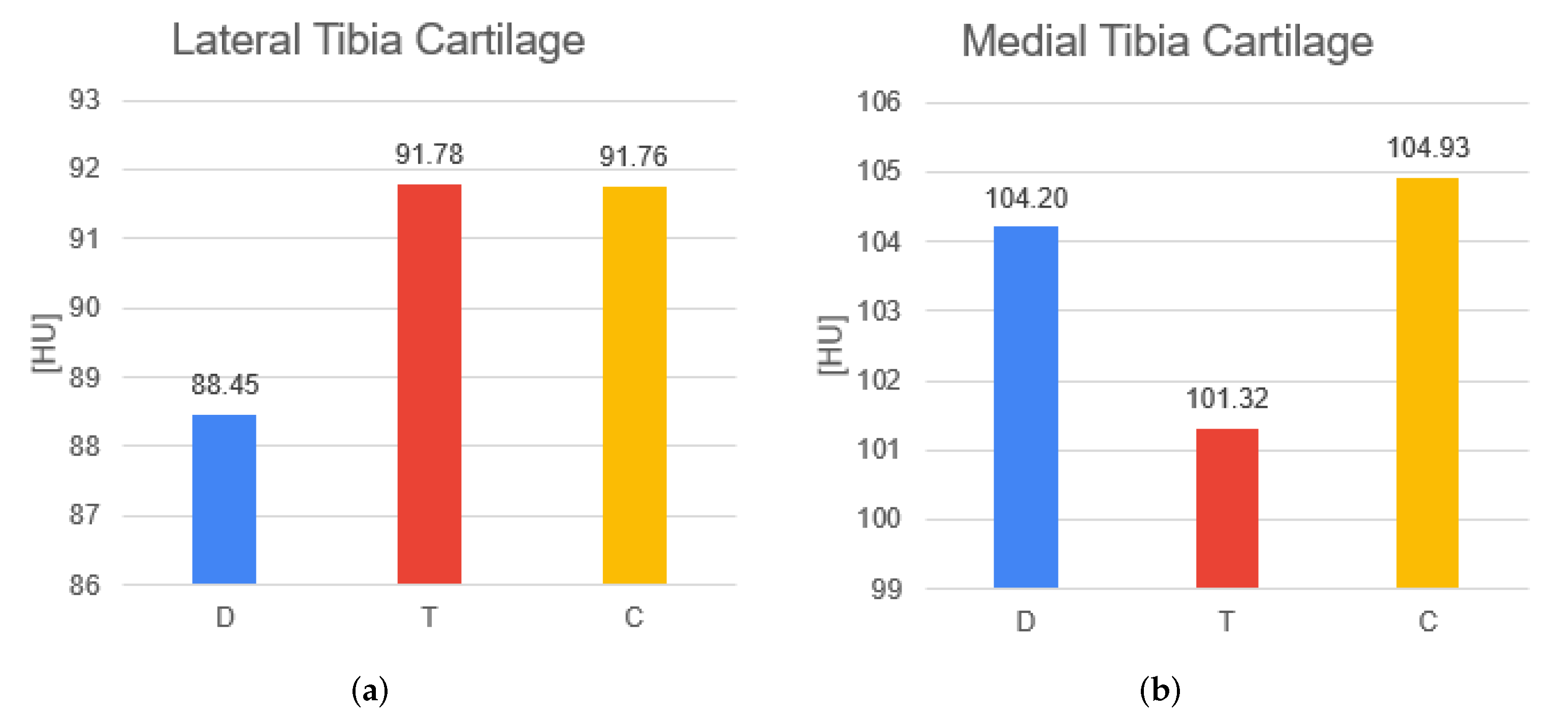
| Lateral Tibia Cartilage | 88.45 (8.30) | 91.78 (19.85) | 91.76 (3.10) |
| 88.20 | 89.53 | 92.21 | |
| Medial Tibia Cartilage | 104.20 (19.94) | 101.32 (16.70) | 104.93 (7.65) |
| 98.90 | 97.33 | 103.74 | |
| Patella Cartilage | 78.98 (17.53) | 79.19 (18.27) | 95.10 (16.47) |
| 74.94 | 77.16 | 88.36 | |
| Cartilage Volume (mm3) | |||
| Femur Cartilage | 20,265.18 (6856.78) | 13,429.59 (2725.04) | 11,764.49 (4479.56) |
| 19,618.83 | 12,956.89 | 9654.09 | |
| Lateral Tibia Cartilage | 2075.11 (1515.56) | 1110.60 (409.59) | 757.59 (380.87) |
| 1371.79 | 1160.67 | 725.18 | |
| Medial Tibia Cartilage | 1526.14 (1226.54) | 981.93 (610.23) | 555.76 (368.51) |
| 1067.28 | 868.62 | 440.17 | |
| Patella Cartilage | 3241.89 (1164.37) | 2778.97 (656.93) | 2866.61 (715.97) |
| 3199.50 | 2734.05 | 2854.35 | |
| Cartilage Surface (mm2) | |||
| Femur Cartilage | 14,737.79 (2866.13) | 12,270.98 (1378.72) | 11,968.50 (2663.58) |
| 14,076.96 | 12,319.56 | 11,541.29 | |
| Lateral Tibia Cartilage | 1809.96 (922.87) | 1299.96 (437.06) | 1082.64 (371.60) |
| 1458.98 | 1238.18 | 1038.20 | |
| Medial Tibia Cartilage | 1702.80 (997.76) | 1233.47 (469.07) | 949.80 (400.35) |
| 1288.59 | 1172.29 | 1001.64 | |
| Patella Cartilage | 2546.89 (469.92) | 2443.72 (480.52) | 2517.28 (409.93) |
| 2588.05 | 2371.60 | 2530.24 | |
| Presence of Holes (%) | 3.76 | 0.47 | 0 |
| Patients | Femoral Cartilage | Lateral Tibia Cartilage | Medial Tibia Cartilage | Patella Cartilage |
|---|---|---|---|---|
| 1 (D) | 3 | 1 | 7 | 0 |
| 2 (D) | 2 | 0 | 0 | 0 |
| 3 (D) | 1 | 0 | 1 | 0 |
| 4 (D) | 1 | 0 | 0 | 1 |
| 5 (D) | 1 | 0 | 0 | 3 |
| 6 (D) | 4 | 0 | 0 | 0 |
| 7 (D) | 1 | 0 | 0 | 0 |
| 8 (D) | 1 | 0 | 0 | 0 |
| 9 (T) | 5 | 0 | 0 | 2 |
| Patients | Femoral Cartilage (mm2) | Lateral Tibia Cartilage (mm2) | Medial Tibia Cartilage (mm2) | Patella Cartilage (mm2) |
|---|---|---|---|---|
| 1 (D) | 50.11 | 1.18 | 53.79 | 0.00 |
| 2 (D) | 186.83 | 0.00 | 0.00 | 0.00 |
| 3 (D) | 29.57 | 0.00 | 22.91 | 0.00 |
| 4 (D) | 3.06 | 0.00 | 0.00 | 2.97 |
| 5 (D) | 10.52 | 0.00 | 0.00 | 4.03 |
| 6 (D) | 19.80 | 0.00 | 0.00 | 0.00 |
| 7 (D) | 0.78 | 0.00 | 0.00 | 0.00 |
| 8 (D) | 4.60 | 0.00 | 0.00 | 0.00 |
| 9 (T) | 488.81 | 0.00 | 0.00 | 1.87 |
| Feat. Selection | Alg. | Acc. | Sens D | Spec D | Sens T | Spec T | Sens C | Spec C |
|---|---|---|---|---|---|---|---|---|
| TOT | RF | 71.7 | 87.5 | 63.6 | 57.1 | 93.8 | 50.0 | 92.1 |
| GB | 67.4 | 87.5 | 68.2 | 64.3 | 84.4 | 12.5 | 92.1 | |
| DT | 63.0 | 79.2 | 59.1 | 71.4 | 78.1 | 0.00 | 97.4 | |
| B-C | RF | 76.1 | 87.5 | 68.2 | 85.7 | 90.6 | 25.0 | 97.4 |
| GB | 69.9 | 79.2 | 72.7 | 71.4 | 90.6 | 37.5 | 86.8 | |
| DT | 58.7 | 66.7 | 77.3 | 78.6 | 65.6 | 0.00 | 92.2 | |
| Bone | RF | 76.1 | 91.7 | 72.7 | 87.5 | 87.5 | 12.5 | 91.4 |
| GB | 67.4 | 83.3 | 68.2 | 78.6 | 78.1 | 0.00 | 97.4 | |
| DT | 60.9 | 79.2 | 63.6 | 50.0 | 81.2 | 25.0 | 89.5 | |
| Cartilage | RF | 69.6 | 87.5 | 59.1 | 50.0 | 90.6 | 50.0 | 94.7 |
| GB | 63.0 | 75.0 | 77.3 | 57.1 | 81.2 | 37.5 | 84.2 | |
| DT | 63.0 | 75.0 | 86.4 | 57.1 | 71.9 | 37.5 | 86.6 | |
| WT-C | RF | 63.0 | 83.3 | 77.3 | 57.1 | 75.0 | 12.5 | 89.5 |
| GB | 60.9 | 75.0 | 77.3 | 57.1 | 75.0 | 28.6 | 86.8 | |
| DT | 60.9 | 75.0 | 72.7 | 57.1 | 81.2 | 25.0 | 84.2 |
| Impo | TOT | % | B-C | % |
|---|---|---|---|---|
| 1 | FemCartVOL | 6.69 | PatellaDENS | 8.54 |
| 2 | PatellaDENS | 5.69 | FemCartVOL | 7.91 |
| 3 | FemWallBelowSTDWeight | 4.22 | FemCartSURF | 7.28 |
| 4 | FemCartDENS | 3.77 | TibiaDENS | 6.16 |
| 5 | PatWallVar | 3.65 | PatCartDENS | 5.91 |
| 6 | LatWallMean | 3.45 | TibCartLatVOL | 5.41 |
| 7 | PatCartDENS | 3.41 | PatellaSTD | 5.35 |
| 8 | FemWallRMS | 3.16 | PatellaSURF | 4.72 |
| 9 | PatWallBelowSTDweight | 3.13 | FemCartDENS | 4.51 |
| 10 | TibiaDENS | 3.06 | TibCartLatSURF | 4.37 |
| 11 | FemCartSTD | 3.01 | PatellaVOL | 4.35 |
| 12 | FemWallMean | 2.82 | TibCartMedSURF | 4.30 |
| Impo | Bone | % | Cart | % |
| 1 | FemurDENS | 17.74 | FemCartVOL | 14.21 |
| 2 | FemurSTD | 16.87 | FemCartDENS | 11.07 |
| 3 | TibiaDENS | 15.20 | PatCartDENS | 9.31 |
| 4 | TibiaSTD | 12.57 | FemCartSURF | 8.14 |
| 5 | PatellaDENS | 12.21 | TibCartLatVOL | 6.84 |
| 6 | PatellaSTD | 10.24 | TibCartMedSTD | 6.51 |
| 7 | PatellaVOL | 7.69 | TibCartLatDENS | 5.96 |
| 8 | PatellaSURF | 7.48 | FemCartSTD | 5.85 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciliberti, F.K.; Guerrini, L.; Gunnarsson, A.E.; Recenti, M.; Jacob, D.; Cangiano, V.; Tesfahunegn, Y.A.; Islind, A.S.; Tortorella, F.; Tsirilaki, M.; et al. CT- and MRI-Based 3D Reconstruction of Knee Joint to Assess Cartilage and Bone. Diagnostics 2022, 12, 279. https://doi.org/10.3390/diagnostics12020279
Ciliberti FK, Guerrini L, Gunnarsson AE, Recenti M, Jacob D, Cangiano V, Tesfahunegn YA, Islind AS, Tortorella F, Tsirilaki M, et al. CT- and MRI-Based 3D Reconstruction of Knee Joint to Assess Cartilage and Bone. Diagnostics. 2022; 12(2):279. https://doi.org/10.3390/diagnostics12020279
Chicago/Turabian StyleCiliberti, Federica Kiyomi, Lorena Guerrini, Arnar Evgeni Gunnarsson, Marco Recenti, Deborah Jacob, Vincenzo Cangiano, Yonatan Afework Tesfahunegn, Anna Sigríður Islind, Francesco Tortorella, Mariella Tsirilaki, and et al. 2022. "CT- and MRI-Based 3D Reconstruction of Knee Joint to Assess Cartilage and Bone" Diagnostics 12, no. 2: 279. https://doi.org/10.3390/diagnostics12020279
APA StyleCiliberti, F. K., Guerrini, L., Gunnarsson, A. E., Recenti, M., Jacob, D., Cangiano, V., Tesfahunegn, Y. A., Islind, A. S., Tortorella, F., Tsirilaki, M., Jónsson, H., Jr., Gargiulo, P., & Aubonnet, R. (2022). CT- and MRI-Based 3D Reconstruction of Knee Joint to Assess Cartilage and Bone. Diagnostics, 12(2), 279. https://doi.org/10.3390/diagnostics12020279











