Immunological Aspects of Chronic Rhinosinusitis
Abstract
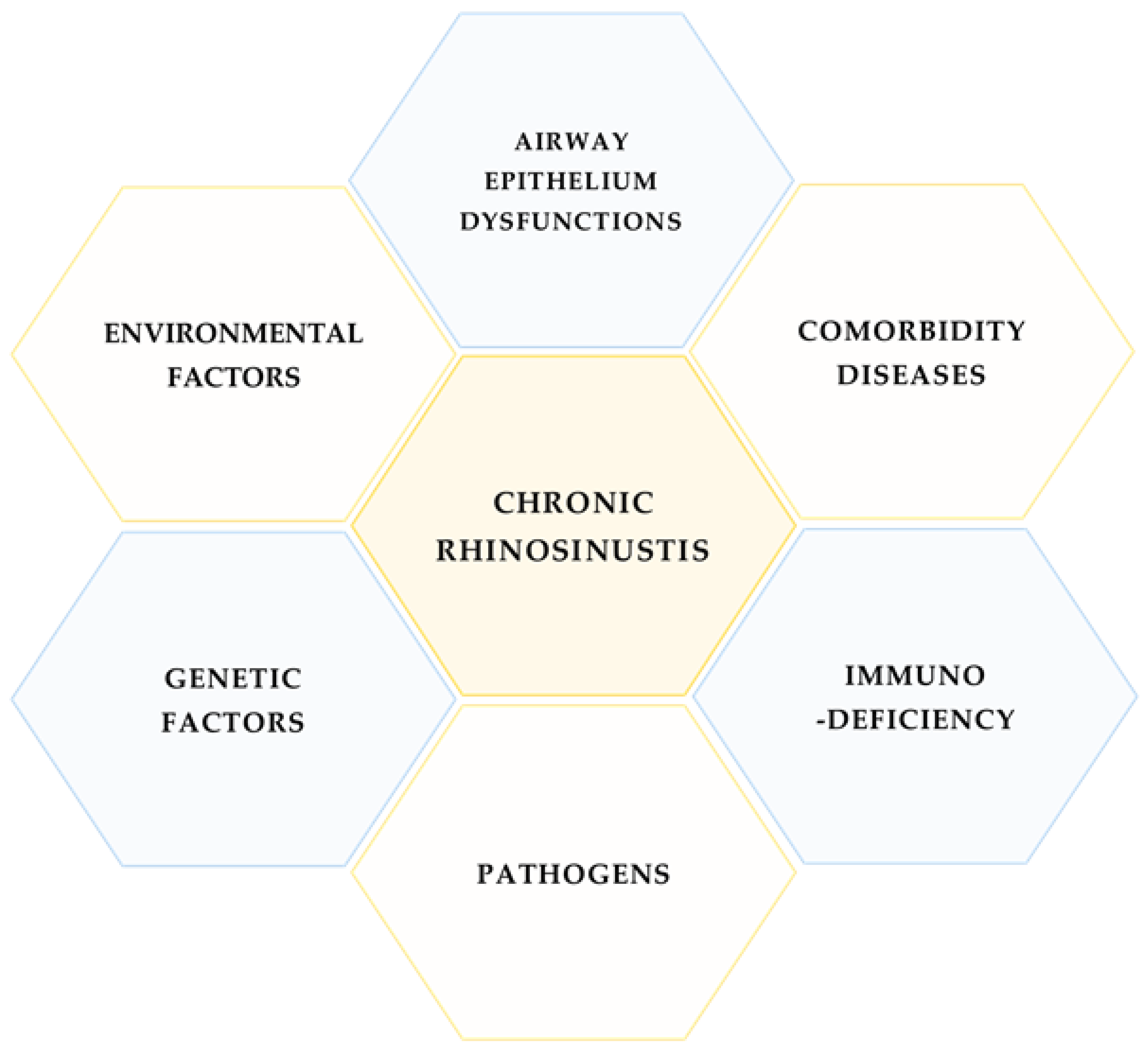
1. Introduction
2. Immunological Response Pathways in CRS
2.1. Airway Epithelium as the First Line of Defense in Sinuses
2.1.1. Structure and Functions of Sinusal Airway Epithelium
2.1.2. Secretory Functions of Airway Epithelium
2.1.3. Chemosensory Cells in the Pathogenesis of CRS
2.1.4. TLRs in the Pathogenesis of CRS
2.1.5. Hypoxic Conditions in the Airway Epithelium
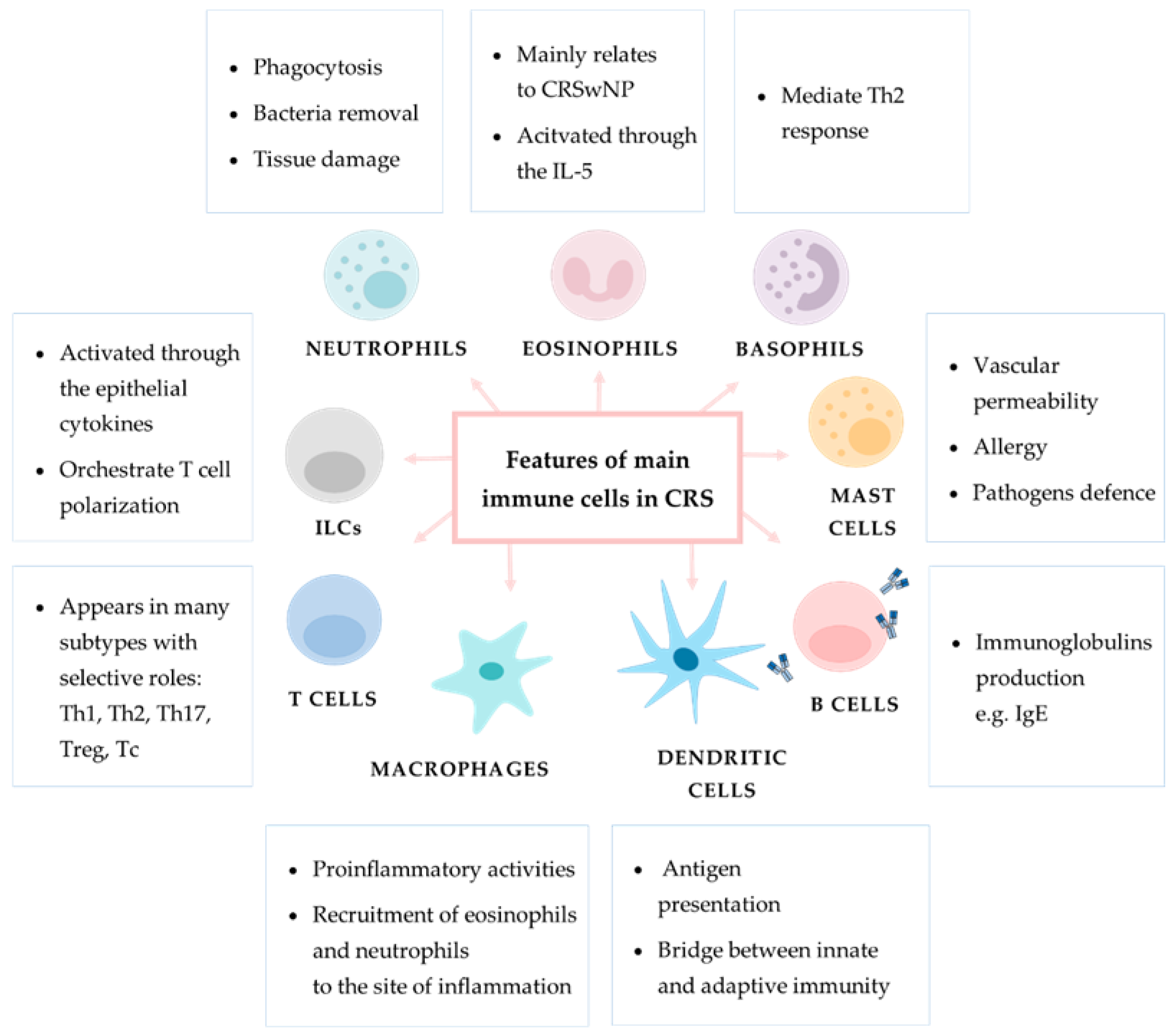
2.2. Innate Immune Cell Response in CRS
2.2.1. Innate Lymphoid Cells
2.2.2. Neutrophils
2.2.3. Monocytes and Macrophages
2.2.4. Basophils
2.2.5. Mast Cells
2.2.6. Eosinophils
2.2.7. Natural Killer Cells
2.3. Adaptive Immune Response Cells in CRS
2.3.1. Dendritic Cells
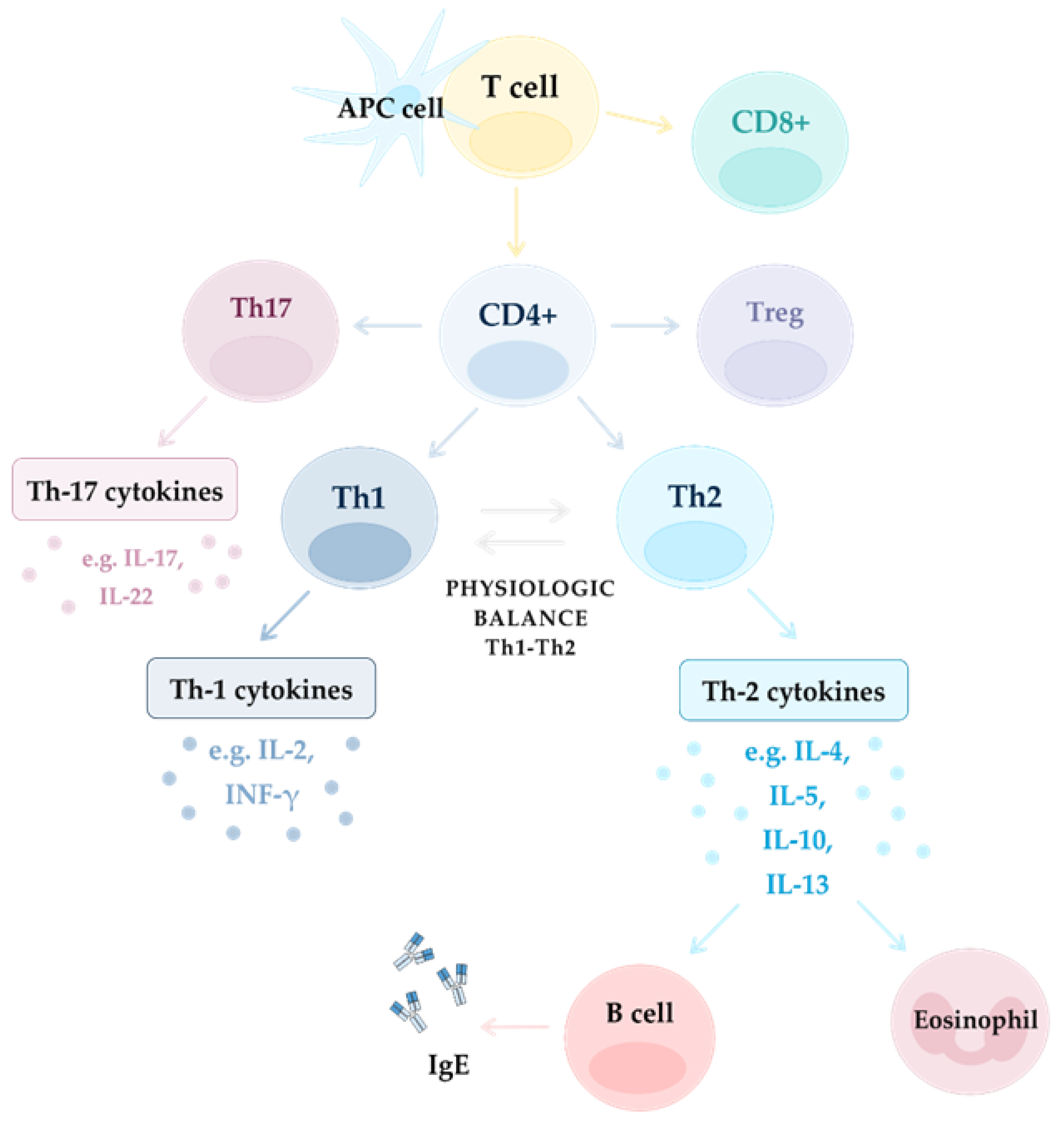
2.3.2. T Cells
2.3.3. B Cells
3. Other Selected Aspects of CRS Pathogenesis
3.1. High Mobility Group Box (HMGB1) Protein and a Receptor for Advanced Glycation and Products (RAGE) Pathway in CRS
3.2. Tissue Remodeling in CRS
3.3. Neuro-Inflammation as an Emerging New Aspect of Airway Inflammatory Diseases
3.4. Small Extracellular Vesicles (sEVs) as a New Promising Aim of Research in the Immunopathology of CRS
4. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AE | airway epithelium |
| AERD | aspirin exacerbated respiratory disease |
| AIR | adaptive immune response |
| BMT | basement membrane thickening |
| CIRP | cold-inducible RNA binding protein |
| CIS | complement immune system |
| CRS | chronic rhinosinusitis |
| CRSsNP | chronic rhinosinusitis without nasal polyps |
| CRSwNP | chronic rhinosinusitis with nasal polyps |
| CTGF | connective tissue growth factor |
| DCs | dendritic cells |
| DUOX | dual oxidase |
| ECM | extracellular matrix |
| EFs | environmental factors |
| EGR2 | early growth response 2 |
| EMT | epithelial to mesenchymal transition |
| GCs | goblet cells |
| HSP47 | heat shock protein 47 |
| HIF-1α | hypoxia-inducible factor 1α |
| HMGB1 | high mobility group box 1 protein |
| IFN-γ | interferon-γ |
| IIS | innate immune system |
| IL | interleukin |
| ILCs | innate lymphoid cells |
| JNK | c-Jun N-terminal kinases |
| MAPK | mitogen-activated protein kinase |
| MCs | mast cells |
| miRNAs | microRNAs |
| MMP | metalloproteinase |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NF-κβ | nuclear factor κβ |
| NKs | natural killer cells |
| NO | nitric oxide |
| NOX4 | NADPH oxidase 4 |
| NPs | nasal polyps |
| OX40L | OX40 ligand |
| PAMPs | pathogen associated molecular patterns |
| PCI | persistent chronic inflammation |
| PD-1 | programmed cell death 1 |
| PPAR-γ | peroxisome proliferator-activated receptor γ |
| RAGE | receptor for advanced glycation end products |
| rhHMGB1 | recombinant human high mobility group box 1 protein |
| ROS | reactive oxygen species |
| SCCs | solitary chemosensory cells |
| sEVs | small extracellular vesicles |
| SM | sinus mucosa |
| T2R38 | taste receptor 2 member 38 |
| T2Rs | taste receptors |
| TGFβ | transforming growth factor β |
| TIMP-2 | tissue inhibitor of metalloproteinase-2 |
| TJs | tight junctions |
| TLRs | Toll-like receptors |
| TNF-α | tumor necrosis factor α |
| Treg | T regulatory cell |
| TSLP | thymic stromal lymphopoietin |
| VEGF-A | vascular endothelial growth factor A |
| ZO-1 | zonula occludens |
References
- Fokkens, W.J.; Lund, V.J.; Hopkins, C.; Hellings, P.W.; Kern, R.; Reitsma, S.; Toppila-Salmi, S.; Bernal-Sprekelsen, M.; Mullol, J.; Alobid, I.; et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology 2020, 58, 1–464. [Google Scholar] [CrossRef] [PubMed]
- Klingler, A.I.; Stevens, W.W.; Tan, B.K.; Peters, A.T.; Poposki, J.A.; Grammer, L.C.; Welch, K.C.; Smith, S.S.; Conley, D.B.; Kern, R.C.; et al. Mechanisms and biomarkers of inflammatory endotypes in chronic rhinosinusitis without nasal polyps. J. Allergy Clin. Immunol. 2021, 147, 1306–1317. [Google Scholar] [CrossRef]
- Kato, A.; Peters, A.T.; Stevens, W.W.; Schleimer, R.P.; Tan, B.K.; Kern, R.C. Endotypes of chronic rhinosinusitis: Relationships to disease phenotypes, pathogenesis, clinical findings, and treatment approaches. Allergy 2022, 77, 812–826. [Google Scholar] [CrossRef]
- Jiao, J.; Duan, S.; Meng, N.; Li, Y.; Fan, E.; Zhang, L. Role of IFN-gamma, IL-13, and IL-17 on mucociliary differentiation of nasal epithelial cells in chronic rhinosinusitis with nasal polyps. Clin. Exp. Allergy 2016, 46, 449–460. [Google Scholar] [CrossRef]
- Bae, C.H.; Na, H.G.; Choi, Y.S.; Song, S.Y.; Kim, Y.D. Clusterin Induces MUC5AC Expression via Activation of NF-kappaB in Human Airway Epithelial Cells. Clin. Exp. Otorhinolaryngol. 2018, 11, 124–132. [Google Scholar] [CrossRef]
- Ito, T.; Ikeda, S.; Asamori, T.; Honda, K.; Kawashima, Y.; Kitamura, K.; Suzuki, K.; Tsutsumi, T. Increased expression of pendrin in eosinophilic chronic rhinosinusitis with nasal polyps. Braz. J. Otorhinolaryngol. 2019, 85, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Derycke, L.; Holtappels, G.; Wang, X.D.; Zhang, L.; Bachert, C.; Zhang, N. Th2 cytokines orchestrate the secretion of MUC5AC and MUC5B in IL-5-positive chronic rhinosinusitis with nasal polyps. Allergy 2019, 74, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Chang, E.H.; Chen, Y.; Lu, W.; Kim, M.M.; Niihori, M.; Hecker, L.; Kim, K.C. MUC1 contributes to goblet cell metaplasia and MUC5AC expression in response to cigarette smoke in vivo. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 319, L82–L90. [Google Scholar] [CrossRef]
- Ebenezer, J.A.; Christensen, J.M.; Oliver, B.G.; Oliver, R.A.; Tjin, G.; Ho, J.; Habib, A.R.; Rimmer, J.; Sacks, R.; Harvey, R.J. Periostin as a marker of mucosal remodelling in chronic rhinosinusitis. Rhinology 2017, 55, 234–241. [Google Scholar] [CrossRef]
- Du, K.; Wang, M.; Zhang, N.; Yu, P.; Wang, P.; Li, Y.; Wang, X.; Zhang, L.; Bachert, C. Involvement of the extracellular matrix proteins periostin and tenascin C in nasal polyp remodeling by regulating the expression of MMPs. Clin. Transl. Allergy 2021, 11, e12059. [Google Scholar] [CrossRef]
- Yan, B.; Lou, H.; Wang, Y.; Li, Y.; Meng, Y.; Qi, S.; Wang, M.; Xiao, L.; Wang, C.; Zhang, L. Epithelium-derived cystatin SN enhances eosinophil activation and infiltration through IL-5 in patients with chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2019, 144, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Yan, B.; Wang, Y.; Wang, C.; Zhang, L. Prognostic and pharmacologic value of cystatin SN for chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2021, 148, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.K.; Wendler, O.; Nocera, A.; Grundtner, P.; Schlegel, P.; Agaimy, A.; Iro, H.; Bleier, B.S. Escalation in mucus cystatin 2, pappalysin-A, and periostin levels over time predict need for recurrent surgery in chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2019, 9, 1212–1219. [Google Scholar] [CrossRef] [PubMed]
- Kao, S.S.; Bassiouni, A.; Ramezanpour, M.; Finnie, J.; Chegeni, N.; Colella, A.D.; Chataway, T.K.; Wormald, P.J.; Vreugde, S.; Psaltis, A.J. Proteomic analysis of nasal mucus samples of healthy patients and patients with chronic rhinosinusitis. J. Allergy Clin. Immunol. 2021, 147, 168–178. [Google Scholar] [CrossRef]
- Soler, Z.M.; Yoo, F.; Schlosser, R.J.; Mulligan, J.; Ramakrishnan, V.R.; Beswick, D.M.; Alt, J.A.; Mattos, J.L.; Payne, S.C.; Storck, K.A.; et al. Correlation of mucus inflammatory proteins and olfaction in chronic rhinosinusitis. Int. Forum Allergy Rhinol. 2020, 10, 343–355. [Google Scholar] [CrossRef]
- Wise, S.K.; Laury, A.M.; Katz, E.H.; Den Beste, K.A.; Parkos, C.A.; Nusrat, A. Interleukin-4 and interleukin-13 compromise the sinonasal epithelial barrier and perturb intercellular junction protein expression. Int. Forum Allergy Rhinol. 2014, 4, 361–370. [Google Scholar] [CrossRef]
- Meng, J.; Zhou, P.; Liu, Y.; Liu, F.; Yi, X.; Liu, S.; Holtappels, G.; Bachert, C.; Zhang, N. The development of nasal polyp disease involves early nasal mucosal inflammation and remodelling. PLoS ONE 2013, 8, e82373. [Google Scholar] [CrossRef]
- Soyka, M.B.; Wawrzyniak, P.; Eiwegger, T.; Holzmann, D.; Treis, A.; Wanke, K.; Kast, J.I.; Akdis, C.A. Defective epithelial barrier in chronic rhinosinusitis: The regulation of tight junctions by IFN-gamma and IL-4. J. Allergy Clin. Immunol. 2012, 130, 1087–1096.e1010. [Google Scholar] [CrossRef]
- Kaneko, Y.; Kohno, T.; Kakuki, T.; Takano, K.I.; Ogasawara, N.; Miyata, R.; Kikuchi, S.; Konno, T.; Ohkuni, T.; Yajima, R.; et al. The role of transcriptional factor p63 in regulation of epithelial barrier and ciliogenesis of human nasal epithelial cells. Sci. Rep. 2017, 7, 10935. [Google Scholar] [CrossRef]
- Boscke, R.; Vladar, E.K.; Konnecke, M.; Husing, B.; Linke, R.; Pries, R.; Reiling, N.; Axelrod, J.D.; Nayak, J.V.; Wollenberg, B. Wnt Signaling in Chronic Rhinosinusitis with Nasal Polyps. Am. J. Respir. Cell Mol. Biol. 2017, 56, 575–584. [Google Scholar] [CrossRef]
- Cho, D.Y.; Nayak, J.V.; Bravo, D.T.; Le, W.; Nguyen, A.; Edward, J.A.; Hwang, P.H.; Illek, B.; Fischer, H. Expression of dual oxidases and secreted cytokines in chronic rhinosinusitis. Int. Forum Allergy Rhinol. 2013, 3, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Jin, H.R.; Eun, K.M.; Mo, J.H.; Cho, S.H.; Oh, S.; Cho, D.; Kim, D.W. The role of interleukin-33 in chronic rhinosinusitis. Thorax 2017, 72, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Johnston, L.K.; Bryce, P.J. Understanding Interleukin 33 and Its Roles in Eosinophil Development. Front. Med. 2017, 4, 51. [Google Scholar] [CrossRef]
- Shin, H.W.; Kim, D.K.; Park, M.H.; Eun, K.M.; Lee, M.; So, D.; Kong, I.G.; Mo, J.H.; Yang, M.S.; Jin, H.R.; et al. IL-25 as a novel therapeutic target in nasal polyps of patients with chronic rhinosinusitis. J. Allergy Clin. Immunol. 2015, 135, 1476–1485.e1477. [Google Scholar] [CrossRef]
- Park, S.K.; Jin, Y.D.; Park, Y.K.; Yeon, S.H.; Xu, J.; Han, R.N.; Rha, K.S.; Kim, Y.M. IL-25-induced activation of nasal fibroblast and its association with the remodeling of chronic rhinosinusitis with nasal polyposis. PLoS ONE 2017, 12, e0181806. [Google Scholar] [CrossRef] [PubMed]
- Liao, B.; Cao, P.P.; Zeng, M.; Zhen, Z.; Wang, H.; Zhang, Y.N.; Hu, C.Y.; Ma, J.; Li, Z.Y.; Song, J.; et al. Interaction of thymic stromal lymphopoietin, IL-33, and their receptors in epithelial cells in eosinophilic chronic rhinosinusitis with nasal polyps. Allergy 2015, 70, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Luo, S.; Li, B.; Dai, H.; Zhang, J. IL-25 contributes to lung fibrosis by directly acting on alveolar epithelial cells and fibroblasts. Exp. Biol. Med. 2019, 244, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Boita, M.; Bucca, C.; Riva, G.; Heffler, E.; Rolla, G. Release of Type 2 Cytokines by Epithelial Cells of Nasal Polyps. J. Immunol. Res. 2016, 2016, 2643297. [Google Scholar] [CrossRef] [PubMed]
- Nagarkar, D.R.; Poposki, J.A.; Tan, B.K.; Comeau, M.R.; Peters, A.T.; Hulse, K.E.; Suh, L.A.; Norton, J.; Harris, K.E.; Grammer, L.C.; et al. Thymic stromal lymphopoietin activity is increased in nasal polyps of patients with chronic rhinosinusitis. J. Allergy Clin. Immunol. 2013, 132, 593–600.e512. [Google Scholar] [CrossRef]
- Soyka, M.B.; Holzmann, D.; Basinski, T.M.; Wawrzyniak, M.; Bannert, C.; Burgler, S.; Akkoc, T.; Treis, A.; Ruckert, B.; Akdis, M.; et al. The Induction of IL-33 in the Sinus Epithelium and Its Influence on T-Helper Cell Responses. PLoS ONE 2015, 10, e0123163. [Google Scholar] [CrossRef]
- Chiappara, G.; Sciarrino, S.; Di Sano, C.; Gallina, S.; Speciale, R.; Lorusso, F.; Di Vincenzo, S.; D’Anna, C.; Bruno, A.; Gjomarkaj, M.; et al. Notch-1 signaling activation sustains overexpression of interleukin 33 in the epithelium of nasal polyps. J. Cell. Physiol. 2019, 234, 4582–4596. [Google Scholar] [CrossRef] [PubMed]
- Poposki, J.A.; Klingler, A.I.; Tan, B.K.; Soroosh, P.; Banie, H.; Lewis, G.; Hulse, K.E.; Stevens, W.W.; Peters, A.T.; Grammer, L.C.; et al. Group 2 innate lymphoid cells are elevated and activated in chronic rhinosinusitis with nasal polyps. Immun. Inflamm. Dis. 2017, 5, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Ma, R.; Zhang, J.; Wu, X.; Yu, G.; Hu, X.; Li, J.; Liu, Z.; Ji, W.; Li, H.; et al. Excessive periostin expression and Th2 response in patients with nasal polyps: Association with asthma. J. Thorac. Dis. 2018, 10, 6585–6597. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Kulka, M.; Jo, A.; Eun, K.M.; Arizmendi, N.; Tancowny, B.P.; Hong, S.N.; Lee, J.P.; Jin, H.R.; Lockey, R.F.; et al. Cross-talk between human mast cells and epithelial cells by IgE-mediated periostin production in eosinophilic nasal polyps. J. Allergy Clin. Immunol. 2017, 139, 1692–1695.e1696. [Google Scholar] [CrossRef][Green Version]
- Barham, H.P.; Cooper, S.E.; Anderson, C.B.; Tizzano, M.; Kingdom, T.T.; Finger, T.E.; Kinnamon, S.C.; Ramakrishnan, V.R. Solitary chemosensory cells and bitter taste receptor signaling in human sinonasal mucosa. Int. Forum Allergy Rhinol. 2013, 3, 450–457. [Google Scholar] [CrossRef]
- Patel, N.N.; Kohanski, M.A.; Maina, I.W.; Triantafillou, V.; Workman, A.D.; Tong, C.C.L.; Kuan, E.C.; Bosso, J.V.; Adappa, N.D.; Palmer, J.N.; et al. Solitary chemosensory cells producing interleukin-25 and group-2 innate lymphoid cells are enriched in chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2018, 8, 900–906. [Google Scholar] [CrossRef]
- Kohanski, M.A.; Workman, A.D.; Patel, N.N.; Hung, L.Y.; Shtraks, J.P.; Chen, B.; Blasetti, M.; Doghramji, L.; Kennedy, D.W.; Adappa, N.D.; et al. Solitary chemosensory cells are a primary epithelial source of IL-25 in patients with chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2018, 142, 460–469.e467. [Google Scholar] [CrossRef]
- Lee, R.J.; Xiong, G.; Kofonow, J.M.; Chen, B.; Lysenko, A.; Jiang, P.; Abraham, V.; Doghramji, L.; Adappa, N.D.; Palmer, J.N.; et al. T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. J. Clin. Investig. 2012, 122, 4145–4159. [Google Scholar] [CrossRef]
- Triantafillou, V.; Workman, A.D.; Kohanski, M.A.; Cohen, N.A. Taste Receptor Polymorphisms and Immune Response: A Review of Receptor Genotypic-Phenotypic Variations and Their Relevance to Chronic Rhinosinusitis. Front. Cell. Infect. Microbiol. 2018, 8, 64. [Google Scholar] [CrossRef]
- Tran, H.T.T.; Herz, C.; Ruf, P.; Stetter, R.; Lamy, E. Human T2R38 Bitter Taste Receptor Expression in Resting and Activated Lymphocytes. Front. Immunol. 2018, 9, 2949. [Google Scholar] [CrossRef]
- Yan, C.H.; Hahn, S.; McMahon, D.; Bonislawski, D.; Kennedy, D.W.; Adappa, N.D.; Palmer, J.N.; Jiang, P.; Lee, R.J.; Cohen, N.A. Nitric oxide production is stimulated by bitter taste receptors ubiquitously expressed in the sinonasal cavity. Am. J. Rhinol. Allergy 2017, 31, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Grassin-Delyle, S.; Salvator, H.; Mantov, N.; Abrial, C.; Brollo, M.; Faisy, C.; Naline, E.; Couderc, L.J.; Devillier, P. Bitter Taste Receptors (TAS2Rs) in Human Lung Macrophages: Receptor Expression and Inhibitory Effects of TAS2R Agonists. Front. Physiol. 2019, 10, 1267. [Google Scholar] [CrossRef] [PubMed]
- Parker, D.; Prince, A. Innate immunity in the respiratory epithelium. Am. J. Respir. Cell Mol. Biol. 2011, 45, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. The role of pattern-recognition receptors in innate immunity: Update on Toll-like receptors. Nat. Immunol. 2010, 11, 373–384. [Google Scholar] [CrossRef]
- Vareille, M.; Kieninger, E.; Edwards, M.R.; Regamey, N. The airway epithelium: Soldier in the fight against respiratory viruses. Clin. Microbiol. Rev. 2011, 24, 210–229. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, C.; Ji, W.; Xu, Y.; Guo, H. Relationship of TLR2, TLR4 and tissue remodeling in chronic rhinosinusitis. Int. J. Clin. Exp. Pathol. 2015, 8, 1199–1212. [Google Scholar]
- Rai, G.; Das, S.; Ansari, M.A.; Singh, P.K.; Dar, S.A.; Haque, S.; Gupta, N.; Sharma, S.; Ramachandran, V.G.; Sharma, S.; et al. TLR-2 expression and dysregulated human Treg/Th17 phenotype in Aspergillus flavus infected patients of chronic rhinosinusitis with nasal polyposis. Microb. Cell Factories 2020, 19, 215. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, B.; Wang, C.; Huang, Q.; Zhang, Q.; Han, Y.; Dai, W.; Fan, E.; Li, Y. Biofilm formation and Toll-like receptor 2, Toll-like receptor 4, and NF-kappaB expression in sinus tissues of patients with chronic rhinosinusitis. Am. J. Rhinol. Allergy 2012, 26, 104–109. [Google Scholar] [CrossRef]
- Xiu, Q.; Kong, C.; Gao, Y.; Gao, Y.; Sha, J.; Cui, N.; Zhu, D. Hypoxia regulates IL-17A secretion from nasal polyp epithelial cells. Oncotarget 2017, 8, 102097–102109. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, Y.; Hu, X.; Xu, H.; Han, M.; Zhang, J.; Wen, W.; Shu, H.; Li, H. Overexpression of hypoxia-inducible factor 1alpha is associated with neutrophilic inflammation in chronic rhinosinusitis with nasal polyps. Auris Nasus Larynx 2020, 47, 401–409. [Google Scholar] [CrossRef]
- Pahl, A.; Szelenyi, S.; Brune, K. Hypoxia induced chemokine expression in nasal epithelial cells: Development of an in vitro model for chronic rhinosinusitis. ALTEX 2006, 23, 59–63. [Google Scholar] [PubMed]
- Yao, T.; Kojima, Y.; Koyanagi, A.; Yokoi, H.; Saito, T.; Kawano, K.; Furukawa, M.; Kusunoki, T.; Ikeda, K. Eotaxin-1, -2, and -3 immunoreactivity and protein concentration in the nasal polyps of eosinophilic chronic rhinosinusitis patients. Laryngoscope 2009, 119, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Cho, H.J.; Shin, W.C.; Song, H.A.; Yoon, J.H.; Kim, C.H. Hypoxia-mediated mechanism of MUC5AC production in human nasal epithelia and its implication in rhinosinusitis. PLoS ONE 2014, 9, e98136. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.I.; Verrier, T.; Vosshenrich, C.A.; Di Santo, J.P. Developmental options and functional plasticity of innate lymphoid cells. Curr. Opin. Immunol. 2017, 44, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Basic Immunology: Functions and Disorders of the Immune System; 6e: Sae-E-Book; Elsevier: New Delhi, India, 2019. [Google Scholar]
- Krabbendam, L.; Bal, S.M.; Spits, H.; Golebski, K. New insights into the function, development, and plasticity of type 2 innate lymphoid cells. Immunol. Rev. 2018, 286, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Krohn, I.K.; Shikhagaie, M.M.; Golebski, K.; Bernink, J.H.; Breynaert, C.; Creyns, B.; Diamant, Z.; Fokkens, W.J.; Gevaert, P.; Hellings, P.; et al. Emerging roles of innate lymphoid cells in inflammatory diseases: Clinical implications. Allergy 2018, 73, 837–850. [Google Scholar] [CrossRef]
- Shikhagaie, M.M.; Germar, K.; Bal, S.M.; Ros, X.R.; Spits, H. Innate lymphoid cells in autoimmunity: Emerging regulators in rheumatic diseases. Nat. Rev. Rheumatol. 2017, 13, 164–173. [Google Scholar] [CrossRef]
- Zhang, N.; Van Zele, T.; Perez-Novo, C.; Van Bruaene, N.; Holtappels, G.; DeRuyck, N.; Van Cauwenberge, P.; Bachert, C. Different types of T-effector cells orchestrate mucosal inflammation in chronic sinus disease. J. Allergy Clin. Immunol. 2008, 122, 961–968. [Google Scholar] [CrossRef]
- Derycke, L.; Zhang, N.; Holtappels, G.; Dutre, T.; Bachert, C. IL-17A as a regulator of neutrophil survival in nasal polyp disease of patients with and without cystic fibrosis. J. Cyst. Fibros. 2012, 11, 193–200. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.Y.; Jiang, W.X.; Liao, B.; Zhai, G.T.; Wang, N.; Zhen, Z.; Ruan, J.W.; Long, X.B.; Wang, H.; et al. The activation and function of IL-36gamma in neutrophilic inflammation in chronic rhinosinusitis. J. Allergy Clin. Immunol. 2018, 141, 1646–1658. [Google Scholar] [CrossRef]
- Pothoven, K.L.; Norton, J.E.; Suh, L.A.; Carter, R.G.; Harris, K.E.; Biyasheva, A.; Welch, K.; Shintani-Smith, S.; Conley, D.B.; Liu, M.C.; et al. Neutrophils are a major source of the epithelial barrier disrupting cytokine oncostatin M in patients with mucosal airways disease. J. Allergy Clin. Immunol. 2017, 139, 1966–1978.e9. [Google Scholar] [CrossRef] [PubMed]
- Carsuzaa, F.; Bequignon, E.; Bainaud, M.; Jegou, J.F.; Dufour, X.; Lecron, J.C.; Favot, L. Oncostatin M Counteracts the Fibrotic Effects of TGF-beta1 and IL-4 on Nasal-Polyp-Derived Fibroblasts: A Control of Fibrosis in Chronic Rhinosinusitis with Nasal Polyps? Int. J. Mol. Sci. 2022, 23, 6308. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Cao, C.; Li, Q.; Wen, X.; Guo, X.; Bao, Q.; Zhou, Y.; Li, L.; Ye, X.; Li, T.; et al. Local IL-17 positive T cells are functionally associated with neutrophil infiltration and their development is regulated by mucosal microenvironment in nasal polyps. Inflamm. Res. 2021, 70, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.W.; Kim, J.H.; Kim, H.J.; Choi, I.H.; Han, H.M.; Lee, K.J.; Kim, T.H.; Lee, S.H. Neutrophil extracellular traps in nasal secretions of patients with stable and exacerbated chronic rhinosinusitis and their contribution to induce chemokine secretion and strengthen the epithelial barrier. Clin. Exp. Allergy 2019, 49, 1306–1320. [Google Scholar] [CrossRef]
- Cao, Y.; Chen, F.; Sun, Y.; Hong, H.; Wen, Y.; Lai, Y.; Xu, Z.; Luo, X.; Chen, Y.; Shi, J.; et al. LL-37 promotes neutrophil extracellular trap formation in chronic rhinosinusitis with nasal polyps. Clin. Exp. Allergy 2019, 49, 990–999. [Google Scholar] [CrossRef]
- Takabayashi, T.; Kato, A.; Peters, A.T.; Hulse, K.E.; Suh, L.A.; Carter, R.; Norton, J.; Grammer, L.C.; Cho, S.H.; Tan, B.K.; et al. Excessive fibrin deposition in nasal polyps caused by fibrinolytic impairment through reduction of tissue plasminogen activator expression. Am. J. Respir. Crit. Care Med. 2013, 187, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Peric, A.; Baletic, N.; Sotirovic, J.; Spadijer-Mirkovic, C. Macrophage inflammatory protein-1 production and eosinophil infiltration in chronic rhinosinusitis with nasal polyps. Ann. Otol. Rhinol. Laryngol. 2015, 124, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, M.; Banaszewski, J.; Leszczynska, M.; Lagiedo-Zelazowska, M.; Nowicka, A.; Romanska, A.; Wierzbicka, M.; Dworacki, G. High frequency of macrophages expressing elevated level of CD80, PD-Ls and TLR1 in nasal polyps of CRS patients. Immunobiology 2019, 224, 154–162. [Google Scholar] [CrossRef]
- Ryu, G.; Bae, J.S.; Kim, J.H.; Kim, E.H.; Lyu, L.; Chung, Y.J.; Mo, J.H. Role of IL-17A in Chronic Rhinosinusitis With Nasal Polyp. Allergy Asthma Immunol. Res. 2020, 12, 507–522. [Google Scholar] [CrossRef]
- Voehringer, D. Protective and pathological roles of mast cells and basophils. Nat. Rev. Immunol. 2013, 13, 362–375. [Google Scholar] [CrossRef]
- Stevens, W.W.; Staudacher, A.G.; Hulse, K.E.; Poposki, J.A.; Kato, A.; Carter, R.G.; Suh, L.A.; Norton, J.E.; Huang, J.H.; Peters, A.T.; et al. Studies of the role of basophils in aspirin-exacerbated respiratory disease pathogenesis. J. Allergy Clin. Immunol. 2021, 148, 439–449.e435. [Google Scholar] [CrossRef] [PubMed]
- Pawankar, R.; Lee, K.H.; Nonaka, M.; Takizawa, R. Role of mast cells and basophils in chronic rhinosinusitis. Clin. Allergy Immunol. 2007, 20, 93–101. [Google Scholar] [PubMed]
- Takabayashi, T.; Kato, A.; Peters, A.T.; Suh, L.A.; Carter, R.; Norton, J.; Grammer, L.C.; Tan, B.K.; Chandra, R.K.; Conley, D.B.; et al. Glandular mast cells with distinct phenotype are highly elevated in chronic rhinosinusitis with nasal polyps. J. Allergy Clin. Immunol. 2012, 130, 410–420.e415. [Google Scholar] [CrossRef] [PubMed]
- Piliponsky, A.M.; Romani, L. The contribution of mast cells to bacterial and fungal infection immunity. Immunol. Rev. 2018, 282, 188–197. [Google Scholar] [CrossRef]
- Zhai, G.T.; Li, J.X.; Zhang, X.H.; Liao, B.; Lu, X.; Liu, Z. Increased accumulation of CD30 ligand-positive mast cells associates with eosinophilic inflammation in nasal polyps. Laryngoscope 2019, 129, E110–E117. [Google Scholar] [CrossRef]
- Zhai, G.T.; Wang, H.; Li, J.X.; Cao, P.P.; Jiang, W.X.; Song, J.; Yao, Y.; Wang, Z.C.; Wang, Z.Z.; Wang, M.C.; et al. IgD-activated mast cells induce IgE synthesis in B cells in nasal polyps. J. Allergy Clin. Immunol. 2018, 142, 1489–1499.e1423. [Google Scholar] [CrossRef]
- Cao, P.P.; Zhang, Y.N.; Liao, B.; Ma, J.; Wang, B.F.; Wang, H.; Zeng, M.; Liu, W.H.; Schleimer, R.P.; Liu, Z. Increased local IgE production induced by common aeroallergens and phenotypic alteration of mast cells in Chinese eosinophilic, but not non-eosinophilic, chronic rhinosinusitis with nasal polyps. Clin. Exp. Allergy 2014, 44, 690–700. [Google Scholar] [CrossRef]
- Cahill, K.N.; Laidlaw, T.M. Pathogenesis of Aspirin-Induced Reactions in Aspirin-Exacerbated Respiratory Disease. Immunol. Allergy Clin. N. Am. 2016, 36, 681–691. [Google Scholar] [CrossRef]
- Borish, L. Aspirin-exacerbated Respiratory Disease: A Syndrome of Mast Cell-mediated PgD2 Overproduction. Am. J. Respir. Crit. Care Med. 2019, 200, 651–652. [Google Scholar] [CrossRef]
- Lou, H.; Zhang, N.; Bachert, C.; Zhang, L. Highlights of eosinophilic chronic rhinosinusitis with nasal polyps in definition, prognosis, and advancement. Int. Forum Allergy Rhinol. 2018, 8, 1218–1225. [Google Scholar] [CrossRef]
- Soler, Z.M.; Sauer, D.; Mace, J.; Smith, T.L. Impact of mucosal eosinophilia and nasal polyposis on quality-of-life outcomes after sinus surgery. Otolaryngol. Head Neck Surg. 2010, 142, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Vlaminck, S.; Vauterin, T.; Hellings, P.W.; Jorissen, M.; Acke, F.; Van Cauwenberge, P.; Bachert, C.; Gevaert, P. The importance of local eosinophilia in the surgical outcome of chronic rhinosinusitis: A 3-year prospective observational study. Am. J. Rhinol. Allergy 2014, 28, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Donnell, N.J.; Marino, M.J.; Zarka, M.A.; Lal, D. Histopathological characteristics of surgical tissue from primary vs recurrent chronic rhinosinusitis with nasal polyposis patients. Laryngoscope Investig. Otolaryngol. 2020, 5, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Feng, T.; Li, T.; Cao, W.; Xu, J.; Guo, Y.; Ren, N.; Liu, D.; Zhou, P.; Liu, B.; Bao, X.; et al. Peripheral blood eosinophil levels in chronic rhinosinusitis and its predictive value in eosinophilic chronic rhinosinusitis. Acta Otolaryngol. 2021, 141, 386–391. [Google Scholar] [CrossRef]
- Kowalski, M.L.; Lewandowska-Polak, A.; Wozniak, J.; Ptasinska, A.; Jankowski, A.; Wagrowska-Danilewicz, M.; Danilewicz, M.; Pawliczak, R. Association of stem cell factor expression in nasal polyp epithelial cells with aspirin sensitivity and asthma. Allergy 2005, 60, 631–637. [Google Scholar] [CrossRef]
- Van Zele, T.; Coppieters, F.; Gevaert, P.; Holtappels, G.; Van Cauwenberge, P.; Bachert, C. Local complement activation in nasal polyposis. Laryngoscope 2009, 119, 1753–1758. [Google Scholar] [CrossRef]
- Smith, S.E.; Schlosser, R.J.; Yawn, J.R.; Mattos, J.L.; Soler, Z.M.; Mulligan, J.K. Sinonasal T-cell expression of cytotoxic mediators granzyme B and perforin is reduced in patients with chronic rhinosinusitis. Am. J. Rhinol. Allergy 2017, 31, 352–356. [Google Scholar] [CrossRef]
- Zhang, X.H.; Zhang, Y.N.; Li, H.B.; Hu, C.Y.; Wang, N.; Cao, P.P.; Liao, B.; Lu, X.; Cui, Y.H.; Liu, Z. Overexpression of miR-125b, a novel regulator of innate immunity, in eosinophilic chronic rhinosinusitis with nasal polyps. Am. J. Respir. Crit. Care Med. 2012, 185, 140–151. [Google Scholar] [CrossRef]
- Bachert, C.; Gevaert, P.; Holtappels, G.; Johansson, S.G.; van Cauwenberge, P. Total and specific IgE in nasal polyps is related to local eosinophilic inflammation. J. Allergy Clin. Immunol. 2001, 107, 607–614. [Google Scholar] [CrossRef]
- Esnault, S.; Kelly, E.A.; Johansson, M.W.; Liu, L.Y.; Han, S.T.; Akhtar, M.; Sandbo, N.; Mosher, D.F.; Denlinger, L.C.; Mathur, S.K.; et al. Semaphorin 7A is expressed on airway eosinophils and upregulated by IL-5 family cytokines. Clin. Immunol. 2014, 150, 90–100. [Google Scholar] [CrossRef]
- Miechowski, W.; Czerwaty, K.; Godlewska, I.; Dzaman, K. Atopy as a specific predictor of response to systemic and local steroid therapy in patients with chronic rhinosinusitis without nasal polyps. Otolaryngol. Polska 2022, 76, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, G.E.; Cho, G.S.; Kwon, H.J.; Joo, C.H.; Kim, H.S.; Jang, Y.J. Natural killer cells from patients with chronic rhinosinusitis have impaired effector functions. PLoS ONE 2013, 8, e77177. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Zhuang, X.; Li, C.; Wang, X. Interactions between dendritic cells and T lymphocytes in pathogenesis of nasal polyps. Exp. Ther. Med. 2018, 15, 5167–5172. [Google Scholar] [CrossRef]
- Kortekaas Krohn, I.; Bobic, S.; Dooley, J.; Lan, F.; Zhang, N.; Bachert, C.; Steelant, B.; Bullens, D.M.; Liston, A.; Ceuppens, J.L.; et al. Programmed cell death-1 expression correlates with disease severity and IL-5 in chronic rhinosinusitis with nasal polyps. Allergy 2017, 72, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Zhang, H.L.; Zhi, L.L.; Jin, P.; Zhao, L.; Li, T.; Zhou, X.M.; Sun, D.S.; Cheng, G.H.; Xin, Q.; et al. CDK5 Regulates PD-L1 Expression and Cell Maturation in Dendritic Cells of CRSwNP. Inflammation 2019, 42, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.L.; Song, J.; Xiong, P.; Cao, P.P.; Liao, B.; Ma, J.; Zhang, Y.N.; Zeng, M.; Liu, Y.; Wang, H.; et al. Disease-specific T-helper cell polarizing function of lesional dendritic cells in different types of chronic rhinosinusitis with nasal polyps. Am. J. Respir. Crit. Care Med. 2014, 190, 628–638. [Google Scholar] [CrossRef]
- Ma, Z.; Shen, Y.; Zeng, Q.; Liu, J.; Yang, L.; Fu, R.; Hu, G. MiR-150-5p regulates EGR2 to promote the development of chronic rhinosinusitis via the DC-Th axis. Int. Immunopharmacol. 2018, 54, 188–197. [Google Scholar] [CrossRef]
- Commins, S.P.; Borish, L.; Steinke, J.W. Immunologic messenger molecules: Cytokines, interferons, and chemokines. J. Allergy Clin. Immunol. 2010, 125, S53–S72. [Google Scholar] [CrossRef]
- Sallusto, F. Heterogeneity of Human CD4(+) T Cells Against Microbes. Annu. Rev. Immunol. 2016, 34, 317–334. [Google Scholar] [CrossRef]
- O’Shea, J.J.; Paul, W.E. Mechanisms underlying lineage commitment and plasticity of helper CD4+ T cells. Science 2010, 327, 1098–1102. [Google Scholar] [CrossRef]
- Henry, E.K.; Inclan-Rico, J.M.; Siracusa, M.C. Type 2 cytokine responses: Regulating immunity to helminth parasites and allergic inflammation. Curr. Pharmacol. Rep. 2017, 3, 346–359. [Google Scholar] [CrossRef] [PubMed]
- Derycke, L.; Eyerich, S.; Van Crombruggen, K.; Perez-Novo, C.; Holtappels, G.; Deruyck, N.; Gevaert, P.; Bachert, C. Mixed T helper cell signatures in chronic rhinosinusitis with and without polyps. PLoS ONE 2014, 9, e97581. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, N.; Bo, M.; Holtappels, G.; Zheng, M.; Lou, H.; Wang, H.; Zhang, L.; Bachert, C. Diversity of TH cytokine profiles in patients with chronic rhinosinusitis: A multicenter study in Europe, Asia, and Oceania. J. Allergy Clin. Immunol. 2016, 138, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.K.; Klingler, A.I.; Poposki, J.A.; Stevens, W.W.; Peters, A.T.; Suh, L.A.; Norton, J.; Carter, R.G.; Hulse, K.E.; Harris, K.E.; et al. Heterogeneous inflammatory patterns in chronic rhinosinusitis without nasal polyps in Chicago, Illinois. J. Allergy Clin. Immunol. 2017, 139, 699–703.e7. [Google Scholar] [CrossRef] [PubMed]
- Biggs, T.C.; Hayes, S.M.; Harries, P.G.; Allan, R.N.; Walls, A.F.; Pender, S.L.F.; Salib, R.J. Immunological profiling of key inflammatory drivers of nasal polyp formation and growth in chronic rhinosinusitis. Rhinology 2019, 57, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Ickrath, P.; Kleinsasser, N.; Ding, X.; Ginzkey, C.; Beyersdorf, N.; Hagen, R.; Kerkau, T.; Hackenberg, S. Characterization of T-cell subpopulations in patients with chronic rhinosinusitis with nasal polyposis. Allergy Rhinol. 2017, 8, 139–147. [Google Scholar] [CrossRef]
- Ickrath, P.; Scherzad, A.; Kleinsasser, N.; Ginzkey, C.; Hagen, R.; Hackenberg, S. Influence of nasal polyp tissue on the differentiation and activation of T lymphocytes in a co-culture system. Biomed. Rep. 2019, 10, 119–126. [Google Scholar] [CrossRef]
- Ma, J.; Shi, L.L.; Deng, Y.K.; Wang, H.; Cao, P.P.; Long, X.B.; Zhang, X.H.; Liu, Y.; Zeng, M.; Liu, Z. CD8(+) T cells with distinct cytokine-producing features and low cytotoxic activity in eosinophilic and non-eosinophilic chronic rhinosinusitis with nasal polyps. Clin. Exp. Allergy 2016, 46, 1162–1175. [Google Scholar] [CrossRef]
- Workman, A.D.; Kohanski, M.A.; Cohen, N.A. Biomarkers in Chronic Rhinosinusitis with Nasal Polyps. Immunol. Allergy Clin. N. Am. 2018, 38, 679–692. [Google Scholar] [CrossRef]
- Kim, D.W.; Eun, K.M.; Roh, E.Y.; Shin, S.; Kim, D.K. Chronic Rhinosinusitis without Nasal Polyps in Asian Patients Shows Mixed Inflammatory Patterns and Neutrophil-Related Disease Severity. Mediat. Inflamm. 2019, 2019, 7138643. [Google Scholar] [CrossRef]
- Palmer, C.; Mulligan, J.K.; Smith, S.E.; Atkinson, C. The role of regulatory T cells in the regulation of upper airway inflammation. Am. J. Rhinol. Allergy 2017, 31, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Watanabe, S.; Sivam, A.; Wang, J.; Neuwirth, S.J.; Perez, R.I.; De Tineo, M.; Baroody, F.M.; Naclerio, R.M.; Pinto, J.M. Peripheral blood and tissue T regulatory cells in chronic rhinosinusitis. Am. J. Rhinol. Allergy 2012, 26, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Munoz, A.; Hwang, P.H.; Nadeau, K.C. Migration of regulatory T cells toward airway epithelial cells is impaired in chronic rhinosinusitis with nasal polyposis. Clin. Immunol. 2010, 137, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Miljkovic, D.; Psaltis, A.; Wormald, P.J.; Vreugde, S. Naive and effector B-cell subtypes are increased in chronic rhinosinusitis with polyps. Am. J. Rhinol. Allergy 2018, 32, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Ickrath, P.; Kleinsasser, N.; Ding, X.; Ginzkey, C.; Beyersdorf, N.; Kerkau, T.; Hagen, R.; Hackenberg, S. Impact and Modulations of Peripheral and Edaphic B Cell Subpopulations in Chronic Rhinosinusitis With Nasal Polyposis. Clin. Exp. Otorhinolaryngol. 2018, 11, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Patadia, M.; Dixon, J.; Conley, D.; Chandra, R.; Peters, A.; Suh, L.A.; Kato, A.; Carter, R.; Harris, K.; Grammer, L.; et al. Evaluation of the presence of B-cell attractant chemokines in chronic rhinosinusitis. Am. J. Rhinol. Allergy 2010, 24, 11–16. [Google Scholar] [CrossRef]
- Xiao, L.; Wei, Y.; Zhang, Y.N.; Luo, X.; Yang, B.Y.; Yu, S.F.; Wu, X.M.; Wu, C.Y.; Li, H.B. Increased IL-21 expression in chronic rhinosinusitis with nasalpolyps. Clin. Exp. Allergy 2015, 45, 404–413. [Google Scholar] [CrossRef]
- Feldman, S.; Kasjanski, R.; Poposki, J.; Hernandez, D.; Chen, J.N.; Norton, J.E.; Suh, L.; Carter, R.G.; Stevens, W.W.; Peters, A.T.; et al. Chronic airway inflammation provides a unique environment for B cell activation and antibody production. Clin. Exp. Allergy 2017, 47, 457–466. [Google Scholar] [CrossRef]
- Xu, J.; Lee, J.W.; Park, S.K.; Lee, S.B.; Yoon, Y.H.; Yeon, S.H.; Rha, K.S.; Choi, J.A.; Song, C.H.; Kim, Y.M. Toll-like receptor 9 ligands increase type I interferon induced B-cell activating factor expression in chronic rhinosinusitis with nasal polyposis. Clin. Immunol. 2018, 197, 19–26. [Google Scholar] [CrossRef]
- Wang, G.; Li, M.; Zheng, J.; Zhan, J.; Zheng, H.; Li, R.; Wei, X. Circulating BAFF as novel biomarker in distinguishing chronic rhinosinusitis with nasal polyps endotypes and predicting postoperative recurrence. Int. Immunopharmacol. 2022, 104, 108515. [Google Scholar] [CrossRef]
- Dzaman, K.; Szczepanski, M.J.; Molinska-Glura, M.; Krzeski, A.; Zagor, M. Expression of the receptor for advanced glycation end products, a target for high mobility group box 1 protein, and its role in chronic recalcitrant rhinosinusitis with nasal polyps. Arch. Immunol. Ther. Exp. 2015, 63, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Dzaman, K.; Zagor, M.; Molinska-Glura, M.; Krzeski, A. High motility group box 1 (HMGB1) protein and its receptor for advanced glycation end products (RAGE) expression in chronic rhinosinusitis without nasal polyps. Folia Histochem. Cytobiol. 2015, 53, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Mao, M.; Bellussi, L.M.; Passali, D.; Chen, L. Increase of high mobility group box chromosomal protein 1 in eosinophilic chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2014, 4, 453–462. [Google Scholar] [CrossRef]
- Taziki, M.H.; Azarhoush, R.; Taziki, M.M.; Naghavi-Alhosseini, M.; Javid, N.; Davoodi, H. Correlation Between HMGB1 and TLR4 Expression in Sinonasal Mucosa in Patients With Chronic Rhinosinusitis. Ear Nose Throat J. 2019, 98, 599–605. [Google Scholar] [CrossRef]
- Bellussi, L.M.; Chen, L.; Chen, D.; Passali, F.M.; Passali, D. The role of High Mobility Group Box 1 chromosomal protein in the pathogenesis of chronic sinusitis and nasal polyposis. Acta Otorhinolaryngol. Ital. 2012, 32, 386–392. [Google Scholar] [PubMed]
- Min, H.J.; Kim, J.H.; Yoo, J.E.; Oh, J.H.; Kim, K.S.; Yoon, J.H.; Kim, C.H. ROS-dependent HMGB1 secretion upregulates IL-8 in upper airway epithelial cells under hypoxic condition. Mucosal Immunol. 2017, 10, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Gouzos, M.; Ramezanpour, M.; Bassiouni, A.; Psaltis, A.J.; Wormald, P.J.; Vreugde, S. Antibiotics Affect ROS Production and Fibroblast Migration in an In-vitro Model of Sinonasal Wound Healing. Front. Cell. Infect. Microbiol. 2020, 10, 110. [Google Scholar] [CrossRef]
- Vetuschi, A.; Pompili, S.; Di Marco, G.P.; Calvaruso, F.; Iacomino, E.; Angelosante, L.; Festuccia, C.; Colapietro, A.; Sferra, R. Can the AGE/RAGE/ERK signalling pathway and the epithelial-to-mesenchymal transition interact in the pathogenesis of chronic rhinosinusitis with nasal polyps? Eur. J. Histochem. 2020, 64, 3079. [Google Scholar] [CrossRef]
- Min, H.J.; Choe, J.W.; Kim, K.S.; Yoon, J.H.; Kim, C.H. High-mobility group box 1 protein induces epithelialmesenchymal transition in upper airway epithelial cells. Rhinology 2020, 58, 495–505. [Google Scholar] [CrossRef]
- Lee, S.H.; Cho, J.H.; Park, J.H.; Cho, J.S.; Lee, H.M. High Mobility Group Box Chromosomal Protein-1 Induces Myofibroblast Differentiation and Extracellular Matrix Production via RAGE, p38, JNK and AP-1 Signaling Pathways in Nasal Fibroblasts. Am. J. Rhinol. Allergy 2021, 35, 774–780. [Google Scholar] [CrossRef]
- Yang, P.; Chen, S.; Zhong, G.; Kong, W.; Wang, Y. Agonist of PPAR-gamma Reduced Epithelial-Mesenchymal Transition in Eosinophilic Chronic Rhinosinusitis with Nasal Polyps via Inhibition of High Mobility Group Box1. Int. J. Med. Sci. 2019, 16, 1631–1641. [Google Scholar] [CrossRef] [PubMed]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Zhou, C.; Ma, C.; Cao, Y.; Hu, G.; Li, H. TGF-beta1 induces epithelial-to-mesenchymal transition in chronic rhinosinusitis with nasal polyps through microRNA-182. Asian Pac. J. Allergy Immunol. 2021, 39, 16–47. [Google Scholar] [CrossRef]
- Kim, H.J.; Park, J.H.; Shin, J.M.; Yang, H.W.; Lee, H.M.; Park, I.H. TGF-beta1-induced HSP47 regulates extracellular matrix accumulation via Smad2/3 signaling pathways in nasal fibroblasts. Sci. Rep. 2019, 9, 15563. [Google Scholar] [CrossRef] [PubMed]
- Shieh, J.M.; Tsai, Y.J.; Chi, J.C.; Wu, W.B. TGFbeta mediates collagen production in human CRSsNP nasal mucosa-derived fibroblasts through Smad2/3-dependent pathway and CTGF induction and secretion. J. Cell. Physiol. 2019, 234, 10489–10499. [Google Scholar] [CrossRef]
- Li, Y.; Tan, G.; Liu, J.; Ke, X.; Shen, Y.; Huang, J.; Hu, G.; Xiang, T.; Yang, Y. The Role of Relaxin-2 in Tissue Remodeling of Chronic Rhinosinusitis With Nasal Polyps. Am. J. Rhinol. Allergy 2019, 33, 490–499. [Google Scholar] [CrossRef]
- Li, X.; Li, C.; Zhu, G.; Yuan, W.; Xiao, Z.A. TGF-beta1 Induces Epithelial-Mesenchymal Transition of Chronic Sinusitis with Nasal Polyps through MicroRNA-21. Int. Arch. Allergy Immunol. 2019, 179, 304–319. [Google Scholar] [CrossRef]
- Shi, L.L.; Ma, J.; Deng, Y.K.; Chen, C.L.; Wang, H.; Cao, P.P.; Long, X.B.; Zeng, M.; Liu, Z. Cold-inducible RNA-binding protein contributes to tissue remodeling in chronic rhinosinusitis with nasal polyps. Allergy 2021, 76, 497–509. [Google Scholar] [CrossRef]
- He, F.; Liu, H.; Luo, W. The PI3K-Akt-HIF-1alpha Pathway Reducing Nasal Airway Inflammation and Remodeling in Nasal Polyposis. Ear Nose Throat J. 2021, 100, NP43–NP49. [Google Scholar] [CrossRef]
- Kang, J.H.; Yang, H.W.; Park, J.H.; Shin, J.M.; Kim, T.H.; Lee, S.H.; Lee, H.M.; Park, I.H. Lipopolysaccharide regulates thymic stromal lymphopoietin expression via TLR4/MAPK/Akt/NF-kappaB-signaling pathways in nasal fibroblasts: Differential inhibitory effects of macrolide and corticosteroid. Int. Forum Allergy Rhinol. 2021, 11, 144–152. [Google Scholar] [CrossRef]
- Park, J.H.; Shin, J.M.; Yang, H.W.; Kim, T.H.; Lee, S.H.; Lee, H.M.; Cho, J.G.; Park, I.H. Cigarette Smoke Extract Stimulates MMP-2 Production in Nasal Fibroblasts via ROS/PI3K, Akt, and NF-kappaB Signaling Pathways. Antioxidants 2020, 9, 739. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.M.; Kang, H.J.; Cho, J.S.; Park, I.H.; Lee, H.M. Nox4 mediates hypoxia-stimulated myofibroblast differentiation in nasal polyp-derived fibroblasts. Int. Arch. Allergy Immunol. 2012, 159, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.H.; Kim, T.H.; Lee, H.M.; Lee, S.H.; Choe, W.; Kim, H.K.; Lee, J.H.; Oh, K.H.; Lee, S.H. Overexpression of the superoxide anion and NADPH oxidase isoforms 1 and 4 (NOX1 and NOX4) in allergic nasal mucosa. Am. J. Rhinol. Allergy 2009, 23, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Hao, J.; Xiao, L.; Wang, M.; Zhao, Y.; Fan, D.; Li, Y.; Wang, X.; Zhang, L. Expression of nicotinamide adenine dinucleotide phosphate oxidase in chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2020, 10, 646–655. [Google Scholar] [CrossRef]
- Chiarella, E.; Lombardo, N.; Lobello, N.; Piazzetta, G.L.; Morrone, H.L.; Mesuraca, M.; Bond, H.M. Deficit in Adipose Differentiation in Mesenchymal Stem Cells Derived from Chronic Rhinosinusitis Nasal Polyps Compared to Nasal Mucosal Tissue. Int. J. Mol. Sci. 2020, 21, 9214. [Google Scholar] [CrossRef]
- Zhang, N.; Xu, J.; Jiang, C.; Lu, S. Neuro-Immune Regulation in Inflammation and Airway Remodeling of Allergic Asthma. Front. Immunol. 2022, 13, 894047. [Google Scholar] [CrossRef]
- Barnes, P.J. Neurogenic inflammation in the airways. Respir. Physiol. 2001, 125, 145–154. [Google Scholar] [CrossRef]
- Saunders, C.J.; Christensen, M.; Finger, T.E.; Tizzano, M. Cholinergic neurotransmission links solitary chemosensory cells to nasal inflammation. Proc. Natl. Acad. Sci. USA 2014, 111, 6075–6080. [Google Scholar] [CrossRef]
- Chen, W.C.; Chang, Y.T.; Chen, S.F.; Lin, W.C.; Su, Y.Y.; Luo, S.D. The symptom burden of autonomic dysfunction is positively associated with chronic rhinosinusitis status. Rhinology 2018, 56, 227–233. [Google Scholar] [CrossRef]
- Lin, J.K.; Wang, H.W.; Su, W.Y. Noradrenergic innervation of nasal polyps and polypoid mucosae. Auris Nasus Larynx 1996, 23, 121–126. [Google Scholar] [CrossRef]
- Cerejeira, R.; Fernandes, S.; Pinto Moura, C. Increased expression of alpha7nAChR in chronic rhinosinusitis: The intranasal cholinergic anti-inflammatory hypothesis. Auris Nasus Larynx 2016, 43, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Lin, W.C.; Yang, C.H.; Wu, C.N.; Luo, S.D. Autonomic dysfunction as an independent risk factor for uncontrolled inflammation in chronic rhinosinusitis following functional endoscopic sinus surgery. Rhinology 2019, 58, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Victores, A.J.; Chen, M.; Smith, A.; Lane, A.P. Olfactory loss in chronic rhinosinusitis is associated with neuronal activation of c-Jun N-terminal kinase. Int. Forum Allergy Rhinol. 2018, 8, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wang, X.; Wang, Y.; Zhao, Y. Exosomal long non-coding RNA GAS5 suppresses Th1 differentiation and promotes Th2 differentiation via downregulating EZH2 and T-bet in allergic rhinitis. Mol. Immunol. 2020, 118, 30–39. [Google Scholar] [CrossRef]
- Mueller, S.K.; Nocera, A.L.; Dillon, S.T.; Gu, X.; Wendler, O.; Otu, H.H.; Libermann, T.A.; Bleier, B.S. Noninvasive exosomal proteomic biosignatures, including cystatin SN, peroxiredoxin-5, and glycoprotein VI, accurately predict chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2019, 9, 177–186. [Google Scholar] [CrossRef]
- Miyake, M.M.; Workman, A.D.; Nocera, A.L.; Wu, D.; Mueller, S.K.; Finn, K.; Amiji, M.M.; Bleier, B.S. Discriminant analysis followed by unsupervised cluster analysis including exosomal cystatins predict presence of chronic rhinosinusitis, phenotype, and disease severity. Int. Forum Allergy Rhinol. 2019, 9, 1069–1076. [Google Scholar] [CrossRef]
- Mueller, S.K.; Nocera, A.L.; Workman, A.; Libermann, T.; Dillon, S.T.; Stegmann, A.; Wurm, J.; Iro, H.; Wendler, O.; Bleier, B.S. Significant polyomic and functional upregulation of the PAPP-A/IGFBP-4/5/IGF-1 axis in chronic rhinosinusitis with nasal polyps. Int. Forum Allergy Rhinol. 2020, 10, 546–555. [Google Scholar] [CrossRef]
- Mueller, S.K.; Nocera, A.L.; Dillon, S.T.; Libermann, T.A.; Wendler, O.; Bleier, B.S. Tissue and Exosomal Serine Protease Inhibitors Are Significantly Overexpressed in Chronic Rhinosinusitis With Nasal Polyps. Am. J. Rhinol. Allergy 2019, 33, 359–368. [Google Scholar] [CrossRef]
- Barham, H.P.; Osborn, J.L.; Snidvongs, K.; Mrad, N.; Sacks, R.; Harvey, R.J. Remodeling changes of the upper airway with chronic rhinosinusitis. Int. Forum Allergy Rhinol. 2015, 5, 565–572. [Google Scholar] [CrossRef]
- Stevens, W.W.; Lee, R.J.; Schleimer, R.P.; Cohen, N.A. Chronic rhinosinusitis pathogenesis. J. Allergy Clin. Immunol. 2015, 136, 1442–1453. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czerwaty, K.; Piszczatowska, K.; Brzost, J.; Ludwig, N.; Szczepański, M.J.; Dżaman, K. Immunological Aspects of Chronic Rhinosinusitis. Diagnostics 2022, 12, 2361. https://doi.org/10.3390/diagnostics12102361
Czerwaty K, Piszczatowska K, Brzost J, Ludwig N, Szczepański MJ, Dżaman K. Immunological Aspects of Chronic Rhinosinusitis. Diagnostics. 2022; 12(10):2361. https://doi.org/10.3390/diagnostics12102361
Chicago/Turabian StyleCzerwaty, Katarzyna, Katarzyna Piszczatowska, Jacek Brzost, Nils Ludwig, Mirosław J. Szczepański, and Karolina Dżaman. 2022. "Immunological Aspects of Chronic Rhinosinusitis" Diagnostics 12, no. 10: 2361. https://doi.org/10.3390/diagnostics12102361
APA StyleCzerwaty, K., Piszczatowska, K., Brzost, J., Ludwig, N., Szczepański, M. J., & Dżaman, K. (2022). Immunological Aspects of Chronic Rhinosinusitis. Diagnostics, 12(10), 2361. https://doi.org/10.3390/diagnostics12102361








