Biomarkers of Muscle Metabolism in Peripheral Artery Disease: A Dynamic NIRS-Assisted Study to Detect Adaptations Following Revascularization and Exercise Training
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Interventions
2.3. Outcome Measures
2.3.1. Hemodynamics
2.3.2. Physical Functioning
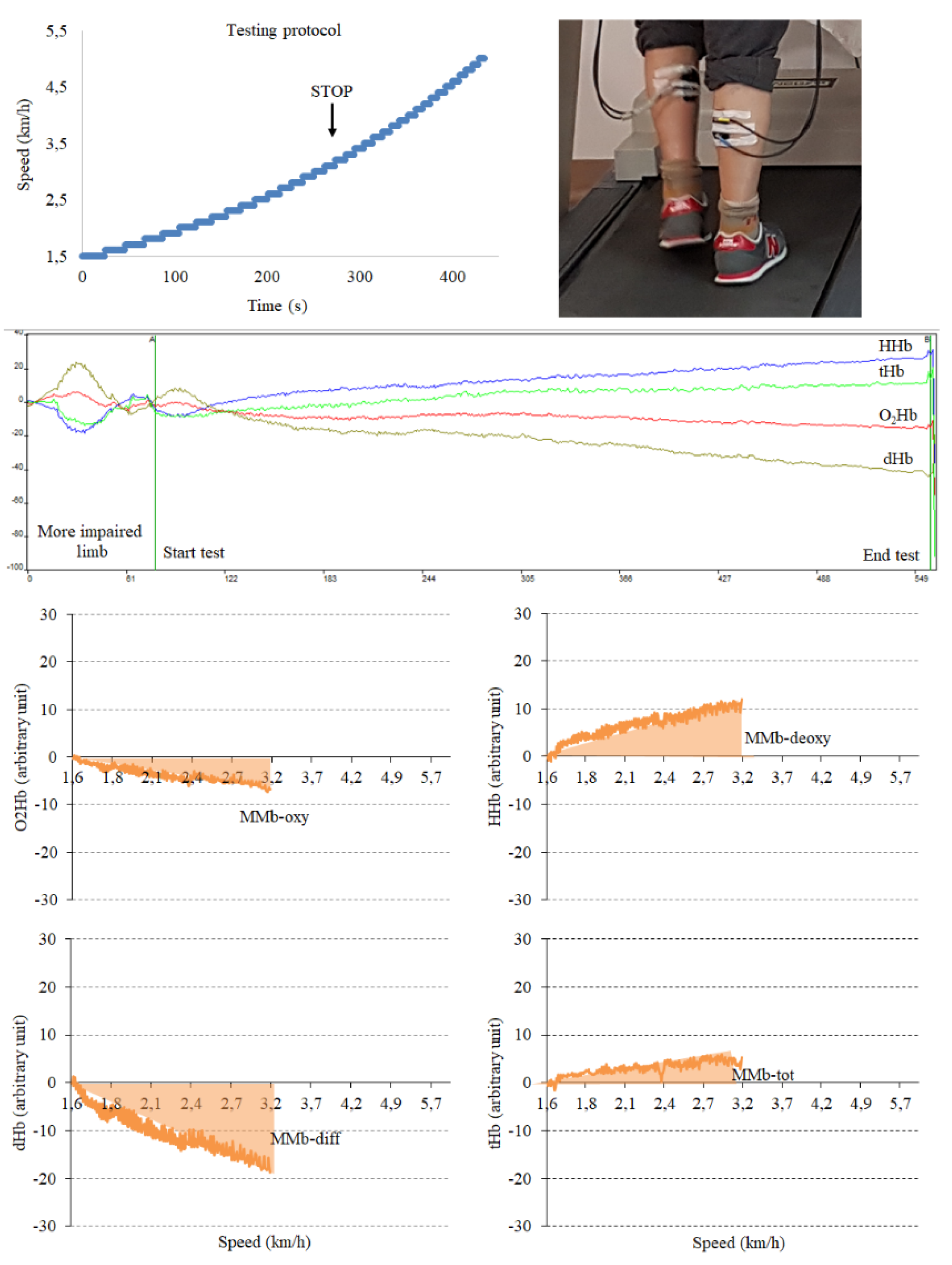
2.3.3. Muscle Metabolism and Related Biomarker Assessment
2.4. Statistical Analysis
3. Results
3.1. Interventions
3.2. Hemodynamics and Functional Capacity
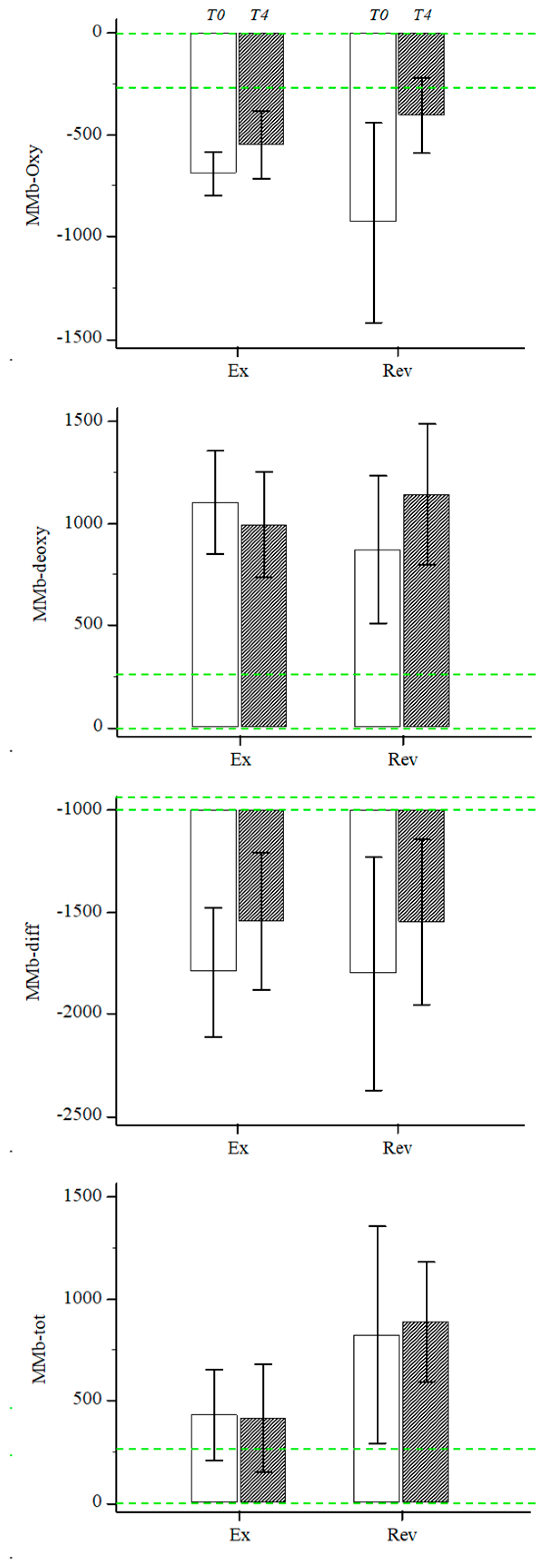
3.3. Muscle Metabolism Biomarkers
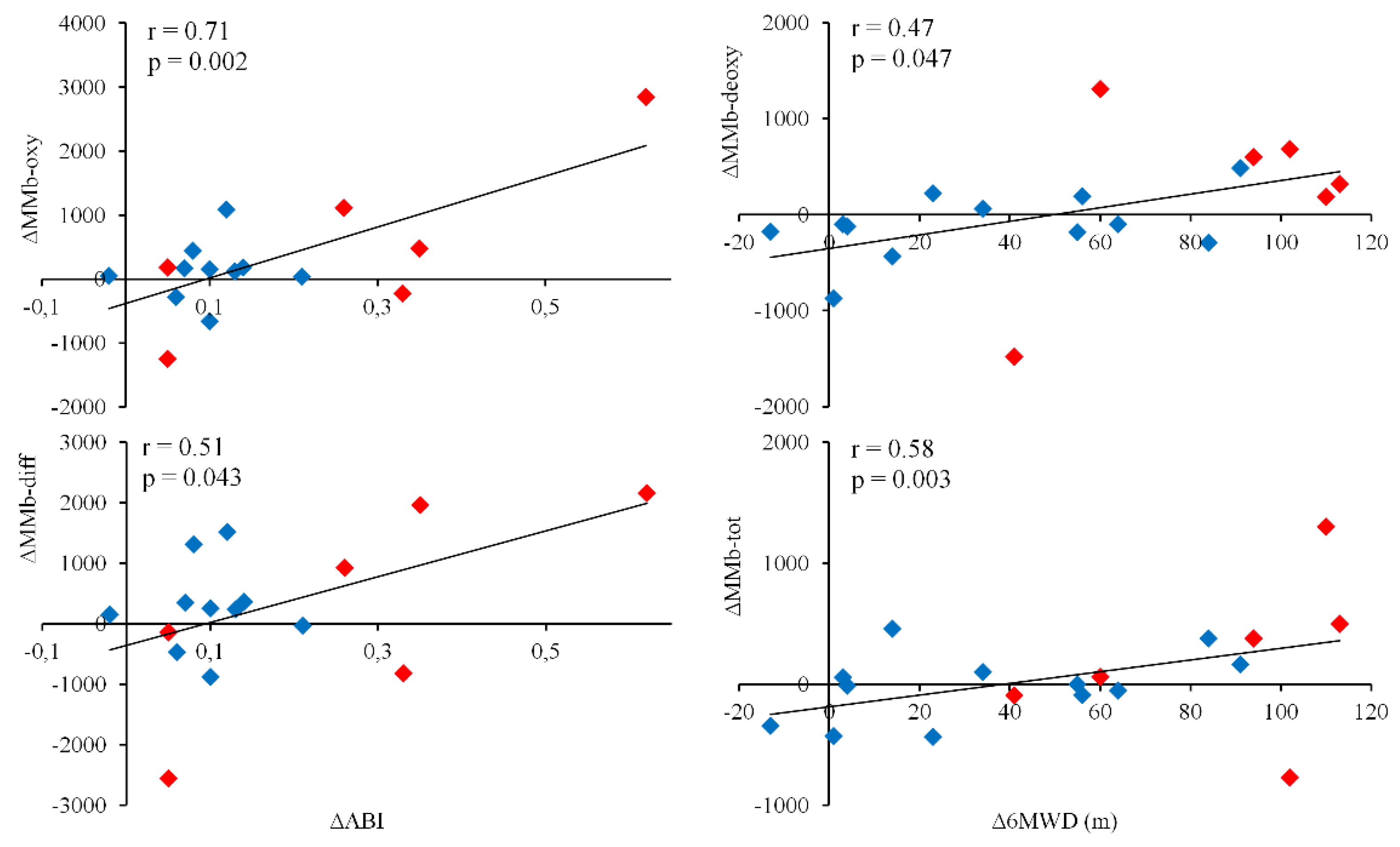
3.4. Relationship between Validated Outcome Measures and MMb
3.5. Relationship between Baseline Values of MMb and Changes in Functional Capacity after the Interventions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation 2019, 139, e56–e528, Erratum in: Circulation 2020, 141, e33. [Google Scholar] [CrossRef] [PubMed]
- Olin, J.W.; White, C.J.; Armstrong, E.J.; Kadian-Dodov, D.; Hiatt, W.R. Peripheral Artery Disease: Evolving Role of Exercise, Medical Therapy, and Endovascular Options. J. Am. Coll. Cardiol. 2016, 67, 1338–1357. [Google Scholar] [CrossRef] [PubMed]
- Grözinger, G.; Pohmann, R.; Schick, F.; Grosse, U.; Syha, R.; Brechtel, K.; Rittig, K.; Martirosian, P. Perfusion measurements of the calf in patients with peripheral arterial occlusive disease before and after percutaneous transluminal angioplasty using MR arterial spin labeling. J. Magn. Reson. Imaging 2014, 40, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Writing Committee Members; Gerhard-Herman, M.D.; Gornik, H.L.; Barrett, C.; Barshes, N.R.; Corriere, M.A.; Drachman, D.E.; Fleisher, L.A.; Fowkes, F.G.R.; Naomi, N.M.H.; et al. 2016 AHA/ACC Guideline on the Management of Patients with Lower Extremity Peripheral Artery Disease: Executive Summary. Vasc. Med. 2017, 22, NP1–NP43. [Google Scholar] [CrossRef]
- Aboyans, V.; Ricco, J.B.; Bartelink, M.E.L.; Björck, M.; Brodmann, M.; Cohnert, T. 2017 ESC Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases, in collaboration with the European Society for Vascular Surgery (ESVS). Eur. J. Vasc. Endovasc. Surg. 2018, 55, 305–368. [Google Scholar] [CrossRef]
- Vieira de Oliveira, G.; Soares, R.N.; Volino-Souza, M.; Murias, J.M.; Alvares, T.S. The association between near-infrared spectroscopy assessment of microvascular reactivity and flow-mediated dilation is disrupted in individuals at high risk for cardiovascular disease. Microcirculation 2019, 26, e12556. [Google Scholar] [CrossRef]
- Manfredini, F.; Lamberti, N.; Rossi, T.; Mascoli, F.; Basaglia, N.; Zamboni, P. A Toe Flexion NIRS assisted Test for Rapid Assessment of Foot Perfusion in Peripheral Arterial Disease: Feasibility, Validity, and Diagnostic Accuracy. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 187–194. [Google Scholar] [CrossRef]
- Casey, S.; Lanting, S.; Oldmeadow, C.; Chuter, V. The reliability of the ankle brachial index: A systematic review. J. Foot. Ankle. Res. 2019, 12, 39. [Google Scholar] [CrossRef]
- Amarteifio, E.; Weber, M.A.; Wormsbecher, S.; Demirel, S.; Krakowski-Roosen, H.; Jöres, A.; Braun, S.; Delorme, S.; Böckler, D.; Kauczor, H.U.; et al. Dynamic contrast-enhanced ultrasound for assessment of skeletal muscle microcirculation in peripheral arterial disease. Investig. Radiol. 2011, 46, 504–508. [Google Scholar] [CrossRef]
- Kalliokoski, K.K.; Kemppainen, J.; Larmola, K.; Takala, T.O.; Peltoniemi, P.; Oksanen, A.; Ruotsalainen, U.; Cobelli, C.; Knuuti, J.; Nuutila, P. Muscle blood flow and flow heterogeneity during exercise studied with positron emission tomography in humans. Eur. J. Appl. Physiol. 2000, 83, 395–401. [Google Scholar] [CrossRef]
- Ledermann, H.P.; Schulte, A.C.; Heidecker, H.G.; Aschwanden, M.; Jäger, K.A.; Scheffler, K.; Steinbrich, W.; Bilecen, D. Blood oxygenation level-dependent magnetic resonance imaging of the skeletal muscle in patients with peripheral arterial occlusive disease. Circulation 2006, 113, 2929–2935. [Google Scholar] [CrossRef] [PubMed]
- Kos, S.; Klarhöfer, M.; Aschwanden, M.; Scheffler, K.; Jacob, A.L.; Bilecen, D. Simultaneous dynamic blood oxygen level-dependent magnetic resonance imaging of foot and calf muscles: Aging effects at ischemia and postocclusive hyperemia in healthy volunteers. Investig. Radiol. 2009, 44, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Garcia, D.; de Bazelaire, C.; Alsop, D.C. Continuous flow-driven inversion for arterial spin labeling using pulsed radio frequency and gradient fields. Magn. Reson. Med. 2008, 60, 1488–1497. [Google Scholar] [CrossRef] [PubMed]
- Malagoni, A.M.; Felisatti, M.; Mandini, S.; Mascoli, F.; Manfredini, R.; Basaglia, N.; Zamboni, P.; Manfredini, F. Resting muscle oxygen consumption by near-infrared spectroscopy in peripheral arterial disease: A parameter to be considered in a clinical setting? Angiology 2010, 61, 530–536. [Google Scholar] [CrossRef]
- Vardi, M.; Nini, A. Near-infrared spectroscopy for evaluation of peripheral vascular disease. A systematic review of literature. Eur. J. Vasc. Endovasc. Surg. 2008, 35, 68–74. [Google Scholar] [CrossRef]
- Ubbink, D.T.; Koopman, B. Near-infrared spectroscopy in the routine diagnostic work-up of patients with leg ischaemia. Eur. J. Vasc. Endovasc. Surg. 2006, 31, 394–400. [Google Scholar] [CrossRef]
- Boezeman, R.P.; Moll, F.L.; Ünlü, Ç.; de Vries, J.P. Systematic review of clinical applications of monitoring muscle tissue oxygenation with near-infrared spectroscopy in vascular disease. Microvasc. Res. 2016, 104, 11–22. [Google Scholar] [CrossRef]
- Jones, S.; Chiesa, S.T.; Chaturvedi, N.; Hughes, A.D. Recent developments in near-infrared spectroscopy (NIRS) for the assessment of local skeletal muscle microvascular function and capacity to utilise oxygen. Artery Res. 2016, 16, 25–33. [Google Scholar] [CrossRef]
- Grassi, B.; Quaresima, V. Near-infrared spectroscopy and skeletal muscle oxidative function in vivo in health and disease: A review from an exercise physiology perspective. J. Biomed. Opt. 2016, 21, 091313. [Google Scholar] [CrossRef]
- Stoner, L.; Hanson, E.D.; Gram, M.; Allen, J.D.; Malin, S.K. Research Toolbox for Peripheral Arterial Disease—Minimally Invasive Assessment of the Vasculature and Skeletal Muscle. Circ. J. 2018, 82, 2462–2469. [Google Scholar] [CrossRef]
- Pedersen, B.L.; Bækgaard, N.; Quistorff, B. A near infrared spectroscopy-based test of calf muscle function in patients with peripheral arterial disease. Int. J. Angiol. 2015, 24, 25–34. [Google Scholar] [PubMed]
- Manfredini, F.; Malagoni, A.M.; Felisatti, M.; Mandini, S.; Mascoli, F.; Manfredini, R.; Basaglia, N.; Zamboni, P. A dynamic objective evaluation of peripheral arterial disease by near-infrared spectroscopy. Eur. J. Vasc. Endovasc. Surg. 2009, 38, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Perrey, S.; Ferrari, M. Muscle Oximetry in Sports Science: A Systematic Review. Sports Med. 2018, 48, 597–616. [Google Scholar] [CrossRef] [PubMed]
- Boezeman, R.P.; Becx, B.P.; van den Heuvel, D.A.; Ünlü, Ç.; Vos, J.A.; de Vries, J.P. Monitoring of Foot Oxygenation with Near-infrared Spectroscopy in Patients with Critical Limb Ischemia Undergoing Percutaneous Transluminal Angioplasty: A Pilot Study. Eur. J. Vasc. Endovasc. Surg. 2016, 52, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, F.; Malagoni, A.M.; Mandini, S.; Felisatti, M.; Mascoli, F.; Basaglia, N.; Manfredini, R.; Mikhailidis, D.P.; Zamboni, P. Near-infrared spectroscopy assessment following exercise training in patients with intermittent claudication and in untrained healthy participants. Vasc. Endovasc. Surg. 2012, 46, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Neidrauer, M.; Zubkov, L.; Weingarten, M.S.; Pourrezaei, K.; Papazoglou, E.S. Near infrared wound monitor helps clinical assessment of diabetic foot ulcers. J. Diabetes Sci. Technol. 2010, 4, 792–798. [Google Scholar] [CrossRef]
- Lamberti, N.; Manfredini, F.; Tessari, M.; Menegatti, E.; Nardi, F.; Basaglia, N.; Zamboni, P. A near-infrared spectroscopy-assisted test discriminates patients with peripheral arterial disease and venous insufficiency with changes of foot oxygenation following light elastic compression therapy. Vasa 2019, 48, 361–367. [Google Scholar] [CrossRef]
- Manfredini, F.; Malagoni, A.M.; Felisatti, M.; Mandini, S.; Lamberti, N.; Manfredini, R.; Mascoli, F.; Basaglia, N.; Zamboni, P. Acute oxygenation changes on ischemic foot of a novel intermittent pneumatic compression device and of an existing sequential device in severe peripheral arterial disease. BMC Cardiovasc. Disord. 2014, 14, 40. [Google Scholar] [CrossRef]
- Lamberti, N.; Malagoni, A.M.; Ficarra, V.; Basaglia, N.; Manfredini, R.; Zamboni, P.; Mascoli, F.; Manfredini, F. Structured Home-Based Exercise Versus Invasive Treatment: A Mission Impossible? A Pilot Randomized Study in Elderly Patients With Intermittent Claudication. Angiology 2016, 67, 772–780. [Google Scholar] [CrossRef]
- Manfredini, F.; Lamberti, N.; Malagoni, A.M.; Zambon, C.; Basaglia, N.; Mascoli, F.; Manfredini, R.; Zamboni, P. Reliability of the vascular claudication reporting in diabetic patients with peripheral arterial disease: A study with near-infrared spectroscopy. Angiology 2015, 66, 365–374. [Google Scholar] [CrossRef]
- Manfredini, F.; Malagoni, A.M.; Mascoli, F.; Mandini, S.; Taddia, M.C.; Basaglia, N.; Manfredini, R.; Conconi, F.; Zamboni, P. Training rather than walking: The test in -train out program for home-based rehabilitation in peripheral arteriopathy. Circ. J. 2008, 72, 946–952. [Google Scholar] [CrossRef] [PubMed]
- Malagoni, A.M.; Vagnoni, E.; Felisatti, M.; Mandini, S.; Heidari, M.; Mascoli, F.; Basaglia, N.; Manfredini, R.; Zamboni, P.; Manfredini, F. Evaluation of patient compliance, quality of life impact and cost-effectiveness of a “test in-train out” exercise-based rehabilitation program for patients with intermittent claudication. Circ. J. 2011, 75, 2128–2134. [Google Scholar] [CrossRef] [PubMed]
- Aboyans, V.; Criqui, M.H.; Abraham, P.; Allison, M.A.; Creager, M.A.; Diehm, C.; Fowkes, F.G.R.; Hiatt, W.R.; Jönsson, B.; Lacroix, P.; et al. Measurement and interpretation of the ankle-brachial index: A scientific statement from the American Heart Association. Circulation 2012, 126, 2890–2909. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, P.S.; Gardner, A.W. The clinical utility of a six-minute walk test in peripheral arterial occlusive disease patients. J. Am. Geriatr. Soc. 1998, 46, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, F.; Conconi, F.; Malagoni, A.M.; Manfredini, R.; Mascoli, F.; Liboni, A.; Zamboni, P. Speed rather than distance: A novel graded treadmill test to assess claudication. Eur. J. Vasc. Endovasc. Surg. 2004, 28, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Lamberti, N.; López-Soto, P.J.; Guerzoni, F.; Napoli, N.; Gasbarro, V.; Zamboni, P.; Tsolaki, E.; Taddia, M.C.; Rodríguez-Borrego, M.A.; Manfredini, R.; et al. Changes in exercise capacity and risk of all-cause mortality in patients with peripheral artery disease: A 10-year retrospective cohort study. Intern. Emerg. Med. 2020, 15, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, F.; Lamberti, N.; Guerzoni, F.; Napoli, N.; Gasbarro, V.; Zamboni, P.; Mascoli, F.; Manfredini, R.; Basaglia, N.; Rodríguez-Borrego, M.A.; et al. Rehabilitative Exercise Reduced the Impact of Peripheral Artery Disease on Vascular Outcomes in Elderly Patients with Claudication: A Three-Year Single Center Retrospective Study. J. Clin. Med. 2019, 8, 210. [Google Scholar] [CrossRef]
- Heiss, E.H.; Dirsch, V.M. Regulation of eNOS enzyme activity by posttranslational modification. Curr. Pharm. Des. 2014, 20, 3503–3513. [Google Scholar] [CrossRef]
- Murrow, J.R.; Brizendine, J.T.; Djire, B.; Young, H.J.; Rathbun, S.; Nilsson, K.R., Jr.; McCully, K.K. Near infrared spectroscopy-guided exercise training for claudication in peripheral arterial disease. Eur. J. Prev. Cardiol. 2019, 26, 471–480. [Google Scholar] [CrossRef]
- Manfredini, F.; Conconi, F.; Malagoni, A.M.; Manfredini, R.; Basaglia, N.; Mascoli, F.; Liboni, A.; Zamboni, P. Training guided by pain threshold speed. Effects of a home-based program on claudication. Int. Angiol. 2004, 23, 379–387. [Google Scholar]
- Boushel, R.; Langberg, H.; Olesen, J.; Gonzales-Alonzo, J.; Bülow, J.; Kjaer, M. Monitoring tissue oxygen availability with near infrared spectroscopy (NIRS) in health and disease. Scand. J. Med. Sci. Sports 2001, 11, 213–222. [Google Scholar] [CrossRef] [PubMed]

| Variable | Rev (n = 6) | Ex (n = 12) | p Value | |
|---|---|---|---|---|
| Age, years | 69 ± 8 | 68 ± 5 | 0.78 | |
| Male sex, n (%) | 6 (100) | 7 (58) | 0.07 | |
| Body mass index, kg/m2 | 25 ± 5 | 27 ± 3 | 0.27 | |
| PAD duration, years | 8 ± 6 | 8 ± 7 | 0.97 | |
| Risk factors, n (%) | ||||
| Hypertension | 6 (100) | 11 (92) | 0.48 | |
| Hyperlipidemia | 4 (67) | 7 (58) | 0.74 | |
| Diabetes | 2 (33) | 5 (42) | 0.74 | |
| Smoking | 6 (100) | 11 (92) | 0.48 | |
| Family history | 2 (33) | 3 (25) | 0.72 | |
| Comorbidities, n (%) | ||||
| Myocardial infarction | 3 (50) | 2 (18) | 0.15 | |
| Coronary heart disease | 4 (67) | 4 (33) | 0.19 | |
| Cerebrovascular disease | 0 (0) | 0 (0) | - | |
| Lung disease | 0 (0) | 1 (8) | 0.48 | |
| Lower limb revascularizations | 2 (40) | 4 (33) | 0.80 | |
| Charlson comorbidity index | 6 ± 2 | 6 ± 2 | 0.82 | |
| Therapy, n (%) | ||||
| Anticoagulants | 1 (20) | 2 (16) | 0.87 | |
| Antiplatelets | 6 (100) | 11 (92) | 0.48 | |
| Cilostazol/pentoxifylline | 0 (0) | 0 (0) | - | |
| Antihypertensive | 6 (100) | 11 (92) | 0.48 | |
| Statins | 4 (67) | 6 (50) | 0.27 | |
| Hypoglycemic agents/insulin | 2 (33) | 5 (42) | 0.74 | |
| Lesion location, n (%) | ||||
| Aortoiliac | 4 (67) | 4 (33) | 0.48 | |
| Femoropopliteal | 6 (100) | 12 (100) | - | |
| Infrapopliteal | 2 (33) | 5 (42) | 0.87 | |
| Outcome measures | ||||
| ABI, more impaired limb | 0.57 ± 0.17 | 0.56 ± 0.19 | 0.99 | |
| ABI, less impaired limb | 0.85 ± 0.27 | 0.81 ± 0.19 | 0.76 | |
| PFWD, m | 137 ± 41 | 173 ± 126 | 0.51 | |
| 6MWD, m | 306 ± 102 | 312 ± 67 | 0.88 | |
| Maximal speed, km/h | 3.1 ± 0.9 | 3.2 ± 1.0 | 0.83 | |
| NIRS parameters | ||||
| MMb-oxy | −930 ± 1204 | −691 ± 372 | 0.83 | |
| MMb-deoxy | 874 ± 886 | 1104 ± 872 | 0.39 | |
| MMb-diff | −1804 ± 1398 | −1795 ± 1096 | 0.83 | |
| MMb-tot | 826 ± 1304 | 432 ± 774 | 0.61 | |
| Variable | Rev (n = 12) | Ex (n = 6) | Between Groups | |||||
|---|---|---|---|---|---|---|---|---|
| T0 | T4 | ∆ | T0 | T4 | ∆ | ∆ | p Value | |
| ABI | 0.57 (0.38–0.75) | 0.84 * (0.70–0.99) | 0.28 (0.05–0.50) | 0.56 (0.43–0.70) | 0.66 * (0.53–0.80) | 0.10 (0.06–0.14) | 0.18 (0.10–0.26) | 0.025 |
| PFWD, m | 137 (94–179) | 248 * (141–355) | 111 (−5 +228) | 173 (92–253) | 275 * (187–362) | 101 (42–160) | 10 (−95 +115) | 0.84 |
| 6MWD, m | 306 (198–413) | 392 * (285–499) | 87 (56–118) | 312 (269–354) | 346 * (292–400) | 35 (13–57) | 52 (17–87) | 0.006 |
| MMb-oxy | −930 (−2117 −89) | −406 (−875 +64) | 525 (−924 +1974) | −691 (−927 −454) | −550 (−915 −184) | 141 (−154 +435) | 384 (−530 +1298) | 0.39 |
| MMb-deoxy | 874 (−56 +1803) | 1143 (261 −2025) | 270 (−718 +1257) | 1104 (550–1658) | 994 (430–1557) | −110 (−329 +109) | 379 (−255 +1014) | 0.22 |
| MMb-diff | −1804 (−3271 −336) | −1549 (−2594 −503) | 255 (−1632 +2142) | −1795 (−2491 −1098) | −1544 (−2280 −807) | 250 (−232 +733) | 5 (−1251 +1262) | 0.99 |
| MMb-tot | 826 (−543 +2194) | 889 (131 −1646) | 63 (−1008 +1134) | 432 (−60 +924) | 415 (−166 +996) | −17 (−198 +164) | 80 (−574 +374) | 0.80 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manfredini, F.; Lamberti, N.; Ficarra, V.; Tsolaki, E.; Straudi, S.; Zamboni, P.; Basaglia, N.; Gasbarro, V. Biomarkers of Muscle Metabolism in Peripheral Artery Disease: A Dynamic NIRS-Assisted Study to Detect Adaptations Following Revascularization and Exercise Training. Diagnostics 2020, 10, 312. https://doi.org/10.3390/diagnostics10050312
Manfredini F, Lamberti N, Ficarra V, Tsolaki E, Straudi S, Zamboni P, Basaglia N, Gasbarro V. Biomarkers of Muscle Metabolism in Peripheral Artery Disease: A Dynamic NIRS-Assisted Study to Detect Adaptations Following Revascularization and Exercise Training. Diagnostics. 2020; 10(5):312. https://doi.org/10.3390/diagnostics10050312
Chicago/Turabian StyleManfredini, Fabio, Nicola Lamberti, Valentina Ficarra, Elpiniki Tsolaki, Sofia Straudi, Paolo Zamboni, Nino Basaglia, and Vincenzo Gasbarro. 2020. "Biomarkers of Muscle Metabolism in Peripheral Artery Disease: A Dynamic NIRS-Assisted Study to Detect Adaptations Following Revascularization and Exercise Training" Diagnostics 10, no. 5: 312. https://doi.org/10.3390/diagnostics10050312
APA StyleManfredini, F., Lamberti, N., Ficarra, V., Tsolaki, E., Straudi, S., Zamboni, P., Basaglia, N., & Gasbarro, V. (2020). Biomarkers of Muscle Metabolism in Peripheral Artery Disease: A Dynamic NIRS-Assisted Study to Detect Adaptations Following Revascularization and Exercise Training. Diagnostics, 10(5), 312. https://doi.org/10.3390/diagnostics10050312









