Comparative Study Regarding the Properties of Methylene Blue and Proflavine and Their Optimal Concentrations for In Vitro and In Vivo Applications
Abstract
1. Introduction
2. Biological Actions of Methylene Blue and Proflavine and Methods
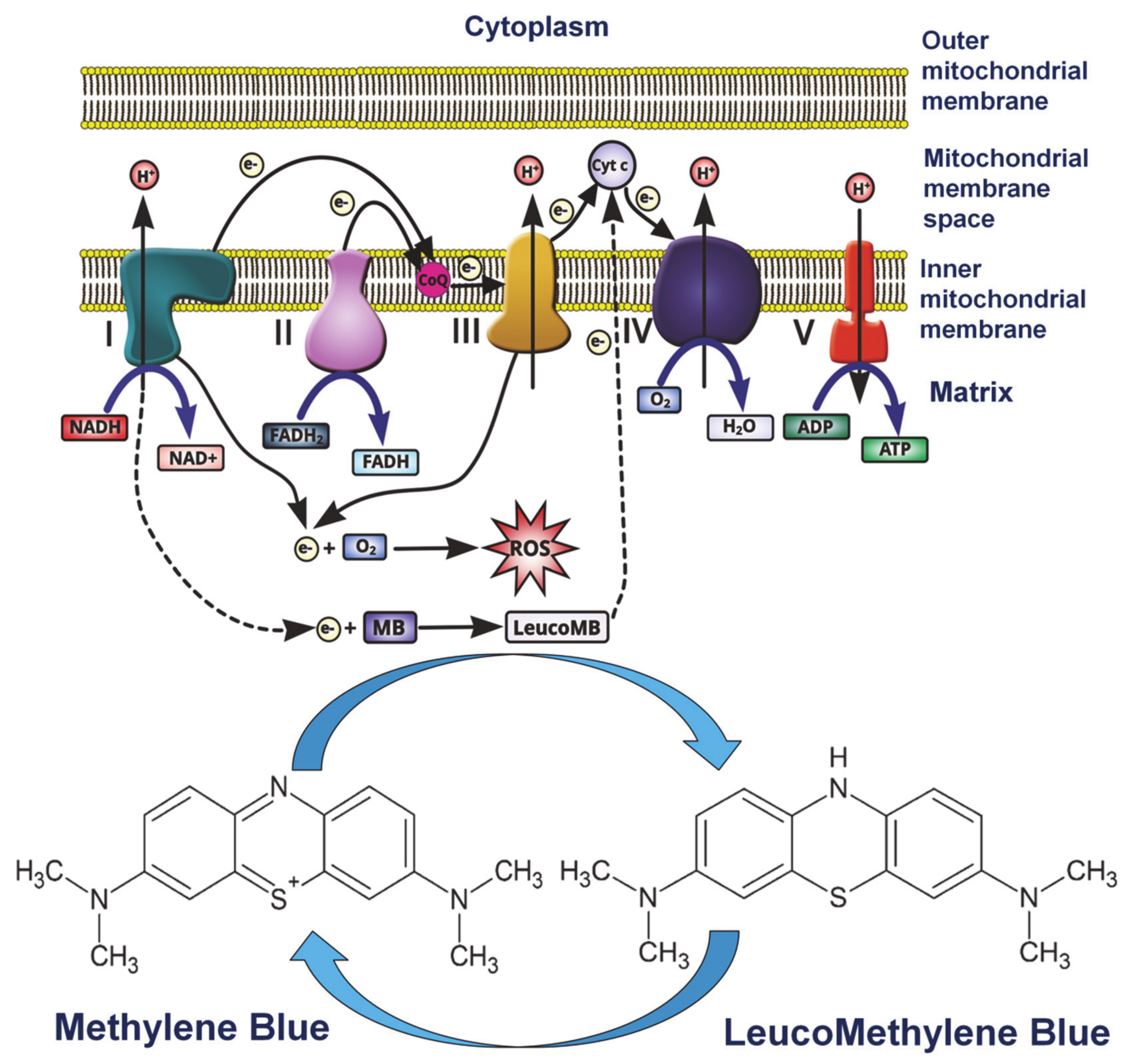
2.1. Methylene Blue
2.2. Proflavine
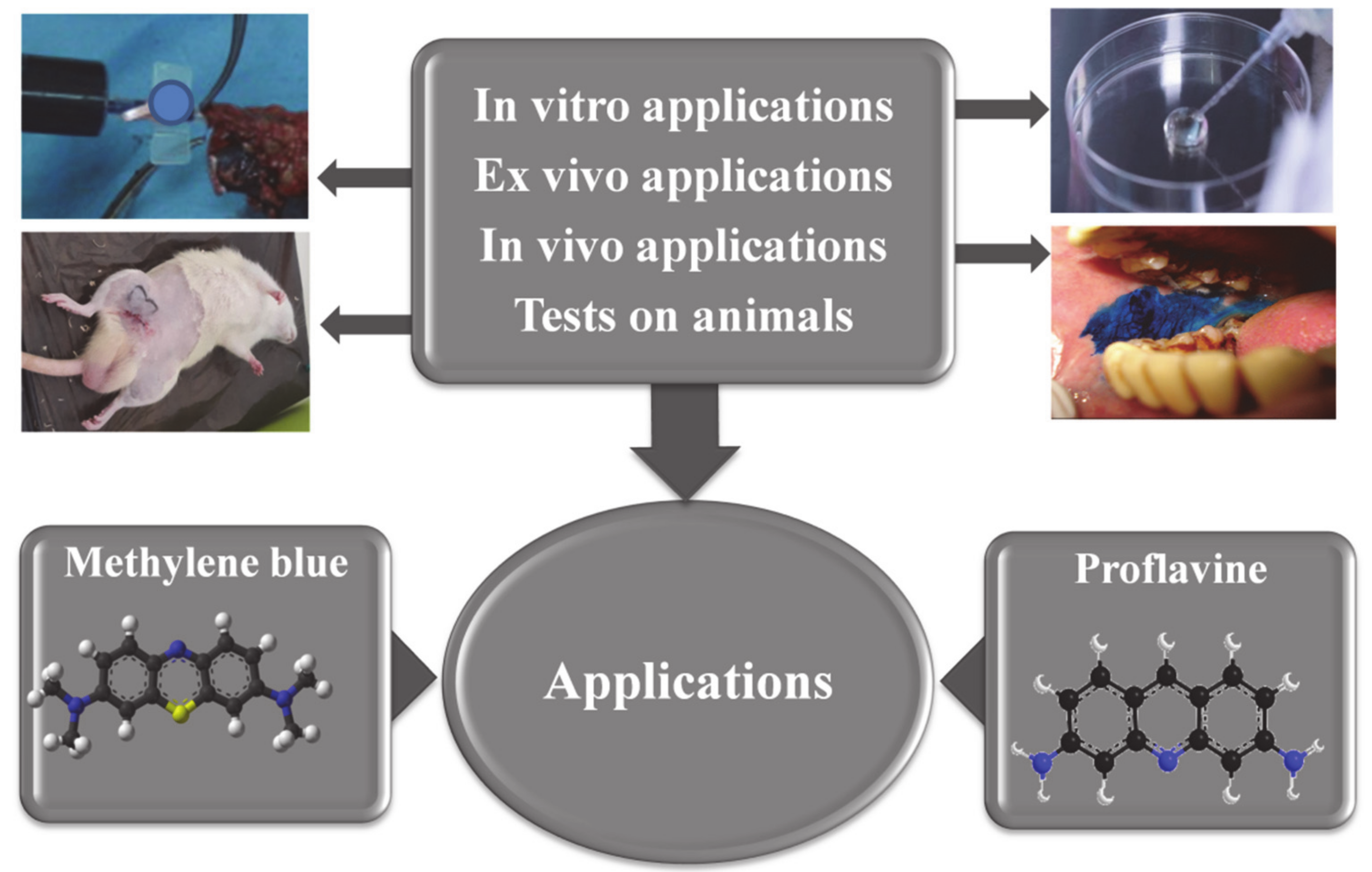
3. In Vitro Studies Using Methylene Blue and Proflavine
3.1. Methylene Blue Uses for In Vitro Studies
3.2. Proflavine Uses for In Vitro Studies
4. In Vivo Studies
4.1. The Use of Methylene Blue on Animals
- Anti-apoptosis as a result of the PI3-Akt activation and GSK3β inhibition in the peri-hematoma brain tissue;
- reduction of the brain tissue edema in ipsilateral basal ganglia;
- the increase of the myeloperoxidase (MPO) number and Iba1 positive after the first 24 h;
- reduction of the pro-inflammatory cytokines (e.g., interleukin (IL)-1β, tumor necrosis factor (TNF)-α, and IL-6) effect and upregulation of IL-10 cytokine that exerts anti-inflammatory and neuroprotective effects;
- reduction of the neutrophil infiltration and microglia activation.
4.2. The Use of Proflavine on Animals
4.3. Clinical Trials of Methylene Blue and Proflavine
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hossain, M.; Suresh Kumar, G. DNA intercalation of methylene blue and quinacrine: New insights into base and sequence specificity from structural and thermodynamic studies with polynucleotides. Mol. Biosyst. 2009, 5, 1311–1322. [Google Scholar] [CrossRef] [PubMed]
- Vardevanyan, P.O.; Antonyan, A.P.; Parsadanyan, M.A.; Shahinyan, M.A.; Mikaelyan, M.S. Study of Methylene Blue Interaction with Human Serum Albumin. Biophys. Rev. Lett. 2019, 14, 17–25. [Google Scholar] [CrossRef]
- Md Sani, N.D.; Ariffin, E.Y.; Sheryn, W.; Shamsuddin, M.A.; Heng, L.Y.; Latip, J.; Hasbullah, S.A.; Hassan, N.I. An Electrochemical DNA Biosensor for Carcinogenicity of Anticancer Compounds Based on Competition between Methylene Blue and Oligonucleotides. Sensors 2019, 19, 5111. [Google Scholar] [CrossRef] [PubMed]
- Tchen, S.; Sullivan, J.B. Clinical utility of midodrine and methylene blue as catecholamine-sparing agents in intensive care unit patients with shock. J. Crit. Care 2020, 57, 146–156. [Google Scholar] [CrossRef]
- Ginimuge, P.R.; Jyothi, S.D. Methylene blue: Revisited. J. Anaesthesiol. Clin. Pharmacol. 2010, 26, 517–520. [Google Scholar]
- Winer, J.H.; Choi, H.S.; Gibbs-Strauss, S.L.; Ashitate, Y.; Colson, Y.L.; Frangioni, J.V. Intraoperative localization of insulinoma and normal pancreas using invisible near-infrared fluorescent light. Ann. Surg. Oncol. 2010, 17, 1094–1100. [Google Scholar] [CrossRef]
- Gatasheh, M.K.; Kannan, S.; Hemalatha, K.; Imrana, N. Proflavine an acridine DNA intercalating agent and strong antimicrobial possessing potential properties of carcinogen. Karbala Int. J. Mod. Sci. 2017, 3, 272–278. [Google Scholar] [CrossRef]
- Louie, J.S.; Richards-Kortum, R.; Anandasabapathy, S. Applications and Advancements in the Use of High-resolution Microendoscopy for Detection of Gastrointestinal Neoplasia. Clin. Gastroenterol. Hepatol. 2014, 12, 1789–1792. [Google Scholar] [CrossRef]
- Pieto, S.P.; Lai, K.K.; Laryea, J.A.; Mizell, J.S.; Mustain, W.C.; Muldoon, T.J. Fluorescein as a topical fluorescent contrast agent for quantitative microendoscopic inspection of colorectal epithelium. Biomed. Opt. Express 2017, 8, 2727–2737. [Google Scholar] [CrossRef]
- Liu, J.-J.; Lu, L.; Hu, F.-Q.; Yuan, H.; Xu, Q.; Qin, Y.-F.; Gong, J.-H. Methylene blue attenuates renal ischemia- reperfusion injury by negative regulation of NLRP3 signaling pathway. Eur. Rev. Med. Phermacol. Sci. 2018, 22, 2847–2853. [Google Scholar] [CrossRef]
- Oz, M.; Lorke, D.E.; Hasan, M.; Petroianu, G.A. Cellular and Molecular Actions of Methylene Blue in the Nervous System. Med. Res. Rev. 2011, 31, 93–117. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Li, W.; Sumien, N.; Forster, M.; Simpkins, J.W.; Liu, R. Alternative mitochondrial electron transfer for the treatment of neurodegenerative diseases and cancers: Methylene blue connects the dots. Prog. Neurobiol. 2017, 157, 273–291. [Google Scholar] [CrossRef] [PubMed]
- Lozano, M.; Cid, J.; Müller, T.H. Plasma treated with methylene blue and light: Clinical efficacy and safety profile. Transfus. Med. Rev. 2013, 27, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Abbaci, M.; Breuskin, I.; Casiraghi, O.; De Leeuw, F.; Ferchiou, M.; Temam, S.; Laplace-Builhé, C. Confocal laser endomicroscopy for non-invasive head and neck cancer imaging: A comprehensive review. Oral Oncol. 2014, 50, 711–716. [Google Scholar] [CrossRef]
- Louie, J.S.; Shukla, R.; Richards-Kortum, R.; Anandasabapathy, S. High-resolution microendoscopy in differentiating neoplastic from non-neoplastic colorectal polyps. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 663–673. [Google Scholar] [CrossRef]
- Atamna, H.; Nguyen, A.; Schultz, C.; Boyle, K.; Newberry, J.; Kato, H.; Ames, B.N. Methylene blue delays cellular senescence and enhances key mitochondrial biochemical pathways. FASEB J. 2008, 22, 703–712. [Google Scholar] [CrossRef]
- Li, J.; Chen, X.; Qi, M.; Id, Y.L. Sentinel lymph node biopsy mapped with methylene blue dye alone in patients with breast cancer: A systematic review and meta- analysis. PLoS ONE 2018, 13, e0204364. [Google Scholar] [CrossRef]
- How, S.-C.; Cheng, Y.-H.; Lo, C.-H.; Lai, J.; Lin, T.-H.; Bednarikova, Z.; Antosova, A.; Gazova, Z.; Wu, J.W.; Wang, S.-S. Exploring the effects of methylene blue on amyloid fi brillogenesis of lysozyme. Int. J. Biol. Macromol. 2018, 119, 1059–1067. [Google Scholar] [CrossRef]
- Yeoh, C.; Tollinche, L. Use of methylene blue in hepatic arterial infusion pump resulting in serotonin syndrome. A case report. Anesthesiol. Case Rep. 2018, 1, 18–19. [Google Scholar]
- Lu, G.; Nagbanshi, M.; Goldau, N.; Mendes Jorge, M.; Meissner, P.; Jahn, A.; Mokenhaupt, F.P.; Muler, O. Efficacy and safety of methylene blue in the treatment of malaria: A systematic review. BMC Med. 2018, 16, 59. [Google Scholar] [CrossRef]
- Baird, K.J.; Maguire, J.D.; Price, R.N. Diagnosis and treatment of Plasmodium vivax malaria. Adv. Parasitol. 2012, 80, 203–270. [Google Scholar] [CrossRef] [PubMed]
- Zoungrana, A.; Coulibaly, B.; Sie, A.; Walter-Sack, I.; Mockenhaupt, F.P.; Kouyate, B.; Schirmer, R.H.; Klose, C.; Mansmann, U.; Meissner, P.; et al. Safety and efficacy of methylene blue combined with artesunate or amodiaquine for uncomplicated falciparum malaria: A randomized controlled trial from Burkina Faso. PLoS ONE 2008, 3, e1630. [Google Scholar] [CrossRef] [PubMed]
- Coulibaly, B.; Zoungrana, A.; Mockenhaupt, F.P.; Schirmer, R.H.; Klose, C.; Mansmann, U.; Meissner, P.E.; Muller, O. Strong gametocytocidal effect of methylene blue-based combination therapy against falciparum malaria: A randomised controlled trial. PLoS ONE. 2009, 4, e5318. [Google Scholar] [CrossRef] [PubMed]
- Krafts, K.; Hempelmann, E.; Skorska-Stania, A. From methylene blue to chloroquine: A brief review of the development of an antimalarial therapy. Parasitol. Res. 2012, 111, 1–6. [Google Scholar] [CrossRef]
- Mekontso-Dessap, A.; Houël, R.; Soustelle, C.; Kirsch, M.; Thébert, D.; Loisance, D.Y. Risk factors for post-cardiopulmonary bypass vasoplegia in patients with preserved left ventricular function. Ann. Thorac. Surg. 2001, 71, 1428–1432. [Google Scholar] [CrossRef]
- Kwok, E.S.H.; Howes, D. Use of methylene blue in sepsis: A systematic review. J. Intensive Care Med. 2006, 21, 359–363. [Google Scholar] [CrossRef]
- Schenk, P.; Madl, C.; Rezaie-Majd, S.; Lehr, S.; Muller, C. Methylene blue improves the hepatopulmonary syndrome. Ann. Intern. Med. 2000, 133, 701–706. [Google Scholar] [CrossRef]
- Meissner, P.E.; Mandi, G.; Coulibaly, B.; Witte, S.; Tapsoba, T.; Mansmann, U.; Rengelshausen, J.; Schiek, W.; Jahn, A.; Walter-Sack, I.; et al. Methylene blue for malaria in Africa: Results from a dose-finding study in combination with chloroquine. Malar. J. 2006, 5, 84. [Google Scholar] [CrossRef]
- Boylston, M.; Beer, D. Methemoglobinemia: A Case Study. Crit. Care Nurse 2002, 22, 50–55. [Google Scholar] [CrossRef]
- Pelgrims, J.; De Vos, F.; Van Den Brande, J.; Schrijvers, D.; Prové, A.; Vermorken, J.B. Methylene blue in the treatment and prevention of ifosfamide-induced encephalopathy: Report of 12 cases and a review of the literature. Br. J. Cancer 2000, 82, 291–294. [Google Scholar] [CrossRef]
- Wondrak, G.T. NQO1-activated phenothiazinium redox cyclers for the targeted bioreductive induction of cancer cell apoptosis. Free Radic. Biol. Med. 2007, 43, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Beretvas, R.I.; Ponsky, J. Endoscopic marking: An adjunct to laparoscopic gastrointestinal surgery. Surg. Endosc. 2001, 15, 1202–1203. [Google Scholar] [CrossRef] [PubMed]
- Simmons, R.M.; Smith, S.M.R.; Osborne, M.P. Methylene Blue Dye as an Alternative to Isosulfan Blue Dye for Sentinel Lymph Node Localization. Breast J. 2001, 7, 181–183. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.; Chowdhury, S.; Sanyal, D.; Chattopadhyay, K.; Kumar, G.S. Comparative Study of Toluidine Blue O and Methylene Blue Binding to Lysozyme and Their Inhibitory Effects on Protein Aggregation. ACS Omega 2018, 3, 2588–2601. [Google Scholar] [CrossRef]
- Pantano, N.; Hunt, B.; Schwarz, R.A.; Parra, S.; Cherry, K.; Possati-Resende, J.C.; Longatto-Filho, A.; Guerreiro Fregnani, J.H.T.; Castle, P.E.; Schmeler, K.; et al. Is Proflavine Exposure Associated with Disease Progression in Women with Cervical Dysplasia ? A Brief Report. Photochem. Photobiol. 2018, 94, 1308–1313. [Google Scholar] [CrossRef]
- Quang, T.; Tran, E.Q.; Schwarz, R.A.; William, M.D.; Gillenwater, A.M.; Richards-Kortum, R. Prospective Evaluation of Multimodal Optical Imaging with Automated Image Analysis to Detect Oral Neoplasia In Vivo. Cancer Prev. Res. 2017, 10, 563–570. [Google Scholar] [CrossRef]
- Vyas, K.; Hughes, M.; Leff, D.R.; Yang, G.-Z. Methylene-blue aided rapid confocal laser endomicroscopy of breast cancer. J. Biomed. Opt. 2017, 22, 020501. [Google Scholar] [CrossRef]
- Mondal, S.B.; Gao, S.; Zhu, N.; Liang, R.; Gruev, V.; Achilefu, S. Real-time fluorescence image-guided oncologic surgery. Adv. Cancer Res. 2014, 124, 171–211. [Google Scholar] [CrossRef]
- Muldoon, T.J.; Thekkek, N.; Roblyer, D. Evaluation of quantitative image analysis criteria for the high-resolution microendoscopic detection of neoplasia in Barrett’s esophagus. J. Biomed. Opt. 2010, 15, 026027. [Google Scholar] [CrossRef]
- Riaz, A.; Shreedhar, B.; Kamboj, M.; Natarajan, S. Methylene blue as an early diagnostic marker for oral precancer and cancer. Springerplus 2013, 2, 95. [Google Scholar] [CrossRef]
- Taghavi, S.A.; Membari, M.E.; Eshraghian, A.; Dehghani, S.M.; Hamidpour, L.; Khademalhoseini, F. Comparison of chromoendoscopy and conventional endoscopy in the detection of premalignant gastric lesions. Can. J. Gastroenterol. 2009, 23, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Dobbs, J.L.; Mueller, J.L.; Krishnamurthy, S.; Shin, D.; Kuerer, H.; Yang, W.; Ramanujam, N.; Richards-Kortum, R. Micro-anatomical quantitative optical imaging: Toward automated assessment of breast tissues. Breast Cancer Res. 2015, 17, 105. [Google Scholar] [CrossRef] [PubMed]
- Dobbs, J.L.; Shin, D.; Krishnamurthy, S.; Kuerer, H.; Yang, W.; Richards-Kortum, R. Confocal fluorescence microscopy to evaluate changes in adipocytes in the tumor microenvironment associated with invasive ductal carcinoma and ductal carcinoma in situ. Int. J. Cancer 2016, 139, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Lang, I.J.; Fisch, M.; Kluth, L.A. Diagnostic and therapeutic concepts for vesicovaginal and ureterovaginal fistulas. Aktuelle Urol. 2018, 49, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Fei, B.; Lu, G.; Wang, X.; Zhang, H.; Little, J.V.; Magliocca, K.R.; Chene, A.Y. Tumor margin assessment of surgical tissue specimen of cancer patients using label-free hyperspectral imaging. Proc. SPIE Int. Soc. Opt. Eng. 2017, 10054, 100540E. [Google Scholar] [PubMed]
- Rosenthal, E.L.; Moore, L.S.; Tipirneni, K.; de Boer, E.; Stevens, T.M.; Hartman, Y.E.; Carroll, W.R.; Zinn, K.R.; Warram, J.M. Sensitivity and Specificity of Cetuximab-IRDye800CW to Identify Regional Metastatic Disease in Head and Neck Cancer. Clin. Cancer Res. 2018, 23, 4744–4752. [Google Scholar] [CrossRef]
- Warram, J.M.; de Boer, E.; Moore, L.S.; Schmalbach, C.E.; Withrow, K.P.; Carroll, W.R.; Richman, J.S.; Morlandt, A.B.; Brandwein-Gensler, M.; Rosenthal, E.L. A Ratiometric Threshold for Determining Presence of Cancer During Fluorescence-guided Surgery. J. Surg. Oncol. 2015, 112, 2–8. [Google Scholar] [CrossRef]
- Schirmer, R.H.; Adler, H.; Pickhardt, M.; Mandelkow, E. Lest we forget you--methylene blue. Neurobiol. Aging 2011, 32, 7–16. [Google Scholar] [CrossRef]
- Salaris, S.C.; Babbs, C.F.; Voorhees, W.D. Methylene blue as an inhibitor generation by xanthine of superoxide oxidase. Biochem. Pharmacol. 1991, 42, 499–506. [Google Scholar] [CrossRef]
- Furian, A.F.; Fighera, M.R.; Oliveira, M.S.; Ferreira, A.P.; Fiorenza, N.G.; de Carvalho Myskiw, J.; Petry, J.C.; Coelho, R.C.; Mello, C.F.; Royes, L.F. Methylene blue prevents methylmalonate-induced seizures and oxidative damage in rat striatum. Neurochem. Int. 2007, 50, 164–171. [Google Scholar] [CrossRef]
- Shah, D.I.; Santani, D.D.; Goswami, S.S. A novel use of methylene blue as a pharmacological tool. J. Pharmacol. Toxicol. Methods 2006, 54, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Stockand, J.D.; Sansom, S.C. Activation by methylene blue of large Ca(2+)-activated K+ channels. Biochim. Biophys. Acta. 1996, 1285, 123–126. [Google Scholar] [CrossRef]
- Tucker, D.; Lu, Y.; Zhang, Q. From Mitochondrial Function to Neuroprotection—An Emerging Role for Methylene Blue. Mol. Neurobiol. 2018, 55, 5137–5153. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, B.; Lin, B.; Ye, Y.; Bao, C.; Zhao, X.; Jion, L.; Xiong, X. Methylene Blue Attenuates Lung Injury Induced by Hindlimb Ischemia Reperfusion in Rats. Mediat. Inflamm. 2018, 2018, 2508620. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; St Denis, T.G.; Huang, Y.Y.; Tanaka, M.; Zadlo, A.; Sarna, T.; Hamblin, M.R. Paradoxical potentiation of methylene blue-mediated antimicrobial photodynamic inactivation by sodium azide: Role of ambient oxygen and azide radicals. Free Radic. Biol. Med. 2012, 53, 2062–2071. [Google Scholar] [CrossRef] [PubMed]
- Trikha, A.; Mohan, V.; Kashyap, L.; Saxena, A. Pulmonary edema following intrauterine methylene blue injection. Acta Anaesthesiol. Scand. 1996, 40, 382–384. [Google Scholar] [CrossRef]
- Kast, R.E. Inhibiting the NLRP3 Inflammasome With Methylene Blue as Treatment Adjunct in Myelodysplasia. Front. Oncol. 2018, 8, 8–11. [Google Scholar] [CrossRef]
- Basu, A.; Kumar, G.S. A biophysical investigation on the binding of proflavine with human hemoglobin: Insights from spectroscopy, thermodynamics and AFM studies. J. Photochem. Photobiol. B Biol. 2016, 165, 42–50. [Google Scholar] [CrossRef]
- Aidas, K.; Olsen, J.M.; Kongsted, J.; Ågren, H. Photoabsorption of Acridine Yellow and Proflavin Bound to Human Serum Albumin Studied by Means of Quantum Mechanics /Molecular Dynamics. J. Phys. Chem. B 2013, 117, 2069–2080. [Google Scholar] [CrossRef]
- Kawada, H.; Inanobe, A.; Kurachi, Y. Isolation of proflavine as a blocker of G protein-gated inward rectifier potassium channels by a cell growth-based screening system. Neuropharmacology 2016, 109, 18–28. [Google Scholar] [CrossRef]
- Sasikala, W.D.; Mukherjee, A. Structure and dynamics of proflavine association around DNA. Phys. Chem. Chem. Phys. 2016, 18, 10383–10391. [Google Scholar] [CrossRef] [PubMed]
- Pépin, G.; Nejad, C.; Thomas, B.J.; Ferrand, J.; McArthur, K.; Bardin, P.G.; Williams, B.R.; Gantier, M.P. Activation of cGAS-dependent antiviral responses by DNA intercalating agents. Nucleic Acids Res. 2017, 45, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Delport, A.; Harvey, B.H.; Petzer, A.; Petzer, J.P. Methylene blue and its analogues as antidepressant compounds. Metab. Brain Dis. 2017, 32, 1357–1382. [Google Scholar] [CrossRef] [PubMed]
- Sasikala, W.D.; Mukherjee, A. Molecular mechanism of direct proflavine-DNA intercalation: Evidence for drug-induced minimum base-stacking penalty pathway. J. Phys. Chem. B 2012, 116, 12208–12212. [Google Scholar] [CrossRef] [PubMed]
- Harrison, D.E.; Strong, R.; Allison, D.B.; Ames, B.N.; Astle, C.M.; Atamna, H.; Fernandez, E.; Flurkey, K.; Javors, M.A.; Nadon, N.L.; et al. Acarbose, 17-α-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging Cell 2014, 13, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Ateş, G.B.; Ak, A.; Garipcan, B.; Gülsoy, M. Methylene blue mediated photobiomodulation on human osteoblast cells. Lasers Med. Sci. 2017, 32, 1847–1855. [Google Scholar] [CrossRef]
- Duran, H.; Üstün, F.; Ba, M.; Karaku, S.; Akta, A.; Tuba, Ş. Investigation of the in vitro cytotoxic effects and wound healing activity of ternary composite substance (hollow silica sphere /gum arabic /methylene blue). Int. J. Biol. Macromol. 2019, 121, 1194–1202. [Google Scholar] [CrossRef]
- Junqueira Garcia, T.M.; Gonçalves, T.P.; São Felix Martins, E.; Martins, T.S.; de Carvalho Abreu Fantini, M.; Regazi Minarini, P.R.; Costa Fernandez, S.; Cassone Salata, G.; Biagini Lopes, L. Improvement of cutaneous delivery of methylene blue by liquid crystals. Int. J. Pharm. 2018, 548, 454–465. [Google Scholar] [CrossRef]
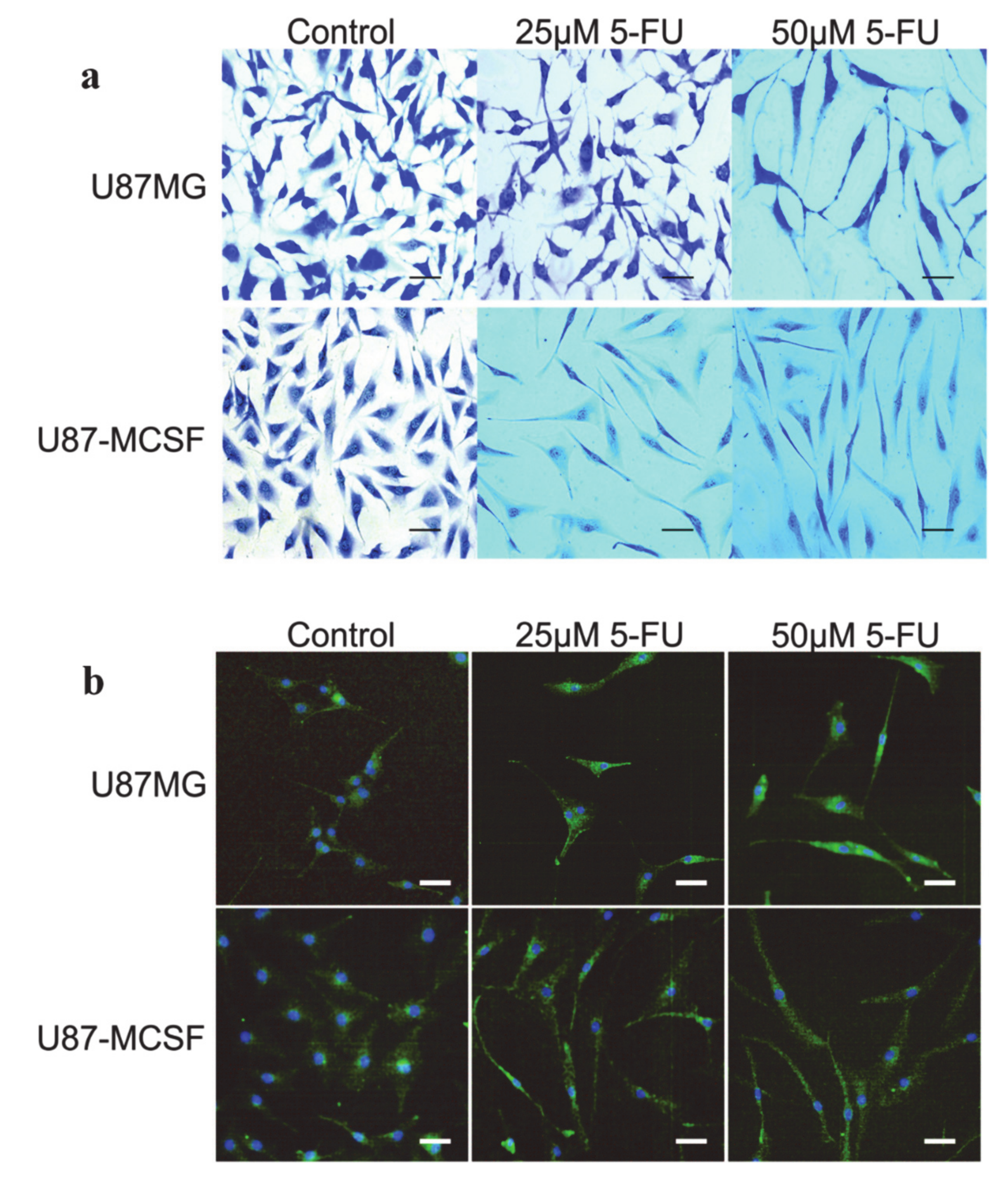
- Chockalingam, S.; Ghosh, S.S. Amelioration of Cancer Stem Cells in Macrophage Colony Stimulating Factor-Expressing U87MG-Human Glioblastoma upon 5-Fluorouracil Therapy. PLoS ONE 2013, 8, 1–9. [Google Scholar] [CrossRef]
- Tian, W.-F.; Zeng, S.; Sheng, Q.; Chen, J.-L.; Weng, P.; Zheng, X.-T.; Yuan, J.-J.; Pang, Q.-F.; Wang, Z.-Q. Methylene Blue Protects the Isolated Rat Lungs from Ischemia—Reperfusion Injury by Attenuating Mitochondrial Oxidative Damage. Lung 2018, 196, 73–82. [Google Scholar] [CrossRef]
- Atamna, H.; Kumar, R. Protective role of methylene blue in Alzheimer’s disease via mitochondria and cytochrome c oxidase. J. Alzheimers Dis. 2010, 20 (Suppl. 2), S439–S552. [Google Scholar] [CrossRef]
- Bean, S.F.; Felber, T.D.; Knox, J.M. Proflavine Staining of the Skin Demonstrated by Fluorescent Microscopy. Arch. Dermatol. 1973, 107, 204–205. [Google Scholar] [CrossRef]
- Prieto, S.P.; Powless, A.J.; Boice, J.W.; Sharma, S.G. Proflavine Hemisulfate as a Fluorescent Contrast Agent for Point-of-Care Cytology. PLoS ONE 2015, 10, e0125598. [Google Scholar] [CrossRef] [PubMed]
- Legault, J.; Larouche, P.L.; Côté, I.; Bouchard, L.; Pichette, A.; Robinson, B.H.; Morin, C. Low-concentration methylene blue maintains energy production and strongly improves survival of Leigh syndrome French Canadian skin fibroblasts. J. Pharm. Pharm. Sci. 2011, 14, 438–449. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bristol, D.W. (Ed.) NTP Technical Report on the Toxicology and Carcinogenesis Studies of Methylene Blue Trihydrate in F344/N Rats and B6C3F 1 Mice; Diane Publishing: Springfield, VA, USA, 2018; p. no. 7220, National Toxicology Program. [Google Scholar]
- Chen, C.; Hu, Z. Methylene blue offers neuroprotection after intracerebral hemorrhage in rats through the PI3K/Akt/GSK3 β signaling pathway. J. Cell Physiol. 2019, 234, 5304–5318. [Google Scholar] [CrossRef] [PubMed]
- Wiklund, L.; Patnaik, R.; Sharma, A.; Miclescu, A.; Sharma, H.S. Cerebral Tissue Oxidative Ischemia-Reperfusion Injury in Connection with Experimental Cardiac Arrest and Cardiopulmonary Resuscitation: Effect of Mild Hypothermia and Methylene Blue. Mol. Neurobiol. 2018, 55, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Sanchala, D.; Bhatt, L.K.; Pethe, P.; Shelat, R.; Kulkarni, Y.A. Anticancer activity of methylene blue via inhibition of heat shock protein 70. Biomed. Pharmacother. 2018, 107, 1037–1045. [Google Scholar] [CrossRef]
- Volpon, L.C.; Barbosa Evora, P.R.; Teixeira, G.D.; Godinho, M.; Scarpelini, S.; Carmona, F.; de Carvalho Panzeri Carlotti, A.P. Methylene blue for refractoryshock in polytraumatized patient: A case report. J. Emerg. Med. 2018, 55, 553–558. [Google Scholar] [CrossRef]
- Hillary, S.L.; Guillermet, S.; Brown, N.J.; Balasubramanian, S.P. Use of methylene blue and near-infrared fluorescence in thyroid and parathyroid surgery. Langenbecks Arch. Surg. 2018, 403, 111–118. [Google Scholar] [CrossRef]
- Park, J.; Mroz, P.; Hamblin, M.R.; Yaroslavsky, A.N. Dye-enhanced multimodal confocal microscopy for noninvasive detection of skin cancers in mouse models. J. Biomed. Opt. 2010, 15, 026023. [Google Scholar] [CrossRef]
- Tanaka, E.; Chen, F.Y.; Flaumenhaft, R.; Graham, G.J.; Laurence, R.G.; Frangioni, J.V. Real-time assessment of cardiac perfusion, coronary angiography, and acute intravascular thrombi using dual-channel near-infrared fluorescence imaging. J. Thorac. Cardiovasc. Surg. 2009, 138, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Ashitate, Y.; Lee, B.T.; Laurence, R.G.; Lunsford, E.; Hutteman, M.; Oketokou, R.; Choi, H.S.; Frangioni, J.V. Intraoperative Prediction of Post-Operative Flap Outcome using the Near-Infrared Fluorophore Methylene Blue. Ann. Plast. Surg. 2013, 70, 360–365. [Google Scholar] [PubMed]
- Zheng, H.; Qiu, L.; Liu, Z.; Su, Y.; Pan, X.; Liu, S.; Yi, C. Exploring the Possibility of a Retrograde Embolism Pathway from the Facial Artery to the Ophthalmic Artery System In Vivo. Aesthet. Plast. Surg. 2017, 41, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Visconti, G.; Brunelli, C.; Mulè, A.; Franceschini, G.; Chen, H.C.; Masetti, R.; Salgarello, M. Septum-based cervical lymph-node free flap in rat: A new model. J. Surg. Res. 2016, 201, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rogers, R.A.; Riviere, J.E. Pharmacologic modulation of the cutaneous vasculature in the isolated perfused porcine skin flap. J. Pharm. Sci. 1994, 83, 1682–1689. [Google Scholar] [CrossRef] [PubMed]
- Magee, N.S.; Wagner, R.F. Surgical Pearl: The use of methylene blue temporary tattoos for tissue orientation in Mohs micrographic surgery. J. Am. Acad. Dermatol. 2004, 50, 640–641. [Google Scholar] [CrossRef]
- Rosique, M.J.; Rosique, R.G.; Faria, F.M.; Oliveira, C.C.; Farina, J.A.; Évora, P.R.B. Methylene blue reduces progression of burn and increases skin survival in an experimental rat model. Burns 2017, 43, 1702–1708. [Google Scholar] [CrossRef]
- Yun, K. Assessing Urinary Tract Junction Obstruction Defects by Methylene Blue Dye Injection. J. Vis. Exp. 2017, e56247. [Google Scholar] [CrossRef]
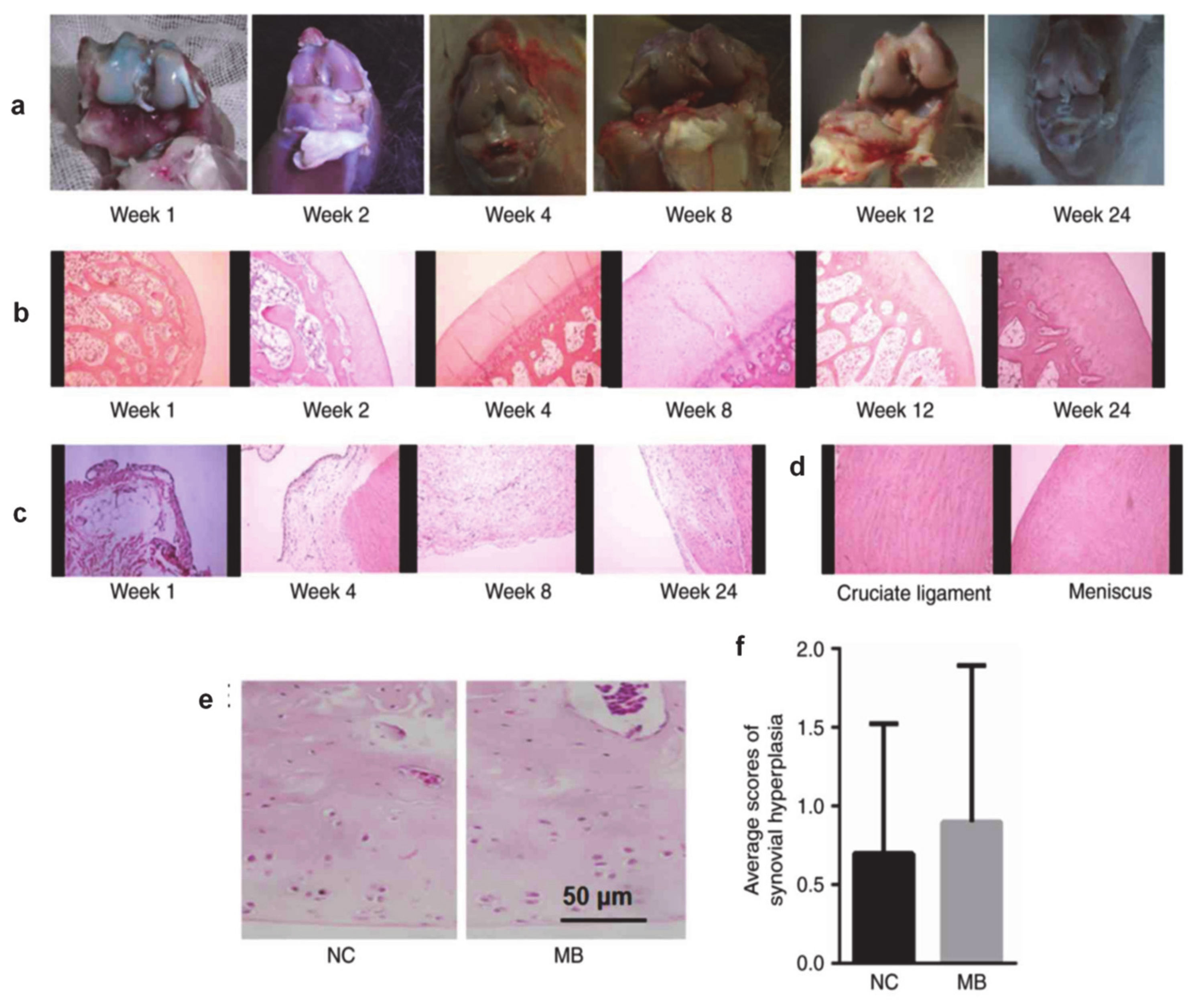
- Li, X.; Tang, C.; Wang, J.; Guo, P.; Wang, C.; Wang, Y.; Zhang, Z.; Wu, H. Methylene blue relieves the development of osteoarthritis by upregulating lncRNA MEG3. Exp. Ther. Med. 2018, 15, 3856–3864. [Google Scholar] [CrossRef]
- Belhassen, S.; Alzahrani, M.; Nader, M.-E.; Gaboury, L.; Saliba, I. Study of Methylene Blue Ototoxicity in the Guinea Pig. J. Clin. Med. Res. 2017, 9, 900–906. [Google Scholar] [CrossRef][Green Version]
- Wirth, D.J.; Snuderl, M.; Curry, W.; Yaroslavsky, A. Comparative evaluation of methylene blue and demeclocycline for enhancing optical contrast of gliomas in optical images. J. Biomed. Opt. 2014, 19, 090504. [Google Scholar] [CrossRef] [PubMed]
- Baran, T.M.; Giesselman, B.R.; Hu, R.; Biel, M.A.; Foster, T.H. Factors Influencing Tumor Response to Photodynamic Therapy Sensitized by Intratumor Administration of Methylene Blue. Lasers Surg. Med. 2010, 42, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.S.; Shin, D.H.; Lee, K.M.; Song, S.; Yoo, H.S.; Moon, D.C.; Hong, J.T.; Chung, Y.B. Effects of guanosine on the pharmacokinetics of acriflavine in rats following the administration of a 1:1 mixture of acriflavine and guanosine, a potential antitumor agent. Arch. Pharm. Res. 2007, 30, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Kwon, O.S.; Chung, Y.B. Pharmacokinetics and metabolism of acriflavine in rats following intravenous or intramuscular administration of AG60, a mixture of acriflavine and guanosine, a potential antitumour agent. Xenobiotica 2005, 35, 755–773. [Google Scholar] [CrossRef]
- Yaroslavsky, A.N.; Feng, X.; Neel, V.A. Optical Mapping of Nonmelanoma Skin Cancers—A Pilot Clinical Study. Lasers Surg. Med. 2017, 49, 803–809. [Google Scholar] [CrossRef]

| Application | Mechanism/Explanations | Dose | Ref. |
|---|---|---|---|
| Preoperative and intraoperative use in cardiac surgery | MB has been used intravenously (i.v.) for over 30 min in patient’s in the intensive care unit (ICU) 1 h before surgery and a decreased incidence and severity of vasoplegic syndrome in high-risk patients was found. | MB solution (1%) | [25] |
| Septic shock | MB is an inhibitor of guanylate cyclase, very effective in improving the arterial pressure and cardiac function in septic shock. | Not reported | [26] |
| Hepatopulmonary syndrome | MB is found to increase arterial O2 pressure and to decrease alveolar-arterial difference for partial pressure of oxygen in all patients with hepatopulmonary syndrome. MB is a potent inhibitor of guanylate cyclase. | Not reported | [27] |
| Antimalarial | MB is an effective and cheap antimalarial agent especially in countries with increasing resistance of Plasmodium falciparum to existing first line antimalarial agents. | Dose of 36–72 mg/kg MB over 3 days is the most effective scheme of treatment | [20,21,22,23,24,28] |
| Methemoglobinemia | MB acts by reacting within red blood cells (RBCs) to form leucomethylene blue, which is a reducing agent of oxidized hemoglobin converting the ferric ion (Fe3+) back to its oxygen carrying ferrous state (Fe2+). | Dose of 1–2 mg/kg of 1% MB solution | [29] |
| Ifosfamide neurotoxicity | MB acts as an alternative electron acceptor and reverses the nicotinamide adenine dinucleotide NADH inhibition of hepatic gluconeogenesis while also inhibiting the transformation of chloroethylamine into chloroacetaldehyde. It also inhibits multiple amine oxidase activities, preventing the formation of chloroacetaldehyde. | Not reported | [30] |
| In cancer | MB and other redox cyclers induce selective cancer cell apoptosis by nicotinamide adenine dinucleotide phosphate NAD(P)H: quinine oxidoreductase (NQO1)-dependent bioreductive generation of cellular oxidative stress. MB is also investigated for the photodynamic treatment of cancer. | Not reported | [31] |
| As dye and stain | MB is used as a safe and effective method of localizing abnormal parathyroid glands, for intraoperative endoscopic marking of intestinal lumen and for location of different lesions. | Concentration from 0.05 μM to 1 M of MB solutions | [32,33] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nedu, M.-E.; Tertis, M.; Cristea, C.; Georgescu, A.V. Comparative Study Regarding the Properties of Methylene Blue and Proflavine and Their Optimal Concentrations for In Vitro and In Vivo Applications. Diagnostics 2020, 10, 223. https://doi.org/10.3390/diagnostics10040223
Nedu M-E, Tertis M, Cristea C, Georgescu AV. Comparative Study Regarding the Properties of Methylene Blue and Proflavine and Their Optimal Concentrations for In Vitro and In Vivo Applications. Diagnostics. 2020; 10(4):223. https://doi.org/10.3390/diagnostics10040223
Chicago/Turabian StyleNedu, Maria-Eliza, Mihaela Tertis, Cecilia Cristea, and Alexandru Valentin Georgescu. 2020. "Comparative Study Regarding the Properties of Methylene Blue and Proflavine and Their Optimal Concentrations for In Vitro and In Vivo Applications" Diagnostics 10, no. 4: 223. https://doi.org/10.3390/diagnostics10040223
APA StyleNedu, M.-E., Tertis, M., Cristea, C., & Georgescu, A. V. (2020). Comparative Study Regarding the Properties of Methylene Blue and Proflavine and Their Optimal Concentrations for In Vitro and In Vivo Applications. Diagnostics, 10(4), 223. https://doi.org/10.3390/diagnostics10040223








