New Approaches in the Study of the Pathogenesis of Urethral Pain Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
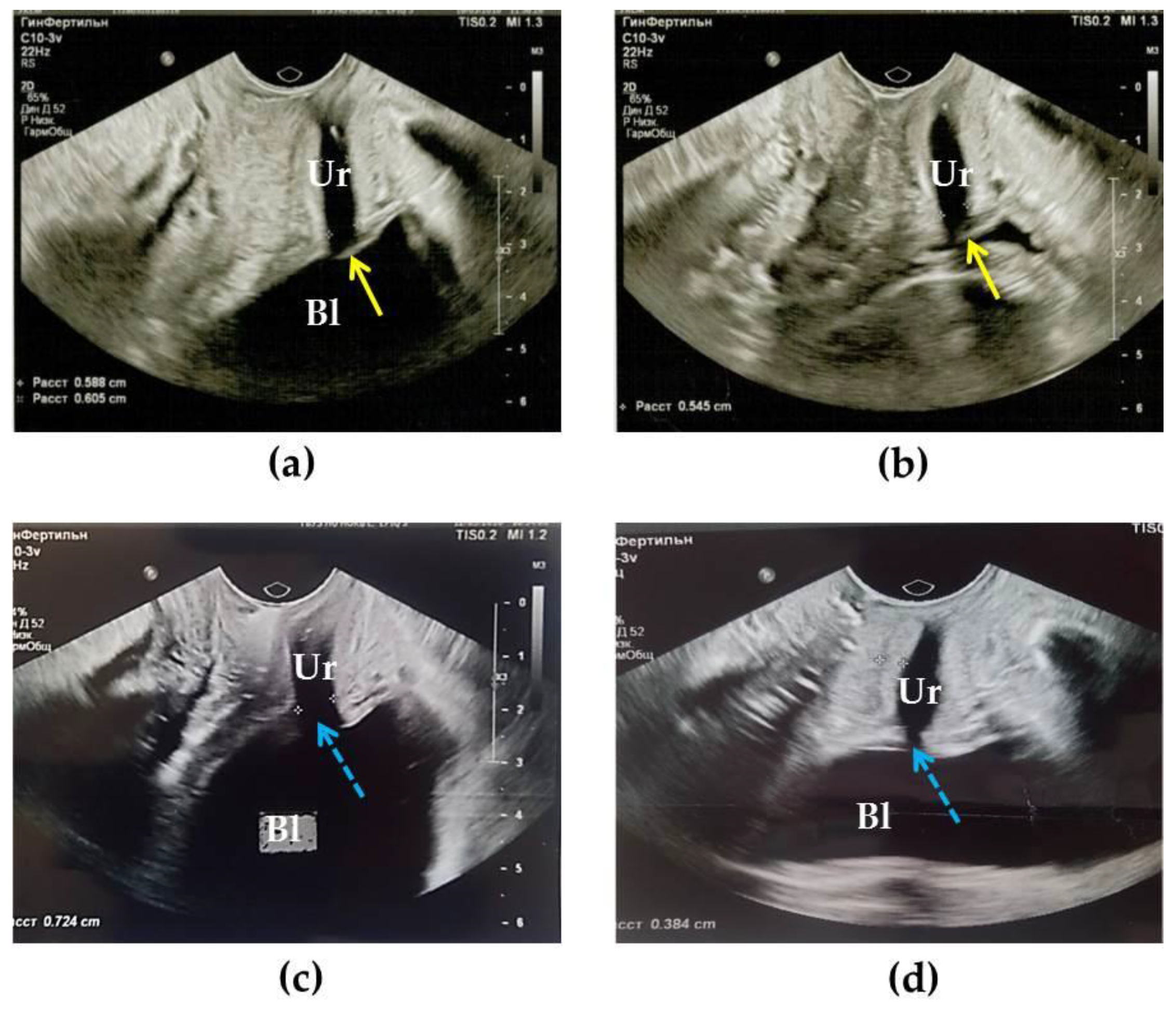
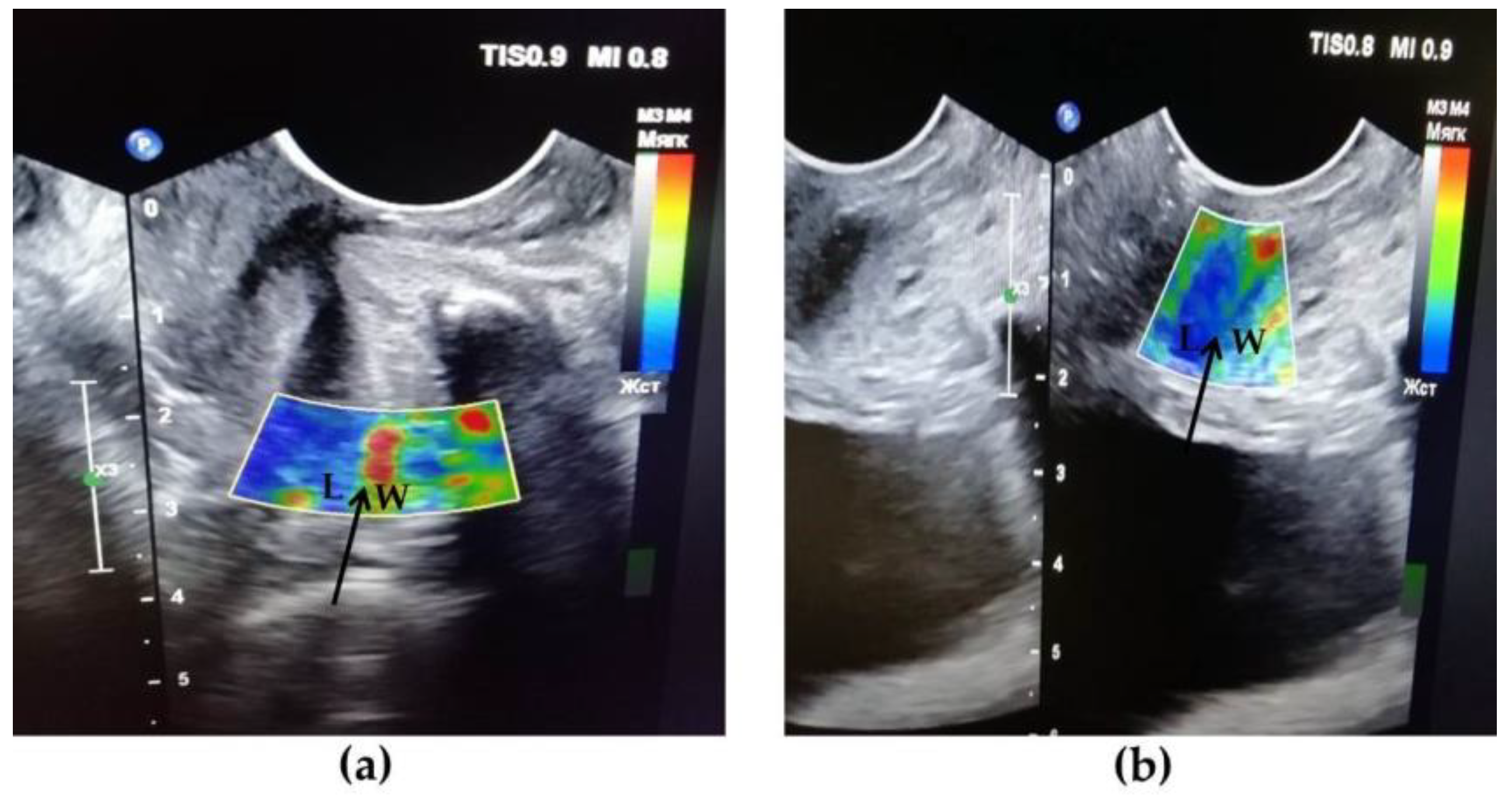
2.2. Transvaginal US
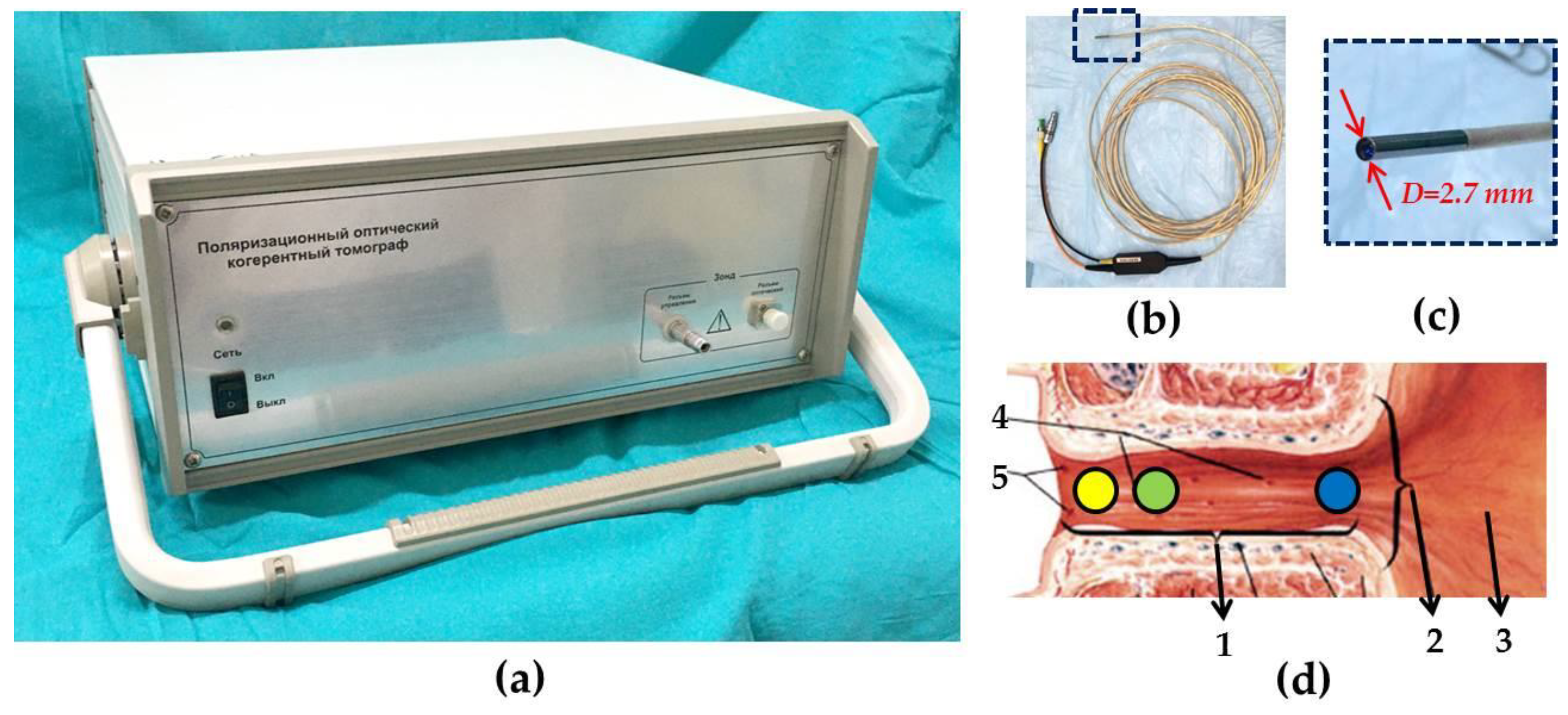
2.3. CP OCT Study and Image Analysis
2.4. Statistical Analysis
3. Results
3.1. The Role of Background Diseases in the Development of UPS
3.2. Results of Cystoscopic Examination
3.3. Uroflowmetry Results
3.4. Results of TVUS Research
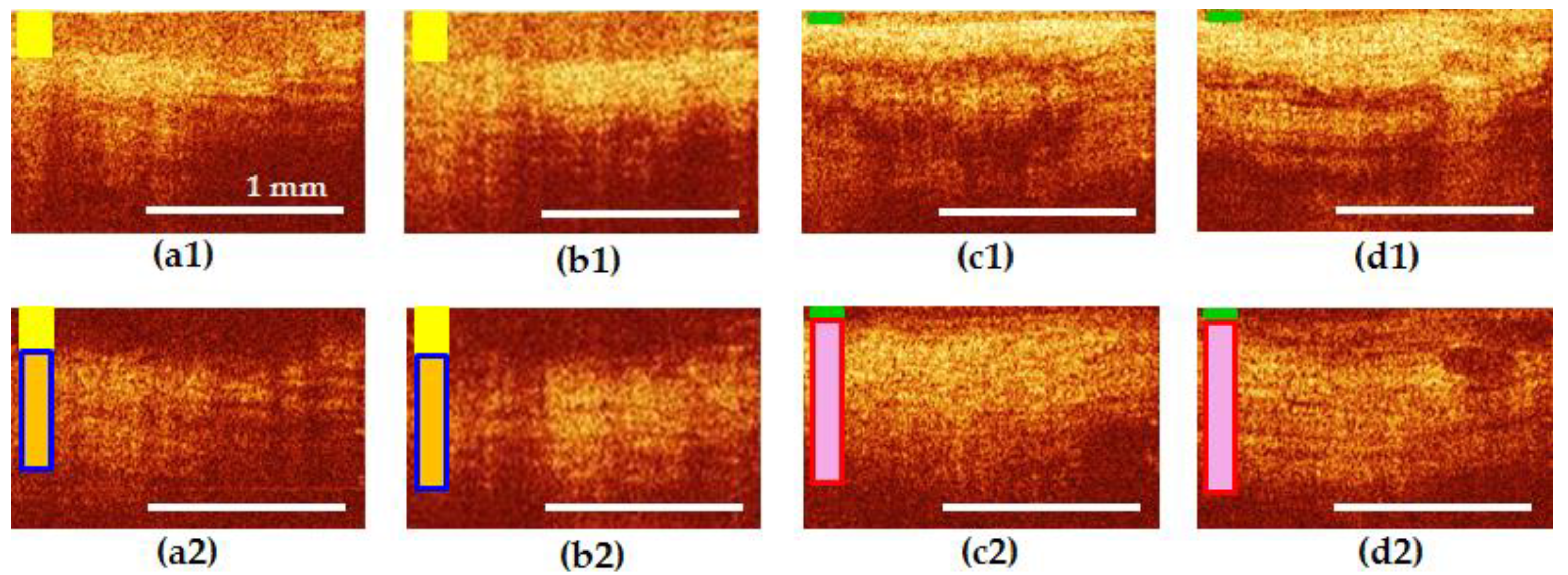
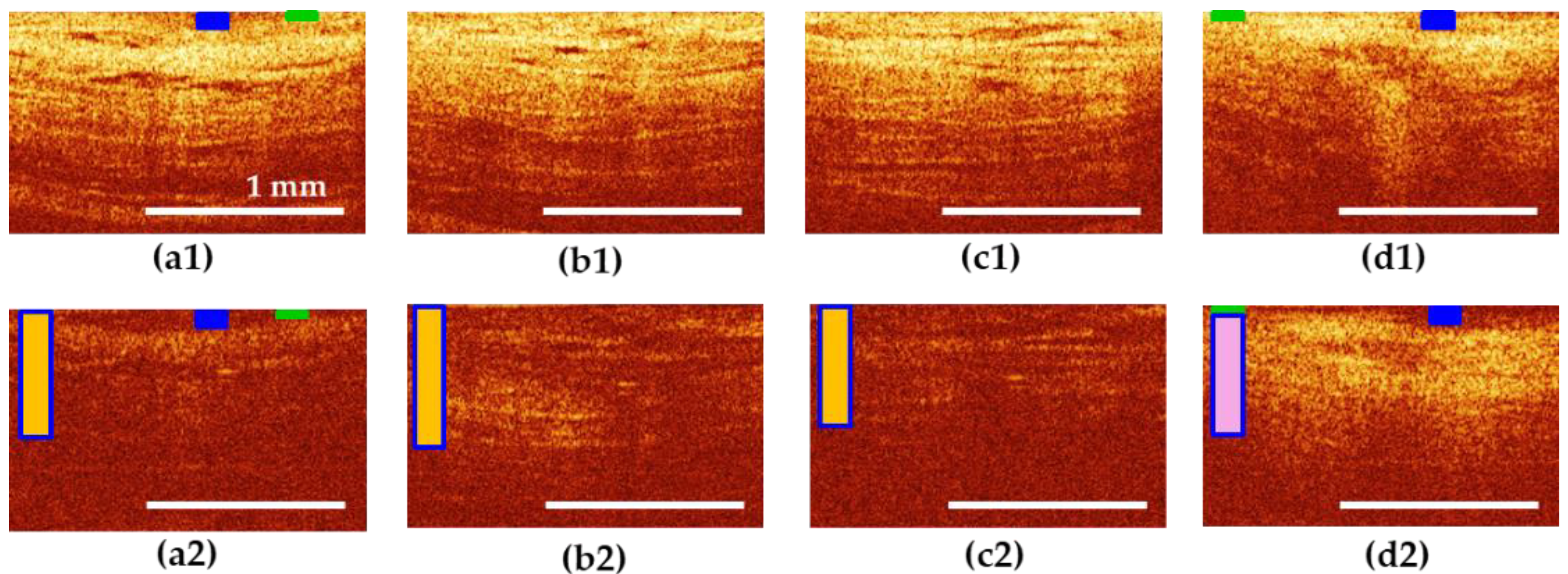
3.5. Results of CP OCT Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rothberg, M.B.; Wong, J.B. All dysuria is local a cost-effectiveness model for designing site-specific management algorithms. J. Gen. Intern. Med. 2004, 19, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.T. Urethral pain syndrome really part of bladder pain syndrome? Urogenit. Tract Infect. 2017, 12, 22–27. [Google Scholar] [CrossRef]
- Phillip, H.; Okewole, I.; Chilaka, V. Enigma of urethral pain syndrome: Why are there so many ascribed etiologies and therapeutic approaches? Int. J. Urol. 2014, 21, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Piontek, K.; Ketels, G.; Albrecht, R.; Schnurr, U.; Dybowski, C.; Brünahl, C.A.; Riegel, B.; Löwe, B. Somatic and psychosocial determinants of symptom severity and quality of life in male and female patients with chronic pelvic pain syndrome. J. Psychosom. Res. 2019, 120, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Passavanti, M.B.; Pota, V.; Sansone, P.; Aurilio, C.; De Nardis, L.; Pace, M.C. Chronic pelvic pain: Assessment, evaluation, and objectivation. Pain Res. Treat. 2017, 2017, 9472925. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.; Arunkalaivanan, A.S. Urethral pain syndrome and its management. Obstet. Gynecol. Surv. 2007, 62, 348–351. [Google Scholar] [CrossRef]
- Elsenbruch, S.; Häuser, W.; Jänig, W. Visceral pain. Schmerz 2015, 29, 496–502. [Google Scholar]
- Fall, M.; Baranowski, A.P.; Elneil, S.; Engeler, D.; Hughes, J.; Messelink, E.J.; Williams, A.D.C. EAU guidelines on chronic pelvic pain syndrome. Europ. Urol. 2010, 57, 35–48. [Google Scholar] [CrossRef]
- Dreger, N.; Degener, S.; Roth, S.; Brandt, A.S.; Lazica, D. Urethral pain syndrome: Fact or fiction—An update. Urol. A 2015, 54, 1248–1255. [Google Scholar] [CrossRef]
- Zaitsev, A.V.; Sharov, M.N.; Pushkar, D.Y.; Khodyreva, L.A.; Dudareva, A.A. Chronic Pelvic Pain Methodical Recommendations № 20; LLC Publishing House ABV-Press: Moscow, Russia, 2006; p. 46. [Google Scholar]
- Flor, H.; Fydrich, T.; Turk, D.C. Efficacy of multidisciplinary pain treatment centers: A meta-analytic review. Pain 1992, 49, 221–230. [Google Scholar] [CrossRef]
- Jarrell, J.F.; Vilos, G.A.; Allaire, C.; Burgess, S.; Fortin, C.; Gerwin, R.; Lapensée, L.; Lea, R.H.; Leyland, N.A.; Martyn, P.; et al. Chronic pelvic pain working group; society of obstetricians and gynaecologists of canada. consensus guidelines for the management of chronic pelvic pain. J. Obstet. Gynaecol. Can. 2005, 27, 869–910. [Google Scholar] [PubMed]
- Grinberg, K.; Sela, Y.; Nissanholtz-Gannot, R. New insights about chronic pelvic pain syndrome (CPPS). Int. J. Environ. Res. Public Health 2020, 17, 3005. [Google Scholar] [CrossRef] [PubMed]
- Persu, C.; Cauni, V.; Gutue, S.; Blaj, I.; Jinga, V.; Geavlete, P. From interstitial cystitis to chronic pelvic pain. J. Med. Life 2010, 3, 167–174. [Google Scholar]
- Aziz, Q.; Giamberardino, M.A.; Barke, A.; Korwisi, B.; Baranowski, A.P.; Wesselmann, U.; Treede, R.D. The IASP Classification of Chronic Pain for ICD-11: Chronic secondary visceral pain. Pain 2019, 160, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Stecco, C. Functional Atlas of the Human Fascial System, 1st ed.; Elsevier Health Sciences: London, UK, 2014; p. 384. [Google Scholar]
- Muiznieks, L.D.; Keeley, F.W. Molecular assembly and mechanical properties of the extracellular matrix: A fibrous protein perspective. Biochim. Biophys. Acta. 2013, 1832, 866–875. [Google Scholar] [CrossRef]
- Langevin, H.M. Connective tissue: A body-wide signaling network? Med. Hypotheses 2006, 66, 1074–1077. [Google Scholar] [CrossRef]
- Shoskes, D.A.; Nickel, J.C.; Kattan, M.W. Phenotypically directed multimodal therapy for chronic prostatitis/chronic pelvic pain syndrome: A prospective study using UPOINT. Urology 2010, 75, 1249–1253. [Google Scholar] [CrossRef]
- Wells, P.N.; Liang, H.D. Medical ultrasound: Imaging of soft tissue strain and elasticity. J. R. Soc. Interface 2011, 8, 1521–1549. [Google Scholar] [CrossRef]
- Bamber, J.; Cosgrove, D.; Dietrich, C.F.; Fromageau, J.; Bojunga, J.; Calliada, F.; Cantisani, V.; Correas, J.M.; D’Onofrio, M.; Drakonaki, E.E.; et al. EFSUMB guidelines and recommendations on the clinical use of ultrasound elastography. Part 1: Basic principles and technology. Ultraschall Med. 2013, 34, 169–184. [Google Scholar] [CrossRef]
- Cosgrove, D.; Piscaglia, F.; Bamber, J.; Bojunga, J.; Correas, J.M.; Gilja, O.H.; Klauser, A.S.; Sporea, I.; Calliada, F.; Cantisani, V.; et al. EFSUMB guidelines and recommendations on the clinical use of ultrasound elastography. Part 2: Clinical applications. Ultraschall Med. 2013, 34, 238–253. [Google Scholar]
- Lupsor-Platon, M. Ultrasound Elastography; IntechOpen: London, UK, 2020; p. 146. [Google Scholar]
- Sigrist, R.; Liau, J.; Kaffas, A.E.; Chammas, M.C.; Willmann, J.K. Ultrasound elastography: Review of techniques and clinical applications. Theranostics 2017, 7, 1303–1329. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Swanson, E.A.; Lin, C.P.; Schuman, J.S.; Stinson, W.G.; Chang, W.; Hee, M.R.; Flotte, T.; Gregory, K.; Puliafito, C.A.; et al. Optical coherence tomography. Science 1991, 254, 1178–1181. [Google Scholar] [CrossRef] [PubMed]
- Drexler, W.; Liu, M.; Kumar, A.; Kamali, T.; Unterhuber, A.; Leitgeb, R.A. Optical coherence tomography today: Speed, contrast, and multimodality. J. Biomed. Opt. 2014, 19, 071412. [Google Scholar] [CrossRef] [PubMed]
- Lamirel, C. Optical Coherence Tomography. In Encyclopedia of the Neurological Sciences, 2nd ed.; Aminoff, M.J., Daroff, R.B., Eds.; Academic Press: Waltham, MA, USA, 2014; pp. 660–668. [Google Scholar]
- Gora, M.J.; Suter, M.J.; Tearney, G.J.; Li, X. Endoscopic optical coherence tomography: Technologies and clinical applications [Invited]. Biomed. Opt. Express 2017, 8, 2405–2444. [Google Scholar] [CrossRef] [PubMed]
- Gelikonov, V.M.; Gelikonov, G.V. New approach to cross-polarized optical coherence tomography based on orthogonal arbitrarily polarized modes. Laser Phys. Let. 2006, 3, 445–451. [Google Scholar] [CrossRef]
- Gladkova, N.; Kiseleva, E.; Robakidze, N.; Balalaeva, I.; Karabut, M.; Gubarkova, E.; Feldchtein, F. Evaluation of oral mucosa collagen condition with cross-polarization optical coherence tomography. J. Biophotonics 2013, 6, 321–329. [Google Scholar] [CrossRef]
- Kiseleva, E.; Kirillin, M.; Feldchtein, F.; Vitkin, A.; Sergeeva, E.; Zagaynova, E.; Gladkova, N. Differential diagnosis of human bladder mucosa pathologies in vivo with cross-polarization optical coherence tomography. Biomed. Optic. Express 2015, 6, 1464–1476. [Google Scholar] [CrossRef]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; van Kerrebroeck, P.; Victor, A.; Wein, A. Standardisation Sub-Committee of the International Continence Society. The standardisation of terminology in lower urinary tract function: Report from the standardisation sub-committee of the International Continence Society. Urology 2003, 61, 37–49. [Google Scholar] [CrossRef]
- Prado-Costa, R.; Rebelo, J.; Monteiro-Barroso, J.; Preto, A.S. Ultrasound elastography: Compression elastography and shear-wave elastography in the assessment of tendon injury. Insights Imaging 2018, 9, 791–814. [Google Scholar] [CrossRef]
- Zahran, M.H.; El-Shafei, M.M.; Emara, D.M.; Eshiba, S.M. Ultrasound elastography: How can it help in differentiating breast lesions? Egypt. J. Radiol. Nucl. Med. 2018, 49, 249–258. [Google Scholar] [CrossRef]
- Gelikonov, V.M.; Gelikonov, G.V. Fibreoptic methods of cross-polarisation optical coherence tomography for endoscopic studies. IEEE J. Quantum Electron. 2008, 38, 634–640. [Google Scholar]
- Kiseleva, E.B.; Moiseev, A.A.; Kuyarov, A.S.; Molvi, M.A.; Gelikonov, G.V.; Maslennikova, A.V.; Streltsova, O.S. In vivo assessment of structural changes of the urethra in lower urinary tract disease using cross-polarization optical coherence tomography. J. Innov. Opt. Health Sci. 2020, 13, 2050024-1-16. [Google Scholar]
- Netter, F.H. Atlas of Human Anatomy, 6th ed.; Elsevier Inc.: Philadelphia, PA, USA, 2014; pp. 339–400. [Google Scholar]
- Kiseleva, E.; Gladkova, N.; Streltzova, O.; Kirillin, M.; Maslennikova, A.; Dudenkova, V.; Sergeeva, E. Cross-polarization OCT for in vivo diagnostics and prediction of bladder cancer. In Bladder Cancer—Management of NMI and Muscle-Invasive Cancer; Ather, M., Ed.; InTech: Rijeka, Croatia, 2017; pp. 43–61. [Google Scholar]
- Afraa, T.A.; Mahfouz, W.; Campeau, L.; Corcos, J. Normal lower urinary tract assessment in women: I. Uroflowmetry and post-void residual, pad tests, and bladder diaries. Int. Urogynecol. J. 2012, 23, 681–685. [Google Scholar]
- Makrushina, N.V.; Fastykovskaya, E.D. Ultrasound diagnosis of pelvic floor muscle insufficiency in women. Sib. Med. J. Tomsk 2012, 27, 91–96. [Google Scholar]
- Schaer, G.N.; Schmid, T.; Peschers, U.; Delancey, J.O. Intraurethral ultrasound correlated with urethral histology. Obstet. Gynecol. 1998, 91, 60–64. [Google Scholar] [PubMed]
- Jung, J.; Ahn, H.K.; Huh, Y. Clinical and functional anatomy of the urethral sphincter. Int. Neurourol. J. 2012, 16, 102–106. [Google Scholar]
- Cakici, Ö.U.; Hamidi, N.; Ürer, E.; Okulu, E.; Kayigil, O. Efficacy of sertraline and gabapentin in the treatment of urethral pain syndrome: Retrospective results of a single institutional cohort. Cent. European J. Urol. 2018, 71, 78–83. [Google Scholar] [PubMed]
- Nickel, J.C. Clinical evaluation of the man with chronic prostatitis/chronic pelvic pain syndrome. Urology 2002, 60, 20–22. [Google Scholar]
- Trinchieri, A.; Magri, V.; Cariani, L.; Bonamore, R.; Restelli, A.; Garlaschi, M.C.; Perletti, G. Prevalence of sexual dysfunction in men with chronic prostatitis/chronic pelvic pain syndrome. Arch. Ital. Urol. Androl. 2007, 79, 67. [Google Scholar]
- Juan, Y.-S.; Shen, J.-T.; Jang, M.-Y.; Huang, C.-H.; Li, C.-C.; Wu, W.-J. Current Management of Male Chronic Pelvic Pain Syndromes. Urol. Sci. 2010, 21, 157–162. [Google Scholar]
- Smith, C.P. Male chronic pelvic pain: An update. Indian J. Urol. 2016, 32, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Archambault-Ezenwa, L.; Markowski, A.; Barral, J.-P. A comprehensive physical therapy evaluation for Male Chronic Pelvic Pain Syndrome: A case series exploring common findings. J. Bodyw. Mov. Ther. 2019, 23, 825–834. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, S.; Lou, S.; Zhang, W.; Cai, H.; Chen, X. Application of optical coherence tomography in clinical diagnosis. J. X-ray Sci. Technol. 2019, 27, 995–1006. [Google Scholar] [CrossRef]
- Carrasco-Zevallos, O.M.; Viehland, C.; Keller, B.; Draelos, M.; Kuo, A.N.; Toth, C.A.; Izatt, J.A. Review of intraoperative optical coherence tomography: Technology and applications. Biomed. Opt. Express 2017, 8, 1607–1637. [Google Scholar] [CrossRef]
- Freund, J.E.; Buijs, M.; Savci-Heijink, C.D.; de Bruin, D.M.; de la Rosette, J.J.; van Leeuwen, T.G.; Laguna, M.P. Optical coherence tomography in urologic oncology: A comprehensive review. SN Compr. Clin. Med. 2019, 1, 67–84. [Google Scholar] [CrossRef]
- Kirillin, M.Y.; Motovilova, T.; Shakhova, N.M. Optical coherence tomography in gynecology: A narrative review. J. Biomed. Opt. 2017, 22, 121709. [Google Scholar] [CrossRef]
- Olsen, J.; Holmes, J.; Jemec, G.B.E. Advances in optical coherence tomography in dermatology—A review. J. Biomed. Opt. 2018, 23, 040901. [Google Scholar] [CrossRef]
- Oliveira, F.R.; Ramos, J.G.L.; Martins-Costa, S. Translabial ultrasonography in the assessment of urethral diameter and intrinsic urethral sphincter deficiency. J. Ultrasound Med. 2006, 25, 1153–1158. [Google Scholar] [CrossRef] [PubMed]
- Petros, P.P.E.; Ulmsten, U.I. An integral theory and its method for the diagnosis and management of female urinary incontinence. Scand. J. Urol. Nephrol. Suppl. 1993, 153, 1–93. [Google Scholar] [PubMed]
- Petros, P.E.P.; Ulmsten, U.I. An integral theory of female urinary incontinence: Experimental and clinical considerations. Acta Obstet. Gynecol. Scand. Suppl. 1990, 153, 7–31. [Google Scholar] [CrossRef]
- Petros, P. The Female Pelvic Floor. Functions, Dysfunctions and Their Treatment in Accordance with the Integral Theory, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010; p. 352. [Google Scholar]
- Streltsova, O.S.; Kiseleva, E.B.; Molvi, M.A.; Lazukin, V.F. Structural features of the urethra in patients with urethral pain syndrome. Exper. Clin. Urol. 2019, 3, 170–177. [Google Scholar] [CrossRef]
- Naboka, Y.L.; Gudima, I.A.; Dzhalagoniya, K.T.; Chernitskaya, M.L.; Ivanov, S.N. Urine and colon microbiota in patients with recurrent uncomplicated lower urinary tract infection. Urol. Herald. 2019, 7, 59–65. [Google Scholar] [CrossRef]
- Crofford, L.J. Chronic pain: Where the body meets the brain. Trans. Am. Clin. Climatol. Assoc. 2015, 126, 167–183. [Google Scholar]
- Bibas, A.G.; Podoleanu, A.G.; Cucu, R.G.; Bonmarin, M.; Dobre, G.M.; Ward, V.M.M.; Odell, E.; Boxer, A.; Gleeson, M.J.; Jackson, D.A. 3-D optical coherence tomography of the laryngeal mucosa. Clin. Otolaryngol. 2004, 29, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Di Stasio, D.; Lauritano, D.; Iquebal, H.; Romano, A.; Gentile, E.; Lucchese, A. Measurement of oral epithelial thickness by optical coherence tomography. Diagnostics 2019, 9, 90. [Google Scholar] [CrossRef]
- Parsons, C.L. The role of a leaky epithelium and potassium in the generation of bladder symptoms in interstitial cystitis/overactive bladder, urethral syndrome, prostatitis and gynecological chronic pelvic pain. BJU Int. 2011, 107, 370–375. [Google Scholar] [CrossRef]

| Type of Study | Purpose of the Study | Number of Patients in the UPS Group | Number of Patients in the Norm Group |
|---|---|---|---|
| 1. History taking | identification of the presence of any previously transferred concomitant pathology | 55 | 14 |
| 2. Laboratory tests of blood and urine | identification of inflammatory processes | 55 | 14 |
| 3. Physical examination and palpation of the urethra and the walls of the vagina (on a gynecological chair) | assessment of the state of the external opening of the urethra, detection of the presence of any myofascial aspect in the disease | 55 | - |
| 4. Uroflowmetry | assessment of the condition of the sphincters of the urethra and bladder | 55 | - |
| 5. Transvaginal US/compression US | assessment of the size, shape, structure of the urethra and bladder neck/mapping of the urethral wall and surrounding tissues stiffness | 24 | 6 |
| 6. Cystoscopy | examination the inside of the bladder in detail; identification and recording of abnormal findings | 33 * | 14 0 |
| 7. CP OCT # | visualization of the internal structure of the bladder neck and urethral wall, evaluation the condition of epithelium and connective tissue layers | 33 | 14 |
| Group | Number of Patients | Number of CP OCT Images | Average Number of CP OCT Images Created from 1 Patient | Number of CP OCT Images of Each Location | |||
|---|---|---|---|---|---|---|---|
| Bladder Neck | Distal Urethra | Medium Urethra | Proximal Urethra | ||||
| UPS | 33 | 169 | 5.12 | 43 | 41 | 40 | 45 |
| Norm | 14 | 58 | 4.14 | 16 | 14 | 12 | 16 |
| Total | 47 | 227 | 4.63 | 59 | 55 | 52 | 61 |
| Organ System with Pathology | n-Abs. (%) | Genesis of Pathology | n-Abs. (%) |
|---|---|---|---|
| 1. Gynecological | 39 (70.9) | Hormonal | 37 (94.8) |
| Inflammatory | 30 (76.9) | ||
| Surgical interventions on the pelvic organs | 12 (30.7) | ||
| 2. Respiratory | 37 (67.2) | Upper (nose, nasal cavity, pharynx, larynx) | 32 (86.4) |
| Lower (trachea, bronchi, lungs) | 5 (13.5) | ||
| Psycho-emotional sphere | 23 (41.8) | ||
| 3. Neurological | 35 (63.6) | Central nervous system | 10 (18.2) |
| Peripheral nervous system | 42 (76.4) | ||
| Psycho-emotional sphere | 23 (41.8) | ||
| 4. Urological | 24 (43.6) | Inflammatory | 10 (41.6) |
| Non-inflammatory | 17 (70.8) | ||
| 5. Gastroenterological | 18 (32.7) | Inflammatory diseases of the stomach, duodenum, biliary tract | 38 (69.0) |
| Bowel disease | 21 (38.2) | ||
| 6. Cardiovascular | 9 (16.3) | Arterial hypertension | 5 (55.5) |
| Other | 4 (44.5) | ||
| Total cases of pathology | 162 |
| Subgroup of Patients by Age, Years | Number of Patients | Number of CP OCT Images | Hyperplasia of the Bladder Neck + Proximal Urethra | Atrophy of the Bladder Neck + Proximal Urethra | Total Matches | % of Changes in the Bladder Neck + Proximal Urethra of the Total Number of Patients |
|---|---|---|---|---|---|---|
| ≤49 | 18 | 72 | 4 | 4 | 8 | 44.4% (8/18) |
| 50≥ | 15 | 60 | 7 | 7 | 14 | 93.3% (14/15) |
| Total (n = 33) | 33 | 132 | 11 | 11 | 22 | 68.8% (22/33) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Streltsova, O.; Kuyarov, A.; Molvi, M.S.A.M.; Zubova, S.; Lazukin, V.; Tararova, E.; Kiseleva, E. New Approaches in the Study of the Pathogenesis of Urethral Pain Syndrome. Diagnostics 2020, 10, 860. https://doi.org/10.3390/diagnostics10110860
Streltsova O, Kuyarov A, Molvi MSAM, Zubova S, Lazukin V, Tararova E, Kiseleva E. New Approaches in the Study of the Pathogenesis of Urethral Pain Syndrome. Diagnostics. 2020; 10(11):860. https://doi.org/10.3390/diagnostics10110860
Chicago/Turabian StyleStreltsova, Olga, Anton Kuyarov, Muhhamad Shuaib Abdul Malik Molvi, Svetlana Zubova, Valery Lazukin, Ekaterina Tararova, and Elena Kiseleva. 2020. "New Approaches in the Study of the Pathogenesis of Urethral Pain Syndrome" Diagnostics 10, no. 11: 860. https://doi.org/10.3390/diagnostics10110860
APA StyleStreltsova, O., Kuyarov, A., Molvi, M. S. A. M., Zubova, S., Lazukin, V., Tararova, E., & Kiseleva, E. (2020). New Approaches in the Study of the Pathogenesis of Urethral Pain Syndrome. Diagnostics, 10(11), 860. https://doi.org/10.3390/diagnostics10110860







