Atherosclerosis Imaging with 18F-Sodium Fluoride PET
Abstract
:1. Introduction
2. Materials and Methods
3. Results
- Disease mechanisms and targeting [10],
3.1. Disease Mechanisms and Targeting
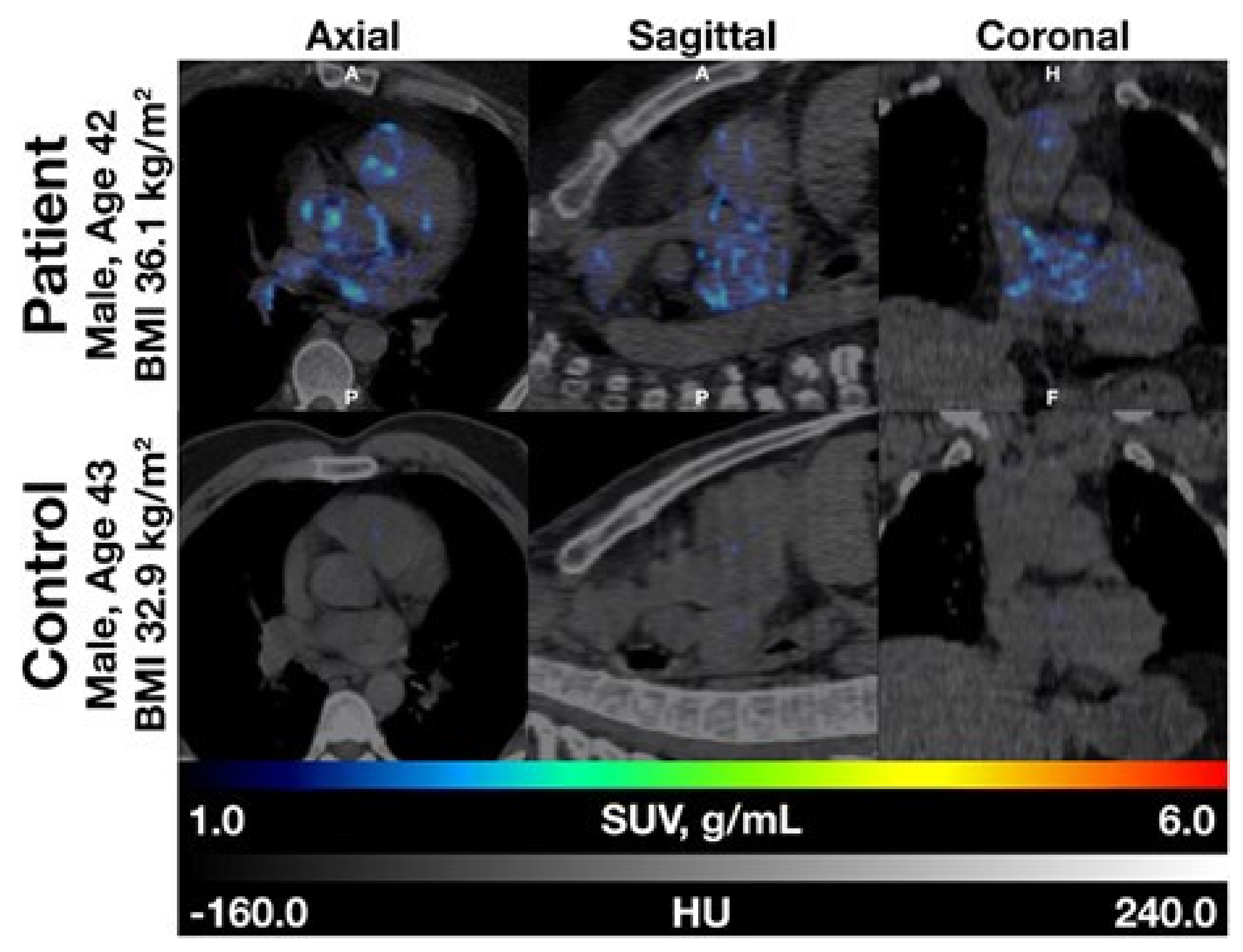
3.2. Early Detection and Prevalence of NaF Uptake in the Heart and Various Arteries
3.3. NaF Uptake in Vulnerable, High-Risk, and Ruptured Plaque
3.4. Influence of Age, Sex, and Other Factors on NaF Uptake
3.5. Association between NaF Uptake and Cardiovascular Risk Factors
3.6. NaF Uptake and Disease Progression
4. Discussion
4.1. Disease Mechanisms and Targeting
4.2. Early Detection and Prevalence of NaF Uptake in the Heart and Various Arteries
4.3. NaF Uptake in Vulnerable, High-Risk, and Ruptured Plaque
4.4. Influence of Age, Sex, and Other Factors on NaF Uptake
4.5. Association between NaF Uptake and Cardiovascular Risk Factors
4.6. NaF Uptake and Disease Progression
4.7. Methodology
4.8. Limitations
4.9. Summary of Information Gleaned from the Literature
- Increased NaF uptake is seen in penile arteries of prostate cancer patients with erectile dysfunction and in the carotid artery ipsilateral to a recent stroke, but may be relatively rare in the renal arteries of high-risk hypertensive patients without cardiovascular symptoms. Slight increases are observed in many cancers and in HIV-positive patients.
- Increased NaF uptake in vulnerable coronary and carotid plaques can characterize these further, but not serve as a single identifying parameter because of the progression from molecular and “spotty” calcification that is an established marker for vulnerable plaque to high density calcification that may confer plaque stability and decreased risk of acute coronary syndromes.
- NaF uptake is positively associated with age and several other factors, but with a wide scatter calling for individual patient assessment.
- NaF is almost consistently associated with CV risk factors, but to what degree assessment of NaF uptake can substitute or enhance CV risk stratification remains unclear.
- Abdominal aorta NaF uptake appears not to progress significantly over a few years despite unchanged or increasing CT-calcification; this calls for studies of other arterial segments recognizing that there could be a steady-state conversion of NaF uptake (mainly micro- calcification) to high density macrocalcification measured by CT [34].
- It is not clear in which compartment of the arterial wall NaF-uptake first occurs; however, the weight of evidence indicates vascular smooth cells in the medial-intimal border [51].
- It is unknown if intervention can prevent or reduce development of arterial microcalcification and later appearance of CT-calcification. Mouse experiments seem to indicate that exercise increases consolidation and density of calcification sites, thereby decreasing surface area and risk of rupture in aorta. Additional experiments in coronary arteries are needed to assess more relevant risks for morbidity and mortality.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Høilund-Carlsen, P.F.; Sturek, M.; Alavi, A.; Gerke, O. Atherosclerosis imaging with 18F-sodium fluoride PET: State-of-the-art review. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1538–1551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raggi, P.; Senior, P.; Shahbaz, S.; Kaul, P.; Hung, R.; Coulden, R.; Yeung, R.; Abele, J. 18F-Sodium Fluoride Imaging of Coronary Atherosclerosis in Ambulatory Patients with Diabetes Mellitus. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 276–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sorci, O.; Batzdorf, A.S.; Mayer, M.; Rhodes, S.; Peng, M.; Jankelovits, A.R.; Hornyak, J.N.; Gerke, O.; Høilund-Carlsen, P.F.; Alavi, A.; et al. 18F-sodium fluoride PET/CT provides prognostic clarity compared to calcium and Framingham risk scoring when addressing whole-heart arterial calcification. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1678–1687. [Google Scholar] [CrossRef] [PubMed]
- Guaraldi, G.; Milic, J.; Prandini, N.; Ligabue, G.; Esposito, F.; Ciusa, G.; Malagoli, A.; Scaglioni, R.; Besutti, G.; Beghetto, B.; et al. 18Fluoride-based molecular imaging of coronary atherosclerosis in HIV infected patients. Atherosclerosis 2020, 297, 127–135. [Google Scholar] [CrossRef]
- Kwiecinski, J.; Dey, D.; Cadet, S.; Lee, S.E.; Otaki, Y.; Huynh, P.T.; Doris, M.K.; Eisenberg, E.; Yun, M.; Jansen, M.A.; et al. Peri-Coronary Adipose Tissue Density Is Associated with 18F-Sodium Fluoride Coronary Uptake in Stable Patients With High-Risk Plaques. JACC Cardiovasc. Imaging 2019, 12, 2000–2010. [Google Scholar] [CrossRef]
- Kwiecinski, J.; Dey, D.; Cadet, S.; Lee, S.E.; Tamarappoo, B.; Otaki, Y.; Huynh, P.T.; Friedman, J.D.; Dweck, M.R.; Newby, D.E.; et al. Predictors of 18F-sodium fluoride uptake in patients with stable coronary artery disease and adverse plaque features on computed tomography angiography. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 58–66. [Google Scholar] [CrossRef] [Green Version]
- Kitagawa, T.; Nakamoto, Y.; Fujii, Y.; Sasaki, K.; Tatsugami, F.; Awai, K.; Hirokawa, Y.; Kihara, Y. Relationship between coronary arterial 18F-sodium fluoride uptake and epicardial adipose tissue analyzed using computed tomography. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1746–1756. [Google Scholar] [CrossRef]
- Youn, T.; Al’Aref, S.J.; Narula, N.; Salvatore, S.; Pisapia, D.; Dweck, M.R.; Narula, J.; Lin, F.Y.; Lu, Y.; Kumar, A.; et al. 18F-Sodium Fluoride Positron Emission Tomography/Computed Tomography in Ex Vivo Human Coronary Arteries With Histological Correlation. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 404–411. [Google Scholar] [CrossRef]
- Creager, M.D.; Hohl, T.; Hutcheson, J.D.; Moss, A.J.; Schlotter, F.; Blaser, M.C.; Park, M.A.; Lee, L.H.; Singh, S.A.; Alcaide-Corral, C.J.; et al. 18F-Fluoride Signal Amplification Identifies Microcalcifications Associated With Atherosclerotic Plaque Instability in Positron Emission Tomography/Computed Tomography Images. Circ. Cardiovasc. Imaging 2019, 12, e007835. [Google Scholar] [CrossRef] [Green Version]
- Moss, A.J.; Dweck, M.R.; Doris, M.K.; Andrews, J.; Bing, R.; Forsythe, R.O.; Cartlidge, T.R.; Pawade, T.A.; Daghem, M.; Raftis, J.B.; et al. Ticagrelor to Reduce Myocardial Injury in Patients With High-Risk Coronary Artery Plaque. JACC Cardiovasc. Imaging 2020, 13, 1549–1560. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.M.; Tarkin, J.M.; Albaghdadi, M.S.; Evans, N.R.; Le, E.; Berrett, T.B.; Sadat, U.; Joshi, F.R.; Warburton, E.A.; Buscombe, J.R.; et al. Vascular Positron Emission Tomography and Restenosis in Symptomatic Peripheral Arterial Disease: A Prospective Clinical Study. JACC Cardiovasc. Imaging 2020, 13, 1008–1017. [Google Scholar] [CrossRef]
- Zwakenberg, S.R.; de Jong, P.A.; Bartstra, J.W.; van Asperen, R.; Westerink, J.; de Valk, H.; Slart, R.; Luurtsema, G.; Wolterink, J.M.; de Borst, G.J.; et al. The effect of menaquinone-7 supplementation on vascular calcification in patients with diabetes: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2019, 110, 883–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Den Harder, A.M.; Wolterink, J.M.; Bartstra, J.W.; Spiering, W.; Zwakenberg, S.R.; Beulens, J.W.; Slart, R.; Luurtsema, G.; Mali, W.P.; de Jong, P.A. Vascular uptake on 18F-sodium fluoride positron emission tomography: Precursor of vascular calcification? J. Nucl. Cardiol. 2020, in press. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takx, R.; van Asperen, R.; Bartstra, J.W.; Zwakenberg, S.R.; Wolterink, J.M.; Celeng, C.; de Jong, P.A.; Beulens, J.W. Determinants of 18F-NaF uptake in femoral arteries in patients with type 2 diabetes mellitus. J. Nucl. Cardiol. 2020, in press. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arani, L.S.; Gharavi, M.H.; Zadeh, M.Z.; Raynor, W.Y.; Seraj, S.M.; Constantinescu, C.M.; Gerke, O.; Werner, T.J.; Høilund-Carlsen, P.F.; Alavi, A. Association between age, uptake of 18F-fluorodeoxyglucose and of 18F-sodium fluoride, as cardiovascular risk factors in the abdominal aorta. Hell. J. Nucl. Med. 2019, 22, 14–19. [Google Scholar] [CrossRef]
- Cecelja, M.; Moore, A.; Fogelman, I.; Frost, M.L.; Blake, G.M.; Chowienczyk, P. Evaluation of aortic 18F-NaF tracer uptake using PET/CT as a predictor of aortic calcification in postmenopausal women: A longitudinal study. JRSM Cardiovasc. Dis. 2019, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakahara, T.; Narula, J.; Fox, J.J.; Jinzaki, M.; Strauss, H.W. Temporal relationship between 18F-sodium fluoride uptake in the abdominal aorta and evolution of CT-verified vascular calcification. J. Nucl. Cardiol. 2019, in press. [Google Scholar] [CrossRef]
- Hop, H.; de Boer, S.A.; Reijrink, M.; Kamphuisen, P.W.; de Borst, M.H.; Pol, R.A.; Zeebregts, C.J.; Hillebrands, J.L.; Slart, R.; Boersma, H.H.; et al. 18F-sodium fluoride positron emission tomography assessed microcalcifications in culprit and non-culprit human carotid plaques. J. Nucl. Cardiol. 2019, 26, 1064–1075. [Google Scholar] [CrossRef] [Green Version]
- Evans, N.R.; Tarkin, J.M.; Chowdhury, M.M.; Le, E.; Coughlin, P.A.; Rudd, J.; Warburton, E.A. Dual-Tracer Positron-Emission Tomography for Identification of Culprit Carotid Plaques and Pathophysiology In Vivo. Circ. Cardiovasc. Imaging 2020, 13, e009539. [Google Scholar] [CrossRef]
- Ryoo, H.G.; Paeng, J.C.; Koo, B.K.; Cheon, G.J.; Lee, D.S.; Kang, K.W. Clinical implication of 18F-NaF PET/computed tomography indexes of aortic calcification in coronary artery disease patients: Correlations with cardiovascular risk factors. Nucl. Med. Commun. 2020, 41, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Al-Zaghal, A.; Mehdizadeh Seraj, S.; Werner, T.J.; Gerke, O.; Høilund-Carlsen, P.F.; Alavi, A. Assessment of Physiological Intracranial Calcification in Healthy Adults Using 18F-NaF PET/CT. J. Nucl. Med. 2019, 60, 267–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira-Santos, M.; McMahon, G.; Castelo-Branco, M.; Silva, R.; Gomes, A.; Chichorro, N.; Abrunhosa, A.; Donato, P.; de Lima, J.P.; Pego, M.; et al. Renal artery wall 18F-NaF activity and glomerular filtration rate: An exploratory analysis in a high cardiovascular risk population. Nucl. Med. Commun. 2020, 41, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, T.; Narula, J.; Tijssen, J.; Agarwal, S.; Chowdhury, M.M.; Coughlin, P.A.; Dweck, M.R.; Rudd, J.; Jinzaki, M.; Mulhall, J.; et al. 18F-Fluoride Positron Emission Tomographic Imaging of Penile Arteries and Erectile Dysfunction. J. Am. Coll. Cardiol. 2019, 73, 1386–1394. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, K.; Norikane, T.; Yamamoto, Y.; Takami, Y.; Mitamura, K.; Okada, M.; Hatakeyama, T.; Kawanishi, M.; Nishiyama, Y. Association between carotid 18F-NaF and 18F-FDG uptake on PET/CT with ischemic vascular brain disease on MRI in patients with carotid artery disease. Ann. Nucl. Med. 2019, 33, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Raggi, P.; Prandini, N.; Ligabue, G.; Braglia, G.; Esposito, F.; Milic, J.; Malagoli, A.; Scaglioni, R.; Besutti, G.; Beghetto, B.; et al. Molecular Imaging of Vascular Calcification with 18F-Sodium-Fluoride in Patients Infected with Human Immunodeficiency Virus. Int. J. Mol. Sci. 2019, 20, 1183. [Google Scholar] [CrossRef] [Green Version]
- Dai, D.; Chuang, H.H.; Macapinlac, H.A.; Li, T.; Pan, T. Correlation of fluorine 18-labeled sodium fluoride uptake and arterial calcification on whole-body PET/CT in cancer patients. Nucl. Med. Commun. 2019, 40, 604–610. [Google Scholar] [CrossRef]
- Hsu, J.J.; Fong, F.; Patel, R.; Qiao, R.; Lo, K.; Soundia, A.; Chang, C.C.; Le, V.; Tseng, C.H.; Demer, L.L.; et al. Changes in microarchitecture of atherosclerotic calcification assessed by 18F-NaF PET and CT after a progressive exercise regimen in hyperlipidemic mice. J. Nucl. Cardiol. 2020, in press. [Google Scholar] [CrossRef]
- Rucher, G.; Cameliere, L.; Fendri, J.; Anfray, A.; Abbas, A.; Kamel, S.; Dupas, Q.; Delcroix, N.; Berger, L.; Manrique, A.; et al. Molecular imaging of endothelial activation and mineralization in a mouse model of accelerated atherosclerosis. EJNMMI Res. 2019, 9, 80. [Google Scholar] [CrossRef] [Green Version]
- Sturek, M.; Alloosh, M.; Sellke, F.W. Swine disease models for optimal vascular engineering. Annu. Rev. Biomed. Eng. 2020, 22, 25–49. [Google Scholar] [CrossRef] [Green Version]
- McKenney-Drake, M.L.; Moghbel, M.C.; Paydary, K.; Alloosh, M.; Houshmand, S.; Moe, S.; Salavati, A.; Sturek, J.M.; Territo, P.R.; Weaver, C.; et al. 18F-NaF and 18F-FDG as molecular probes in the evaluation of atherosclerosis. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 2190–2200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Rosendael, A.R.; Narula, J.; Lin, F.Y.; van den Hoogen, I.J.; Gianni, U.; Al Hussein Alawamlh, O.; Dunham, P.C.; Peña, J.M.; Lee, S.E.; Andreini, D.; et al. Association of High-Density Calcified 1K Plaque With Risk of Acute Coronary Syndrome. JAMA Cardiol. 2020, 5, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Stabley, J.N.; Towler, D.A. Arterial Calcification in Diabetes Mellitus: Preclinical Models and Translational Implications. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 205–217. [Google Scholar] [CrossRef] [Green Version]
- Moss, A.J.; Doris, M.K.; Andrews, J.; Bing, R.; Daghem, M.; van Beek, E.; Forsyth, L.; Shah, A.; Williams, M.C.; Sellers, S.; et al. Molecular Coronary Plaque Imaging Using 18F-Fluoride. Circ. Cardiovasc. Imaging 2019, 12, e008574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrews, J.P.M.; Fayad, Z.A.; Dweck, M.R. New methods to image unstable atherosclerotic plaques. Atherosclerosis 2018, 272, 118–128. [Google Scholar] [CrossRef] [Green Version]
- MacAskill, M.G.; Newby, D.E.; Tavares, A.A.S. Frontiers in positron emission tomography imaging of the vulnerable atherosclerotic plaque. Cardiovasc. Res. 2019, 115, 1952–1962. [Google Scholar] [CrossRef] [PubMed]
- Salarian, M.; Sadeghi, M.M. Hype or hope: 18F-NaF positron emission tomography for vulnerable coronary plaque imaging. Circ. Cardiovasc. Imaging 2019, 12, e009591. [Google Scholar] [CrossRef]
- Irkle, A.; Vesey, A.T.; Lewis, D.Y.; Skepper, J.N.; Bird, J.L.; Dweck, M.R.; Joshi, F.R.; Gallagher, F.A.; Warburton, E.A.; Bennett, M.R.; et al. Identifying active vascular microcalcification by 18F-sodium fluoride positron emission tomography. Nat. Commun. 2015, 6, 7495. [Google Scholar] [CrossRef] [Green Version]
- Kwiecinski, J.; Cadet, S.; Daghem, M.; Lassen, M.L.; Dey, D.; Dweck, M.R.; Berman, D.S.; Newby, D.E.; Slomka, P.J. Whole-vessel coronary 18F-sodium fluoride PET for assessment of the global coronary microcalcification burden. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1736–1745. [Google Scholar] [CrossRef]
- Blomberg, B.A.; Thomassen, A.; de Jong, P.A.; Simonsen, J.A.; Lam, M.G.; Nielsen, A.L.; Mickley, H.; Mali, W.P.; Alavi, A.; Høilund-Carlsen, P.F. Impact of personal characteristics and technical factors on quantification of [18F]-sodium fluoride uptake in human arteries: Prospective evaluation of healthy subjects. J. Nucl. Med. 2015, 56, 1534–1540. [Google Scholar] [CrossRef] [Green Version]
- Lassen, M.L.; Kwiecinski, J.; Dey, D.; Cadet, S.; Germano, G.; Berman, D.S.; Adamson, P.D.; Moss, A.J.; Dweck, M.R.; Newby, D.E.; et al. Triple-gated motion and blood pool clearance corrections improve reproducibility of coronary 18F-NaF PET. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2610–2620. [Google Scholar] [CrossRef] [PubMed]
- Kwiecinski, J.; Slomka, P.J.; Dweck, M.R.; Newby, D.E.; Berman, D.S. Vulnerable plaque imaging using 18F-sodium fluoride positron emission tomography. Br. J. Radiol. 2020, 93, 20190797. [Google Scholar] [CrossRef] [PubMed]
- Alavi, A.; Werner, T.J.; Høilund-Carlsen, P.F. What can be and what cannot be accomplished with PET to detect and characterize atherosclerotic plaques. J. Nucl. Cardiol. 2018, 25, 2012–2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Høilund-Carlsen, P.F.; Edenbrandt, L.; Alavi, A. Global disease score (GDS) is the name of the game! Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1768–1772. [Google Scholar] [CrossRef] [Green Version]
- Arbab-Zadeh, A.; Fuster, V. The myth of the “vulnerable plaque”: Transitioning from a focus on individual lesions to atherosclerotic disease burden for coronary artery disease risk assessment. J. Am. Coll. Cardiol. 2015, 65, 846–855. [Google Scholar] [CrossRef] [Green Version]
- Arbab-Zadeh, A. Does “Vulnerable” Atherosclerotic Plaque Modify Coronary Blood Flow: How Myths Perpetuate. JACC Cardiovasc. Imaging 2020, 13, 757–759. [Google Scholar] [CrossRef]
- Gerke, O.; Ehlers, K.; Motschall, E.; Høilund-Carlsen, P.F.; Vach, W. PET/CT-based response evaluation in cancer-a systematic review of design issues. Mol. Imaging Biol. 2020, 22, 33–46. [Google Scholar] [CrossRef]
- McKenney-Drake, M.L.; Rodenbeck, S.D.; Owen, M.K.; Schultz, K.A.; Alloosh, M.; Tune, J.D.; Sturek, M. Biphasic alterations in coronary smooth muscle Ca2+ regulation in a repeat cross-sectional study of coronary artery disease severity in metabolic syndrome. Atherosclerosis 2016, 249, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Meijers, W.C.; de Boer, R.A. Common risk factors for heart failure and cancer. Cardiovasc. Res. 2019, 115, 844–853. [Google Scholar] [CrossRef]
- Oishi, Y.; Manabe, I. Organ System Crosstalk in Cardiometabolic Disease in the Age of Multimorbidity. Front. Cardiovasc. Med. 2020, 7, 64. [Google Scholar] [CrossRef]
- Durham, A.L.; Speer, M.Y.; Scatena, M.; Giachelli, C.M.; Shanahan, C.M. Role of smooth muscle cells in vascular calcification: Implications in atherosclerosis and arterial stiffness. Cardiovasc. Res. 2018, 114, 590–600. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Høilund-Carlsen, P.F.; Piri, R.; Constantinescu, C.; Iversen, K.K.; Werner, T.J.; Sturek, M.; Alavi, A.; Gerke, O. Atherosclerosis Imaging with 18F-Sodium Fluoride PET. Diagnostics 2020, 10, 852. https://doi.org/10.3390/diagnostics10100852
Høilund-Carlsen PF, Piri R, Constantinescu C, Iversen KK, Werner TJ, Sturek M, Alavi A, Gerke O. Atherosclerosis Imaging with 18F-Sodium Fluoride PET. Diagnostics. 2020; 10(10):852. https://doi.org/10.3390/diagnostics10100852
Chicago/Turabian StyleHøilund-Carlsen, Poul F., Reza Piri, Caius Constantinescu, Kasper Karmark Iversen, Thomas J. Werner, Michael Sturek, Abass Alavi, and Oke Gerke. 2020. "Atherosclerosis Imaging with 18F-Sodium Fluoride PET" Diagnostics 10, no. 10: 852. https://doi.org/10.3390/diagnostics10100852
APA StyleHøilund-Carlsen, P. F., Piri, R., Constantinescu, C., Iversen, K. K., Werner, T. J., Sturek, M., Alavi, A., & Gerke, O. (2020). Atherosclerosis Imaging with 18F-Sodium Fluoride PET. Diagnostics, 10(10), 852. https://doi.org/10.3390/diagnostics10100852








