Long-Term Survivability of Tardigrade Paramacrobiotus experimentalis (Eutardigrada) at Increased Magnesium Perchlorate Levels: Implications for Astrobiological Research
Abstract
1. Introduction
2. Materials and Methods
2.1. Culturing Procedure
2.2. Experimental Design
2.3. Microscopy Measurements
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yamagishi, A.; Kakegawa, T.; Usui, T. (Eds.) Astrobiology; Springer: Singapore, 2019; ISBN 9789811336386. [Google Scholar]
- Dunér, D. Introduction: The History and Philosophy of Astrobiology. Astrobiology 2012, 12, 901–905. [Google Scholar] [CrossRef]
- Jagadeesh, M.K.; Roszkowska, M.; Kaczmarek, Ł. Tardigrade Indexing Approach on Exoplanets. Life Sci. Space Res. 2018, 19, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Hecht, M.H.; Kounaves, S.P.; Quinn, R.C.; West, S.J.; Young, S.M.M.; Ming, D.W.; Catling, D.C.; Clark, B.C.; Boynton, W.V.; Hoffman, J.; et al. Detection of Perchlorate and the Soluble Chemistry of Martian Soil at the Phoenix Lander Site. Science 2009, 325, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Kounaves, S.P.; Hecht, M.H.; Kapit, J.; Gospodinova, K.; DeFlores, L.; Quinn, R.C.; Boynton, W.V.; Clark, B.C.; Catling, D.C.; Hredzak, P.; et al. Wet Chemistry Experiments on the 2007 Phoenix Mars Scout Lander Mission: Data Analysis and Results. J. Geophys. Res. 2010, 115. [Google Scholar] [CrossRef]
- Kounaves, S.P.; Chaniotakis, N.A.; Chevrier, V.F.; Carrier, B.L.; Folds, K.E.; Hansen, V.M.; McElhoney, K.M.; O’Neil, G.D.; Weber, A.W. Identification of the Perchlorate Parent Salts at the Phoenix Mars Landing Site and Possible Implications. Icarus 2014, 232, 226–231. [Google Scholar] [CrossRef]
- Clark, B.C.; Kounaves, S.P. Evidence for the Distribution of Perchlorates on Mars. Int. J. Astrobiol. 2016, 15, 311–318. [Google Scholar] [CrossRef]
- Martin, P.E.; Farley, K.A.; Douglas Archer, P., Jr.; Hogancamp, J.V.; Siebach, K.L.; Grotzinger, J.P.; McLennan, S.M. Reevaluation of Perchlorate in Gale Crater Rocks Suggests Geologically Recent Perchlorate Addition. J. Geophys. Res. Planets 2020, 125, e2019JE006156. [Google Scholar] [CrossRef]
- Navarro-Gonzalez, R.; Sutter, B.; Archer, D.; Ming, D.; Eigenbrode, J.; Franz, H.; Glavin, D.; McAdam, A.; Stern, J.; McKay, C.; et al. Possible Detection of Perchlorates by the Sample Analysis at Mars (SAM) Instrument: Comparison with Previous Missions. In EGU General Assembly Conference Abstracts; EGU General Assembly: Vienna, Austria, 2013; p. EGU2013-6529. [Google Scholar]
- Kumarathilaka, P.; Oze, C.; Indraratne, S.P.; Vithanage, M. Perchlorate as an Emerging Contaminant in Soil, Water and Food. Chemosphere 2016, 150, 667–677. [Google Scholar] [CrossRef]
- Pleus, R.C.; Corey, L.M. Environmental Exposure to Perchlorate: A Review of Toxicology and Human Health. Toxicol. Appl. Pharmacol. 2018, 358, 102–109. [Google Scholar] [CrossRef]
- Davila, A.F.; Willson, D.; Coates, J.D.; McKay, C.P. Perchlorate on Mars: A Chemical Hazard and a Resource for Humans. Int. J. Astrobiol. 2013, 12, 321–325. [Google Scholar] [CrossRef]
- Navarro-González, R.; Vargas, E.; de la Rosa, J.; Raga, A.C.; McKay, C.P. Reanalysis of the Viking Results Suggests Perchlorate and Organics at Midlatitudes on Mars. J. Geophys. Res. 2010, 115. [Google Scholar] [CrossRef]
- Seckbach, J.; Stan-Lotter, H. Extremophiles as Astrobiological Models; John Wiley & Sons: Hoboken, NJ, USA, 2020; ISBN 9781119593102. [Google Scholar]
- Sijimol, M.R.; Jyothy, S.; Pradeepkumar, A.P.; Chandran, M.S.S.; Ghouse, S.S.; Mohan, M. Review on Fate, Toxicity, and Remediation of Perchlorate. Environ. Forensics 2015, 16, 125–134. [Google Scholar] [CrossRef]
- Rzymski, P.; Poniedziałek, B.; Hippmann, N.; Kaczmarek, Ł. Screening the Survival of Cyanobacteria under Perchlorate Stress. Potential Implications for Mars in Situ Resource Utilization. Astrobiology 2022, 22, 672–684. [Google Scholar] [CrossRef]
- Fagliarone, C.; Fernandez, B.G.; Di Stefano, G.; Mosca, C.; Billi, D. Insights into the Chaotropic Tolerance of the Desert Cyanobacterium Chroococcidiopsis sp. 029 (Chroococcidiopsales, Cyanobacteria). J. Phycol. 2023, 60, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Alsaleh, S.A.; Barron, L.; Sturzenbaum, S. Perchlorate Detection via an Invertebrate Biosensor. Anal. Methods 2021, 13, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, S.; Meyer, T.L.; Ferreira, A.L.; Amorim, M.J.; Soares, A.M.V.M. Single and Joint Effects of Perchlorates to Daphnia Magna: Additivity and Interaction Patterns. Fresenius Environ. Bull. 2012, 21, 844–852. [Google Scholar]
- Hespeels, B.; Fontaneto, D.; Cornet, V.; Penninckx, S.; Berthe, J.; Bruneau, L.; Larrick, J.W.; Rapport, E.; Bailly, J.; Debortoli, N.; et al. Back to the Roots, Desiccation and Radiation Resistances Are Ancestral Characters in Bdelloid Rotifers. BMC Biol. 2023, 21, 72. [Google Scholar] [CrossRef] [PubMed]
- Carrero, D.; Pérez-Silva, J.G.; Quesada, V.; López-Otín, C. Differential Mechanisms of Tolerance to Extreme Environmental Conditions in Tardigrades. Sci. Rep. 2019, 9, 14938. [Google Scholar] [CrossRef] [PubMed]
- Copley, J. Indestructible. New Scientist (1971), 23 October 1999; 44–46. [Google Scholar]
- Nelson, D.R.; Guidetti, R.; Rebecchi, L.; Kaczmarek, Ł.; McInnes, S. Phylum Tardigrada. In Thorp and Covich’s Freshwater Invertebrates; Elsevier: Amsterdam, The Netherlands, 2020; pp. 505–522. [Google Scholar]
- Degma, P.; Bertolani, R.; Guidetti, R. Actual Checklist of Tardigrada Species. 2019. Available online: https://iris.unimore.it/retrieve/bf8e14a4-625f-4cdd-8100-347e5cbc5f63/Actual%20checklist%20of%20Tardigrada%2042th%20Edition%2009-01-23.pdf (accessed on 27 February 2024).
- Jönsson, K.I.; Guidetti, R. Effects of Methyl Bromide Fumigation on Anhydrobiotic Micrometazoans. Ecotoxicol. Environ. Saf. 2001, 50, 72–75. [Google Scholar] [CrossRef]
- Horikawa, D.D.; Sakashita, T.; Katagiri, C.; Watanabe, M.; Kikawada, T.; Nakahara, Y.; Hamada, N.; Wada, S.; Funayama, T.; Higashi, S.; et al. Radiation Tolerance in the Tardigrade Milnesium Tardigradum. Int. J. Radiat. Biol. 2006, 82, 843–848. [Google Scholar] [CrossRef]
- Møbjerg, N.; Halberg, K.A.; Jørgensen, A.; Persson, D.; Bjørn, M.; Ramløv, H.; Kristensen, R.M. Survival in Extreme Environments—On the Current Knowledge of Adaptations in Tardigrades. Acta Physiol. 2011, 202, 409–420. [Google Scholar] [CrossRef]
- Guidetti, R.; Rizzo, A.M.; Altiero, T.; Rebecchi, L. What Can We Learn from the Toughest Animals of the Earth? Water Bears (Tardigrades) as Multicellular Model Organisms in Order to Perform Scientific Preparations for Lunar Exploration. Planet. Space Sci. 2012, 74, 97–102. [Google Scholar] [CrossRef]
- Kaczmarek, Ł.; Roszkowska, M.; Fontaneto, D.; Jezierska, M.; Pietrzak, B.; Wieczorek, R.; Poprawa, I.; Kosicki, J.Z.; Karachitos, A.; Kmita, H. Staying Young and Fit? Ontogenetic and Phylogenetic Consequences of Animal Anhydrobiosis. J. Zool. 2019, 309, 1–11. [Google Scholar] [CrossRef]
- Arakawa, K. Examples of Extreme Survival: Tardigrade Genomics and Molecular Anhydrobiology. Annu. Rev. Anim. Biosci. 2022, 10, 17–37. [Google Scholar] [CrossRef] [PubMed]
- Roszkowska, M.; Gołdyn, B.; Wojciechowska, D.; Księżkiewicz, Z.; Fiałkowska, E.; Pluskota, M.; Kmita, H.; Kaczmarek, Ł. How Long Can Tardigrades Survive in the Anhydrobiotic State? A Search for Tardigrade Anhydrobiosis Patterns. PLoS ONE 2023, 18, e0270386. [Google Scholar] [CrossRef] [PubMed]
- Schill, R.O.; Hengherr, S. Environmental Adaptations: Desiccation Tolerance. In Water Bears: The Biology of Tardigrades; Springer International Publishing: Cham, Switzerland, 2018; pp. 273–293. ISBN 9783319957012. [Google Scholar]
- Roszkowska, M.; Gołdyn, B.; Wojciechowska, D.; Kosicki, J.Z.; Fiałkowska, E.; Kmita, H.; Kaczmarek, Ł. Tolerance to Anhydrobiotic Conditions among Two Coexisting Tardigrade Species Differing in Life Strategies. Zool. Stud. 2021, 60, e74. [Google Scholar]
- Kasianchuk, N.; Rzymski, P.; Kaczmarek, Ł. The Biomedical Potential of Tardigrade Proteins: A Review. Biomed. Pharmacother. 2023, 158, 114063. [Google Scholar] [CrossRef] [PubMed]
- Bertolani, R.; Rebecchi, L.; Jönsson, K.I.; Borsari, S.; Guidetti, R.; Altieri, T. Tardigrades as a Model for Experiences of Animal Survival in the Spaces. Microgravity Q. 2001, 2, 211–212. [Google Scholar]
- Horikawa, D.D.; Kunieda, T.; Abe, W.; Watanabe, M.; Nakahara, Y.; Yukuhiro, F.; Sakashita, T.; Hamada, N.; Wada, S.; Funayama, T.; et al. Establishment of a Rearing System of the Extremotolerant Tardigrade Ramazzottius Varieornatus: A New Model Animal for Astrobiology. Astrobiology 2008, 8, 549–556. [Google Scholar] [CrossRef]
- Jönsson, K.I. Tardigrades—Evolutionary Explorers in Extreme Environments. In Extremophiles as Astrobiological Models; Wiley: Hoboken, NJ, USA, 2020; pp. 255–274. [Google Scholar]
- Kayastha, P.; Rzymski, P.; Gołdyn, B.; Nagwani, A.K.; Fiałkowska, E.; Pajdak-Stós, A.; Sobkowiak, R.; Robotnikowski, G.; Kaczmarek, Ł. Tolerance against Exposure to Solution of Magnesium Perchlorate in Microinvertebrates. Zool. J. Linn. Soc. 2023, 200, 239–257. [Google Scholar] [CrossRef]
- Kaczmarek, Ł.; Roszkowska, M.; Poprawa, I.; Janelt, K.; Kmita, H.; Gawlak, M.; Fiałkowska, E.; Mioduchowska, M. Integrative Description of Bisexual Paramacrobiotus experimentalis sp. Nov. (Macrobiotidae) from Republic of Madagascar (Africa) with Microbiome Analysis. Mol. Phylogenet. Evol. 2020, 145, 106730. [Google Scholar] [CrossRef] [PubMed]
- Roszkowska, M.; Wojciechowska, D.; Kmita, H.; Cerbin, S.; Dziuba, M.K.; Fiałkowska, E.; Sobkowiak, R.; Szydło, W.; Kaczmarek, Ł. Tips and Tricks How to Culture Water Bears: Simple Protocols for Culturing Eutardigrades (Tardigrada) under Laboratory Conditions. Eur. Zool. J. 2021, 88, 449–465. [Google Scholar] [CrossRef]
- Ericksen, G.E. The Chilean Nitrate Deposits: The Origin of the Chilean Nitrate Deposits, Which Contain a Unique Group of Saline Minerals, Has Provoked Lively Discussion for More than 100 Years. Am. Sci. 1983, 71, 366–374. [Google Scholar]
- Calderón, R.; Palma, P.; Parker, D.; Molina, M.; Godoy, F.A.; Escudey, M. Perchlorate Levels in Soil and Waters from the Atacama Desert. Arch. Environ. Contam. Toxicol. 2014, 66, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Steinmaus, C.M. Perchlorate in Water Supplies: Sources, Exposures, and Health Effects. Curr. Environ. Health Rep. 2016, 3, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Naidu, R. A Review of Perchlorate Contamination: Analysis and Remediation Strategies. Chemosphere 2023, 338, 139562. [Google Scholar] [CrossRef] [PubMed]
- Wadsworth, J.; Cockell, C.S. Perchlorates on Mars Enhance the Bacteriocidal Effects of UV Light. Sci. Rep. 2017, 7, 4662. [Google Scholar] [CrossRef]
- Mooney, T.J.; McCullough, C.D.; Jansen, A.; Chandler, L.; Douglas, M.; Harford, A.J.; van Dam, R.; Humphrey, C. Elevated Magnesium Concentrations Altered Freshwater Assemblage Structures in a Mesocosm Experiment. Environ. Toxicol. Chem. 2020, 39, 1973–1987. [Google Scholar] [CrossRef]
- Szklarek, S.; Górecka, A.; Salabert, B.; Wojtal-Frankiewicz, A. Acute Toxicity of Seven De-Icing Salts on Four Zooplankton Species–Is There an “Eco-Friendly” Alternative? Int. J. Ecohydrol. Hydrobiol. 2022, 22, 589–597. [Google Scholar] [CrossRef]
- Urbansky, E.T.; Brown, S.K. Perchlorate Retention and Mobility in Soils. J. Environ. Monit. 2003, 5, 455–462. [Google Scholar] [CrossRef]
- Kumar, M.; Kumari, P.; Reddy, C.R.K.; Jha, B. Salinity and Desiccation Induced Oxidative Stress Acclimation in Seaweeds. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2014; pp. 91–123. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine. J. Free Radic. Biol. Med. 1985, 1, 331–332. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.A.; Pérez de la Lastra, J.M.; Plou, F.J.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef] [PubMed]
- Hand, S.C.; Menze, M.A.; Toner, M.; Boswell, L.; Moore, D. LEA Proteins during Water Stress: Not Just for Plants Anymore. Annu. Rev. Physiol. 2011, 73, 115–134. [Google Scholar] [CrossRef] [PubMed]
- Hering, L.; Bouameur, J.-E.; Reichelt, J.; Magin, T.M.; Mayer, G. Novel Origin of Lamin-Derived Cytoplasmic Intermediate Filaments in Tardigrades. eLife 2016, 5, e11117. [Google Scholar] [CrossRef] [PubMed]
- Hesgrove, C.; Boothby, T.C. The Biology of Tardigrade Disordered Proteins in Extreme Stress Tolerance. Cell Commun. Signal. 2020, 18, 178. [Google Scholar] [CrossRef] [PubMed]
- Mínguez-Toral, M.; Cuevas-Zuviría, B.; Garrido-Arandia, M.; Pacios, L.F. A Computational Structural Study on the DNA-Protecting Role of the Tardigrade-Unique Dsup Protein. Sci. Rep. 2020, 10, 13424. [Google Scholar] [CrossRef] [PubMed]
- Boothby, T.C.; Tapia, H.; Brozena, A.H.; Piszkiewicz, S.; Smith, A.E.; Giovannini, I.; Rebecchi, L.; Pielak, G.J.; Koshland, D.; Goldstein, B. Tardigrades Use Intrinsically Disordered Proteins to Survive Desiccation. Mol. Cell 2017, 65, 975–984.e5. [Google Scholar] [CrossRef]
- Nguyen, K.; Kc, S.; Gonzalez, T.; Tapia, H.; Boothby, T.C. Trehalose and Tardigrade CAHS Proteins Work Synergistically to Promote Desiccation Tolerance. Commun. Biol. 2022, 5, 1046. [Google Scholar] [CrossRef]
- Lim, S.; Reilly, C.B.; Barghouti, Z.; Marelli, B.; Way, J.C.; Silver, P.A. Tardigrade Secretory Proteins Protect Biological Structures from Desiccation. bioRxiv 2023. [Google Scholar] [CrossRef]
- Rzymski, P.; Klimaszyk, P.; Kasianchuk, N.; Jakubiak, P.; Proch, J.; Niedzielski, P. Blue on Red: Chemical Conditions of Liquid Water Emerging on Simulated Martian Regolith. Icarus 2023, 389, 115263. [Google Scholar] [CrossRef]
- Heidemann, N.W.T.; Smith, D.K.; Hygum, T.L.; Stapane, L.; Clausen, L.K.B.; Jørgensen, A.; Hélix-Nielsen, C.; Møbjerg, N. Osmotic Stress Tolerance in Semi-Terrestrial Tardigrades. Zool. J. Linn. Soc. 2016, 178, 912–918. [Google Scholar] [CrossRef]
- Møbjerg, N.; Neves, R.C. New Insights into Survival Strategies of Tardigrades. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2021, 254, 110890. [Google Scholar] [CrossRef]
- Moore, M.; Folt, C. Zooplankton Body Size and Community Structure: Effects of Thermal and Toxicant Stress. Trends Ecol. Evol. 1993, 8, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Hagelbäck, P.; Jönsson, K.I. An Experimental Study on Tolerance to Hypoxia in Tardigrades. Front. Physiol. 2023, 14, 1249773. [Google Scholar] [CrossRef] [PubMed]
- Neves, R.C.; Hvidepil, L.K.B.; Sørensen-Hygum, T.L.; Stuart, R.M.; Møbjerg, N. Thermotolerance Experiments on Active and Desiccated States of Ramazzottius Varieornatus Emphasize That Tardigrades Are Sensitive to High Temperatures. Sci. Rep. 2020, 10, 94. [Google Scholar] [CrossRef]
- Schill, R.O.; Mali, B.; Dandekar, T.; Schnölzer, M.; Reuter, D.; Frohme, M. Molecular Mechanisms of Tolerance in Tardigrades: New Perspectives for Preservation and Stabilization of Biological Material. Biotechnol. Adv. 2009, 27, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, K.I. Radiation Tolerance in Tardigrades: Current Knowledge and Potential Applications in Medicine. Cancers 2019, 11, 1333. [Google Scholar] [CrossRef]
- Horikawa, D.D.; Cumbers, J.; Sakakibara, I.; Rogoff, D.; Leuko, S.; Harnoto, R.; Arakawa, K.; Katayama, T.; Kunieda, T.; Toyoda, A.; et al. Analysis of DNA Repair and Protection in the Tardigrade Ramazzottius Varieornatus and Hypsibius Dujardini after Exposure to UVC Radiation. PLoS ONE 2013, 8, e64793. [Google Scholar] [CrossRef] [PubMed]
- Ricci, C.; Riolo, G.; Marzocchi, C.; Brunetti, J.; Pini, A.; Cantara, S. The Tardigrade Damage Suppressor Protein Modulates Transcription Factor and DNA Repair Genes in Human Cells Treated with Hydroxyl Radicals and UV-C. Biology 2021, 10, 970. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Xu, R.; Nie, H.; Yan, Q.; Liu, J.; Sun, Y. Mechanism of Interaction between Ammonium Perchlorate and Aluminum. J. Phys. Chem. A 2023, 127, 6532–6540. [Google Scholar] [CrossRef] [PubMed]
- Glavin, D.P.; Freissinet, C.; Miller, K.E.; Eigenbrode, J.L.; Brunner, A.E.; Buch, A.; Sutter, B.; Archer, P.D., Jr.; Atreya, S.K.; Brinckerhoff, W.B.; et al. Evidence for Perchlorates and the Origin of Chlorinated Hydrocarbons Detected by SAM at the Rocknest Aeolian Deposit in Gale Crater. J. Geophys. Res. Planets 2013, 118, 1955–1973. [Google Scholar] [CrossRef]
- Hassler, D.M.; Zeitlin, C.; Wimmer-Schweingruber, R.F.; Ehresmann, B.; Rafkin, S.; Eigenbrode, J.L.; Brinza, D.E.; Weigle, G.; Böttcher, S.; Böhm, E.; et al. Mars’ Surface Radiation Environment Measured with the Mars Science Laboratory’s Curiosity Rover. Science 2014, 343, 1244797. [Google Scholar] [CrossRef] [PubMed]
- Ojha, L.; Wilhelm, M.B.; Murchie, S.L.; McEwen, A.S.; Wray, J.J.; Hanley, J.; Massé, M.; Chojnacki, M. Spectral Evidence for Hydrated Salts in Recurring Slope Lineae on Mars. Nat. Geosci. 2015, 8, 829–832. [Google Scholar] [CrossRef]
- Alzahrani, S.M.; Ebert, P.R. Stress Pre-Conditioning with Temperature, UV and Gamma Radiation Induces Tolerance against Phosphine Toxicity. PLoS ONE 2018, 13, e0195349. [Google Scholar] [CrossRef]
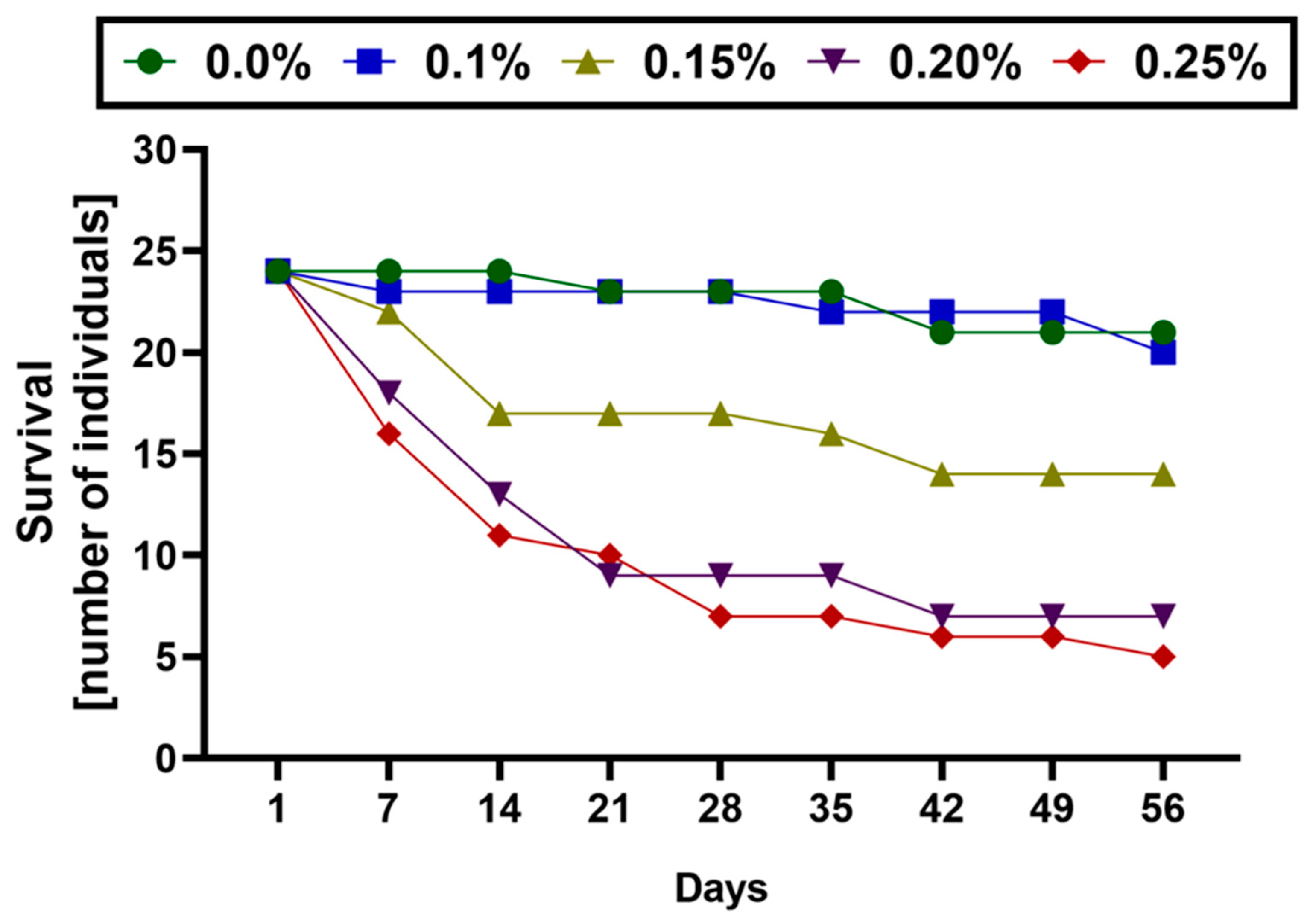
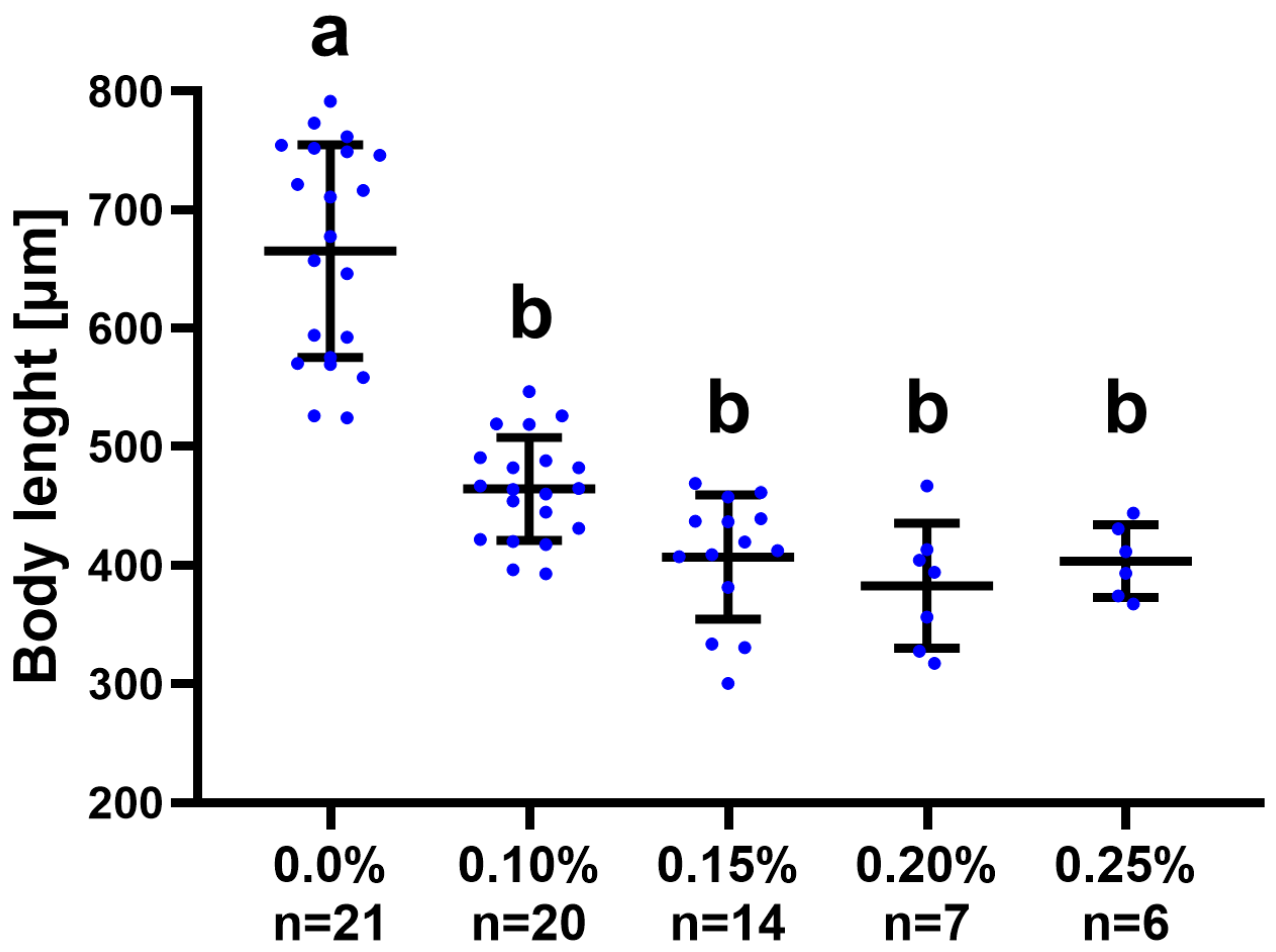
| ER/Days | 1 | 7 | 14 | 21 | 28 | 35 | 42 | 49 | 56 | Survivability |
|---|---|---|---|---|---|---|---|---|---|---|
| control | 24 | 24 | 24 | 23 | 23 | 23 | 21 | 21 | 21 | 87.5% |
| 0.10% | 24 | 23 | 23 | 23 | 23 | 22 | 22 | 22 | 20 | 83.3% |
| 0.15% | 24 | 22 | 17 | 17 | 17 | 16 | 14 | 14 | 14 | 58.3% |
| 0.20% | 24 | 18 | 13 | 9 | 9 | 9 | 7 | 7 | 7 | 29.2% |
| 0.25% | 24 | 16 | 11 | 10 | 7 | 7 | 6 | 6 | 5 | 20.8% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilanowska, P.A.; Rzymski, P.; Kaczmarek, Ł. Long-Term Survivability of Tardigrade Paramacrobiotus experimentalis (Eutardigrada) at Increased Magnesium Perchlorate Levels: Implications for Astrobiological Research. Life 2024, 14, 335. https://doi.org/10.3390/life14030335
Wilanowska PA, Rzymski P, Kaczmarek Ł. Long-Term Survivability of Tardigrade Paramacrobiotus experimentalis (Eutardigrada) at Increased Magnesium Perchlorate Levels: Implications for Astrobiological Research. Life. 2024; 14(3):335. https://doi.org/10.3390/life14030335
Chicago/Turabian StyleWilanowska, Paulina Anna, Piotr Rzymski, and Łukasz Kaczmarek. 2024. "Long-Term Survivability of Tardigrade Paramacrobiotus experimentalis (Eutardigrada) at Increased Magnesium Perchlorate Levels: Implications for Astrobiological Research" Life 14, no. 3: 335. https://doi.org/10.3390/life14030335
APA StyleWilanowska, P. A., Rzymski, P., & Kaczmarek, Ł. (2024). Long-Term Survivability of Tardigrade Paramacrobiotus experimentalis (Eutardigrada) at Increased Magnesium Perchlorate Levels: Implications for Astrobiological Research. Life, 14(3), 335. https://doi.org/10.3390/life14030335








