Influence of Lemongrass Essential Oil (Cymbopogon flexuosus) Supplementation on Diabetes in Rat Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Essential Oil
2.2. Gas Chromatography-Mass Spectrometry (GC-MS)
2.3. Determination of Antioxidant Activity
2.4. Animals
2.5. Induction of Diabetes Mellitus
2.6. Experimental Groups
2.7. Supplementation
2.8. Sample Collection
2.9. Determination of Serum Biochemical Markers
2.10. Determination of Oxidative Stress Markers in Tissues
2.11. Statistical Analysis
3. Results
3.1. Chemical Composition of EOCF
3.2. Antioxidant Activity of EOCF
3.2.1. DPPH Radical Scavenging
3.2.2. ABTS Radical Scavenging
3.2.3. FRAP (Ferric Reducing Antioxidant Power)
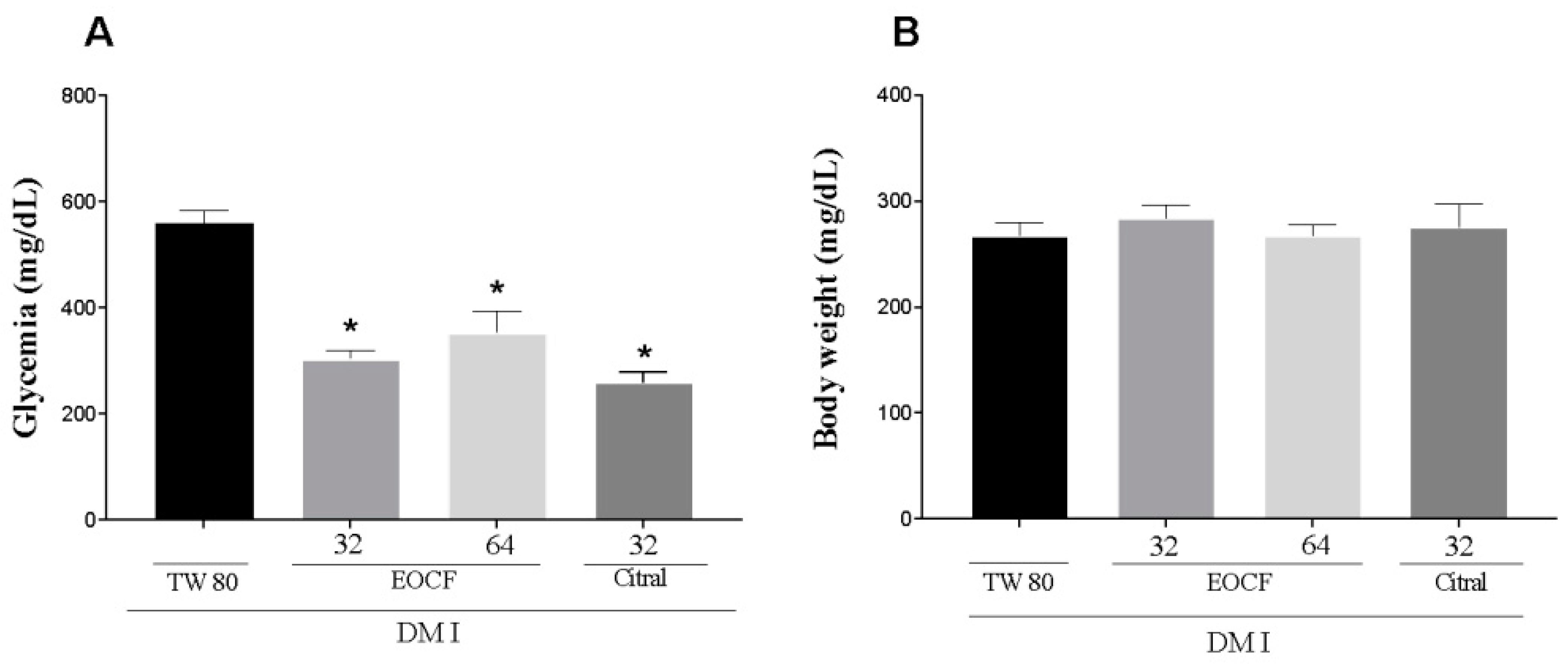
3.3. Anti Hyperglycemic Activity
3.4. Effect of EOCF on Hepatic and Renal Functions
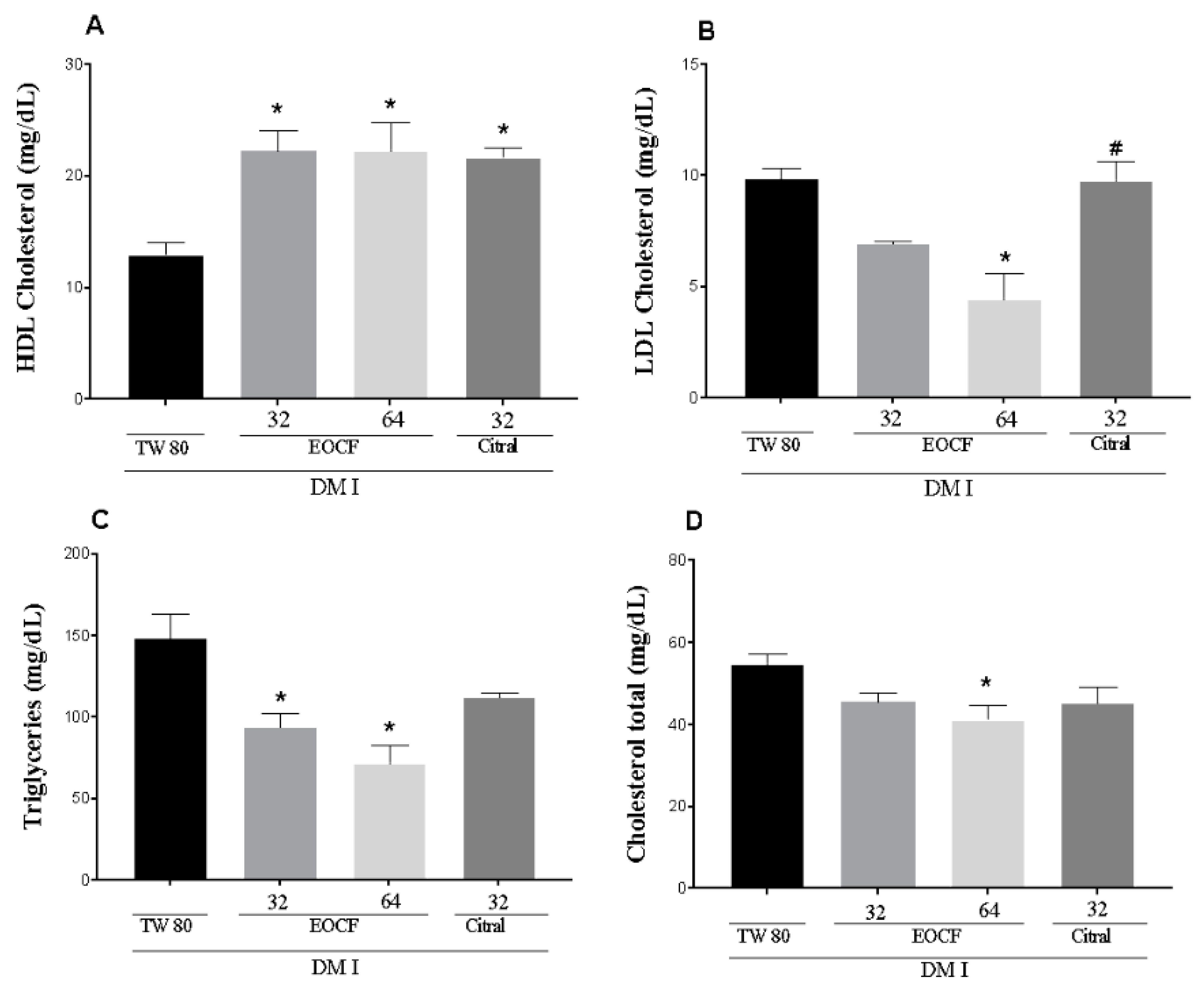
3.5. Effect of EOCF on Metabolic Lipid Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qi, J.; He, P.; Yao, H.; Xue, Y.; Sun, W.; Lu, P.; Qi, X.; Zhang, Z.; Jing, R.; Cui, B.; et al. Developing a Prediction Model for All-Cause Mortality Risk among Patients with Type 2 Diabetes Mellitus in Shanghai, China. J. Diabetes 2023, 15, 27–35. [Google Scholar] [CrossRef]
- International Diabetes Federation. IDF Diabetes Atlas; International Diabetes Federation: Brussels, Belgium, 2021. [Google Scholar]
- American Diabetes Association. Standards of Care in Diabetes—2023 Abridged for Primary Care Providers. Clin. Diabetes 2023, 41, 4–31. [Google Scholar] [CrossRef]
- Siahbalaei, R.; Kavoosi, G.; Shakeri, R. In Vitro Antioxidant and Antidiabetic Activity of Essential Oils Encapsulated in Gelatin-Pectin Particles against Sugar, Lipid and Protein Oxidation and Amylase and Glucosidase Activity. Food Sci. Nutr. 2020, 8, 6457–6466. [Google Scholar] [CrossRef]
- Zhang, P.; Li, T.; Wu, X.; Nice, E.C.; Huang, C.; Zhang, Y. Oxidative Stress and Diabetes: Antioxidative Strategies. Front. Med. 2020, 14, 583–600. [Google Scholar] [CrossRef]
- Rodacki, M.; Teles, M.; Gabbay, M.; Montenegro, R.; Bertoluci, M. Classificação Do Diabetes. In Diretriz Oficial da Sociedade Brasileira de Diabetes; Conectando Pessoas: Brasília, Brazil, 2022. [Google Scholar] [CrossRef]
- Tan, S.Y.; Mei Wong, J.L.; Sim, Y.J.; Wong, S.S.; Mohamed Elhassan, S.A.; Tan, S.H.; Ling Lim, G.P.; Rong Tay, N.W.; Annan, N.C.; Bhattamisra, S.K.; et al. Type 1 and 2 Diabetes Mellitus: A Review on Current Treatment Approach and Gene Therapy as Potential Intervention. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 364–372. [Google Scholar] [CrossRef]
- Bellido, V.; Pinés-Corrales, P.J.; Villar-Taibo, R.; Ampudia-Blasco, F.J. Time-in-Range for Monitoring Glucose Control: Is It Time for a Change? Diabetes Res. Clin. Pract. 2021, 177, 108917. [Google Scholar] [CrossRef]
- Magalhães, D.B.; Castro, I.; Lopes-Rodrigues, V.; Pereira, J.M.; Barros, L.; Ferreira, I.C.F.R.; Xavier, C.P.R.; Vasconcelos, M.H. Melissa officinalis L. Ethanolic Extract Inhibits the Growth of a Lung Cancer Cell Line by Interfering with the Cell Cycle and Inducing Apoptosis. Food Funct. 2018, 9, 3134–3142. [Google Scholar] [CrossRef]
- Bhatnagar, A. Chemical Composition and Antioxidant Activity of Essential Oil of Cymbopogon flexuosus. J. Appl. Nat. Sci. 2020, 12, 25–29. [Google Scholar] [CrossRef]
- da Silva, D.H.A.; Barbosa, H.M.; da Silva, J.F.; Moura, C.A.; Gomes, D.A.; Almeida, J.R.G.S.; Lira, E.C. Antidiabetic Properties of Oral Treatment of Hexane and Chloroform Fractions of Morus nigra Leaves in Streptozotocin-Induced Rats. An. Acad. Bras. Ciênc. 2021, 93, e20210744. [Google Scholar] [CrossRef]
- Li, J.; Bai, L.; Wei, F.; Zhao, J.; Wang, D.; Xiao, Y.; Yan, W.; Wei, J. Therapeutic Mechanisms of Herbal Medicines against Insulin Resistance: A Review. Front. Pharmacol. 2019, 10, 661. [Google Scholar] [CrossRef]
- Kifle, Z.D.; Bayleyegn, B.; Tadesse, T.Y.; Woldeyohanins, A.E. Prevalence and Associated Factors of Herbal Medicine Use among Adult Diabetes Mellitus Patients at Government Hospital, Ethiopia: An Institutional-Based Cross-Sectional Study. Metab. Open 2021, 11, 100120. [Google Scholar] [CrossRef]
- Ghafouri, A.; Karegar, S.J.; Hajiluian, G.; Hosseini, S.; Shidfar, S.; Kamalinejad, M.; Hosseini, A.F.; Heydari, I.; Shidfar, F. The Effects of Aqueous and Ethanolic Extracts of Rheum ribes on Insulin-Resistance and Apolipoproteins in Patients with Type 2 Diabetes Mellitus: A Randomized Controlled Trial. BMC Complement. Med. Ther. 2023, 23, 46. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Habib, S.; Sahu, D.; Gupta, J. Chemical Properties and Therapeutic Potential of Citral, a Monoterpene Isolated from Lemongrass. Med. Chem. 2021, 17, 2–12. [Google Scholar] [CrossRef]
- Kumar, K.; Malhotra, J.; Kumar, S.; Sood, V.; Singh, D.; Sharma, M.; Joshi, R. Citral Enrichment in Lemongrass (Cymbopogon flexuosus) Oil Using Spinning Band Equipped High Vacuum Distillation Column and Sensory Evaluation of Fractions. Food Chem. Adv. 2023, 2, 100291. [Google Scholar] [CrossRef]
- Mansour, H.A.; Newairy, A.S.A.; Yousef, M.I.; Sheweita, S.A. Biochemical study on the effects of some Egyptian herbs in alloxan-induced diabetic rats. Toxicology 2002, 170, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Ghadyale, V.; Takalikar, S.; Haldavnekar, V.; Arvindekar, A. Effective Control of Postprandial Glucose Level through Inhibition of Intestinal Alpha Glucosidase by Cymbopogon martinii (Roxb.). Evid. Based Complement. Alternat. Med. 2012, 2012, 372909. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Kumar, A.; Prakash, O.; Krishnan, S.; Gupta, A.K. Essential Oil of Cymbopogon citratus against Diabetes: Validation by in vivo Experiments and Computational Studies. J. Bioanal. Biomed. 2013, 5, 194–203. [Google Scholar] [CrossRef]
- Khan, S.J.; Afroz, S.; Khan, R.A. Antihyperlipidemic and anti-hyperglycemic effects of Cymbopogon jwarancusa in high-fat high-sugar diet model. Pak. J. Pharm. Sci. 2018, 31, 1341–1345. [Google Scholar]
- Itankar, P.R.; Tauqeer, M.; Dalal, J.S. Toxicological and pharmacological profiling of organically and non-organically cultivated Cymbopogon citratus. J. Ayurveda Integr. Med. 2019, 10, 233–240. [Google Scholar] [CrossRef]
- Garba, H.A.; Mohammed, A.; Ibrahim, M.A.; Shuaibu, M.N. Effect of lemongrass (Cymbopogon citratus Stapf) tea in a type 2 diabetes rat model. Clin. Phytosci. 2020, 6, 19. [Google Scholar] [CrossRef]
- Dobhal, S.; Singh, M.F.; Setya, S.; Bisht, S. Comparative Assessment of the Effect of Lemongrass (Cymbopogon citratus) Ethanolic Extract, Aqueous Extract and Essential Oil in High Fat Diet and Fructose Induced Metabolic Syndrome in Rats. Indian J. Pharm. Educ. Res. 2022, 56, S281–S293. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on Products of Browning Reaction: Antioxidative Activities of Product of Browning Reaction Prepared from Glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Wang, X.; Li, H.; Fan, Z.; Liu, Y. Effect of Zinc Supplementation on Type 2 Diabetes Parameters and Liver Metallothionein Expressions in Wistar Rats. J. Physiol. Biochem. 2012, 68, 563–572. [Google Scholar] [CrossRef]
- Barman, S.; Srinivasan, K. Ameliorative Effect of Zinc Supplementation on Compromised Small Intestinal Health in Streptozotocin-Induced Diabetic Rats. Chem. Biol. Interact. 2019, 307, 37–50. [Google Scholar] [CrossRef]
- Gross, J.L.; Silveiro, S.P.; Camargo, J.L.; Reichelt, A.J.; de Azevedo, M.J. Diabetes Melito: Diagnóstico, Classificação e Avaliação Do Controle Glicêmico. Arq. Bras. Endocrinol. Metabol. 2002, 46, 16–26. [Google Scholar] [CrossRef]
- Ewenighi, C.O.; Dimkpa, U.; Adejumo, B.I.; Onyeanusi, J.C.; Onoh, L.U.M.; Ezeugwu, U.; Onoh, G.O.; Uzor, S.; Orji, E.; Anojulu, A. Estimation of Lipid Profile and Glucose Level in Alloxan-Induced Diabetic Rats Treated with Cymbopogon citratus (Lemongrass). J. Exp. Integr. Med. 2013, 3, 249–253. [Google Scholar] [CrossRef]
- Falode, J.A.; Olofinlade, T.B.; Fayeun, G.S.; Adeoye, A.O.; Bamisaye, F.A.; Ajuwon, O.R.; Obafemi, T.O. Free and Bound Phenols from Cymbopogon citratus Mitigated Hepatocellular Injury in Streptozotocin-Induced Type 1 Diabetic Male Rats via Decrease in Oxidative Stress, Inflammation, and Other Risk Markers. Pharmacol. Res.—Mod. Chin. Med. 2023, 7, 100234. [Google Scholar] [CrossRef]
- Mishra, C.; Khalid, M.A.; Nazmin, F.; Singh, B.; Tripathi, D.; Waseem, M.; Mahdi, A.A. Effects of Citral on Oxidative Stress and Hepatic Key Enzymes of Glucose Metabolism in Streptozotocin/High-Fat-Diet Induced Diabetic Dyslipidemic Rats. Iran. J. Basic Med. Sci. 2019, 22, 49–57. [Google Scholar] [CrossRef]
- Anaruma, N.D.; Schmidt, F.L.; Duarte, M.C.T.; Figueira, G.M.; Delarmelina, C.; Benato, E.A.; Sartoratto, A. Control of Colletotrichum gloeosporioides (Penz.) Sacc. in Yellow Passion Fruit Using Cymbopogon citratus Essential Oil. Braz. J. Microbiol. 2010, 41, 66–73. [Google Scholar] [CrossRef]
- Vera, S.S.; Zambrano, D.F.; Méndez-Sanchez, S.C.; Rodríguez-Sanabria, F.; Stashenko, E.E.; Duque Luna, J.E. Essential Oils with Insecticidal Activity against Larvae of Aedes aegypti (Diptera: Culicidae). Parasitol. Res. 2014, 113, 2647–2654. [Google Scholar] [CrossRef]
- Mata, A.T.; Proença, C.; Ferreira, A.R.; Serralheiro, M.L.M.; Nogueira, J.M.F.; Araújo, M.E.M. Antioxidant and Antiacetylcholinesterase Activities of Five Plants Used as Portuguese Food Spices. Food Chem. 2007, 103, 778–786. [Google Scholar] [CrossRef]
- Ganjewala, D. Cymbopogon Essential Oils: Chemical Compositions and Bioactivities. Int. J. Essent. Oil Ther. 2009, 3, 56–65. [Google Scholar]
- Devi, M.A.; Sahoo, D.; Singh, T.B.; Rajashekar, Y. Antifungal Activity and Volatile Organic Compounds Analysis of Essential Oils from Cymbopogon Species Using Solid-Phase Microextraction-Gas Chromatography-Mass Spectrometry. J. Agric. Food Res. 2021, 3, 100110. [Google Scholar] [CrossRef]
- Pietta, P.-G. Flavonoids as Antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Lin, H.S. Antioxidant Activity in Fruits and Leaves of Blackberry, Raspberry, and Strawberry Varies with Cultivar and Developmental Stage. J. Agric. Food Chem. 2000, 48, 140–146. [Google Scholar] [CrossRef] [PubMed]
- de Sá, A.Á.M.; dos Santos, E.W.P.; Santana, M.H.D.S.; Santos, A.D.J.; de Araujo, G.R.S.; Santana, D.G.; Maria de Lara, P.D.M.; e Silva, A.M.D.O.; Correa, C.B.; Nunes, R.D.S.; et al. Evaluation of the Incorporation of Essential Oils in Microemulsions as a Promising Formulation in the Inhibition of Tyrosinase. Ind. Crops Prod. 2020, 154, 112654. [Google Scholar] [CrossRef]
- Poznyak, A.; Grechko, A.V.; Poggio, P.; Myasoedova, V.A.; Alfieri, V.; Orekhov, A.N. The Diabetes Mellitus—Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism and Chronic Inflammation. Int. J. Mol. Sci. 2020, 21, 1835. [Google Scholar] [CrossRef]
- Zanetti, M.; Barazzoni, R.; Kiwanuka, E.; Vettore, M.; Vedovato, M.; Tessari, P. Accelerated Whole-Body Protein Catabolism in Subjects with Type 2 Diabetes Mellitus and Albuminuria. PLoS ONE 2020, 15, e0243638. [Google Scholar] [CrossRef] [PubMed]
- Akinlade, O.M.; Owoyele, B.V.; Soladoye, A.O. Streptozotocin-Induced Type 1 and 2 Diabetes in Rodents: A Model for Studying Diabetic Cardiac Autonomic Neuropathy. Afr. Health Sci. 2021, 21, 719–727. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. 14. Management of Diabetes in Pregnancy: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019, 42, S165–S172. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Diagnosis and Management of Type 2 Diabetes (HEARTS-D); World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Jiang, N.; Chen, Z.; Yin, X.; Liu, L.; Yang, H.; Tan, X.; Wang, J.; Li, H.; Tian, M.; Lu, Z.; et al. Association of Metformin with Mortality or ARDS in Patients with COVID-19 and Type 2 Diabetes: A Retrospective Cohort Study. Diabetes Res. Clin. Pract. 2021, 173, 108619. [Google Scholar] [CrossRef]
- Ighodaro, O.M. Molecular Pathways Associated with Oxidative Stress in Diabetes Mellitus. Biomed. Pharmacother. 2018, 108, 656–662. [Google Scholar] [CrossRef]
- Finsen, S.H.; Hansen, M.R.; Hansen, P.B.L.; Mortensen, S.P. Aldosterone Induces Vasoconstriction in Individuals with Type 2 Diabetes: Effect of Acute Antioxidant Administration. J. Clin. Endocrinol. Metab. 2021, 106, E1262–E1270. [Google Scholar] [CrossRef] [PubMed]
- Grabež, M.; Škrbić, R.; Stojiljković, M.P.; Vučić, V.; Grujić, V.R.; Jakovljević, V.; Djuric, D.M.; Suručić, R.; Šavikin, K.; Bigović, D.; et al. A Prospective, Randomized, Double-Blind, Placebo-Controlled Trial of Polyphenols on the Outcomes of Inflammatory Factors and Oxidative Stress in Patients with Type 2 Diabetes Mellitus. Rev. Cardiovasc. Med. 2022, 23, 057. [Google Scholar] [CrossRef] [PubMed]
- Sarabhai, T.; Mastrototaro, L.; Kahl, S.; Bönhof, G.J.; Jonuscheit, M.; Bobrov, P.; Katsuyama, H.; Guthoff, R.; Wolkersdorfer, M.; Herder, C.; et al. Hyperbaric Oxygen Rapidly Improves Tissue-Specific Insulin Sensitivity and Mitochondrial Capacity in Humans with Type 2 Diabetes: A Randomised Placebo-Controlled Crossover Trial. Diabetologia 2023, 66, 57–69. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of Medical Care in Diabetes—2022 Abridged for Primary Care Providers. Clin. Diabetes 2022, 40, 10–38. [Google Scholar] [CrossRef]
- American Diabetes Association Professional Practice Committee. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2022. Diabetes Care 2022, 45, S17–S38. [Google Scholar] [CrossRef]
- Liu, L.; Swift, S.; Tollemache, C.; Perera, J.; Kilmartin, P.A. Antimicrobial and Antioxidant AIE Chitosan-Based Films Incorporating a Pickering Emulsion of Lemon Myrtle (Backhousia citriodora) Essential Oil. Food Hydrocoll. 2022, 133, 107971. [Google Scholar] [CrossRef]
- Modak, T.; Mukhopadhaya, A. Effects of Citral, a Naturally Occurring Antiadipogenic Molecule, on an Energy-Intense Diet Model of Obesity. Indian J. Pharmacol. 2011, 43, 300–305. [Google Scholar] [CrossRef]
- Vaezi, M.; Yaghmaei, P.; Hayati-Roodbari, N.; Irani, S.; Ebrahim-Habibi, A. Citral Effect in Male NMRI Mice Nonalcoholic Steatosis Model: Assessing Biochemical and Histological Parameters and PPARα Gene Expression. Braz. J. Pharm. Sci. 2018, 54, e17596. [Google Scholar] [CrossRef]
- Zarandi, M.H.; Sharifiyazdi, H.; Nazifi, S.; Ghaemi, M.; Bakhtyari, M.K. Effects of Citral on Serum Inflammatory Factors and Liver Gene Expression of IL-6 and TNF-Alpha in Experimental Diabetes. Comp. Clin. Pathol. 2021, 30, 351–361. [Google Scholar] [CrossRef]
- Groussard, C.; Machefer, G.; Rannou, F.; Faure, H.; Zouhal, H.; Sergent, O.; Chevanne, M.; Cillard, J.; Gratas-Delamarche, A. Physical Fitness and Plasma Non-Enzymatic Antioxidant Status at Rest and after a Wingate Test. Can. J. Appl. Physiol. 2003, 28, 79–92. [Google Scholar] [CrossRef]
- Santos, J.L.; Araújo, S.S.; Silva, A.M.O.; Lima, C.A.; Vieira Souza, L.M.; Costa, R.A.; Aidar, F.J.A.; Voltarelli, F.A.; Estevam, C.D.S.; Marçal, A.C. Ethanolic extract and ethyl acetate fraction of Coutoubea spicata attenuate hyperglycemia, oxidative stress, and muscle damage in alloxan-induced diabetic rats subjected to resistance exercise training program. Appl. Physiol. Nutr. Metab. 2020, 45, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Newsholme, P.; Keane, K.N.; Carlessi, R.; Cruzat, V. Oxidative Stress Pathways in Pancreatic-Cells and Insulin-Sensitive Cells and Tissues: Importance to Cell Metabolism, Function, and Dysfunction. Am. J. Physiol. Cell Physiol. 2019, 317, 420–433. [Google Scholar] [CrossRef] [PubMed]


| Experimental Group | Species | Number of Groups | Initial Number of Animals/Final Numbers of Animals | Total Number |
|---|---|---|---|---|
| TW 80 | Rats Wistar | 1 | 5/5 | 5 |
| EOCF 32 | Rats Wistar | 1 | 5/5 | 5 |
| EOCF 64 | Rats Wistar | 1 | 5/5 | 5 |
| CT | Rats Wistar | 1 | 5/5 | 5 |
| Total: | 20 |
| Peak | Time of Retention (min) | Compound | Experimental IR | Literary IR | Relative Area (%) |
|---|---|---|---|---|---|
| 1 | 10.61 | Tricyclene | 928 | 926 | 0.07 |
| 2 | 10.98 | Pinene <α-> | 937 | 939 | 0.11 |
| 3 | 11.69 | Camphene | 956 | 954 | 0.64 |
| 4 | 16.67 | Nonanone | 1074 | 1090 | 0.52 |
| 5 | 17.96 | Linalool | 1103 | 1096 | 0.65 |
| 6 | 20.50 | Isogeraniol | 1158 | 1229 | 0.30 |
| 7 | 20.88 | Isoneral | 1166 | 1164 | 0.38 |
| 8 | 21.76 | Isocitral | 1185 | 1180 | 0.74 |
| 9 | 24.66 | Neral | 1248 | 1238 | 19.42 |
| 10 | 24.99 | Geraniol | 1256 | 1252 | 12.58 |
| 11 | 26.02 | α-citral (3,7-dimethyl-2,6-octadienal) | 1278 | 1318 | 53.21 |
| 12 | 30.63 | Geranyl acetate | 1381 | 1381 | 4.90 |
| 13 | 32.82 | Caryophyllene | 1432 | 1419 | 1.35 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Júnior, A.S.S.; Aidar, F.J.; Silva, L.A.S.; de B. Silva, T.; de Almeida, S.F.M.; Teles, D.C.S.; de L. Junior, W.; Schimieguel, D.M.; de Souza, D.A.; Nascimento, A.C.S.; et al. Influence of Lemongrass Essential Oil (Cymbopogon flexuosus) Supplementation on Diabetes in Rat Model. Life 2024, 14, 336. https://doi.org/10.3390/life14030336
Júnior ASS, Aidar FJ, Silva LAS, de B. Silva T, de Almeida SFM, Teles DCS, de L. Junior W, Schimieguel DM, de Souza DA, Nascimento ACS, et al. Influence of Lemongrass Essential Oil (Cymbopogon flexuosus) Supplementation on Diabetes in Rat Model. Life. 2024; 14(3):336. https://doi.org/10.3390/life14030336
Chicago/Turabian StyleJúnior, Ailton S. S., Felipe J. Aidar, Luiz A. S. Silva, Thais de B. Silva, Sara F. M. de Almeida, Daiane C. S. Teles, Waldecy de L. Junior, Dulce M. Schimieguel, Daniel A. de Souza, Ana C. S. Nascimento, and et al. 2024. "Influence of Lemongrass Essential Oil (Cymbopogon flexuosus) Supplementation on Diabetes in Rat Model" Life 14, no. 3: 336. https://doi.org/10.3390/life14030336
APA StyleJúnior, A. S. S., Aidar, F. J., Silva, L. A. S., de B. Silva, T., de Almeida, S. F. M., Teles, D. C. S., de L. Junior, W., Schimieguel, D. M., de Souza, D. A., Nascimento, A. C. S., Camargo, E. A., dos Santos, J. L., de O. e Silva, A. M., de S. Nunes, R., Borges, L. P., & Lira, A. A. M. (2024). Influence of Lemongrass Essential Oil (Cymbopogon flexuosus) Supplementation on Diabetes in Rat Model. Life, 14(3), 336. https://doi.org/10.3390/life14030336








