Parasitism Shifts the Effects of Native Soil Microbes on the Growth of the Invasive Plant Alternanthera philoxeroides
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Species
2.2. Containers and Substrate
2.3. Experimental Design
2.4. Harvest and Measurements
2.5. Data Analysis
3. Results
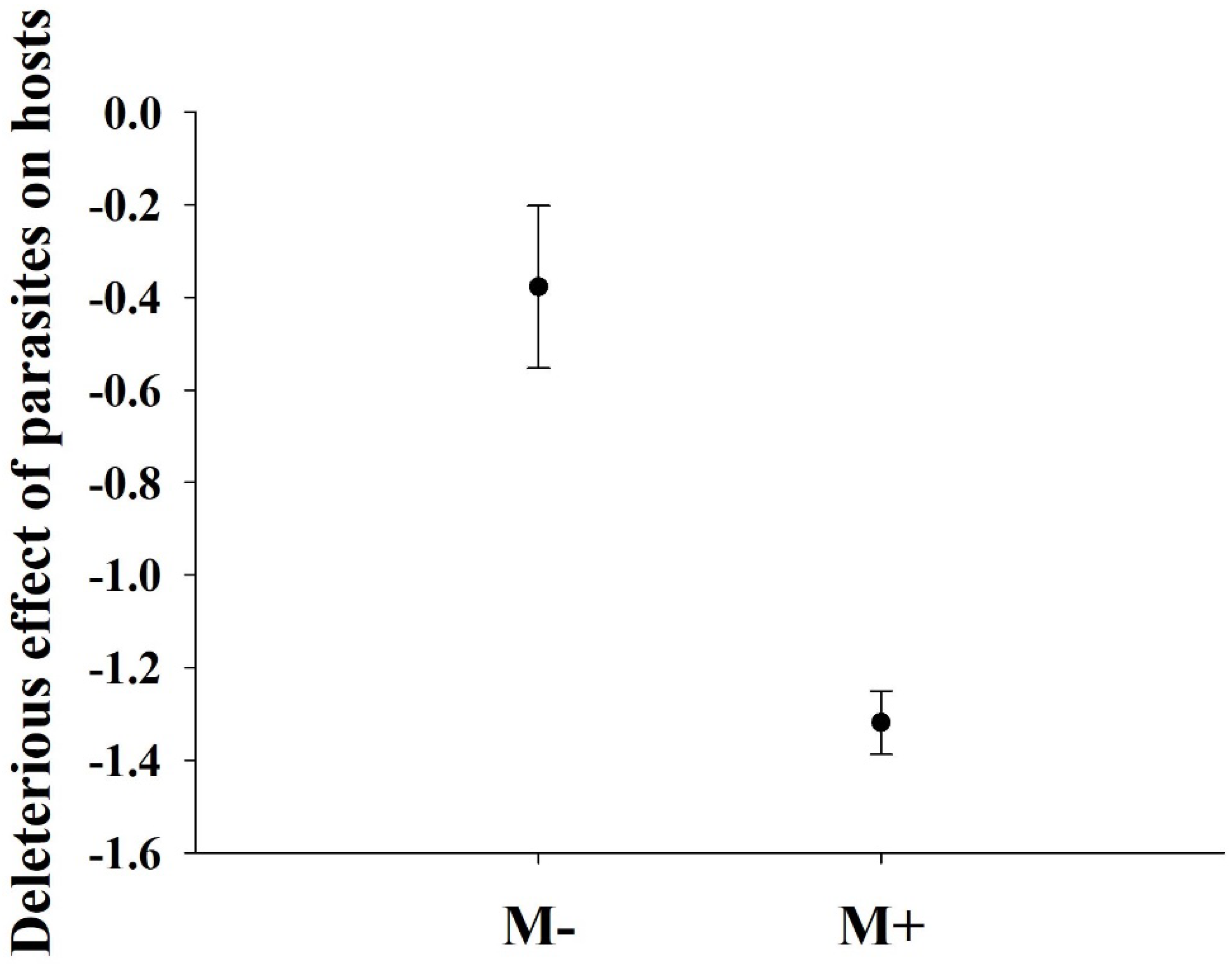
3.1. Effects of Parasites and Soil Microbes on the Biomass of A. philoxeroides
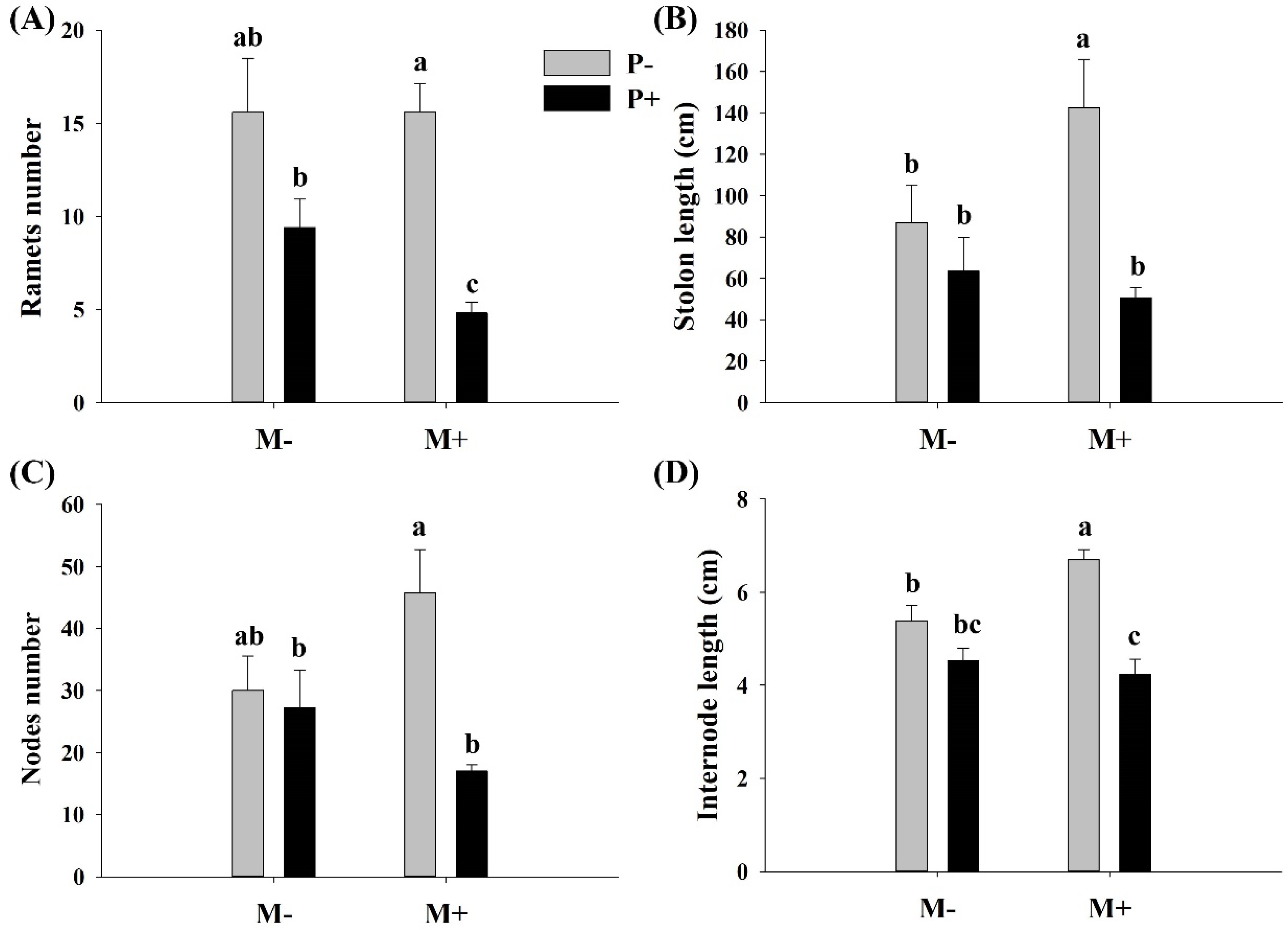
3.2. Effects of Parasites and Soil Microbes on the Clonal Propagation of A. philoxeroides
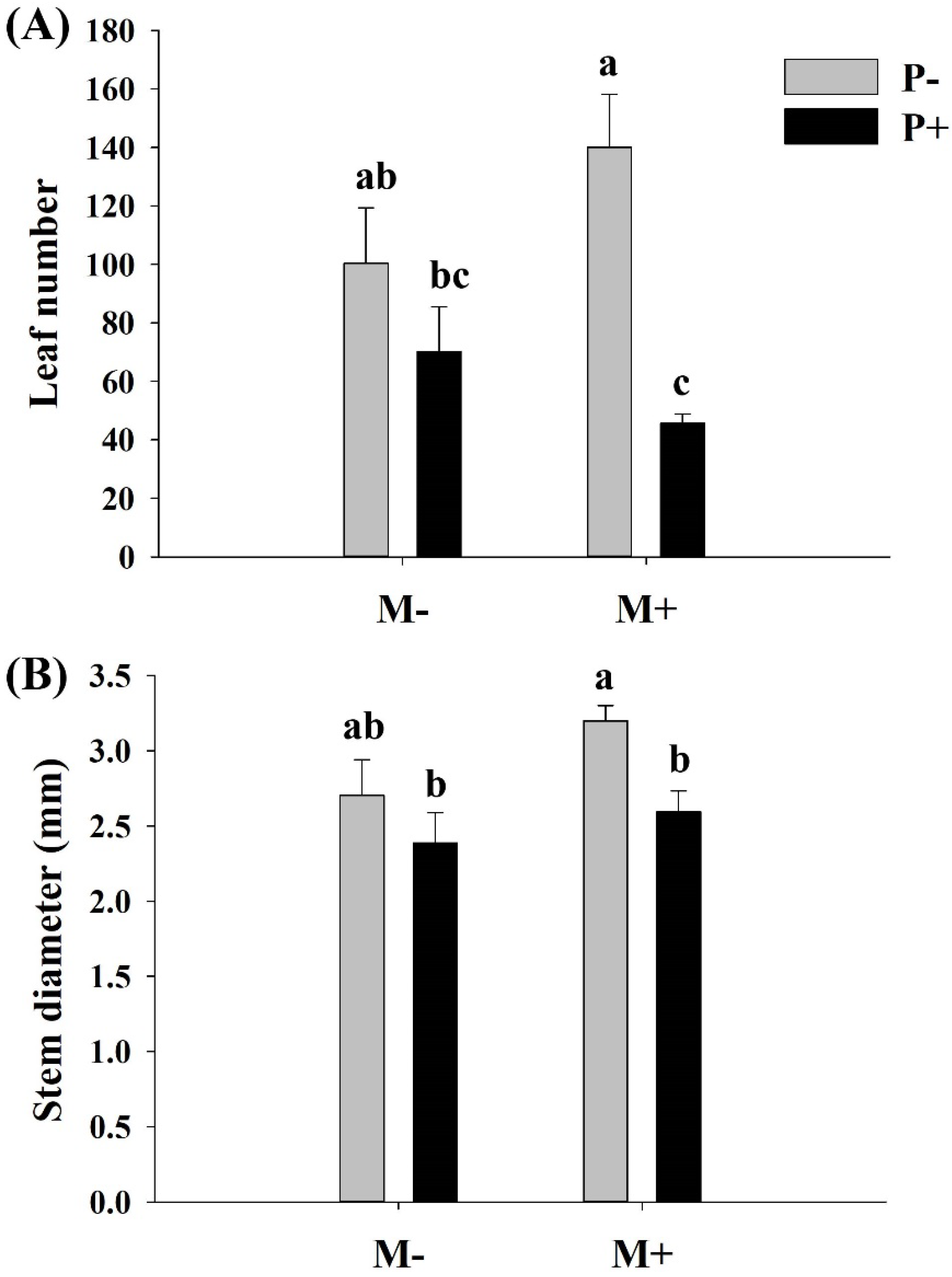
3.3. Effects of Parasites and Soil Microbes on the Morphology of A. philoxeroides
3.4. Effects of Soil Microbes on the Biomass of Cuscuta grovonii
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mack, R.N.; Simberloff, D.; Lonsdale, W.M.; Evans, H.; Clout, M.; Bazzaz, F.A. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecol. Appl. 2000, 10, 689–710. [Google Scholar] [CrossRef]
- Vilà, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarošík, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pyšek, P. Ecological impacts of invasive alien plants: A meta-analysis of their effects on species.; communities and ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Oduor, A.M.O.; Zhang, Z.; Manea, A.; Tooth, I.M.; Leishman, M.R.; Xu, X.L.; van Kleunen, M. Do invasive alien plants benefit more from global environmental change than native plants? Glob. Chang. Biol. 2017, 23, 3363–3370. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Li, B.; Wu, J.H.; Hu, S.J. Invasive plants differentially affect soil biota through litter and rhizosphere pathways: A meta-analysis. Ecol. Lett. 2019, 22, 200–210. [Google Scholar] [CrossRef]
- Zhou, Y.; Staver, A.C. Enhanced activity of soil nutrient releasing enzymes after plant invasion: A meta-analysis. Ecology 2019, 100, e02830. [Google Scholar] [CrossRef]
- Pei, Y.; Siemann, E.; Tian, B.; Ding, J. Root flavonoids are related to enhanced AMF colonization of an invasive tree. AoB Plants 2020, 12, plaa002. [Google Scholar] [CrossRef]
- Yu, H.; He, Y.; Zhang, W.; Chen, L.; Zhang, J.; Zhang, X.; Dawson, W.; Ding, J. Greater chemical signaling in root exudates enhances soil mutualistic associations in invasive plants compared to natives. New Phytol. 2022, 236, 1140–1153. [Google Scholar] [CrossRef]
- Reinhart, K.O.; Callaway, R.M. Soil biota facilitate exotic Acer invasions in Europe and North America. Ecol. Appl. 2004, 14, 1737–1745. [Google Scholar] [CrossRef]
- Reinhart, K.O.; Callaway, R.M. Soil biota and invasive plants. New Phytol. 2006, 170, 445–457. [Google Scholar] [CrossRef]
- Stinson, K.A.; Campbell, S.A.; Powell, J.R.; Wolfe, B.E.; Callaway, R.M.; Thelen, G.C.; Hallett, S.G.; Prati, D.; Klironomos, J.N. Invasive plant suppresses the growth of native tree seedlings by disrupting belowground mutualisms. PLoS Biol. 2006, 4, 727–731. [Google Scholar] [CrossRef]
- Vogelsang, K.M.; Bever, J.D. Mycorrhizal densities decline in association with nonnative plants and contribute to plant invasion. Ecology 2009, 90, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Eppinga, M.B.; Rietkerk, M.; Dekker, S.C.; de Ruiter, P.C.; van der Putten, W.H. Accumulation of local pathogens: A new hypothesis to explain exotic plant invasions. Oikos 2006, 114, 168–176. [Google Scholar] [CrossRef]
- Mangla, S.; Callaway, R.M. Exotic invasive plant accumulates native soil pathogens which inhibit native plants. J. Ecol. 2008, 96, 58–67. [Google Scholar] [CrossRef]
- Adomako, M.O.; Xue, W.; Tang, M.; Du, D.L.; Yu, F.H. Synergistic effects of soil microbes on Solidago canadensis depend on water and nutrient availability. Microb. Ecol. 2020, 80, 837–845. [Google Scholar] [CrossRef]
- Sveen, T.R.; Netherway, T.; Juhanson, J.; Oja, J.; Borgström, P.; Viketoft, M.; Strengbom, J.; Bommarco, R.; Clemmensen, K.; Hallin, S.; et al. Plant-microbe interactions in response to grassland herbivory and nitrogen eutrophication. Soil Biol. Biochem. 2021, 156, 108208. [Google Scholar] [CrossRef]
- Penning, S.C.; Callaway, R.M. Parasitic plants: Parallels and contrasts with herbivores. Oecologia 2002, 131, 479–489. [Google Scholar] [CrossRef]
- Press, M.C.; Phoenix, G.K. Impacts of parasitic plants on natural communities. New Phytol. 2005, 166, 737–751. [Google Scholar] [CrossRef]
- Li, J.M.; Jin, Z.X.; Song, W.J. Do native parasitic plants cause more damage to exotic invasive hosts than native non-invasive hosts? An implication for biocontrol. PLoS ONE 2012, 7, e34577. [Google Scholar] [CrossRef][Green Version]
- Yu, H.; Yu, F.H.; Miao, S.L.; Dong, M. Holoparasitic Cuscuta campestris suppresses invasive Mikania micrantha and contributes to native community recovery. Biol. Conserv. 2008, 141, 2653–2661. [Google Scholar] [CrossRef]
- Bardgett, R.D.; Smith, R.S.; Shiel, R.S.; Peacock, S.; Simkin, J.M.; Quirk, H.; Hobbs, P.J. Parasitic plants indirectly regulate below-ground properties in grassland ecosystems. Nature 2006, 439, 969–972. [Google Scholar] [CrossRef]
- Li, J.M.; Jin, Z.X.; Hagedorn, F.; Li, M.H. Short-term parasite-infection alters already the biomass, activity and function diversity of soil microbial communities. Sci. Rep. 2014, 4, 6895–6902. [Google Scholar] [CrossRef] [PubMed]
- Brunel, C.; Yang, B.F.; Pouteau, R.; Li, J.M.; van Kleunen, M. Responses of rhizospheric microbial communities of native and alien plant species to Cuscuta parasitism. Microb. Ecol. 2020, 79, 617–630. [Google Scholar] [CrossRef]
- Li, J.M.; Oduor, A.M.O.; Yu, F.H.; Dong, M. A native parasitic plant and soil microorganisms facilitate a native plant co-occurrence with an invasive plant. Ecol. Evol. 2019, 9, 8652–8663. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.H.; Li, J.; Cao, H.L.; Ge, X.J. Genetic uniformity of Alternanthera philoxeroides in South China. Weed Res. 2003, 43, 297–302. [Google Scholar] [CrossRef]
- Yin, J.L.; Hou, L.; Jiang, X.C.; Yang, J.; He, Y.; Zhou, X.K.; Zhu, X.M.; Gong, A.D.; Zhu, Y.X.; Chen, Z.Y. Identification and validation of reference genes for quantitative real-time PCR studies in alligatorweed (Alternanthera philoxeroides). Weed Sci. 2021, 69, 404–411. [Google Scholar] [CrossRef]
- Yang, B.F.; Zhang, X.; Zagorchev, L.; Li, J.M.; Frey, B.; Li, M.H. Parasitism changes rhizospheric soil microbial communities of invasive Alternanthera philoxeroides, benefitting the growth of neighboring plants. Appl. Soil Ecol. 2019, 143, 1–9. [Google Scholar] [CrossRef]
- Dong, B.C.; Alpert, P.; Guo, W.; Yu, F.H. Effects of fragmentation on the survival and growth of the invasive, clonal plant Alternanthera philoxeroides. Biol. Invasions 2012, 14, 1101–1110. [Google Scholar] [CrossRef]
- Arriola, L.; Niemira, B.A.; Safir, G.R. Border cells and arbuscular mycorrhizae in four Amaranthaceae species. Phytopathology 1997, 87, 1240–1242. [Google Scholar] [CrossRef][Green Version]
- Sainty, G.; McCorkelle, G.; Julien, M. Control and spread of alligator weed Alternanthera philoxeroides (Mart.) Griseb., in Australia: Lessons for other regions. Wetl. Ecol. Manag. 1997, 5, 195–201. [Google Scholar] [CrossRef]
- Holm, L.G.; Weldon, L.W.; Blackburn, R.D. Aquatic weeds. Science 1969, 166, 699–709. [Google Scholar] [CrossRef]
- Fang, R.C.; Lytton, J.M.; Uzi, P. Cuscuta Linnaeus. In Flora of China; Wu, Z.Y., Raven, P.H., Hong, D.Y., Eds.; Science Press: Beijing, China; Missouri Botanical Garden Press: St. Louis, MO, USA, 1995; Volume 16, pp. 322–325. [Google Scholar]
- Lau, J.A.; Lennon, J.T. Evolutionary ecology of plant-microbe interactions: Soil microbial structure alters selection on plant traits. New Phytol. 2011, 192, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Borda, V.; Longo, S.; Marro, N.; Urcelay, C. The global invader Ligustrum lucidum accumulates beneficial arbuscular mycorrhizal fungi in a novel range. Plant Ecol. 2021, 222, 397–408. [Google Scholar] [CrossRef]
- Zhang, W.T.; Dong, W. SPSS Statistical Analysis Advanced Course; Higher Education Press: Beijing, China, 2004; pp. 42–43. [Google Scholar]
- Parepa, M.; Schaffner, U.; Bossdorf, O. Help from under ground: Soil biota facilitate knotweed invasion. Ecosphere 2013, 4, 31. [Google Scholar] [CrossRef]
- Callaway, R.M.; Cipollini, D.; Barto, K.; Thelen, G.C.; Hallett, S.G.; Prati, D.; Stinson, K.; Klironomos, J. Novel weapons: Invasive plant suppresses fungal mutualists in America but not in its native Europe. Ecology 2008, 89, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Bais, H.P.; Vepachedu, R.; Gilroy, S.; Callaway, R.M.; Vivanco, J.M. Allelopathy and exotic plant invasion: From molecules and genes to species interactions. Science 2003, 301, 1377–1380. [Google Scholar] [CrossRef]
- Beckstead, J.; Parker, I.M. Invasiveness of Ammophila arenaria: Release from soil-borne pathogens? Ecology 2003, 84, 2824–2831. [Google Scholar] [CrossRef]
- Whipps, J.M. Microbial interactions and biocontrol in the rhizosphere. J. Exp. Bot. 2001, 52, 487–511. [Google Scholar] [CrossRef]
- Gao, F.L.; Alpert, P.; Yu, F.H. Parasitism induces negative effects of physiological integration in a clonal plant. New Phytol. 2021, 229, 585–592. [Google Scholar] [CrossRef]
- Mishev, K.; Dobrev, P.I.; Lacek, J.; Filepová, R.; Yuperlieva-Mateeva, B.; Kostadinova, A.; Hristeva, T. Hormonomic changes driving the negative impact of broomrape on plant host interactions with arbuscular mycorrhizal fungi. Int. J. Mol. Sci. 2021, 22, 13677. [Google Scholar] [CrossRef]
- Beare, M.H.; Neely, C.L.; Coleman, D.C.; Hargrove, W.L. A substrate induced respiration (SIR) method for measurement of fungal and bacterial biomass on plant residues. Soil Biol. Biochem. 1990, 22, 585–594. [Google Scholar] [CrossRef]
- Quested, H.M.; Cornelissen, J.H.C.; Press, M.C.; Callaghan, T.V.; Aerts, R.; Trosien, F.; Riemann, P.; Gwynn-Jones, D.; Kondratchuk, A.; Jonasson, S.E. Decomposition of sub-arctic plants with differing nitrogen economies: A functional role for hemiparasites. Ecology 2003, 84, 3209–3221. [Google Scholar] [CrossRef]
- Wise, M.J.; Abrahamson, W.G. Effects of resource availability on tolerance of herbivory: A review and assessment of three opposing models. Am. Nat. 2007, 169, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Matzek, V. Superior performance and nutrient-use efficiency of invasive plants over non-invasive congeners in a resource limited environment. Biol. Invasions 2011, 13, 3005–3014. [Google Scholar] [CrossRef]

| Variables | Source of Variation | d.f. | F Value | p Value |
|---|---|---|---|---|
| Total biomass | P | 1 | 0.045 | 0.512 |
| M | 1 | 0.000 | 0.998 | |
| P × M | 1 | 0.024 | 0.879 | |
| Aboveground biomass | P | 1 | 4.315 | 0.054 |
| M | 1 | 0.731 | 0.405 | |
| P × M | 1 | 0.468 | 0.504 | |
| Belowground biomass | P | 1 | 6.507 | 0.021 |
| M | 1 | 0.257 | 0.619 | |
| P × M | 1 | 0.577 | 0.459 | |
| Leaf biomass | P | 1 | 12.707 | 0.003 |
| M | 1 | 3.116 | 0.097 | |
| P × M | 1 | 4.478 | 0.050 | |
| Stem biomass | P | 1 | 10.875 | 0.005 |
| M | 1 | 4.873 | 0.042 | |
| P × M | 1 | 4.159 | 0.058 | |
| Root: shoot biomass | P | 1 | 0.167 | 0.688 |
| M | 1 | 2.795 | 0.114 | |
| P × M | 1 | 0.702 | 0.414 | |
| Ramets number | P | 1 | 26.817 | 0.000 |
| M | 1 | 3.292 | 0.088 | |
| P × M | 1 | 4.898 | 0.042 | |
| Stolon length | P | 1 | 11.498 | 0.004 |
| M | 1 | 1.560 | 0.230 | |
| P × M | 1 | 4.057 | 0.061 | |
| Internode length | P | 1 | 31.464 | 0.000 |
| M | 1 | 3.009 | 0.102 | |
| P × M | 1 | 7.363 | 0.015 | |
| Nodes number | P | 1 | 8.529 | 0.010 |
| M | 1 | 0.268 | 0.612 | |
| P × M | 1 | 5.774 | 0.029 | |
| Leaf number | P | 1 | 16.586 | 0.001 |
| M | 1 | 0.240 | 0.631 | |
| P × M | 1 | 4.403 | 0.052 | |
| Stem diameter | P | 1 | 6.694 | 0.020 |
| M | 1 | 3.915 | 0.065 | |
| P × M | 1 | 0.650 | 0.432 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, C.; Zhao, Y.; Yuan, Y.; Li, J. Parasitism Shifts the Effects of Native Soil Microbes on the Growth of the Invasive Plant Alternanthera philoxeroides. Life 2023, 13, 150. https://doi.org/10.3390/life13010150
Cai C, Zhao Y, Yuan Y, Li J. Parasitism Shifts the Effects of Native Soil Microbes on the Growth of the Invasive Plant Alternanthera philoxeroides. Life. 2023; 13(1):150. https://doi.org/10.3390/life13010150
Chicago/Turabian StyleCai, Chaonan, Yingying Zhao, Yongge Yuan, and Junmin Li. 2023. "Parasitism Shifts the Effects of Native Soil Microbes on the Growth of the Invasive Plant Alternanthera philoxeroides" Life 13, no. 1: 150. https://doi.org/10.3390/life13010150
APA StyleCai, C., Zhao, Y., Yuan, Y., & Li, J. (2023). Parasitism Shifts the Effects of Native Soil Microbes on the Growth of the Invasive Plant Alternanthera philoxeroides. Life, 13(1), 150. https://doi.org/10.3390/life13010150







