Effect of Smut Infection on the Photosynthetic Physiological Characteristics and Related Defense Enzymes of Sugarcane
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials, Growth Conditions, and Treatments
2.2. Measurement of Leaf Gas Exchange and Chlorophyll Fluorescence
2.3. Enzyme Extraction
2.3.1. Determination of Peroxidase and Superoxide Dismutase Activity
2.3.2. Quantification of Polyphenol Oxidase and Phenylalanine Ammonia-Lyase Activity
2.4. Data Processing
3. Results
3.1. Effect of Smut Infection on Photosynthetic Leaf Gas Exchange
3.2. Effect of Smut Infection on Chlorophyll Fluorescence Variables
3.3. Changes in Photochemical Quenching Coefficient and Non-Photochemical Quenching Coefficient
3.4. Correlation Coefficient between Photosynthesis, Stomatal Conductance, Intercellular CO2 Concentration, Transpiration Rate, and Water-Use Efficiency
3.5. Correlation Coefficient between Leaf Photosynthetic Parameters and Chlorophyll Fluorescence Variables
3.6. Effects of Smut Infection on Enzymatic Activities in Sugarcane Plants
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Su, Y.; Wang, Z.; Xu, L.; Peng, Q.; Liu, F.; Li, Z.; Que, Y. Early selection for smut resistance in sugarcane using pathogen proliferation and changes in physiological and biochemical indices. Front. Plant Sci. 2016, 7, e84426. [Google Scholar] [CrossRef] [Green Version]
- Verma, K.K.; Song, X.P.; Yadav, G.; Degu, H.D.; Parvaiz, A.; Singh, M.; Huang, H.H.; Mustafa, G.; Xu, L.; Li, Y.R. Impact of agroclimatic variables on proteogenomics in sugar cane (Saccharum spp.) plant productivity. ACS Omega 2022, 7, 22997–23008. [Google Scholar] [CrossRef] [PubMed]
- Que, Y.; Su, Y.; Guo, J.; Wu, Q.; Xu, L. A global view of transcriptome dynamics during Sporisorium scitamineum challenge in sugarcane by RNA-seq. PLoS ONE 2014, 9, e106476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, B. The occurrence status and the research progress of sugarcane disease in China. Sugar Crops China 2007, 3, 48–51. [Google Scholar]
- Rajput, M.A.; Rajput, N.A.; Syed, R.N.; Lodhi, A.M.; Que, Y. Sugarcane smut: Current knowledge and the way forward for management. J. Fungi 2021, 7, 1095. [Google Scholar] [CrossRef]
- Sun, G.; Wang, W. Effects of leaf mosaic infection on photosynthesis and transpiration of papaya leaves. Acta Phytophylacica Sin. 1985, 15, 230–234. [Google Scholar]
- Pinon, D.; de Armas, R.; Vicente, C.; Legaz, M.E. Role of polyamines in the infection of sugarcane buds by Ustilago scitaminea spores. Plant Physiol. Biochem. 1999, 37, 57–64. [Google Scholar] [CrossRef]
- De Armas, R.; Santiago, R.; Legaz, M.-E.; Vicente, C. Levels of phenolic compounds and enzyme activity can be used to screen for resistance of sugarcane to smut (Ustilago scitaminea). Australas Plant Path. 2007, 36, 32–38. [Google Scholar] [CrossRef]
- Kim, Y.C.; Kim, S.Y.; Choi, D.; Ryu, C.-M.; Park, J.M. Molecular characterization of a pepper C2 domain-containing SRC2 protein implicated in resistance against host and non-host pathogens and abiotic stresses. Planta 2008, 227, 1169–1179. [Google Scholar] [CrossRef]
- Torres, M.A.; Jones, J.D.; Dangl, J.L. Reactive oxygen species signaling in response to pathogens. Plant Physiol. 2006, 141, 373–378. [Google Scholar] [CrossRef] [Green Version]
- Wawrzynska, A.; Christiansen, K.M.; Lan, Y.; Rodibaugh, N.L.; Innes, R.W. Powdery mildew resistance conferred by loss of the enhanced disease resistance1 protein kinase is suppressed by a missense mutation in keep on going, a regulator of abscisic acid signaling. Plant Physiol. 2008, 148, 1510–1522. [Google Scholar] [CrossRef] [Green Version]
- Hao, L.; Wang, L.; Ma, C.; Yan, Z.F.; Wang, J.B. Research advances in interaction between pathogenic fungi and plant host. J. Hebei Agric. 2001, 5, 73–77. [Google Scholar]
- Girousse, C.; Faucher, M.; Kleinpeter, C.; Bonnemain, J.L. Dissection of the effects of the pea aphid Acyrthosiphon pisum feeding on assimilate partitioning in Medicago sativa. New Phytol. 2003, 157, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Li, C.X.; Li, P.; Su, Y.F.; Zheng, P.Q.; Zhang, F.Q.; Zhang, Y. Effect of salicylic acid on permeability of plasma membrane and activities of protect enzymes of maize (Zea mays L.) seedlings under cadmium stress. Plant Physiol Comm. 2006, 45, 882–884. [Google Scholar]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-sepecific peroxidase in spinach choloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I occurrence in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shao, X. Characterisation of polyphenol oxidase and peroxidase and the role in browning of loquat fruit. Czech J. Food Sci. 2016, 33, 109–117. [Google Scholar] [CrossRef] [Green Version]
- Aoki, S.; Araki, C.; Kaneko, K.; Katayama, O. Occurrence of L-phenylalanine ammonia-lyase activity in peach fruit during growth. Agric. Biol. Chem. 1971, 35, 784–787. [Google Scholar] [CrossRef]
- Song, X.P.; Verma, K.K.; Tian, D.D.; Zhang, X.Q.; Liang, Y.J.; Huang, X.; Li, C.L.; Li, Y.R. Exploration of silicon functions to integrate with biotic stress tolerance and crop improvement. Biol. Res. 2021, 54, 19. [Google Scholar] [CrossRef] [PubMed]
- Lal, A.; Ku, M.; Edwards, G.E. Analysis of inhibition of photosynthesis due to water stress in the C3 species Hordeum vulgare and Vicia faba: Electron transport, CO2 fixation and carboxylation capacity. Photosynth. Res. 1996, 49, 57–69. [Google Scholar] [CrossRef]
- Yu, W.Y.; Pan, T.G.; Ke, Y.Q. Studies on photosynthesis of sweet potato under the stress of sweet potato scab. Chin. J. Eco-Agric. 2006, 14, 161–164. [Google Scholar]
- Schnettger, B.; Critchley, C.; Santore, U.J.; Graf, M.; Krause, G.H. Relationship between photoinhibition of photosynthesis, D1 protein turnover and chloroplast structure: Effects of protein synthesis inhibitors. Plant Cell Environ. 1994, 17, 55–64. [Google Scholar] [CrossRef]
- Song, X.P.; Mo, F.; Verma, K.K.; Wei, J.; Zhang, X.; Yang, L.; Li, Y.R. Effect of sugarcane smut (Ustilago scitaminea Syd.) on ultrastructure and biochemical indices of sugarcane. Biomed. J. Sci. Tech. Res. 2019, 17, 12546–12551. [Google Scholar]
- Wang, K.; Xu, C. The effects of water stress on some in vivo chlorophyll: A fluorescence parameters of wheat flag leaves. Acta Biophys. Sin. 1997, 13, 273–278. [Google Scholar]
- Fu, D.Y.; Hong, J.; Chen, J.S.; Wu, J.X. Accumulation of coat protein of turnip mosaic virus in host chloroplasts and its effect on PSII activity. J. Plant Physiol. Mol. Biol. 2004, 30, 34–40. [Google Scholar]
- Feng, Y.; Zhang, Y.; Zhu, C. Relationship between photo-inhibition of photosynthesis and reactive oxygen species in leaves of poplars suffering root osmotic stress. Chin. J. Appl. Ecol. 2003, 14, 1213–1217. [Google Scholar]
- Tao, Z.Y.; Zou, Q. Roles of mehler reaction in dissipating excess light energy in soybean leaves. Acta Phytophysiol. Sin. 2001, 27, 66–72. [Google Scholar]
- Zou, Q.; Xu, C.; Zhao, S.; Meng, Q. The role of SOD in protecting the photosynthetic apparatus of soybean leaves from midday high light stress. Acta Phytophysiol. Sin. 1995, 21, 397–401. [Google Scholar]
- Chen, H.X.; Gao, H.Y.; An, S.Z.; Li, W.J. Dissipation of excess energy in Mehler-peroxidase reactioin in Rumex leaves during salt shock. Photosynthetica 2004, 42, 117–122. [Google Scholar] [CrossRef]
- Mehler, A.H. Studies on reactions of illuminated chloroplasts. I. Mechanism of the reduction of oxygen and other Hill reagents. Arch. Biochem. Biophys. 1951, 33, 65–77. [Google Scholar] [CrossRef]
- Song, R.; Ding, Y.; Gong, C.; Xu, G.; Han, X. Research advances in relationship between tobacco resistance and protective enzymes activity. Chin. Agric. Sci. Bull. 2007, 23, 309–314. [Google Scholar]
- Wang, M.L.; Hu, Z.L.; Zhou, M.Q.; Song, Y.C. Advances in research of polyphenol oxidase in plants. Chin. Bull. Bot. 2005, 22, 215–222. [Google Scholar]
- Trebst, A.; Dep, K.B. Polyphenol oxidase and photosynthesis research. Photosynth. Res. 1995, 46, 414–432. [Google Scholar] [CrossRef]
- Huang, M.; Peng, S. Progress on polyphenol oxidases in plants. J. Guangxi Norm. Univ. 1998, 16, 66–70. [Google Scholar]
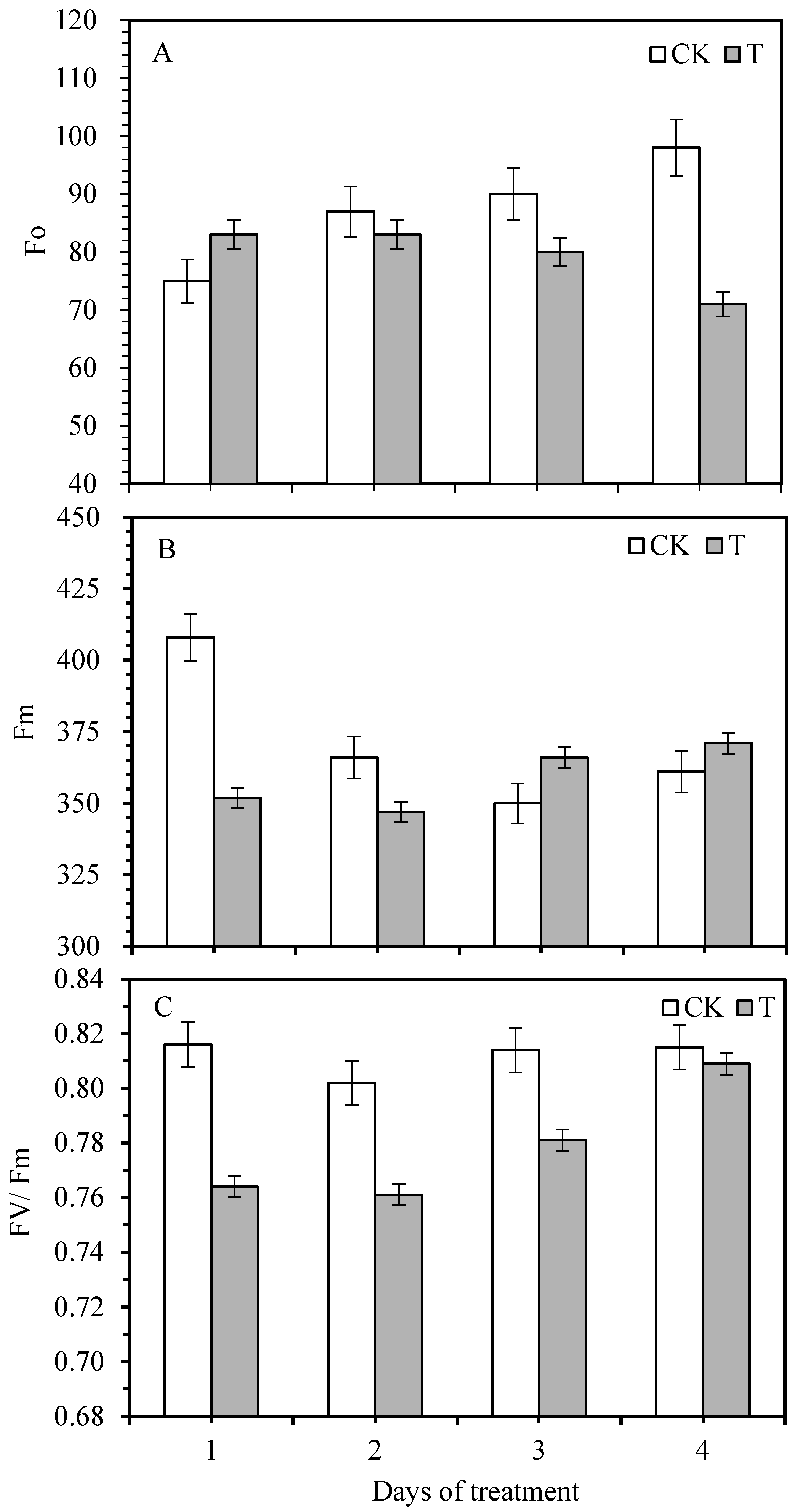
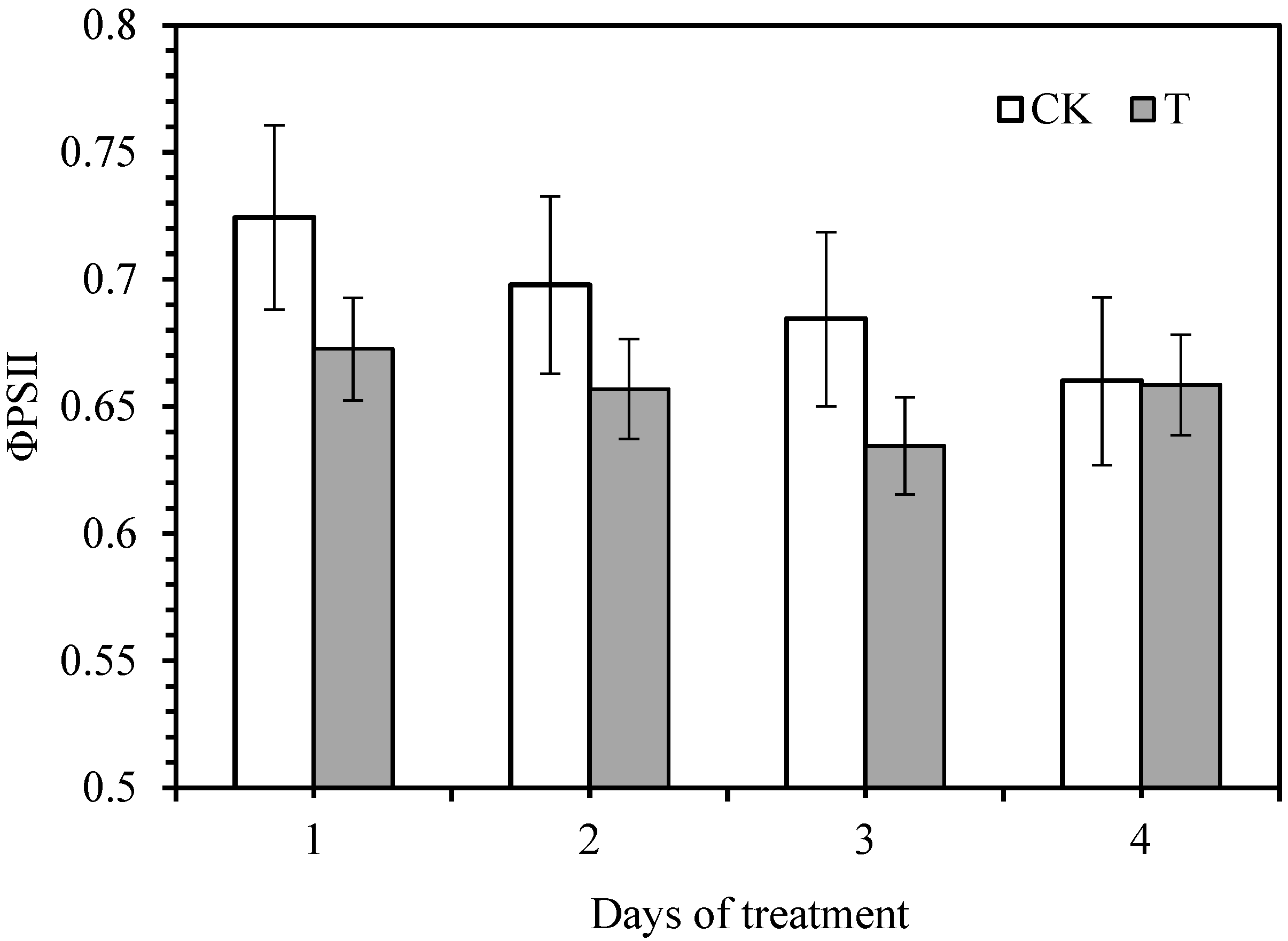
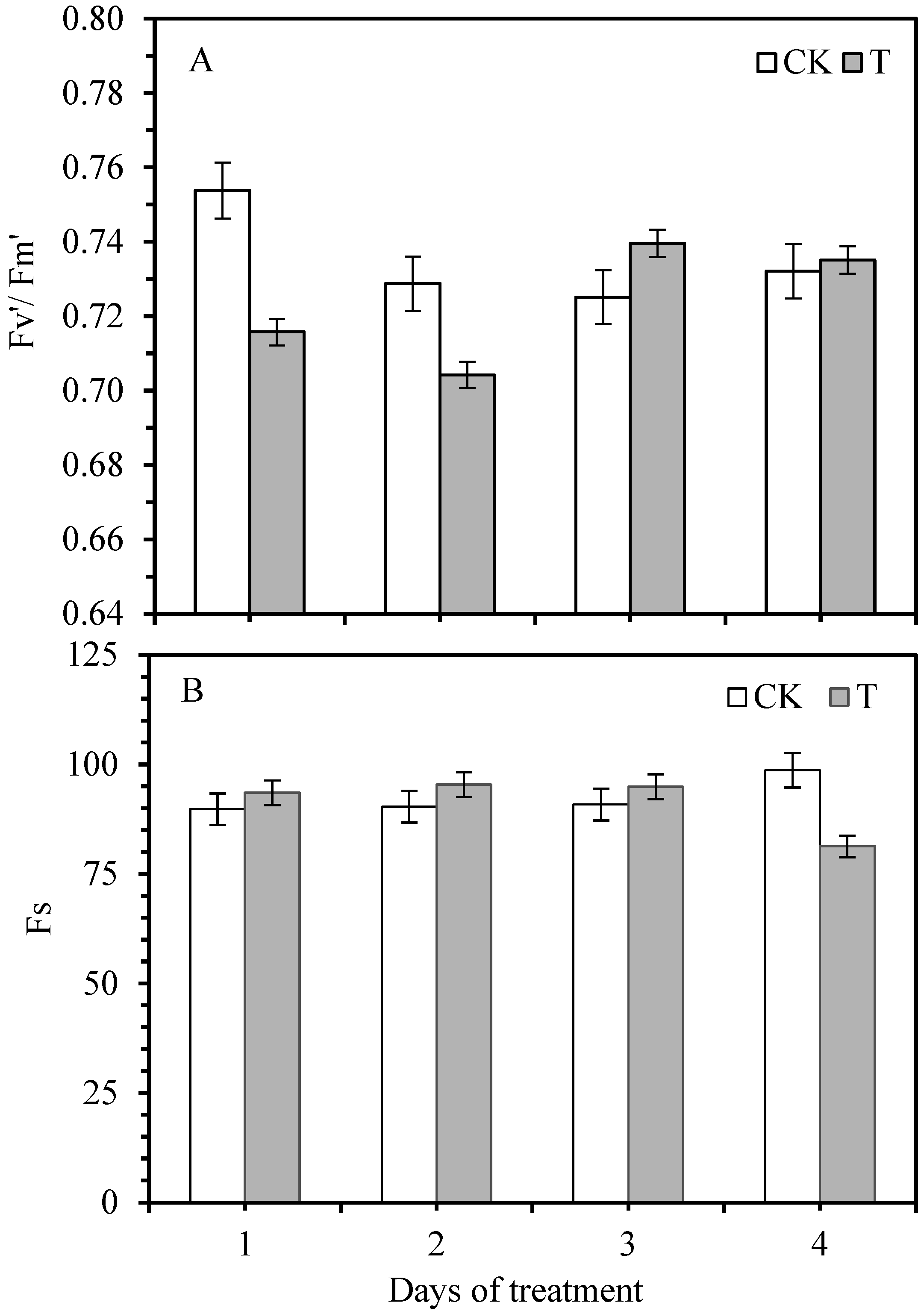

| Photosynthetic Response | Treatment Condition | Days of Smut Inoculation | S | R | |||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||||
| Pn (μmol m−2 s−1) | CK | 14.9 ± 0.72 a | 18.9 ± 0.10 a | 15.4 ± 0.35 a | 19.6 ± 0.62 b | 2.331 | 0.978 |
| T | 15.9 ± 0.74 a | 17.7 ± 0.18 b | 10.7 ± 1.23 b | 25.2 ± 0.92 a | 7.233 | 0.837 | |
| gs (mol m−2 s−1) | CK | 0.077 ± 0.005 a | 0.133 ± 0.002 a | 0.081 ± 0.003 a | 0.128 ± 0.010 a | 0.031 | 0.912 |
| T | 0.092 ± 0.008 a | 0.127 ± 0.004 a | 0.078 ± 0.006 a | 0.130 ± 0.029 a | 0.030 | 0.917 | |
| Ci (µmol mol−1) | CK | 54.93 ± 2.93 a | 108.33 ± 0.67 a | 55.27 ± 8.03 b | 100.53 ± 1.85 a | 31.063 | 0.863 |
| T | 72.23 ± 11.15 a | 113.33 ± 3.84 a | 131.67 ± 7.69 a | 97.06 ± 7.55 a | 20.155 | 0.960 | |
| Tr (mmol m−2 s−1) | CK | 2.410 ± 0.153 a | 3.610 ± 0.063 a | 2.523 ± 0.121 a | 3.397 ± 0.271 a | 0.623 | 0.952 |
| T | 2.950 ± 1.182 a | 3.497 ± 0.029 a | 2.367 ± 0.152 a | 3.607 ± 0.711 a | 0.689 | 0.944 | |
| PWUE (μmol CO2 mmol H2O−1) | CK | 6.185 ± 1.021 a | 5.230 ± 0.801 a | 6.104 ± 0.871 a | 5.432 ± 0.832 a | 1.601 | 0.901 |
| T | 5.391 ± 0.924 a | 5.147 ± 1.023 a | 4.472 ± 0.591 a | 5.315 ± 1.201 a | 1.623 | 0.867 | |
| Fluorescence Parameters | Treatment | Days of Smut Inoculation | Loss or Gain (%) | |||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |||
| qP | CK | 0.959 ± 0.003 a | 0.949 ± 0.003 a | 0.903 ± 0.007 b | 0.923 ± 0.008 b | −0.04 |
| T | 0.948 ± 0.004 b | 0.930 ± 0.004 b | 0.956 ± 0.003 a | 0.948 ± 0.002 a | −0.01 | |
| qNP | CK | 0.253 ± 0.017 a | 0.230 ± 0.019 b | 0.321 ± 0.021 a | 0.137 ± 0.020 b | −0.46 |
| T | 0.245 ± 0.011 a | 0.286 ± 0.011 a | 0.167 ± 0.009 b | 0.278 ± 0.017 a | 0.13 | |
| Variable | Pn | gs | Ci | Tr | WUE |
|---|---|---|---|---|---|
| Pn | 1.000 | ||||
| gs | 0.926 * | 1.000 | |||
| Ci | −0.461 ** | −0.093 | 1.000 | ||
| Tr | 0.984 ** | 0.978 ** | −0.295 | 1.000 | |
| WUE | 0.838 ** | 0.577 ** | −0.864 ** | 0.727 ** | 1.000 |
| Variable | Pn | gs | Ci | Fo | Fv/Fm | Fs | Fv′/Fm′ | ΦPSII | qP | qNP | ETR |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pn | 1.000 | ||||||||||
| gs | 0.995 | 1.000 | |||||||||
| Ci | 0.301 | 0.283 | 1.000 | ||||||||
| Fo | 0.457 | 0.525 | 0.554 | 1.000 | |||||||
| Fv/Fm | −0.484 | −0.537 | −0.693 | −0.984 | 1.000 | ||||||
| Fs | 0.360 | 0.416 | 0.714 | 0.971 | −0.990 | 1.000 | |||||
| Fv′/Fm′ | −0.265 | −0.267 | −0.978 | −0.691 | 0.804 | −0.834 | 1.000 | ||||
| ΦPSII | −0.359 | −0.345 | −0.997 | −0.603 | 0.736 | −0.750 | 0.982 | 1.000 | |||
| qP | −0.757 | −0.698 | −0.662 | −0.181 | 0.317 | −0.241 | 0.533 | 0.677 | 1.000 | ||
| qNP | 0.062 | −0.020 | 0.706 | −0.191 | 0.012 | 0.009 | −0.554 | −0.667 | −0.699 | 1.000 | |
| ETR | 0.114 | 0.211 | 0.091 | 0.846 | −0.744 | 0.756 | −0.290 | −0.137 | 0.352 | −0.634 | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, X.; Mo, F.; Yan, M.; Zhang, X.; Zhang, B.; Huang, X.; Huang, D.; Pan, Y.; Verma, K.K.; Li, Y.-R. Effect of Smut Infection on the Photosynthetic Physiological Characteristics and Related Defense Enzymes of Sugarcane. Life 2022, 12, 1201. https://doi.org/10.3390/life12081201
Song X, Mo F, Yan M, Zhang X, Zhang B, Huang X, Huang D, Pan Y, Verma KK, Li Y-R. Effect of Smut Infection on the Photosynthetic Physiological Characteristics and Related Defense Enzymes of Sugarcane. Life. 2022; 12(8):1201. https://doi.org/10.3390/life12081201
Chicago/Turabian StyleSong, Xiupeng, Fenglian Mo, Meixin Yan, Xiaoqiu Zhang, Baoqing Zhang, Xing Huang, Dongmei Huang, Yangfei Pan, Krishan K. Verma, and Yang-Rui Li. 2022. "Effect of Smut Infection on the Photosynthetic Physiological Characteristics and Related Defense Enzymes of Sugarcane" Life 12, no. 8: 1201. https://doi.org/10.3390/life12081201
APA StyleSong, X., Mo, F., Yan, M., Zhang, X., Zhang, B., Huang, X., Huang, D., Pan, Y., Verma, K. K., & Li, Y.-R. (2022). Effect of Smut Infection on the Photosynthetic Physiological Characteristics and Related Defense Enzymes of Sugarcane. Life, 12(8), 1201. https://doi.org/10.3390/life12081201







