Outcome and Midterm Survival after Heart Transplantation Is Independent from Donor Length of Stay in the Intensive Care Unit
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Recipients and Study Design
2.3. Data Collection
2.4. Statistical Analysis and Figure Making
3. Results
3.1. Recipient Data
3.2. Donor Data
3.3. Perioperative Morbidity
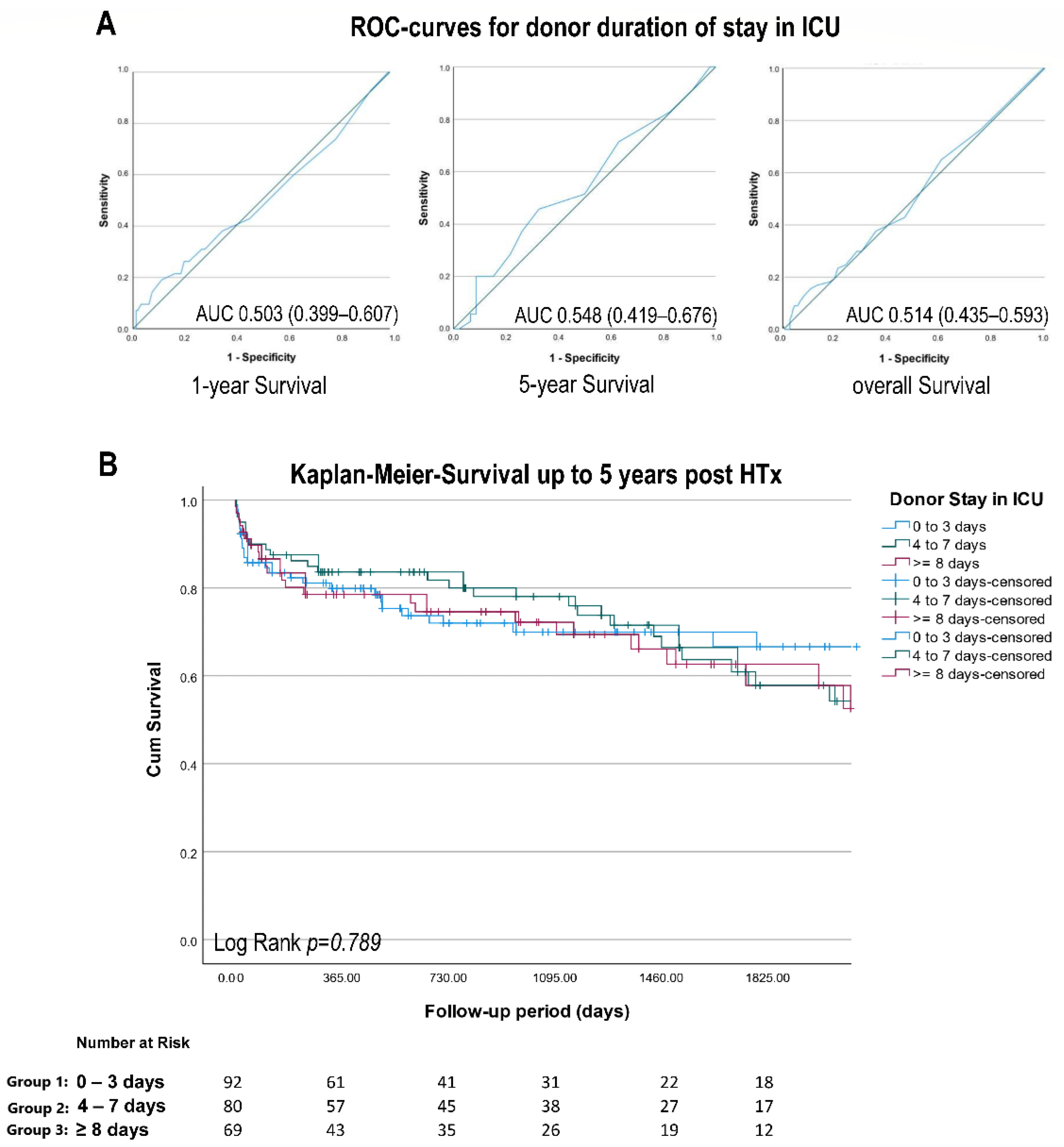
3.4. Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sorabella, R.A.; Guglielmetti, L.; Kantor, A.; Castillero, E.; Takayama, H.; Schulze, P.C.; Mancini, D.; Naka, Y.; George, I. Cardiac Donor Risk Factors Predictive of Short-Term Heart Transplant Recipient Mortality: An Analysis of the United Network for Organ Sharing Database. In Transplantation Proceedings; Elsevier: Amsterdam, The Netherlands, 2015; Volume 47, pp. 2944–2951. [Google Scholar] [CrossRef] [Green Version]
- Murana, G.; Fiorentino, M.; Gliozzi, G.; Di Marco, L.; Potena, L.; Martin Suarez, S.; Pacini, D.; Loforte, A. Donor risk analysis and validation in heart transplants: A single-centre experience. Interact. Cardiovasc. Thorac. Surg. 2020, 31, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Smits, J.M.; De Pauw, M.; de Vries, E.; Rahmel, A.; Meiser, B.; Laufer, G.; Zuckermann, A. Donor scoring system for heart transplantation and the impact on patient survival. J. Heart Lung Transplant. 2012, 31, 387–397. [Google Scholar] [CrossRef]
- Oehler, D.; Immohr, M.B.; Erbel-Khurtsidze, S.; Aubin, H.; Bruno, R.R.; Holst, H.T.; Westenfeld, R.; Horn, P.; Kelm, M.; Tudorache, I.; et al. Intracerebral bleeding in donors is associated with reduced short-term to midterm survival of heart transplant recipients. ESC Heart Fail 2022, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Immohr, M.B.; Akhyari, P.; Bottger, C.; Mehdiani, A.; Dalyanoglu, H.; Westenfeld, R.; Oehler, D.; Tudorache, I.; Aubin, H.; Lichtenberg, A.; et al. Cytomegalovirus mismatch after heart transplantation: Impact of antiviral prophylaxis and intravenous hyperimmune globulin. Immun. Inflamm. Dis. 2021, 9, 1554–1562. [Google Scholar] [CrossRef] [PubMed]
- Immohr, M.B.; Aubin, H.; Westenfeld, R.; Erbel-Khurtsidze, S.; Tudorache, I.; Akhyari, P.; Lichtenberg, A.; Boeken, U. Heart Transplantation of the Elderly-Old Donors for Old Recipients: Can We Still Achieve Acceptable Results? J. Clin. Med. 2022, 11, 929. [Google Scholar] [CrossRef] [PubMed]
- Sugimura, Y.; Immohr, M.B.; Aubin, H.; Mehdiani, A.; Rellecke, P.; Tudorache, I.; Lichtenberg, A.; Boeken, U.; Akhyari, P. Impact of Reported Donor Ejection Fraction on Outcome after Heart Transplantation. Thorac. Cardiovasc. Surg. 2021, 69, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Moitra, V.K.; Guerra, C.; Linde-Zwirble, W.T.; Wunsch, H. Relationship Between ICU Length of Stay and Long-Term Mortality for Elderly ICU Survivors. Crit. Care Med. 2016, 44, 655–662. [Google Scholar] [CrossRef]
- Marshall, D.C.; Hatch, R.A.; Gerry, S.; Young, J.D.; Watkinson, P. Conditional Survival With Increasing Duration of ICU Admission: An Observational Study of Three Intensive Care Databases. Crit. Care Med. 2020, 48, 91–97. [Google Scholar] [CrossRef]
- Akbas, T. Long length of stay in the ICU associates with a high erythrocyte transfusion rate in critically ill patients. J. Int. Med. Res. 2019, 47, 1948–1957. [Google Scholar] [CrossRef] [Green Version]
- Mahesh, B.; Choong, C.K.; Goldsmith, K.; Gerrard, C.; Nashef, S.A.; Vuylsteke, A. Prolonged stay in intensive care unit is a powerful predictor of adverse outcomes after cardiac operations. Ann. Thorac. Surg. 2012, 94, 109–116. [Google Scholar] [CrossRef]
- Nixon, J.L.; Kfoury, A.G.; Brunisholz, K.; Horne, B.D.; Myrick, C.; Miller, D.V.; Budge, D.; Bader, F.; Everitt, M.; Saidi, A.; et al. Impact of high-dose inotropic donor support on early myocardial necrosis and outcomes in cardiac transplantation. Clin. Transplant. 2012, 26, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Verzelloni Sef, A.; Sef, D.; Garcia Saez, D.; Trkulja, V.; Walker, C.; Mitchell, J.; McGovern, I.; Stock, U. Heart Transplantation in Adult Congenital Heart Disease with the Organ Care System Use: A 4-Year Single-Center Experience. ASAIO J. 2021, 67, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Briceno, J.; Marchal, T.; Padillo, J.; Solorzano, G.; Pera, C. Influence of marginal donors on liver preservation injury. Transplantation 2002, 74, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Deschenes, M.; Belle, S.H.; Krom, R.A.; Zetterman, R.K.; Lake, J.R. Early allograft dysfunction after liver transplantation: A definition and predictors of outcome. National Institute of Diabetes and Digestive and Kidney Diseases Liver Transplantation Database. Transplantation 1998, 66, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Cuende, N.; Miranda, B.; Canon, J.F.; Garrido, G.; Matesanz, R. Donor characteristics associated with liver graft survival. Transplantation 2005, 79, 1445–1452. [Google Scholar] [CrossRef] [Green Version]
- Markmann, J.F.; Markmann, J.W.; Markmann, D.A.; Bacquerizo, A.; Singer, J.; Holt, C.D.; Gornbein, J.; Yersiz, H.; Morrissey, M.; Lerner, S.M.; et al. Preoperative factors associated with outcome and their impact on resource use in 1148 consecutive primary liver transplants. Transplantation 2001, 72, 1113–1122. [Google Scholar] [CrossRef]
- Hoofnagle, J.H.; Lombardero, M.; Zetterman, R.K.; Lake, J.; Porayko, M.; Everhart, J.; Belle, S.H.; Detre, K.M. Donor age and outcome of liver transplantation. Hepatology 1996, 24, 89–96. [Google Scholar] [CrossRef]
- Ghobrial, R.M.; Steadman, R.; Gornbein, J.; Lassman, C.; Holt, C.D.; Chen, P.; Farmer, D.G.; Yersiz, H.; Danino, N.; Collisson, E.; et al. A 10-year experience of liver transplantation for hepatitis C: Analysis of factors determining outcome in over 500 patients. Ann. Surg. 2001, 234, 384–393; discussion 393–394. [Google Scholar] [CrossRef]
- Cignarella, A.; Redley, B.; Bucknall, T. Organ donation within the intensive care unit: A retrospective audit. Aust. Crit. Care 2020, 33, 167–174. [Google Scholar] [CrossRef]
- Hunter, A.; Johnson, L.; Coustasse, A. Reduction of intensive care unit length of stay: The case of early mobilization. Health Care Manag. 2014, 33, 128–135. [Google Scholar] [CrossRef]
- Angleitner, P.; Kaider, A.; Gokler, J.; Moayedifar, R.; Osorio-Jaramillo, E.; Zuckermann, A.; Laufer, G.; Aliabadi-Zuckermann, A. High-dose catecholamine donor support and outcomes after heart transplantation. J. Heart Lung Transplant. 2018, 37, 596–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiorelli, A.I.; Branco, J.N.; Dinkhuysen, J.J.; Oliveira Junior, J.L.; Pereira, T.V.; Dinardi, L.F.; Santos, M.M.; Dias, R.R.; Pereira, L.A.; Stolf, N.A. Risk factor analysis of late survival after heart transplantation according to donor profile: A multi-institutional retrospective study of 512 transplants. In Transplantation Proceedings; Elsevier: Amsterdam, The Netherlands, 2012; Volume 44, pp. 2469–2472. [Google Scholar] [CrossRef]
- D’Aragon, F.; Lamontagne, F.; Cook, D.; Dhanani, S.; Keenan, S.; Chasse, M.; English, S.; Burns, K.E.A.; Frenette, A.J.; Ball, I.; et al. Variability in deceased donor care in Canada: A report of the Canada-DONATE cohort study. Can. J. Anaesth 2020, 67, 992–1004. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.; Thomas, M. Cardiac arrest in ICU. J. Intensive Care Soc. 2017, 18, 173. [Google Scholar] [CrossRef] [Green Version]
- Thongprayoon, C.; Cheungpasitporn, W.; Petnak, T.; Miao, J.; Qian, Q. Increased short-term and long-term mortality in community- and hospital-acquired hypernatraemia and in patients with delayed serum sodium correction. Int. J. Clin. Pract. 2021, 75, e14590. [Google Scholar] [CrossRef] [PubMed]
- Boeken, U.; Albert, A.; Mehdiani, A.; Petrov, G.; Westenfeld, R.; Saeed, D.; Akhyari, P.; Lichtenberg, A. Impact of Donor Hypernatremia on Outcome after Cardiac Transplantation. Thorac. Cardiovasc. Surg. 2016, 64, ePP96. [Google Scholar] [CrossRef]
- Hoefer, D.; Ruttmann-Ulmer, E.; Smits, J.M.; Devries, E.; Antretter, H.; Laufer, G. Donor hypo- and hypernatremia are predictors for increased 1-year mortality after cardiac transplantation. Transpl. Int. 2010, 23, 589–593. [Google Scholar] [CrossRef]
- Finger, M.A.; Cipullo, R.; Rossi Neto, J.M.; Dos Santos, C.C.; Contreras, C.A.; Chaccur, P.; Dinkhuysen, J.J.; de Souza, R.; Dias Franca, J.I.; Lin-Wang, H.T. Donor hypernatremia and smoking addiction contribute to primary graft failure in heart transplantation. Clin. Transplant. 2019, 33, e13693. [Google Scholar] [CrossRef]
- Misar, A.; McLin, V.A.; Calinescu, A.M.; Wildhaber, B.E. Impact of length of donor ICU stay on outcome of patients after pediatric liver transplantation with whole and ex situ split liver grafts. Pediatr. Transplant. 2022, 26, e14186. [Google Scholar] [CrossRef]
- Rizvi, S.A.; Luc, J.G.Y.; Choi, J.H.; Phan, K.; Moncho Escriva, E.; Patel, S.; Massey, H.T.; Tchantchaleishvili, V. Outcomes and survival following heart retransplantation for cardiac allograft failure: A systematic review and meta-analysis. Ann. Cardiothorac. Surg. 2018, 7, 12–18. [Google Scholar] [CrossRef] [Green Version]
- Rivinius, R.; Helmschrott, M.; Ruhparwar, A.; Schmack, B.; Darche, F.F.; Thomas, D.; Bruckner, T.; Doesch, A.O.; Katus, H.A.; Ehlermann, P. Elevated pre-transplant pulmonary vascular resistance is associated with early post-transplant atrial fibrillation and mortality. ESC Heart Fail. 2020, 7, 176–187. [Google Scholar] [CrossRef] [Green Version]
| Recipient Variables | All Patients | Gr 1 0–3 Days | Gr 2 4–7 Days | Gr 3 ≥8 Days | p | ||
|---|---|---|---|---|---|---|---|
| n = 241 | Total n = 92 | Total n = 80 | Total n = 69 | 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | |
| Age (y) | 58 (50–62) | 59 (51–62) | 57 (45–63) | 59 (53–63) | 0.18 | 0.47 | 0.06 |
| Gender (% male) | 72.2 | 73.9 | 75.0 | 66.7 | >0.99 | 0.38 | 0.28 |
| Height (cm) | 175 (169–180) | 175 (168–182) | 175 (170–180) | 173 (168–178) | 0.66 | 0.29 | 0.50 |
| Weight (kg) | 78 (68–87) | 80 (69–90) | 79 (70–87) | 74 (65–83) | 0.95 | 0.06 | 0.07 |
| Body mass index (kg/m2) | 25 (23–28) | 26 (23–29) | 26 (23–29) | 24 (21–28) | 0.82 | 0.10 | 0.09 |
| Predicted heart mass ratio (%) | 0.98 (0.87–1.10) | 0.99 (0.89–1.07) | 0.98 (0.84–1.14) | 0.97 (0.88–1.12) | 0.71 | 0.47 | 0.76 |
| Cardiac reoperation (%) | 63.1 | 62.0 | 65.0 | 62.3 | 0.75 | >0.99 | 0.86 |
| High-urgency waiting list (%) | 44.8 | 37.0 | 48.8 | 50.7 | 0.13 | 0.11 | 0.87 |
| Ventricular assist device (%) | 50.6 | 50.0 | 50.0 | 52.2 | >0.99 | 0.87 | 0.87 |
| CPR pre HTx (%) | 7.3 | 13.0 | 10.1 | 15.9 | 0.72 | 0.83 | 0.27 |
| Diabetes mellitus (%) | 22.6 | 17.6 | 24.1 | 27.5 | 0.34 | 0.18 | 0.71 |
| Arterial hypertension (%) | 57.1 | 59.8 | 60.8 | 49.3 | >0.99 | 0.20 | 0.19 |
| ICM (%) | 43.5 | 53.3 | 37.2 | 37.7 | 0.06 | 0.06 | >0.99 |
| Pulmonary hypertension (%) | 9.1 | 12.0 | 8.9 | 5.8 | 0.62 | 0.27 | 0.54 |
| PRA pre HTx (% as mean) | 2.33 (±13.7) | 0.82 (±5.2) | 3.8 (±19.3) | 2.63 (±13.5) | 0.18 | 0.30 | 0.68 |
| PRA >10% pre HTx (n, %) | 7/241 (2.9 | 1/92 (1.1) | 3/80 (3.7) | 3/69 (4.3) | 0.34 | 0.31 | >0.99 |
| Laboratory values | |||||||
| Hemoglobin (g/dL) | 12 (10–14) | 12 (10–14) | 12 (10–14) | 12 (10–13) | 0.85 | 0.78 | 0.93 |
| Creatinine (mg/dL) | 1.2 (1.0–1.6) | 1.2 (1.0–1.6) | 1.1 (1.0–1.5) | 1.2 (0.9–1.7) | 0.47 | 0.37 | 0.87 |
| GFR pre HTx (mL/min) | 62 (45–82) | 63 (47–81) | 65 (48–81) | 60 (39–85) | 0.71 | 0.53 | 0.35 |
| Bilirubin (mg/dL) | 0.6 (0.4–1.0) | 0.6 (0.4–1.0) | 0.6 (0.4–1.1) | 0.6 (0.4–1.0) | 0.37 | 0.48 | 0.86 |
| Lactate dehydrogenase (U/L) | 254 (213–314) | 248 (212–307) | 259 (211–328) | 258 (224–316) | 0.47 | 0.44 | 0.14 |
| Sodium (mmol/L) | 138 (136–141) | 139 (136–141) | 138 (136–140) | 138 (137–141) | 0.37 | 0.66 | 0.20 |
| Potassium (mmol/L) | 4.3 (3.9–4.6) | 4.3 (3.9–4.6) | 4.3 (3.9–4.6) | 4.2 (3.9–4.6) | 0.25 | 0.25 | 0.99 |
| Donor Variables | All Patients | Gr 1 0–3 Days | Gr 2 4–7 Days | Gr 3 ≥8 Days | p | ||
|---|---|---|---|---|---|---|---|
| n = 241 | Total n = 92 | Total n = 80 | Total n = 69 | 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | |
| Age (y) | 46 (35–53) | 43 (32–51) | 46 (33–54) | 46 (38–54) | 0.35 | 0.06 | 0.33 |
| Gender (% male) | 54.8 | 57.6 | 57.5 | 47.8 | >0.99 | 0.26 | 0.25 |
| Height (cm) | 175 (168–180) | 175 (170–183) | 175 (168–180) | 173 (166–180) | 0.43 | 0.09 | 0.35 |
| Weight (kg) | 80 (70–85) | 80 (70–87) | 79 (70–85) | 75 (70–81) | 0.56 | 0.28 | 0.12 |
| Body mass index (kg/m2) | 25 (23–28) | 26 (23–28) | 26 (23–29) | 25 (23–28) | 0.56 | 0.56 | 0.21 |
| Left ventricular ejection fraction (%) | 60 (55–65) | 60 (55–62) | 61 (57–68) | 61 (55–65) | 0.31 | 0.31 | 0.32 |
| CPR pre brain death (%) | 28.6 | 29.3 | 38.8 | 15.9 | 0.20 | 0.06 | 0.003 |
| Donor duration on IMC/ICU (d) | 4 (3–8) | 2 (2–3) | 5 (4–5) | 12 (10–16) | <0.0001 | <0.0001 | <0.0001 |
| Arterial hypertension (%) | 49.6 | 38.9 | 57.9 | 57.1 | 0.09 | 0.13 | >0.99 |
| Diabetes mellitus (%) | 16.5 | 15.9 | 18.2 | 16.0 | >0.99 | >0.99 | >0.99 |
| History of smoking (%) | 62.7 | 65.0 | 65.9 | 56.1 | >0.99 | 0.37 | 0.35 |
| History of alcohol abuse (%) | 41.8 | 42.9 | 49.2 | 33.3 | 0.39 | 0.45 | 0.09 |
| History of drug abuse (%) | 11.8 | 13.7 | 10.2 | 10.9 | 0.54 | 0.79 | >0.99 |
| Norepinephrine, peak dose (µg/kg/min) | 0.12 (0.05–0.26) | 0.15 (0.05–0.28) | 0.11 (0.04–0.26) | 0.12 (0.05–0.24) | 0.79 | 0.16 | 0.33 |
| Donor cause of death | |||||||
| ICB (%) | 46.1 | 50.0 | 28.7 | 60.9 | 0.005 | 0.20 | 0.0001 |
| Trauma (%) | 22.4 | 26.1 | 26.3 | 13.0 | >0.99 | 0.05 | 0.06 |
| Hypoxic (%) | 16.2 | 15.2 | 20.0 | 13.0 | 0.43 | 0.82 | 0.28 |
| Vascular (%) | 6.2 | 3.3 | 10.0 | 5.8 | 0.12 | 0.70 | 0.38 |
| Other (%) | 9.1 | 5.4 | 15.0 | 7.2 | 0.12 | 0.18 | >0.99 |
| Laboratory values | |||||||
| White blood cell count, peak (109/L) | 18 (14–23) | 17 (13–23) | 18 (15–22) | 19 (14–23) | 0.29 | 0.70 | 0.34 |
| C-reactive protein (mg/L) | 163 (63–249) | 118 (24–218) | 185 (103–259) | 187 (100–284) | 0.40 | 0.73 | 0.43 |
| Hemoglobin (g/dL) | 10 (8–12) | 11 (8–14) | 10 (8–12) | 9 (8–10) | 0.12 | 0.00011 | 0.025 |
| Lactate dehydrogenase (U/L) | 323 (238–531) | 300 (234–519) | 323 (248–509) | 333 (242–586) | 0.22 | 0.56 | 0.36 |
| Sodium (mmol/L) | 149 (144–154) | 147 (142–151) | 153 (145–158) | 149 (143–154) | <0.0001 | 0.11 | 0.017 |
| Potassium (mmol/L) | 4 (4–5) | 4 (4–5) | 4 (4–4) | 4 (4–4) | 0.14 | 0.64 | 0.25 |
| Outcome and Survival | All Patients | Gr 1 0–3 Days | Gr 2 4–7 Days | Gr 3 ≥8 Days | p | ||
|---|---|---|---|---|---|---|---|
| n = 241 | Total n = 92 | Total n = 80 | Total n = 69 | 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | |
| Total graft ischemia time (min) | 213 (187–237) | 214 (193–242) | 215 (185–215) | 207 (176–207) | 0.41 | 0.24 | 0.72 |
| Graft cold ischemia time (min) | 149 (127–172) | 151 (134–176) | 147 (119–147) | 148 (121–148) | 0.39 | 0.14 | 0.58 |
| Postoperative hospital stay (d) | 36 (27–51) | 37 (26–48) | 34 (26–34) | 39 (28–39) | 0.32 | 0.41 | 0.08 |
| Postoperative IMC/ICU stay (d) | 16 (10–28) | 17 (10–30) | 15 (10–15) | 16 (10–16) | 0.21 | 0.51 | 0.09 |
| Mechanical ventilation (h) | 66 (25–175) | 92 (28–201) | 54 (26–54) | 51 (21–51) | 0.30 | 0.98 | 0.37 |
| Duration of surgery (min) | 412 (339–505) | 415 (346–491) | 403 (333–403) | 411 (341–411) | 0.74 | 0.68 | 0.49 |
| Blood transfusions surgery | |||||||
| Packed red blood cells, ml | 2970 (1620–4590) | 2970 (1620–4455) | 2430 (1350–2430) | 3375 (1890–3375) | 0.69 | 0.49 | 0.29 |
| Platelets, ml | 880 (660–1540) | 880 (660–1540) | 880 (660–880) | 880 (440–880) | 0.33 | 0.65 | 0.65 |
| Fresh frozen plasma, ml | 1000 (0–2000) | 1250 (500–2125) | 1000 (0–1000) | 1250 (0–1250) | 0.20 | 0.80 | 0.35 |
| Blood transfusions IMC/ICU | |||||||
| Packed red blood cells, ml | 1890 (810–4320) | 1620 (810–4320) | 1890 (540–1890) | 2160 (810–2160) | 0.56 | 0.62 | 0.95 |
| Platelets, ml | 220 (0–1100) | 220 (0–1100) | 220 (0–220) | 220 (0–220) | 0.44 | 0.18 | 0.54 |
| Fresh frozen plasma, ml | 3500 (2000–6750) | 4250 (2250–8000) | 3000 (1750–3000) | 3500 (2000–3500) | 0.76 | 0.25 | 0.54 |
| Postoperative morbidity | |||||||
| Infection/sepsis (%) | 23.2 | 29.7 | 18.8 | 19.7 | 0.11 | 0.12 | >0.99 |
| Rejection within stay (%) | 6.8 | 3.3 | 11.3 | 6.1 | 0.07 | 0.46 | 0.38 |
| Hemodialysis post HTx (%) | 57.4 | 60.7 | 50.6 | 61.3 | 0.21 | >0.99 | 0.23 |
| Neurological complications (%) | 14.8 | 13.2 | 12.5 | 19.7 | >0.99 | 0.38 | 0.26 |
| Re-thoracotomy post HTx (%) | 30.0 | 29.7 | 28.8 | 31.8 | >0.99 | 0.86 | 0.72 |
| ECLS post HTx (%) | 28.7 | 31.9 | 25.0 | 28.8 | 0.40 | 0.73 | 0.71 |
| Survival | |||||||
| 30-day survival n (%) | 216/238 (91%) | 79/91 (87%) | 75/80 (94%) | 62/67 (93%) | 0.20 | 0.31 | >0.99 |
| 1-year survival n (%) | 161/203 (79%) | 61/78 (78%) | 57/69 (82%) | 43/56 (77%) | 0.54 | >0.99 | 0.51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oehler, D.; Böttger, C.; Immohr, M.B.; Bruno, R.R.; Haschemi, J.; Scheiber, D.; Horn, P.; Aubin, H.; Tudorache, I.; Westenfeld, R.; et al. Outcome and Midterm Survival after Heart Transplantation Is Independent from Donor Length of Stay in the Intensive Care Unit. Life 2022, 12, 1053. https://doi.org/10.3390/life12071053
Oehler D, Böttger C, Immohr MB, Bruno RR, Haschemi J, Scheiber D, Horn P, Aubin H, Tudorache I, Westenfeld R, et al. Outcome and Midterm Survival after Heart Transplantation Is Independent from Donor Length of Stay in the Intensive Care Unit. Life. 2022; 12(7):1053. https://doi.org/10.3390/life12071053
Chicago/Turabian StyleOehler, Daniel, Charlotte Böttger, Moritz Benjamin Immohr, Raphael Romano Bruno, Jafer Haschemi, Daniel Scheiber, Patrick Horn, Hug Aubin, Igor Tudorache, Ralf Westenfeld, and et al. 2022. "Outcome and Midterm Survival after Heart Transplantation Is Independent from Donor Length of Stay in the Intensive Care Unit" Life 12, no. 7: 1053. https://doi.org/10.3390/life12071053
APA StyleOehler, D., Böttger, C., Immohr, M. B., Bruno, R. R., Haschemi, J., Scheiber, D., Horn, P., Aubin, H., Tudorache, I., Westenfeld, R., Akhyari, P., Kelm, M., Lichtenberg, A., & Boeken, U. (2022). Outcome and Midterm Survival after Heart Transplantation Is Independent from Donor Length of Stay in the Intensive Care Unit. Life, 12(7), 1053. https://doi.org/10.3390/life12071053








