Tea Tree Oil Nanoemulsion-Based Hydrogel Vehicle for Enhancing Topical Delivery of Neomycin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material
2.2. Designing the Experiment
2.3. Development of NEO-NE
2.4. Characterization of Developed NE
Globule Size and Polydispersity Index (PDI) Determination
2.5. In Vitro Study
2.6. Zeta Potential
2.7. Development of NEO-NE-Based Hydrogel
2.8. Characterizing the Developed NEO-NE-Based Hydrogel
2.8.1. Visual Examination
2.8.2. pH Measurement
2.8.3. Viscosity
2.8.4. Spreadability
2.9. Scanning Electron Microscopy (SEM)
2.10. In Vitro Release of NEO from Different Developed Formulations
2.11. Kinetic Study
2.12. Stability Test
2.13. Animal
2.13.1. Animals
2.13.2. Statement of Animal Ethics
2.13.3. Skin Irritation Test
2.14. Microbiological Study
2.15. Statistics
3. Results
3.1. Model Fitting and Statistical Data Analysis
3.2. Characterization of Developed NEs
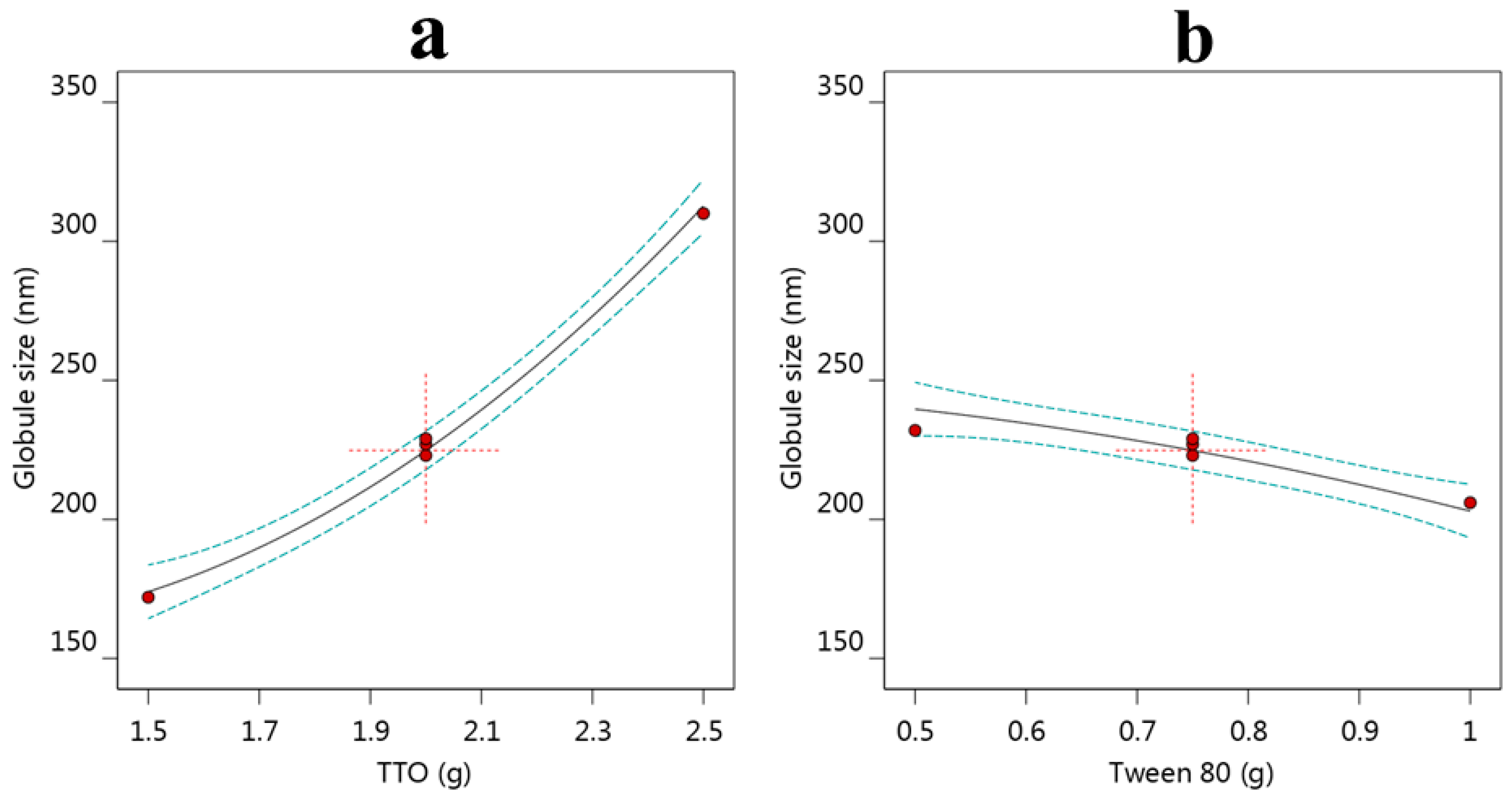
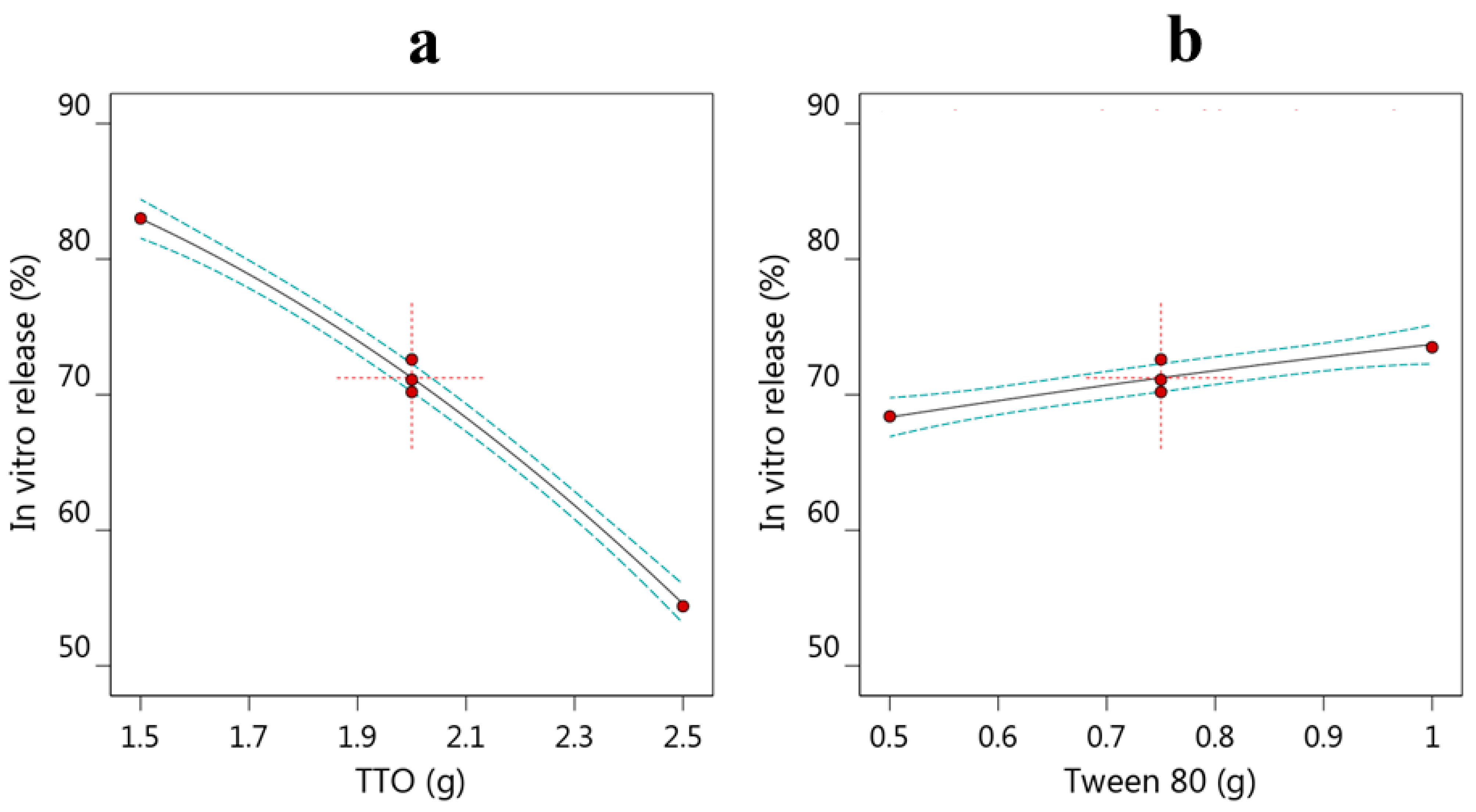
3.2.1. Effect of Variables A and B on Y1
3.2.2. Effect of Variables A and B on Y2
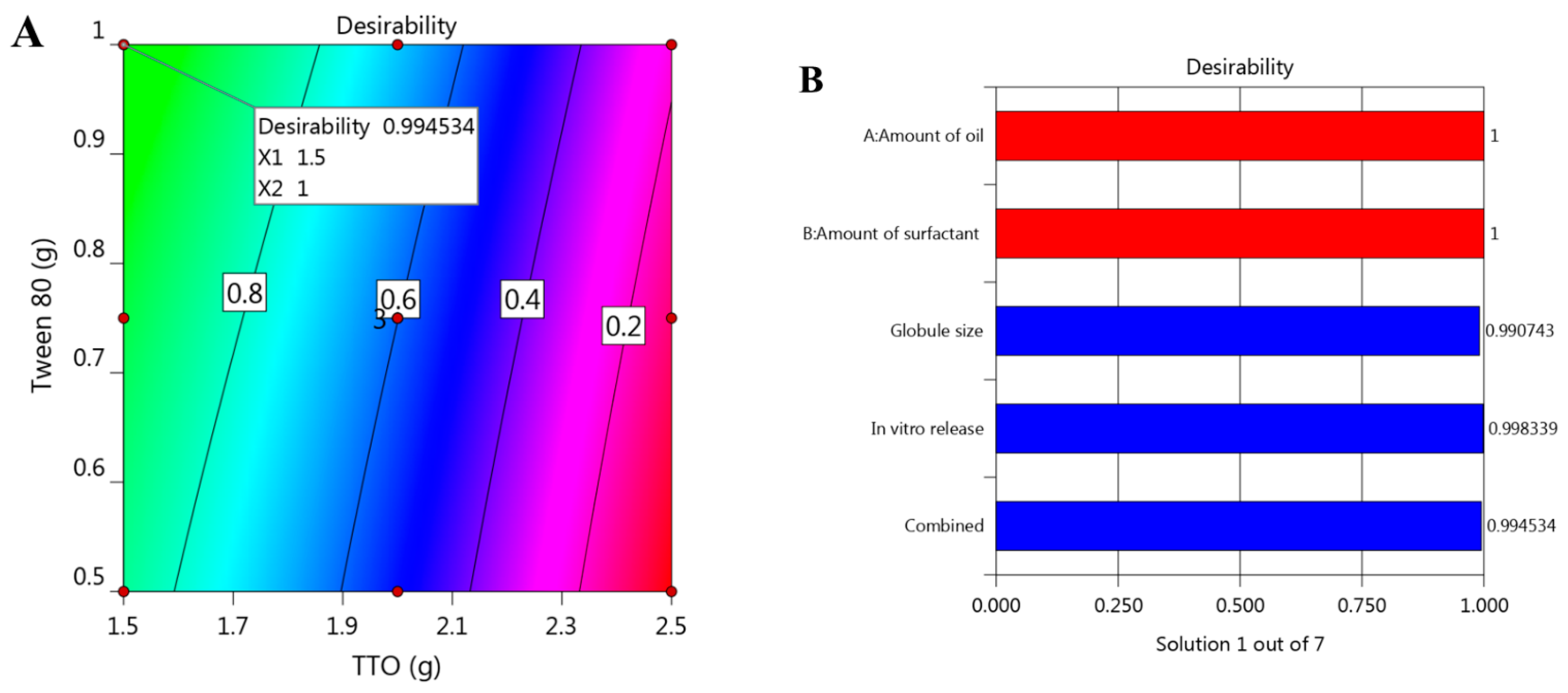
3.3. Optimization and Validation of Variables
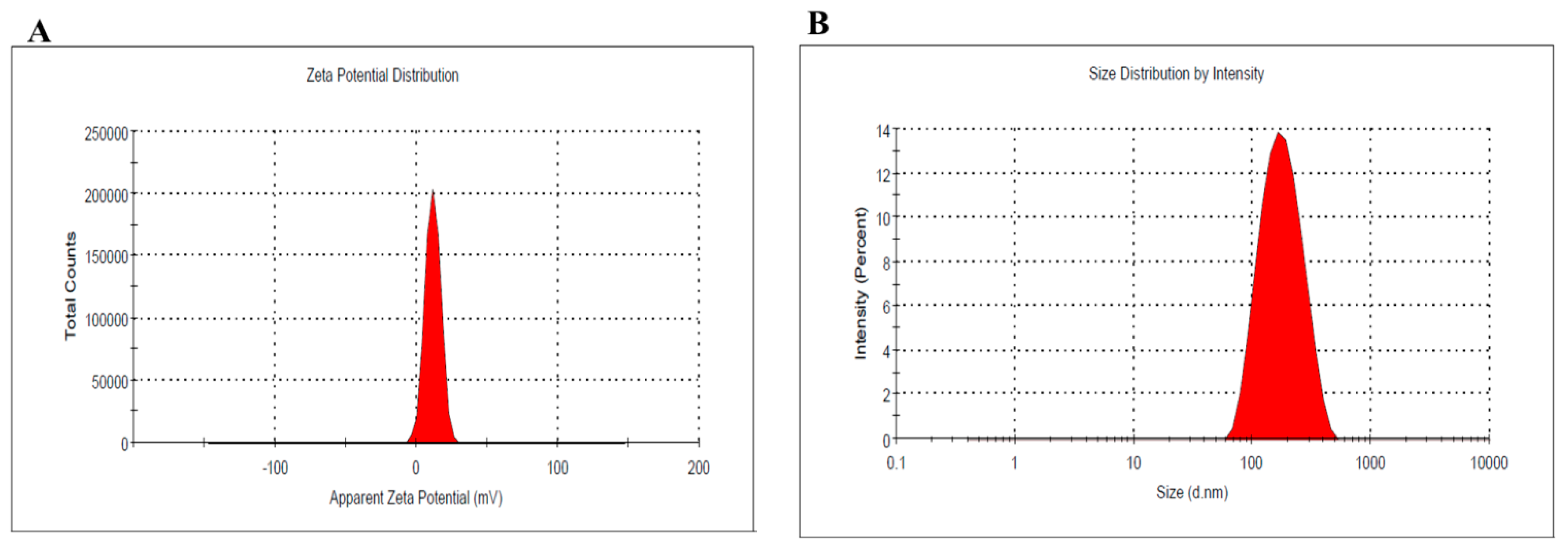
3.4. Zeta Potential
3.5. Characterizing the Developed NEO-NE-Based Hydrogel
3.5.1. Visual Examination
3.5.2. pH Measurement
3.5.3. Viscosity
3.5.4. Spreadability
3.6. SEM
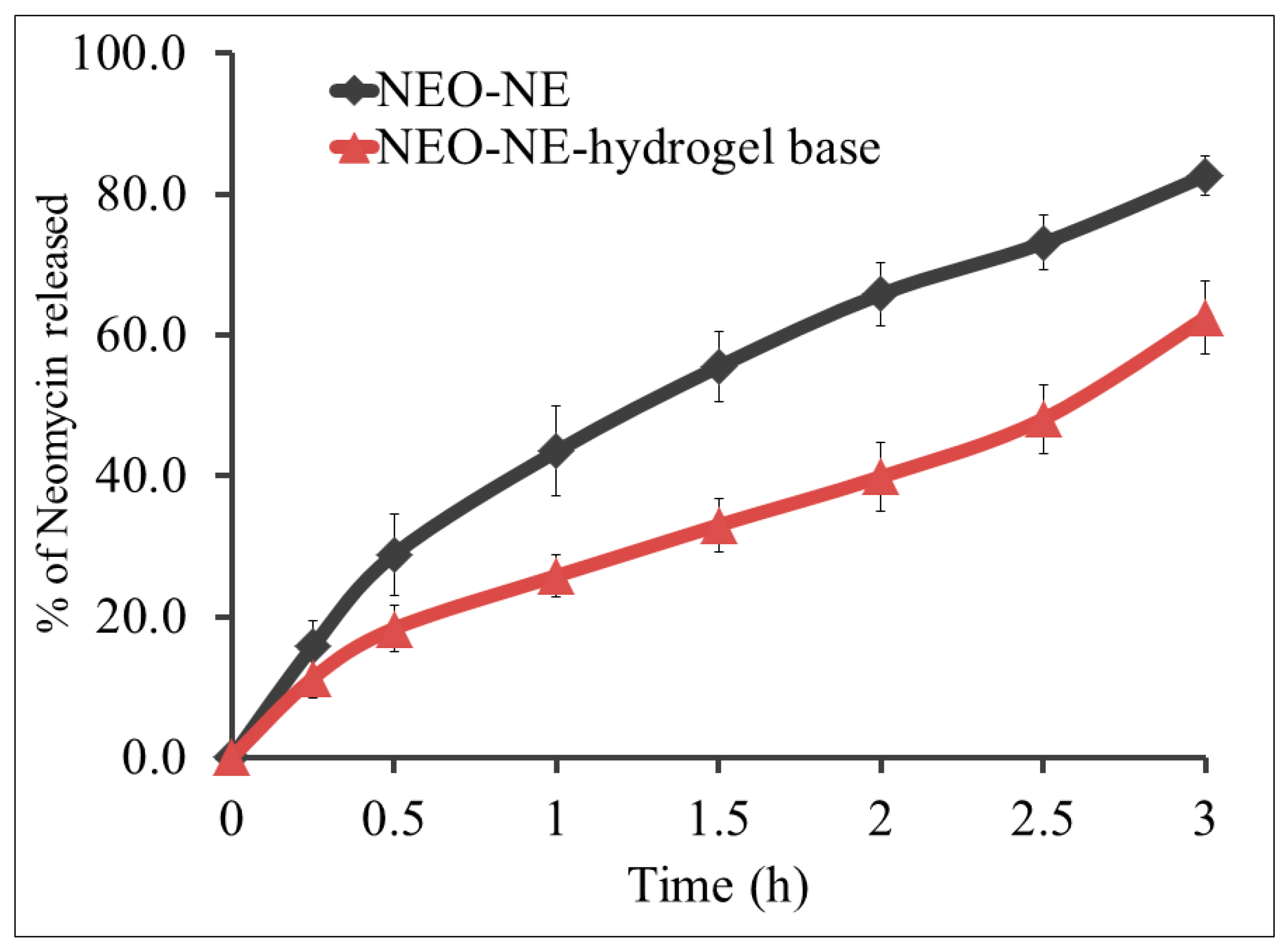
3.7. In Vitro Release of NEO from Different Developed Formulations
3.8. Kinetic Study
3.9. Stability Test
3.10. In Vivo Study
In Vivo Skin Irritation Test
3.11. Microbiological Study
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alkilani, A.Z.; McCrudden, M.T.C.; Donnelly, R.F. Transdermal Drug Delivery: Innovative Pharmaceutical Developments Based on Disruption of the Barrier Properties of the Stratum Corneum. Pharmaceutics 2015, 7, 438–470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benson, H.A.; Grice, J.E.; Mohammed, Y.; Namjoshi, S.; Roberts, M.S. Topical and Transdermal Drug Delivery: From Simple Potions to Smart Technologies. Curr. Drug Deliv. 2019, 16, 444–460. [Google Scholar] [CrossRef] [PubMed]
- Rungseevijitprapa, W.; Yingngam, B.; Chaiyasut, C. Improvement of Biophysical Skin Parameters of Topically Applied Fermented Soybean Extract-Loaded Niosomes with No Systemic Toxicity in Ovariectomized Rats. Pharmaceutics 2021, 13, 1068. [Google Scholar] [CrossRef] [PubMed]
- Boztepe, H.; Ozdemir, H.; Karababa, C.; Yildiz, O. Difficulties experienced during preparation and administration of oral drugs. Turk. Pediatri. Ars. 2014, 49, 231–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khullar, R.; Kumar, D.; Seth, N.; Saini, S. Formulation and evaluation of mefenamic acid emulgel for topical delivery. Saudi Pharm. J. 2011, 20, 63–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaur, I.P.; Kakkar, S. Topical delivery of antifungal agents. Expert Opin. Drug Deliv. 2010, 7, 1303–1327. [Google Scholar] [CrossRef] [PubMed]
- Shehata, T.M.; Elnahas, H.M.; Elsewedy, H.S. Development, Characterization and Optimization of the Anti-Inflammatory Influence of Meloxicam Loaded into a Eucalyptus Oil-Based Nanoemulgel. Gels 2022, 8, 262. [Google Scholar] [CrossRef]
- Md, S.; Alhakamy, N.; Aldawsari, H.; Husain, M.; Khan, N.; Alfaleh, M.; Asfour, H.; Riadi, Y.; Bilgrami, A.; Akhter, H. Plumbagin-Loaded Glycerosome Gel as Topical Delivery System for Skin Cancer Therapy. Polymers 2021, 13, 923. [Google Scholar] [CrossRef]
- Pinzaru, I.; Tanase, A.; Enatescu, V.; Coricovac, D.; Bociort, F.; Marcovici, I.; Watz, C.; Vlaia, L.; Soica, C.; Dehelean, C. Proniosomal Gel for Topical Delivery of Rutin: Preparation, Physicochemical Characterization and In Vitro Toxicological Profile Using 3D Reconstructed Human Epidermis Tissue and 2D Cells. Antioxidants 2021, 10, 85. [Google Scholar] [CrossRef]
- Almostafa, M.M.; Elsewedy, H.S.; Shehata, T.M.; Soliman, W.E. Novel Formulation of Fusidic Acid Incorporated into a Myrrh-Oil-Based Nanoemulgel for the Enhancement of Skin Bacterial Infection Treatment. Gels 2022, 8, 245. [Google Scholar] [CrossRef]
- Bashir, M.; Ahmad, J.; Asif, M.; Khan, S.-U.; Irfan, M.; Ibrahim, A.Y.; Asghar, S.; Khan, I.U.; Iqbal, M.S.; Haseeb, A.; et al. Nanoemulgel, an Innovative Carrier for Diflunisal Topical Delivery with Profound Anti-Inflammatory Effect: In vitro and in vivo Evaluation. Int. J. Nanomed. 2021, 16, 1457–1472. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, Q.-Y.; Xu, Y.-M.; Lau, A.T.Y. Recent Progress of Nanocarrier-Based Therapy for Solid Malignancies. Cancers 2020, 12, 2783. [Google Scholar] [CrossRef] [PubMed]
- Weiss, J.; Gaysinsky, S.; Davidson, M.; McClements, J. Nanostructured encapsulation systems: Food antimicrobials. In Global Issues in Food Science and Technology; Elsevier: Amsterdam, The Netherlands, 2009; pp. 425–479. [Google Scholar]
- Elsewedy, H.S.; Al-Dhubiab, B.E.; Mahdy, M.A.; Elnahas, H.M. Basic Concepts of Nanoemulsion and its Potential application in Pharmaceutical, Cosmeceutical and Nutraceutical fields. Res. J. Pharm. Technol. 2021, 14, 3938–3946. [Google Scholar] [CrossRef]
- Yen, C.-C.; Chen, Y.-C.; Wu, M.-T.; Wang, C.-C.; Wu, Y.-T. Nanoemulsion as a strategy for improving the oral bioavailability and anti-inflammatory activity of andrographolide. Int. J. Nanomed. 2018, 13, 669–680. [Google Scholar] [CrossRef] [Green Version]
- Harun, S.N.; Amin Nordin, S.; Abd Gani, S.S.; Shamsuddin, A.F.; Basri, M.; Bin Basri, H. Development of nanoemulsion for efficient brain parenteral delivery of cefuroxime: Designs, characterizations, and pharmacokinetics. Int. J. Nanomed. 2018, 13, 2571–2584. [Google Scholar] [CrossRef] [Green Version]
- Sharma, A.; Singh, A.P.; Harikumar, S.L.; Sl, H. Development and optimization of nanoemulsion based gel for enhanced transdermal delivery of nitrendipine using box-behnken statistical design. Drug Dev. Ind. Pharm. 2020, 46, 329–342. [Google Scholar] [CrossRef]
- Ansari, M.N.; Soliman, G.A.; Rehman, N.U.; Anwer, K. Crisaborole Loaded Nanoemulsion Based Chitosan Gel: Formulation, Physicochemical Characterization and Wound Healing Studies. Gels 2022, 8, 318. [Google Scholar] [CrossRef]
- Bokhtia, R.M.; Girgis, A.S.; Ibrahim, T.S.; Rasslan, F.; Nossier, E.S.; Barghash, R.F.; Sakhuja, R.; Abdel-Aal, E.H.; Panda, S.S.; Al-Mahmoudy, A.M.M. Synthesis, Antibacterial Evaluation, and Computational Studies of a Diverse Set of Linezolid Conjugates. Pharmaceuticals 2022, 15, 191. [Google Scholar] [CrossRef]
- Yu, F.; Zhang, M.; Sun, J.; Wang, F.; Li, X.; Liu, Y.; Wang, Z.; Zhao, X.; Li, J.; Chen, J.; et al. Improved Neomycin Sulfate Potency in Streptomyces fradiae Using Atmospheric and Room Temperature Plasma (ARTP) Mutagenesis and Fermentation Medium Optimization. Microorganisms 2022, 10, 94. [Google Scholar] [CrossRef]
- A Manuel, M.; Kurtz, I.; Saiphoo, C.S.; Nedzelski, J.M. Nephrotoxicity and ototoxicity following irrigation of wounds with neomycin. Can. J. Surg. 1979, 22, 274–277. [Google Scholar] [PubMed]
- Hosny, K.M.; Sindi, A.M.; Bakhaidar, R.B.; Zaki, R.M.; Abualsunun, W.A.; Alkhalidi, H.M.; Bahmdan, R.H.; Md, S.; Hassan, A.H. Formulation and optimization of neomycin Sulfate–Thioctic acid loaded in a eucalyptus oil self-nanoemulsion to enhance the beneficial activity of the substances and limit the side effects associated with the treatment of hepatic coma. J. Drug Deliv. Sci. Technol. 2020, 61, 102108. [Google Scholar] [CrossRef]
- Bandyopadhyay, D. Topical antibacterials in dermatology. Indian J. Dermatol. 2021, 66, 117–125. [Google Scholar] [CrossRef]
- Wolverton, S.E.; Wu, J. Comprehensive Dermatologic Drug Therapy; Elsevier Health Sciences: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Bergen, P.J.; Landersdorfer, C.B.; Lee, H.J.; Li, J.; Nation, R.L. ‘Old’ antibiotics for emerging multidrug-resistant bacteria. Curr. Opin. Infect. Dis. 2012, 25, 626–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Keller, K.; Takemoto, J.Y.; Bensaci, M.; Litke, A.; Czyryca, P.G.; Chang, C.-W.T. Synthesis and combinational antibacterial study of 5″-modified neomycin. J. Antibiot. 2009, 62, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chiang, F.-I.; Wu, L.; Czyryca, P.G.; Li, D.; Chang, C.-W.T. Surprising Alteration of Antibacterial Activity of 5′′-Modified Neomycin against Resistant Bacteria. J. Med. Chem. 2008, 51, 7563–7573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanchard, C.; Brooks, L.; Beckley, A.; Colquhoun, J.; Dewhurst, S.; Dunman, P.M. Neomycin Sulfate Improves the Antimicrobial Activity of Mupirocin-Based Antibacterial Ointments. Antimicrob. Agents Chemother. 2016, 60, 862–872. [Google Scholar] [CrossRef] [Green Version]
- Coates, A.; Hu, Y.; Holt, J.; Yeh, P. Antibiotic combination therapy against resistant bacterial infections: Synergy, rejuvenation and resistance reduction. Expert Rev. Anti-Infective Ther. 2020, 18, 5–15. [Google Scholar] [CrossRef]
- Yasin, M.; Younis, A.; Ramzan, F.; Javed, T.; Shabbir, R.; Noushahi, H.A.; Skalicky, M.; Ondrisik, P.; Brestic, M.; Hassan, S.; et al. Extraction of Essential Oil from River Tea Tree (Melaleuca bracteata F. Muell.): Antioxidant and Antimicrobial Properties. Sustainability 2021, 13, 4827. [Google Scholar] [CrossRef]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (Tea Tree) Oil: A Review of Antimicrobial and Other Medicinal Properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar] [CrossRef] [Green Version]
- Cox, S.D.; Mann, C.M.; Markham, J.L.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. Determining the Antimicrobial Actions of Tea Tree Oil. Molecules 2001, 6, 87–91. [Google Scholar] [CrossRef] [Green Version]
- Yasin, M.; Younis, A.; Javed, T.; Akram, A.; Ahsan, M.; Shabbir, R.; Ali, M.M.; Tahir, A.; El-Ballat, E.M.; Sheteiwy, M.S.; et al. River Tea Tree Oil: Composition, Antimicrobial and Antioxidant Activities, and Potential Applications in Agriculture. Plants 2021, 10, 2105. [Google Scholar] [CrossRef] [PubMed]
- Shehata, T.M.; Khalil, H.E.; Elsewedy, H.S.; Soliman, W.E. Myrrh essential oil-based nanolipid formulation for enhancement of the antihyperlipidemic effect of atorvastatin. J. Drug Deliv. Sci. Technol. 2020, 61, 102277. [Google Scholar] [CrossRef]
- Shehata, T.M.; Elsewedy, H.S. Paclitaxel and Myrrh oil Combination Therapy for Enhancement of Cytotoxicity against Breast Cancer; QbD Approach. Processes 2022, 10, 907. [Google Scholar] [CrossRef]
- Haroun, M.; Elsewedy, H.S.; Shehata, T.M.; Tratrat, C.; Al Dhubiab, B.E.; Venugopala, K.N.; Almostafa, M.M.; Kochkar, H.; Elnahas, H.M. Significant of injectable brucine PEGylated niosomes in treatment of MDA cancer cells. J. Drug Deliv. Sci. Technol. 2022, 71, 103322. [Google Scholar] [CrossRef]
- Abdallah, M.H.; Elsewedy, H.S.; AbuLila, A.S.; Almansour, K.; Unissa, R.; Elghamry, H.A.; Soliman, M.S. Quality by Design for Optimizing a Novel Liposomal Jojoba Oil-Based Emulgel to Ameliorate the Anti-Inflammatory Effect of Brucine. Gels 2021, 7, 219. [Google Scholar] [CrossRef]
- Ayoub, A.M.; Ibrahim, M.M.; Abdallah, M.H.; Mahdy, M.A. Sulpiride microemulsions as antipsychotic nasal drug delivery systems: In-vitro and pharmacodynamic study. J. Drug Deliv. Sci. Technol. 2016, 36, 10–22. [Google Scholar] [CrossRef]
- Binder, L.; Mazál, J.; Petz, R.; Klang, V.; Valenta, C. The role of viscosity on skin penetration from cellulose ether-based hydrogels. Ski. Res. Technol. 2019, 25, 725–734. [Google Scholar] [CrossRef] [Green Version]
- Elsewedy, H.S.; Al Dhubiab, B.E.; Mahdy, M.A.; Elnahas, H.M. Brucine PEGylated nanoemulsion: In vitro and in vivo evaluation. Coll. Surfaces A Physicochem. Eng. Asp. 2020, 608, 125618. [Google Scholar] [CrossRef]
- Abdallah, M.H.; Abu Lila, A.S.; Unissa, R.; Elsewedy, H.S.; Elghamry, H.A.; Soliman, M.S. Brucine-Loaded Ethosomal Gel: Design, Optimization, and Anti-inflammatory Activity. AAPS PharmSciTech 2021, 22, 1–15. [Google Scholar] [CrossRef]
- Husseiny, R.A.; Abu Lila, A.S.; Abdallah, M.H.; Hamed, E.E.; El-Ghamry, H.A. Design, in vitro/in vivo evaluation of meclizine HCl-loaded floating microspheres targeting pregnancy-related nausea and vomiting. J. Drug Deliv. Sci. Technol. 2018, 47, 395–403. [Google Scholar] [CrossRef]
- Elsewedy, H.S.; Younis, N.S.; Shehata, T.M.; Mohamed, M.E.; Soliman, W.E. Enhancement of Anti-Inflammatory Activity of Optimized Niosomal Colchicine Loaded into Jojoba Oil-Based Emulgel Using Response Surface Methodology. Gels 2021, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Abdelnabi, D.M.; Abdallah, M.H.; Elghamry, H.A. Buspirone Hydrochloride Loaded In Situ Nanovesicular Gel as an Anxiolytic Nasal Drug Delivery System: In Vitro and Animal Studies. AAPS PharmSciTech 2019, 20, 134. [Google Scholar] [CrossRef] [PubMed]
- Soliman, W.E.; Shehata, T.M.; Mohamed, M.E.; Younis, N.S.; Elsewedy, H.S. Enhancement of Curcumin Anti-Inflammatory Effect via Formulation into Myrrh Oil-Based Nanoemulgel. Polymers 2021, 13, 577. [Google Scholar] [CrossRef]
- Zainol, S.; Basri, M.; Bin Basri, H.; Shamsuddin, A.F.; Gani, S.S.A.; Karjiban, R.A.; Abdul-Malek, E. Formulation Optimization of a Palm-Based Nanoemulsion System Containing Levodopa. Int. J. Mol. Sci. 2012, 13, 13049–13064. [Google Scholar] [CrossRef]
- Jaiswal, M.; Dudhe, R.; Sharma, P.K. Nanoemulsion: An advanced mode of drug delivery system. 3 Biotech 2015, 5, 123–127. [Google Scholar] [CrossRef] [Green Version]
- Sarheed, O.; Dibi, M.; Ramesh, K.V.R.N.S. Studies on the Effect of Oil and Surfactant on the Formation of Alginate-Based O/W Lidocaine Nanocarriers Using Nanoemulsion Template. Pharmaceutics 2020, 12, 1223. [Google Scholar] [CrossRef]
- Singh, G.; Ahuja, N.; Sharma, P.; Capalash, N. Response surface methodology for the optimized production of an alkalophilic laccase from gamma-proteobacterium JB. BioResources 2009, 4, 544–553. [Google Scholar]
- Wu, B.; Liu, H.; Chen, G.; Zhang, Y.; Ma, Z. Effects of PEGylated lipid nanoparticles on the oral absorption of one BCS II drug: A mechanistic investigation. Int. J. Nanomed. 2014, 9, 5503–5514. [Google Scholar] [CrossRef] [Green Version]
- Cieślak, A.; Wauthoz, N.; Orellana, A.N.; Lautram, N.; Béjaud, J.; Hureaux, J.; Lafleur, M.; Benoit, J.-P.; Salomon, C.J.; Bastiat, G. Stealth nanocarriers based sterosomes using PEG post-insertion process. Eur. J. Pharm. Biopharm. 2017, 115, 31–38. [Google Scholar] [CrossRef]
- Shi, L.; Zhang, J.; Zhao, M.; Tang, S.; Cheng, X.; Zhang, W.; Li, W.; Liu, X.; Peng, H.; Wang, Q. Effects of polyethylene glycol on the surface of nanoparticles for targeted drug delivery. Nanoscale 2021, 13, 10748–10764. [Google Scholar] [CrossRef]
- Islam, M.T.; Rodríguez-Hornedo, N.; Ciotti, S.; Ackermann, C. Rheological Characterization of Topical Carbomer Gels Neutralized to Different pH. Pharm. Res. 2004, 21, 1192–1199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harish, N.; Prabhu, P.; Charyulu, R.; Gulzar, M.; Subrahmanyam, E. Formulation and evaluation of in situ gels containing clotrimazole for oral candidiasis. Indian J. Pharm. Sci. 2009, 71, 421–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Md, S.; Alhakamy, N.; Aldawsari, H.M.; Kotta, S.; Ahmad, J.; Akhter, S.; Alam, S.; Khan, M.A.; Awan, Z.; Sivakumar, P.M. Improved Analgesic and Anti-Inflammatory Effect of Diclofenac Sodium by Topical Nanoemulgel: Formulation Development—In Vitro and In Vivo Studies. J. Chem. 2020, 2020, 4071818. [Google Scholar] [CrossRef] [Green Version]
- Yen, W.F.; Basri, M.; Ahmad, M.; Ismail, M. Formulation and Evaluation of Galantamine Gel as Drug Reservoir in Transdermal Patch Delivery System. Sci. World J. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Arora, R.; Aggarwal, G.; Harikumar, S.L.; Kaur, K. Nanoemulsion Based Hydrogel for Enhanced Transdermal Delivery of Ketoprofen. Adv. Pharm. 2014, 2014, 1–12. [Google Scholar] [CrossRef]
- Pinheiro, M.; Ribeiro, R.; Vieira, A.; Andrade, F.; Reis, S. Design of a nanostructured lipid carrier intended to improve the treatment of tuberculosis. Drug Des. Dev. Ther. 2016, 10, 2467–2475. [Google Scholar] [CrossRef] [Green Version]
- Mircioiu, C.; Voicu, V.; Anuta, V.; Tudose, A.; Celia, C.; Paolino, D.; Fresta, M.; Sandulovici, R.; Mircioiu, I. Mathematical Modeling of Release Kinetics from Supramolecular Drug Delivery Systems. Pharmaceutics 2019, 11, 140. [Google Scholar] [CrossRef] [Green Version]
- Sainy, J.; Atneriya, U.; Kori, J.L.; Maheshwari, R. Development of an Aloe vera-based Emulgel for the Topical Delivery of Desoximetasone. Turk. J. Pharm. Sci. 2021, 18, 465–475. [Google Scholar] [CrossRef]
- Osel, N.; Parfant, T.P.; Kristl, A.; Roškar, R. Stability-Indicating Analytical Approach for Stability Evaluation of Lactoferrin. Pharmaceutics 2021, 13, 1065. [Google Scholar] [CrossRef]
- Cox, S.D.; Mann, C.M.; Markham, J.L.; Bell, H.C.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. The mode of antimicrobial action of the essential oil of Melaleuca alternifolia (tea tree oil). J. Appl. Microbiol. 2001, 88, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Southwell, I.; Hayes, A.; Markham, J.; Leach, D. The search for optimally bioactive Australian tea tree oil. Acta Hortic. 1993, 344, 256–265. [Google Scholar] [CrossRef]




| Formula | Space Type | Independent Variables | Response Values | PDI | ||
|---|---|---|---|---|---|---|
| A (g) | B (g) | Y1 (nm) | Y2 (%) | |||
| F1 | Factorial | 1.5 | 1 | 153 ± 2.0 | 84.8 ± 4.0 | 0.29 ± 0.062 |
| F2 | Axial | 1.5 | 0.75 | 172 ± 3.0 | 83.0 ± 3.6 | 0.32 ± 0.020 |
| F3 | Center | 2 | 0.75 | 227 ± 4.4 | 70.2 ± 3.2 | 0.41 ± 0.011 |
| F4 | Factorial | 1.5 | 0.5 | 190 ± 3.6 | 80.7 ± 4.1 | 0.34 ± 0.028 |
| F5 | Factorial | 2.5 | 0.5 | 334 ± 4.5 | 51.0 ± 3.0 | 0.28 ± 0.029 |
| F6 | Center | 2 | 0.75 | 229 ± 4.6 | 72.6 ± 3.2 | 0.39 ± 0.034 |
| F7 | Axial | 2.5 | 0.75 | 310 ± 4.4 | 54.4 ± 3.7 | 0.32 ± 0.020 |
| F8 | Axial | 2 | 1 | 206 ± 3.1 | 73.5 ± 3.1 | 0.28 ± 0.014 |
| F9 | Axial | 2 | 0.5 | 232 ± 4.2 | 68.4 ± 2.9 | 0.30 ± 0.015 |
| F10 | Center | 2 | 0.75 | 223 ± 3.0 | 71.1 ± 3.1 | 0.31 ± 0.014 |
| F11 | Factorial | 2.5 | 1 | 287 ± 3.5 | 57.9 ± 2.8 | 0.26 ± 0.012 |
| Source | Y1 | Y2 | ||
|---|---|---|---|---|
| F-Value | p-Value | F-Value | p-Value | |
| Model | 229.65 | <0.0001 * | 415.48 | <0.0001 * |
| A | 1042.77 | <0.0001 * | 1975.05 | <0.0001 * |
| B | 72.91 | 0.0004 * | 70.53 | 0.0004 * |
| AB | 0.9038 | 0.3854 | 3.20 | 0.1337 |
| A2 | 31.44 | 0.0025 * | 25.20 | 0.0040 * |
| B2 | 1.11 | 0.3413 | 0.1973 | 0.6755 |
| Lack of Fit | 4.27 | 0.1955 | 0.0278 | 0.9920 |
| Dependent Variable | Source | R2 | Adjusted R2 | Predicted R2 | SD | Adequate Precision | Remark |
|---|---|---|---|---|---|---|---|
| Y1 | Linear | 0.9674 | 0.9593 | 0.9321 | 11.39 | - | - |
| 2FI | 0.9682 | 0.9546 | 0.8773 | 12.03 | - | - | |
| Quadratic | 0.9957 | 0.9913 | 0.9633 | 5.26 | 45.1400 | Suggested | |
| Cubic | 0.9984 | 0.9945 | 0.8636 | 4.19 | - | - | |
| Y2 | Linear | 0.9823 | 0.9779 | 0.9654 | 1.68 | - | - |
| 2FI | 0.9839 | 0.9769 | 0.9419 | 1.72 | - | - | |
| Quadratic | 0.9976 | 0.9952 | 0.9949 | 0.7827 | 58.4163 | Suggested | |
| Cubic | 0.9977 | 0.9922 | 0.9918 | 0.9966 | - | - |
| Independent Variable | Symbol | Criteria |
|---|---|---|
| Amount of TTO | A | In range |
| Amount of tween 80 | B | In range |
| Dependent response | Predicted values | Observed values |
| Y1 (nm) | 154.675 ± 5.25 | 161.3 ± 3.2 |
| Y2 (%) | 84.743 ± 0.78 | 82.63 ± 2.41 |
| Kinetic Model | NEO-NE | NEO-NE-Hydrogel Base |
|---|---|---|
| Zero-order kinetic (R2) | 0.9533 | 0.9765 |
| First-order kinetic (R2) | 0.8548 | 0.9409 |
| Higuchi kinetic (R2) | 0.9989 | 0.9533 |
| Korsmeyer–Peppas kinetic (R2) | 0.9924 | 0.9856 |
| Inhibition Zone Diameter (cm) | Bacterial Type | |||
|---|---|---|---|---|
| Bacillus subtilis | Staphylococcus aureus | klebsiella pneumoniae | E. coli | |
| NEO-NE-hydrogel | 4.42 ± 0.13 * # | 4.26 ±0.11* # | 4.54 ± 0.11 * # | 4.38 ± 0.11 * # |
| Blank NE-hydrogel | 2.86 ± 0.12 * | 3.09 ± 012 * | 3.02 ±0.14 * | 2.95 ± 0.13 * |
| NEO solution | 4.14 ± 0.11 # | 4.01 ± 0.11 # | 4.29 ± 0.12 # | 4.12 ± 0.12 # |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsewedy, H.S.; Shehata, T.M.; Soliman, W.E. Tea Tree Oil Nanoemulsion-Based Hydrogel Vehicle for Enhancing Topical Delivery of Neomycin. Life 2022, 12, 1011. https://doi.org/10.3390/life12071011
Elsewedy HS, Shehata TM, Soliman WE. Tea Tree Oil Nanoemulsion-Based Hydrogel Vehicle for Enhancing Topical Delivery of Neomycin. Life. 2022; 12(7):1011. https://doi.org/10.3390/life12071011
Chicago/Turabian StyleElsewedy, Heba S., Tamer M. Shehata, and Wafaa E. Soliman. 2022. "Tea Tree Oil Nanoemulsion-Based Hydrogel Vehicle for Enhancing Topical Delivery of Neomycin" Life 12, no. 7: 1011. https://doi.org/10.3390/life12071011
APA StyleElsewedy, H. S., Shehata, T. M., & Soliman, W. E. (2022). Tea Tree Oil Nanoemulsion-Based Hydrogel Vehicle for Enhancing Topical Delivery of Neomycin. Life, 12(7), 1011. https://doi.org/10.3390/life12071011







