Promising Effects of 3-Month Period of Quercetin Phytosome® Supplementation in the Prevention of Symptomatic COVID-19 Disease in Healthcare Workers: A Pilot Study
Abstract
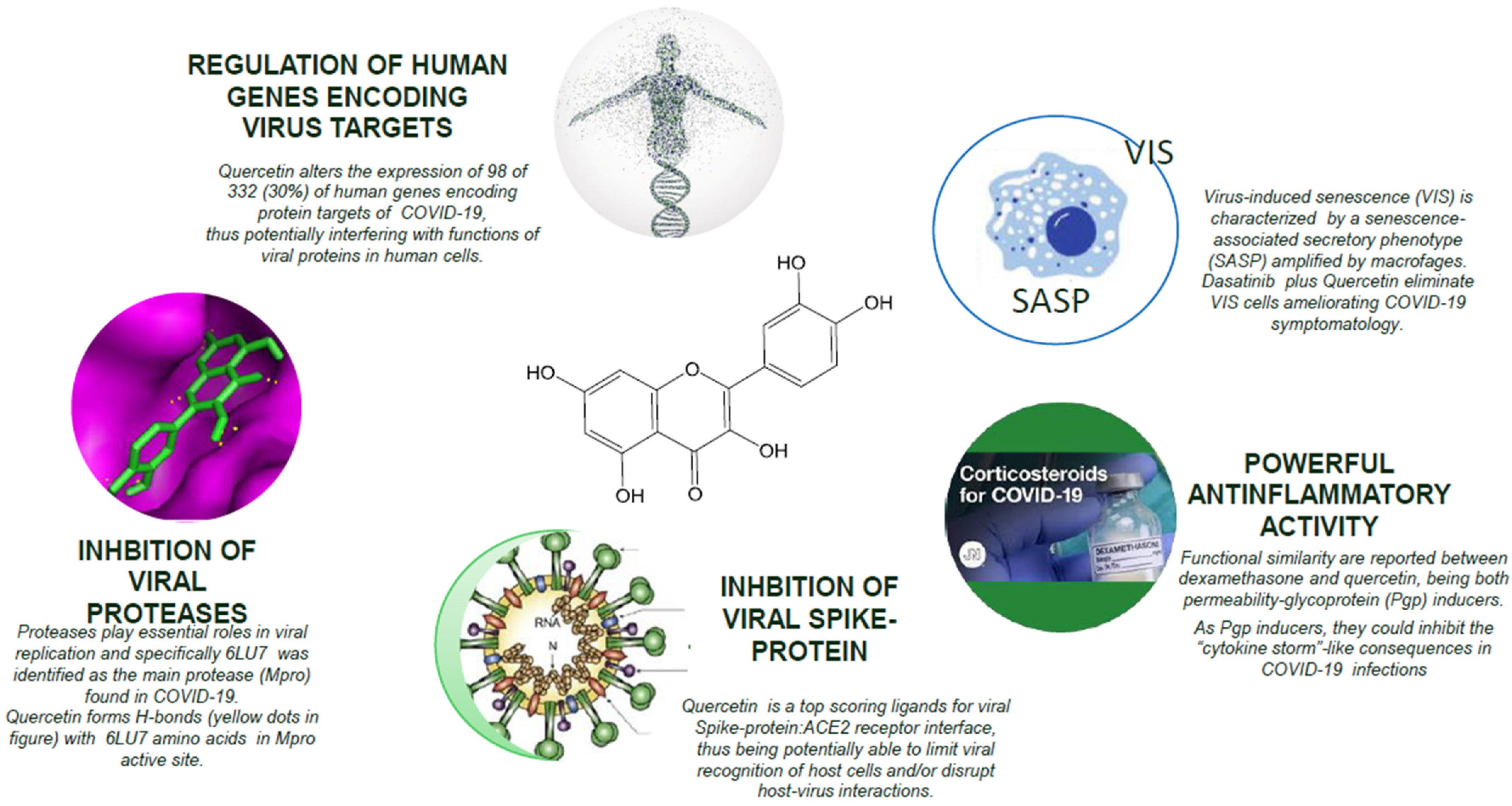
:1. Introduction
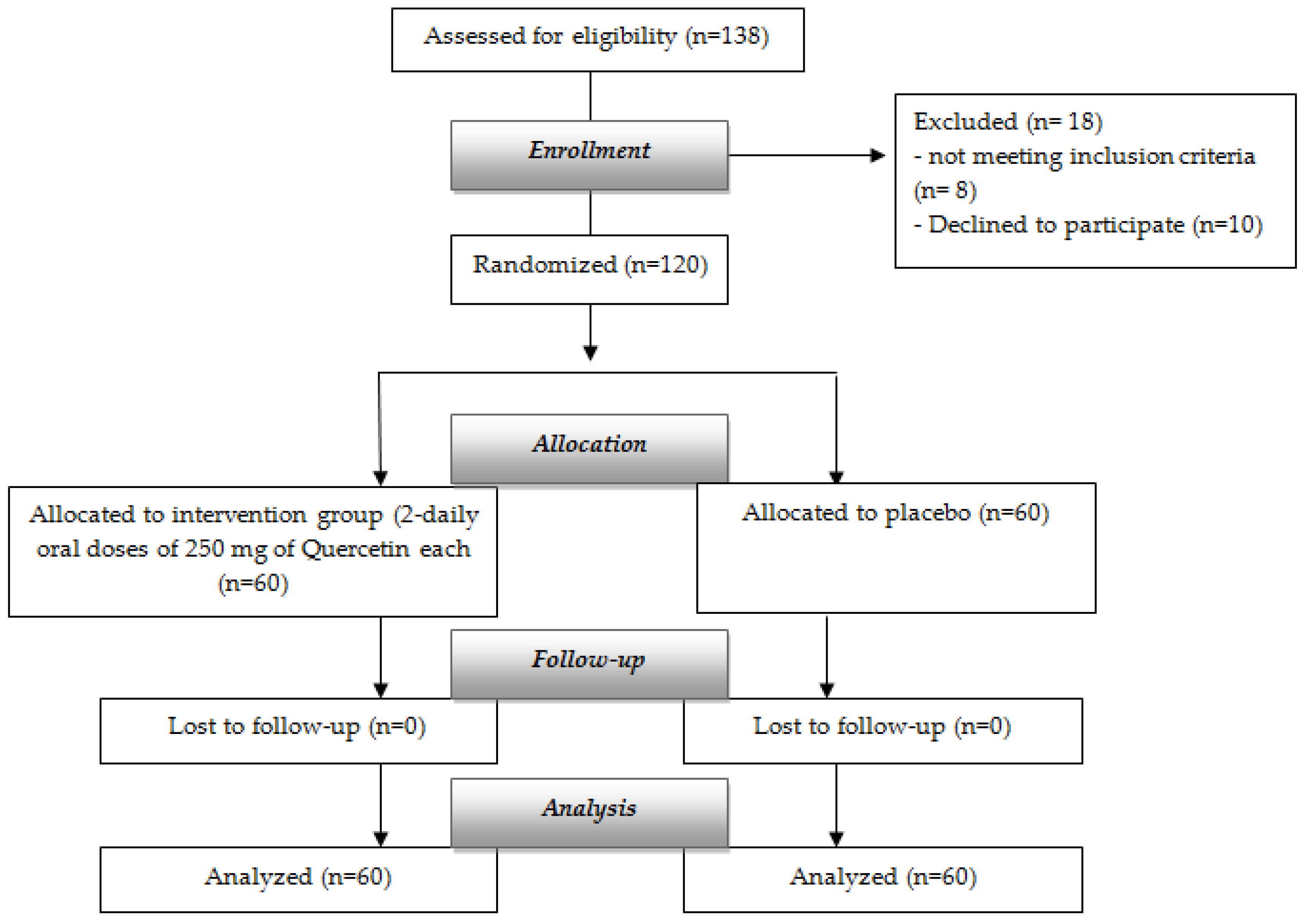
2. Materials and Methods
2.1. Participants
2.2. Dietary Supplement
2.3. Statistical Analysis
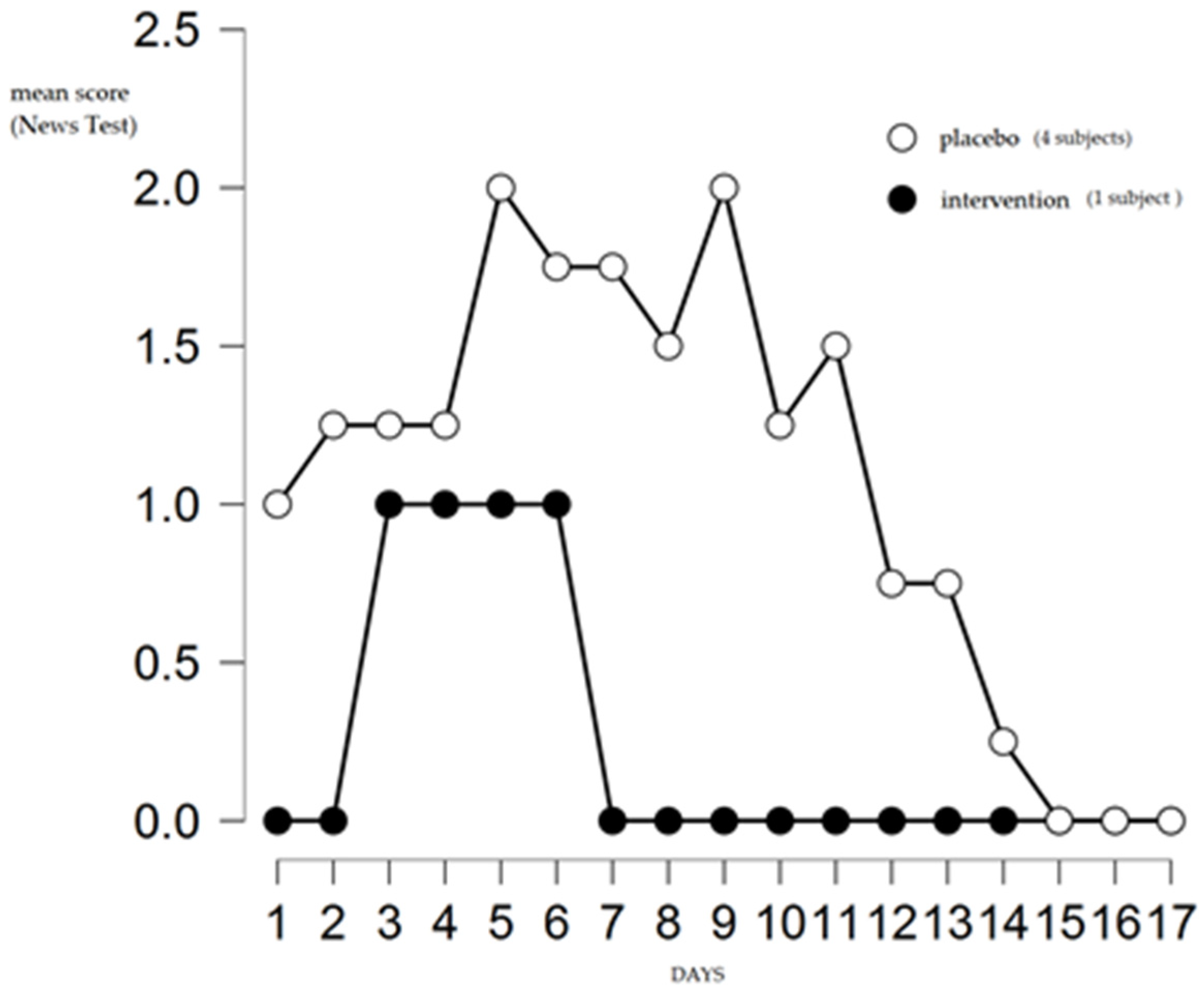
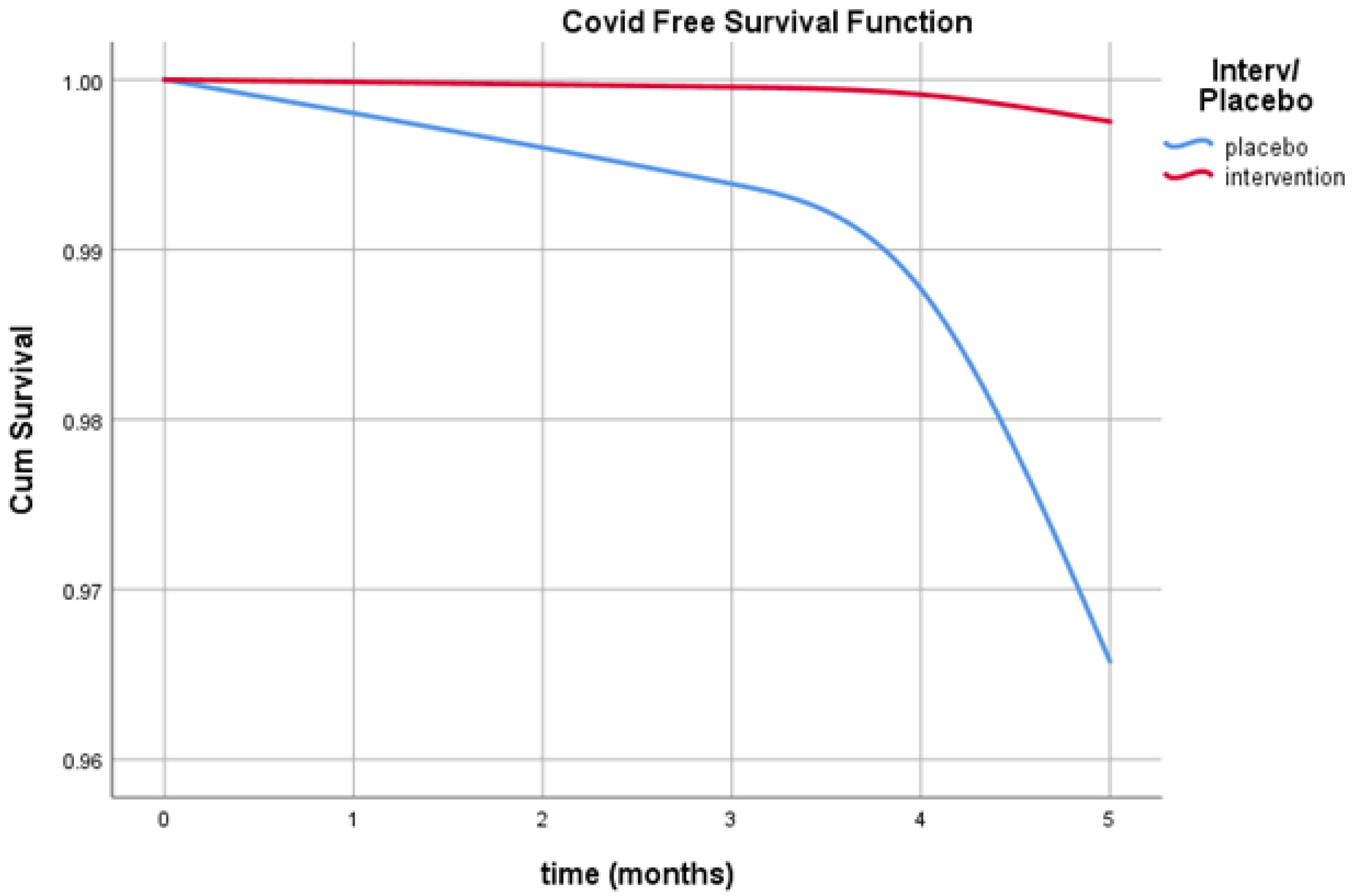
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coronavirus Disease (COVID-19). Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019?adgroupsurvey=%7Badgroupsurvey%7D&gclid=EAIaIQobChMImZnf_puL9AIVFNN3Ch0FJgp2EAAYASAAEgIGlfD_BwE (accessed on 9 November 2021).
- Lee, S.; Yu, Y.; Trimpert, J.; Benthani, F.; Mairhofer, M.; Richter-Pechanska, P.; Wyler, E.; Belenki, D.; Kaltenbrunner, S.; Pammer, M.; et al. Virus-induced senescence is a driver and therapeutic target in COVID-19. Nature 2021, 599, 283–289. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, Y.; Shao, C.; Huang, J.; Gan, J.; Huang, X.; Bucci, E.; Piacentini, M.; Ippolito, G.; Melino, G. COVID-19 infection: The perspectives on immune responses. Cell Death Differ. 2020, 27, 1451–1454. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Geng, M.; Peng, Y.; Meng, L.; Lu, S. Molecular immune pathogenesis and diagnosis of COVID-19. J. Pharm. Anal. 2020, 10, 102–108. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus infections and immune responses. J. Med. Virol. 2020, 92, 424–432. [Google Scholar] [CrossRef]
- Gralinski, L.E.; Baric, R.S. Molecular pathology of emerging coronavirus infections. J. Pathol. 2015, 235, 185–195. [Google Scholar] [CrossRef] [Green Version]
- Majumder, J.; Minko, T. Recent Developments on Therapeutic and Diagnostic Approaches for COVID-19. AAPS J. 2021, 23, 14. [Google Scholar] [CrossRef]
- Brendler, T.; Al-Harrasi, A.; Bauer, R.; Gafner, S.; Hardy, M.L.; Heinrich, M.; Hosseinzadeh, H.; Izzo, A.A.; Michaelis, M.; Nassiri-Asl, M.; et al. Botanical drugs and supplements affecting the immune response in the time of COVID-19: Implications for research and clinical practice. Phytother. Res. 2021, 35, 3013–3031. [Google Scholar] [CrossRef]
- Aucoin, M.; Cooley, K.; Saunders, P.R.; Cardozo, V.; Remy, D.; Cramer, H.; Neyre Abad, C.; Hannan, N. The effect of quercetin on the prevention or treatment of COVID-19 and other respiratory tract infections in humans: A rapid review. Adv. Integr. Med. 2020, 7, 247–251. [Google Scholar] [CrossRef]
- Russo, M.; Moccia, S.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Roles of flavonoids against coronavirus infection. Chem. Biol. Interact. 2020, 328, 109211. [Google Scholar] [CrossRef]
- Riva, A.; Vitale, J.A.; Belcaro, G.; Hu, S.; Feragalli, B.; Vinciguerra, G.; Cacchio, M.; Bonanni, E.; Giacomelli, L.; Eggenhöffner, R.; et al. Quercetin phytosome® in triathlon athletes: A pilot registry study. Minerva Med. 2018, 109, 285–289. [Google Scholar] [CrossRef]
- Cesarone, M.R.; Belcaro, G.; Hu, S.; Dugall, M.; Hosoi, M.; Ledda, A.; Feragalli, B.; Maione, C.; Cotellese, R. Supplementary prevention and management of asthma with quercetin phytosome: A pilot registry. Minerva Med. 2019, 110, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Pawar, A.; Pal, A. Molecular and functional resemblance of dexamethasone and quercetin: A paradigm worth exploring in dexamethasone-nonresponsive COVID-19 patients. Phytother. Res. 2020, 34, 3085–3088. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Smith, J. Repurposing Therapeutics for COVID-19: Supercomputer-Based Docking to the SARS-CoV-2 Viral Spike Protein and Viral Spike Protein-Human ACE2 Interface; ChemRxiv: Cambridge, UK, 2020. [Google Scholar]
- Khaerunnisa, S.; Kurniawan, H.; Awaluddin, R.; Suhartati, S.; Soetjipto, S. Potential Inhibitor of COVID-19 Main Protease (Mpro) From Several Medicinal Plant Compounds by Molecular Docking Study. Preprints 2020, 2020, 2020030226. [Google Scholar]
- Glinsky, G.V. Tripartite Combination of Candidate Pandemic Mitigation Agents: Vitamin D, Quercetin, and Estradiol Manifest Properties of Medicinal Agents for Targeted Mitigation of the COVID-19 Pandemic Defined by Genomics-Guided Tracing of SARS-CoV-2 Targets in Human. Biomedicines 2020, 8, 129. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Raghuvanshi, R.; Ceylan, F.D.; Bolling, B.W. Quercetin and Its Metabolites Inhibit Recombinant Human Angiotensin-Converting Enzyme 2 (ACE2) Activity. J. Agric. Food Chem. 2020, 68, 13982–13989. [Google Scholar] [CrossRef]
- Arslan, B.; Ucuncu Ergun, N.; Topuz, S.; Yilmaz Semerci, S.; Suner, N.; Kocatas, A.; Onal, H. Synergistic Effect of Quercetin and Vitamin C Against COVID-19: Is a Possible Guard for Front Liners. SSRN Electron. J. 2020, 1–19. [Google Scholar] [CrossRef]
- Di Pierro, F.; Iqtadar, S.; Khan, A.; Ullah Mumtaz, S.; Masud Chaudhry, M.; Bertuccioli, A.; Derosa, G.; Maffioli, P.; Togni, S.; Riva, A.; et al. Potential Clinical Benefits of Quercetin in the Early Stage of COVID-19: Results of a Second, Pilot, Randomized, Controlled and Open-Label Clinical Trial. Int. J. Gen. Med. 2021, 14, 2807–2816. [Google Scholar] [CrossRef]
- Di Pierro, F.; Derosa, G.; Maffioli, P.; Bertuccioli, A.; Togni, S.; Riva, A.; Allegrini, P.; Khan, A.; Khan, S.; Khan, B.A.; et al. Possible Therapeutic Effects of Adjuvant Quercetin Supplementation Against Early-Stage COVID-19 Infection: A Prospective, Randomized, Controlled, and Open-Label Study. Int. J. Gen. Med. 2021, 14, 2359–2366. [Google Scholar] [CrossRef]
- Dziadzko, M.A.; Novotny, P.J.; Sloan, J.; Gajic, O.; Herasevich, V.; Mirhaji, P.; Wu, Y.; Gong, M.N. Multicenter derivation and validation of an early warning score for acute respiratory failure or death in the hospital. Crit. Care 2018, 22, 286. [Google Scholar] [CrossRef] [Green Version]
- Smith, G.B.; Prytherch, D.R.; Meredith, P.; Schmidt, P.E.; Featherstone, P.I. The ability of the National Early Warning Score (NEWS) to discriminate patients at risk of early cardiac arrest, unanticipated intensive care unit admission, and death. Resuscitation 2013, 84, 465–470. [Google Scholar] [CrossRef]
- Margolin, L.; Luchins, J.; Margolin, D.; Margolin, M.; Lefkowitz, S. 20-Week Study of Clinical Outcomes of Over-the-Counter COVID-19 Prophylaxis and Treatment. J. Evid.-Based Integr. Med. 2021, 26, 1–13. [Google Scholar] [CrossRef]
- Gomes da Silva, A.P. Fighting coronaviruses with natural polyphenols. Biocatal. Agric. Biotechnol. 2021, 37, 102179. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P.; D’Angelo, A.; Di Pierro, F. A role for quercetin in coronavirus disease 2019 (COVID-19). Phytother. Res. 2021, 35, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- DI Pierro, F.; Khan, A.; Bertuccioli, A.; Maffioli, P.; Derosa, G.; Khan, S.; Khan, B.A.; Nigar, R.; Ujjan, I.; Devrajani, B.R. Quercetin Phytosome® as a potential candidate for managing COVID-19. Minerva Gastroenterol. 2021, 67, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Riva, A.; Ronchi, M.; Petrangolini, G.; Bosisio, S.; Allegrini, P. Improved Oral Absorption of Quercetin from Quercetin Phytosome®, a New Delivery System Based on Food Grade Lecithin. Eur. J. Drug Metab. Pharmacokinet. 2019, 44, 169–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riva, A.; Corti, A.; Belcaro, G.; Cesarone, M.R.; Dugall, M.; Vinciguerra, G.; Feragalli, B.; Zuccarini, M.; Eggenhoffner, R.; Giacomelli, L. Interaction study between antiplatelet agents, anticoagulants, diabetic therapy and a novel delivery form of quercetin. Minerva Cardioangiol. 2019, 67, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Di Pede, G.; Bresciani, L.; Calani, L.; Petrangolini, G.; Riva, A.; Allegrini, P.; Del Rio, D.; Mena, P. The human microbial metabolism of quercetin in different formulations: An in vitro evaluation. Foods 2020, 9, 1121. [Google Scholar] [CrossRef] [PubMed]
| Variable | Placebo Group (N = 60) | Quercetin Group (N = 60) | Total (N = 120) |
|---|---|---|---|
| Age (years) | 47.70 ± 13.67 | 50.88 ± 12.08 | 49.29 ± 12.94 |
| Height (m) | 1.74 ± 0.10 | 1.69 ± 0.10 | 1.71 ± 0.11 |
| Weight (kg) | 75.39 ± 12.02 | 69.19 ± 11.53 | 72.29 ± 12.13 |
| BMI (kg/m2) | 24.88 ± 2.74 | 24.10 ± 2.95 | 24.49 ± 2.86 |
| Variable | Number (Percentage) |
|---|---|
| Gender | |
| Male | 63 (52.50%) |
| Female | 57 (47.50%) |
| Smoker | |
| Yes | 21 (17.50%) |
| No | 99 (82.50%) |
| Physical activity | |
| Yes | 44 (36.67%) |
| No | 76 (63.33%) |
| Profession | |
| Job in contact with other people | 77 (64.17%) |
| Job not in contact with other people | 43 (35.83%) |
| Degree of education | |
| Middle-school diploma | 19 (15.83%) |
| High School diploma | 56 (46.67%) |
| University degree | 45 (37.50%) |
| Swab after 10 days | Placebo Group | Quercetin Group | Total |
|---|---|---|---|
| Negative | 0 | 1 | 1 |
| Positive | 4 | 0 | 4 |
| Total | 4 | 1 | 5 |
| Swab after 10 + 7 days | |||
| Negative | 4 | 0 | 4 |
| Total | 4 | 0 | 4 |
| B | SE | Wald | Sig. | Exp(B) | 95% CI for Exp(B) | ||
|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||
| /PLACEBO/INTEGR | 2.646 | 1.304 | 4.117 | 0.042 * | 14.097 | 1.094 | 181.596 |
| BMI | −0.214 | 0.205 | 1.083 | 0.298 | 0.808 | 0.540 | 1.208 |
| Smoker | 2.447 | 1.260 | 3.771 | 0.052 | 11.552 | 0.978 | 136.514 |
| Physical activity | 0.617 | 1.012 | 0.372 | 0.542 | 1.854 | 0.255 | 13.468 |
| Profession | −0.565 | 1.210 | 0.218 | 0.640 | 0.568 | 0.053 | 6.090 |
| Education | 2.068 | 1.094 | 3.570 | 0.059 | 7.908 | 0.926 | 67.559 |
| Age | 0.099 | 0.059 | 2.843 | 0.092 | 1.104 | 0.984 | 1.239 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rondanelli, M.; Perna, S.; Gasparri, C.; Petrangolini, G.; Allegrini, P.; Cavioni, A.; Faliva, M.A.; Mansueto, F.; Patelli, Z.; Peroni, G.; et al. Promising Effects of 3-Month Period of Quercetin Phytosome® Supplementation in the Prevention of Symptomatic COVID-19 Disease in Healthcare Workers: A Pilot Study. Life 2022, 12, 66. https://doi.org/10.3390/life12010066
Rondanelli M, Perna S, Gasparri C, Petrangolini G, Allegrini P, Cavioni A, Faliva MA, Mansueto F, Patelli Z, Peroni G, et al. Promising Effects of 3-Month Period of Quercetin Phytosome® Supplementation in the Prevention of Symptomatic COVID-19 Disease in Healthcare Workers: A Pilot Study. Life. 2022; 12(1):66. https://doi.org/10.3390/life12010066
Chicago/Turabian StyleRondanelli, Mariangela, Simone Perna, Clara Gasparri, Giovanna Petrangolini, Pietro Allegrini, Alessandro Cavioni, Milena Anna Faliva, Francesca Mansueto, Zaira Patelli, Gabriella Peroni, and et al. 2022. "Promising Effects of 3-Month Period of Quercetin Phytosome® Supplementation in the Prevention of Symptomatic COVID-19 Disease in Healthcare Workers: A Pilot Study" Life 12, no. 1: 66. https://doi.org/10.3390/life12010066
APA StyleRondanelli, M., Perna, S., Gasparri, C., Petrangolini, G., Allegrini, P., Cavioni, A., Faliva, M. A., Mansueto, F., Patelli, Z., Peroni, G., Tartara, A., & Riva, A. (2022). Promising Effects of 3-Month Period of Quercetin Phytosome® Supplementation in the Prevention of Symptomatic COVID-19 Disease in Healthcare Workers: A Pilot Study. Life, 12(1), 66. https://doi.org/10.3390/life12010066






