Constituent Characteristics and Functional Properties of Passion Fruit Seed Extract
Abstract
:1. Introduction
2. Passiflora edulis Seed Compounds
2.1. Nutritional Composition of P. edulis Seeds
2.2. Polyphenol Components in P. edulis Seeds
3. Health Benefits of P. edulis Seed Components
3.1. Antioxidant Activity
3.2. Effect on Skin
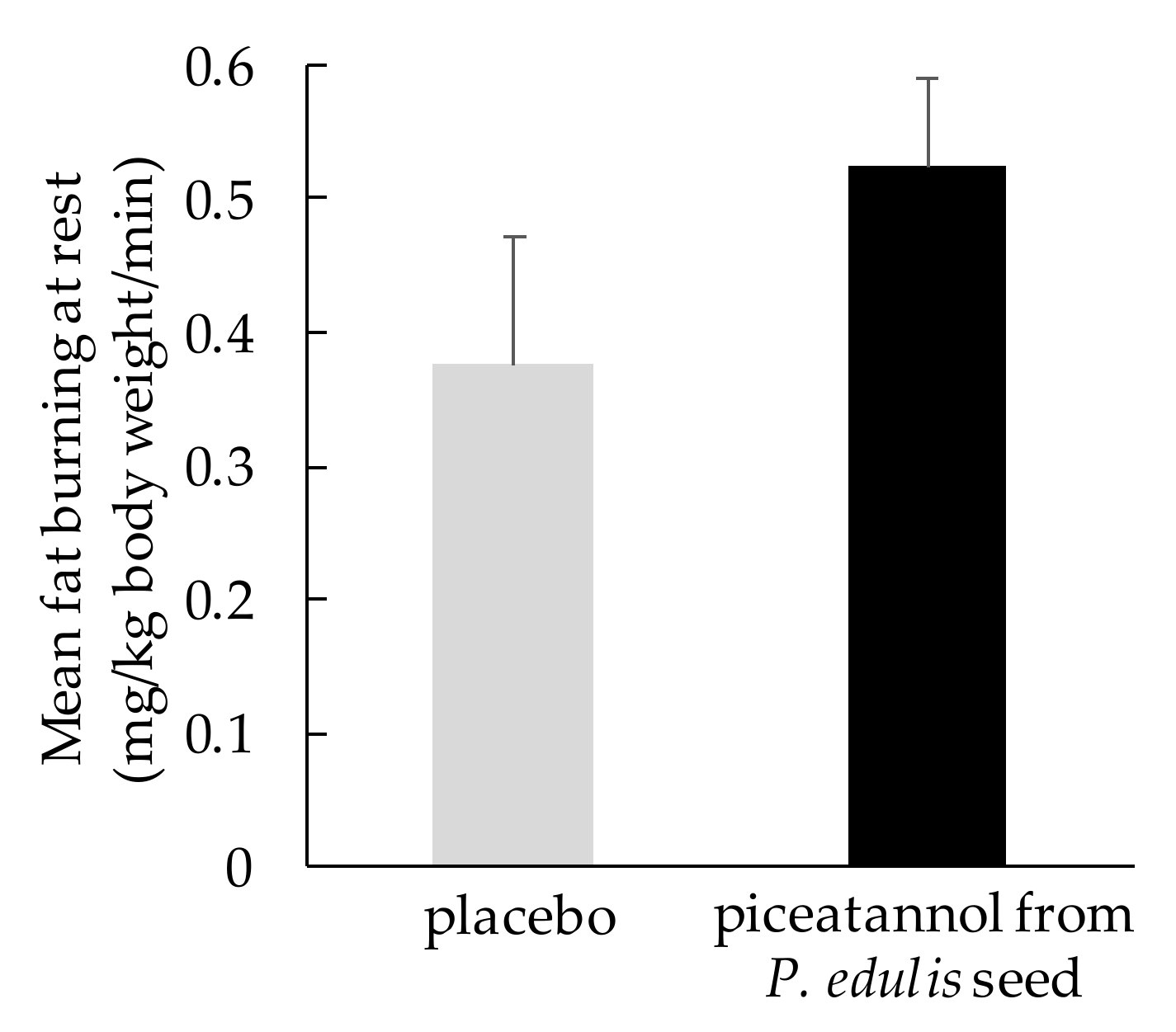
3.3. Effect on Fat Metabolism
3.4. Hypoglycemic Effect
3.5. Other Physiological Effects
4. Safety of P. edulis Seed Extract
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ulmer, T.; MacDougal, J.M. Passiflora: Passionflowers of the World; Timber Press: Portland, OR, USA, 2004. [Google Scholar]
- Cerqueira-Silva, C.B.; Jesus, O.N.; Santos, E.S.; Corrêa, R.X.; Souza, A.P. Genetic breeding and diversity of the genus Passiflora: Progress and perspectives in molecular and genetic studies. Int. J. Mol. Sci. 2014, 15, 14122–14152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reis, C.C.; Mamede, A.M.G.N.; Soares, A.; Freitas, S.P. Production of lipids and natural antioxidants from passion fruit seeds. Grasas Y Aceites 2020, 71, e385. [Google Scholar] [CrossRef]
- Qiu, W.; Su, W.; Cai, Z.; Dong, L.; Li, C.; Xin, M.; Fang, W.; Liu, Y.; Wang, X.; Huang, Z.; et al. Combined Analysis of Transcriptome and Metabolome Reveals the Potential Mechanism of Coloration and Fruit Quality in Yellow and Purple Passiflora edulis Sims. J. Agric. Food Chem. 2020, 68, 12096–12106. [Google Scholar] [CrossRef] [PubMed]
- FAO. Food Outlook—Biannual Report on Global Food Markets; FAO: Rome, Italy, 2018; pp. 67–74. [Google Scholar]
- IBGE. Instituto Brasileiro de Geografia E Estatística. Available online: https://sidra.ibge.gov.br/tabela/5457 (accessed on 15 December 2021).
- Da Silva, J.L.; da Silva, W.; Lopes, L.E.M.; Silva, M.; Silva-Cabral, J.R.A.; Costa, J.F.O.; Lima, G.S.A.; Assuncao, I.P. First report of Colletotrichum tropicale causing anthracnose on Passiflora edulis in Brazil. Plant Dis. 2021. [Google Scholar] [CrossRef]
- Mandal, G. Production Preference and importance of passion fruit (Passiflora Edulis): A review. J. Agric. Eng. Food Technol. 2017, 4, 27–30. [Google Scholar]
- He, X.; Luan, F.; Yang, Y.; Wang, Z.; Zhao, Z.; Fang, J.; Wang, M.; Zuo, M.; Li, Y. Passiflora edulis: An Insight into Current Researches on Phytochemistry and Pharmacology. Front. Pharmacol. 2020, 11, 617. [Google Scholar] [CrossRef]
- Malacrida, C.R.; Jorge, N. Yellow passion fruit seed oil (Passiflora edulis f. flavicarpa): Physical and chemical characteristics. Braz. Arch. Biol. Technol. 2012, 55, 127–134. [Google Scholar] [CrossRef] [Green Version]
- Sarris, J. Herbal medicines in the treatment of psychiatric disorders: 10-year updated review. Phytother. Res. 2018, 32, 1147–1162. [Google Scholar] [CrossRef]
- Da Fonseca, L.R.; Rodrigues, R.A.; Ramos, A.S.; da Cruz, J.D.; Ferreira, J.L.P.; Silva, J.R.A.; Amaral, A.C.F. Herbal Medicinal Products from Passiflora for Anxiety: An Unexploited Potential. Sci. World J. 2020, 2020, 6598434. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Zhou, T.; Wang, F.; Zhou, Y.; Li, Y.; Zhang, J.J.; Zheng, J.; Xu, D.P.; Li, H.B. The Effects of Syzygium samarangense, Passiflora edulis and Solanum muricatum on Alcohol-Induced Liver Injury. Int. J. Mol. Sci. 2016, 17, 1616. [Google Scholar] [CrossRef] [Green Version]
- Urrego, N.; Sepúlveda, P.; Aragón, M.; Ramos, F.A.; Costa, G.M.; Ospina, L.F.; Castellanos, L. Flavonoids and saponins from Passiflora edulis f. edulis leaves (purple passion fruit) and its potential anti-inflammatory activity. J. Pharm. Pharmacol. 2021, 73, 1530–1538. [Google Scholar] [CrossRef]
- Do Carmo, M.C.L.; Martins, I.M.; Magalhães, A.E.R.; Júnior, M.R.M.; Macedo, J.A. Passion fruit (Passiflora edulis) leaf aqueous extract ameliorates intestinal epithelial barrier dysfunction and reverts inflammatory parameters in Caco-2 cells monolayer. Food Res. Int. 2020, 133, 109162. [Google Scholar] [CrossRef]
- Soares, R.D.F.; Campos, M.G.N.; Ribeiro, G.P.; Salles, B.C.C.; Cardoso, N.S.; Ribeiro, J.R.; Souza, R.M.; Leme, K.C.; Soares, C.B.; de Oliveira, C.M.; et al. Development of a chitosan hydrogel containing flavonoids extracted from Passiflora edulis leaves and the evaluation of its antioxidant and wound healing properties for the treatment of skin lesions in diabetic mice. J. Biomed. Mater. Res. A 2020, 108, 654–662. [Google Scholar] [CrossRef]
- Salles, B.C.C.; da Silva, M.A.; Taniguthi, L.; Ferreira, J.N.; da Rocha, C.Q.; Vilegas, W.; Dias, P.H.; Pennacchi, P.C.; Duarte, S.; Rodrigues, M.R.; et al. Passiflora edulis Leaf Extract: Evidence of Antidiabetic and Antiplatelet Effects in Rats. Biol. Pharm. Bull. 2020, 43, 169–174. [Google Scholar] [CrossRef]
- Alves, J.S.F.; Silva, A.; da Silva, R.M.; Tiago, P.R.F.; de Carvalho, T.G.; de Araújo Júnior, R.F.; de Azevedo, E.P.; Lopes, N.P.; Ferreira, L.S.; Gavioli, E.C.; et al. In Vivo Antidepressant Effect of Passiflora edulis f. flavicarpa into Cationic Nanoparticles: Improving Bioactivity and Safety. Pharmaceutics 2020, 12, 383. [Google Scholar] [CrossRef]
- Devaki, K.; Beulah, U.; Akila, G.; Gopalakrishnan, V.K. Effect of Aqueous Extract of Passiflora edulis on Biochemical and Hematological Parameters of Wistar Albino Rats. Toxicol. Int. 2012, 19, 63–67. [Google Scholar] [CrossRef] [Green Version]
- Cabral, B.; Gonçalves, T.A.F.; Abreu, L.S.; Andrade, A.W.L.; de Azevedo, F.; de Castro, F.D.; Tavares, J.F.; Guerra, G.C.B.; de Rezende, A.A.; de Medeiros, I.A.; et al. Cardiovascular Effects Induced by Fruit Peels from Passiflora edulis in Hypertensive Rats and Fingerprint Analysis by HPLC-ESI-MSn spectrometry. Planta Med. 2021. [Google Scholar] [CrossRef]
- Guan, Y.; Sun, H.; Chen, H.; Li, P.; Shan, Y.; Li, X. Physicochemical characterization and the hypoglycemia effects of polysaccharide isolated from Passiflora edulis Sims peel. Food Funct. 2021, 12, 4221–4230. [Google Scholar] [CrossRef]
- Goss, M.J.; Nunes, M.L.O.; Machado, I.D.; Merlin, L.; Macedo, N.B.; Silva, A.M.O.; Bresolin, T.M.B.; Santin, J.R. Peel flour of Passiflora edulis Var. Flavicarpa supplementation prevents the insulin resistance and hepatic steatosis induced by low-fructose-diet in young rats. Biomed. Pharmacother. 2018, 102, 848–854. [Google Scholar] [CrossRef]
- Vuolo, M.M.; Lima, G.C.; Batista, Â.G.; Carazin, C.B.B.; Cintra, D.E.; Prado, M.A.; Júnior, M.R.M. Passion fruit peel intake decreases inflammatory response and reverts lipid peroxidation and adiposity in diet-induced obese rats. Nutr. Res. 2020, 76, 106–117. [Google Scholar] [CrossRef]
- De Faveri, A.; De Faveri, R.; Broering, M.F.; Bousfield, I.T.; Goss, M.J.; Muller, S.P.; Pereira, R.O.; de Oliveira, E.S.A.M.; Machado, I.D.; Quintão, N.L.M.; et al. Effects of passion fruit peel flour (Passiflora edulis f. flavicarpa O. Deg.) in cafeteria diet-induced metabolic disorders. J. Ethnopharmacol. 2020, 250, 112482. [Google Scholar] [CrossRef]
- Panelli, M.F.; Pierine, D.T.; de Souza, S.L.B.; Ferron, A.J.T.; Garcia, J.L.; Santos, K.C.D.; Belin, M.A.F.; Lima, G.P.P.; Borguini, M.G.; Minatel, I.O.; et al. Bark of Passiflora edulis Treatment Stimulates Antioxidant Capacity, and Reduces Dyslipidemia and Body Fat in db/db Mice. Antioxidants 2018, 7, 120. [Google Scholar] [CrossRef] [Green Version]
- Devi, S.R.; Sidik, J.; Zakaria, M. Nutritive values of passion fruit (Passiflora Species) seeds and its role in human health. J. Agric. Food Dev. 2018, 4, 23–30. [Google Scholar]
- Chau, C.F.; Huang, Y.L. Characterization of passion fruit seed fibres—A potential fibre source. Food Chem. 2004, 85, 189–194. [Google Scholar] [CrossRef]
- Oliveira, D.A.; Angonese, M.; Gomes, C.; Ferreira, S.R.S. Valorization of passion fruit (Passiflora edulis sp.) by-products: Sustainable recovery and biological activities. J. Supercrit. Fluids 2016, 111, 55–62. [Google Scholar] [CrossRef]
- De Santana, F.C.; de Oliveira Torres, L.R.; Shinagawa, F.B.; de Oliveira, E.S.A.M.; Yoshime, L.T.; de Melo, I.L.P.; Marcellini, P.S.; Mancini-Filho, J. Optimization of the antioxidant polyphenolic compounds extraction of yellow passion fruit seeds (Passiflora edulis Sims) by response surface methodology. J. Food Sci. Technol. 2017, 54, 3552–3561. [Google Scholar] [CrossRef]
- Dos Reis, L.C.R.; Facco, E.M.P.; Salvador, M.; Flôres, S.H.; de Oliveira Rios, A. Antioxidant potential and physicochemical characterization of yellow, purple and orange passion fruit. J. Food Sci. Technol. 2018, 55, 2679–2691. [Google Scholar] [CrossRef]
- Lam, S.K.; Ng, T.B. Passiflin, a novel dimeric antifungal protein from seeds of the passion fruit. Phytomedicine 2009, 16, 172–180. [Google Scholar] [CrossRef]
- Djuricic, I.; Calder, P.C. Beneficial Outcomes of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Human Health: An Update for 2021. Nutrients 2021, 13, 2421. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Lucci, P.; Núñez, O.; Tundis, R.; Balzano, M.; Frega, N.G.; Conte, L.; Moret, S.; Filatova, D.; Moyano, E.; et al. Native Colombian Fruits and Their by-Products: Phenolic Profile, Antioxidant Activity and Hypoglycaemic Potential. Foods 2019, 8, 89. [Google Scholar] [CrossRef] [Green Version]
- Serra, J.L.; Rodrigues, A.; de Freitas, R.A.; Meirelles, A.J.A.; Darnet, S.H.; Silva, L. Alternative sources of oils and fats from Amazonian plants: Fatty acids, methyl tocols, total carotenoids and chemical composition. Food Res. Int. 2019, 116, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Takam, P.N.; Djikeng, F.T.; Kuate, D.; Kengne, A.P.N.; Tsafack, H.D.; Makamwé, I.; Oben, J.E. Passiflora edulis seed oil from west Cameroon: Chemical characterization and assessment of its hypolipidemic effect in high-fat diet-induced rats. Food Sci. Nutr. 2019, 7, 3751–3758. [Google Scholar] [CrossRef] [PubMed]
- Abu-Ouf, N.M.; Jan, M.M. The impact of maternal iron deficiency and iron deficiency anemia on child’s health. Saudi Med. J. 2015, 36, 146–149. [Google Scholar] [CrossRef] [PubMed]
- De Santana, F.C.; Shinagawa, F.B.; Araujo Eda, S.; Costa, A.M.; Mancini-Filho, J. Chemical Composition and Antioxidant Capacity of Brazilian Passiflora Seed Oils. J. Food Sci. 2015, 80, C2647–C2654. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.M.R.; de Figueiredo, E.A.T.; Ricardo, N.M.P.S.; Vieira, I.G.P.; de Figueiredo, R.W.; Brasil, I.M.; Gomes, C.L. Quantification of bioactive compounds in pulps and by-products of tropical fruits from Brazil. Food Chem. 2014, 143, 398–404. [Google Scholar] [CrossRef] [Green Version]
- Matsui, Y.; Sugiyama, K.; Kamei, M.; Takahashi, T.; Suzuki, T.; Katagata, Y.; Ito, T. Extract of passion fruit (Passiflora edulis) seed containing high amounts of piceatannol inhibits melanogenesis and promotes collagen synthesis. J. Agric. Food Chem. 2010, 58, 11112–11118. [Google Scholar] [CrossRef]
- Uchida-Maruki, H.; Inagaki, H.; Ito, R.; Kurita, I.; Sai, M.; Ito, T. Piceatannol lowers the blood glucose level in diabetic mice. Biol. Pharm. Bull. 2015, 38, 629–633. [Google Scholar] [CrossRef] [Green Version]
- Kawakami, S.; Kinoshita, Y.; Maruki-Uchida, H.; Yanae, K.; Sai, M.; Ito, T. Piceatannol and its metabolite, isorhapontigenin, induce SIRT1 expression in THP-1 human monocytic cell line. Nutrients 2014, 6, 4794–4804. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos, L.C.; Mendiola, J.A.; Sánchez-Camargo, A.D.P.; Álvarez-Rivera, G.; Viganó, J.; Cifuentes, A.; Ibáñez, E.; Martínez, J. Selective Extraction of Piceatannol from Passiflora edulis by-Products: Application of HSPs Strategy and Inhibition of Neurodegenerative Enzymes. Int. J. Mol. Sci. 2021, 22, 6248. [Google Scholar] [CrossRef]
- Pan, Z.H.; Ning, D.S.; Fu, Y.X.; Li, D.P.; Zou, Z.Q.; Xie, Y.C.; Yu, L.L.; Li, L.C. Preparative Isolation of Piceatannol Derivatives from Passion Fruit (Passiflora edulis) Seeds by High-Speed Countercurrent Chromatography Combined with High-Performance Liquid Chromatography and Screening for α-Glucosidase Inhibitory Activities. J. Agric. Food Chem. 2020, 68, 1555–1562. [Google Scholar] [CrossRef]
- Krambeck, K.; Oliveira, A.; Santos, D.; Pintado, M.M.; Baptista Silva, J.; Sousa Lobo, J.M.; Amaral, M.H. Identification and Quantification of Stilbenes (Piceatannol and Resveratrol) in Passiflora edulis By-Products. Pharmaceuticals 2020, 13, 73. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, T.; Sato, A.; Takai, Y.; Yoshimori, A.; Umehara, M.; Ogino, Y.; Inada, M.; Shimada, N.; Nishida, A.; Ichida, R.; et al. Effect of piceatannol-rich passion fruit seed extract on human glyoxalase I-mediated cancer cell growth. Biochem. Biophys. Rep. 2019, 20, 100684. [Google Scholar] [CrossRef]
- Sano, S.; Sugiyama, K.; Ito, T.; Katano, Y.; Ishihata, A. Identification of the strong vasorelaxing substance scirpusin B, a dimer of piceatannol, from passion fruit (Passiflora edulis) seeds. J. Agric. Food Chem. 2011, 59, 6209–6213. [Google Scholar] [CrossRef]
- Lourith, N.; Kanlayavattanakul, M. Antioxidant activities and phenolics of Passiflora edulis seed recovered from juice production residue. J. Oleo Sci. 2013, 62, 235–240. [Google Scholar] [CrossRef]
- Taborda, J.A.V.; Arango, W.M.; Méndez Arteaga, J.J.; Guerra Almonacid, C.M. Encapsulation of bioactive compounds from byproducts of two species of passionflowers: Evaluation of the physicochemical properties and controlled release in a gastrointestinal model. Heliyon 2021, 7, e07627. [Google Scholar] [CrossRef]
- Jiménez, Á.A.R.; Méndez, J.J.A.; Murillo, W.A.; Guerrero, M.F.P. Vasodilator effect of ethanolic extracts of Passiflora vitifolia and Passiflora edulis f. edulis seeds. J. Appl. Pharm. Sci. 2021, 11, 61–69. [Google Scholar] [CrossRef]
- Oliver, J.M.; Burg, D.L.; Wilson, B.S.; McLaughlin, J.L.; Geahlen, R.L. Inhibition of mast cell Fc epsilon R1-mediated signaling and effector function by the Syk-selective inhibitor, piceatannol. J. Biol. Chem. 1994, 269, 29697–29703. [Google Scholar] [CrossRef]
- Howitz, K.T.; Bitterman, K.J.; Cohen, H.Y.; Lamming, D.W.; Lavu, S.; Wood, J.G.; Zipkin, R.E.; Chung, P.; Kisielewski, A.; Zhang, L.L.; et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature 2003, 425, 191–196. [Google Scholar] [CrossRef]
- Dai, H.; Sinclair, D.A.; Ellis, J.L.; Steegborn, C. Sirtuin activators and inhibitors: Promises, achievements, and challenges. Pharmacol. Ther. 2018, 188, 140–154. [Google Scholar] [CrossRef]
- Cantos, E.; Espín, J.C.; Fernández, M.J.; Oliva, J.; Tomás-Barberán, F.A. Postharvest UV-C-irradiated grapes as a potential source for producing stilbene-enriched red wines. J. Agric. Food Chem. 2003, 51, 1208–1214. [Google Scholar] [CrossRef]
- Yu, S.Y.; Lee, Y.J.; Kim, J.D.; Kang, S.N.; Lee, S.K.; Jang, J.Y.; Lee, H.K.; Lim, J.H.; Lee, O.H. Phenolic composition, antioxidant activity and anti-adipogenic effect of hot water extract from safflower (Carthamus tinctorius L.) seed. Nutrients 2013, 5, 4894–4907. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Batalla, L.; Widholm, J.M.; Fahey, G.C., Jr.; Castaño-Tostado, E.; Paredes-López, O. Chemical components with health implications in wild and cultivated Mexican common bean seeds (Phaseolus vulgaris L.). J. Agric. Food Chem. 2006, 54, 2045–2052. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Salehi, B.; Sharifi-Rad, J.; Aslam Gondal, T.; Saeed, F.; Imran, A.; Shahbaz, M.; Tsouh Fokou, P.V.; Umair Arshad, M.; Khan, H.; et al. Kaempferol: A Key Emphasis to Its Anticancer Potential. Molecules 2019, 24, 2277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Da Silva Francischini, D.; Lopes, A.P.; Segatto, M.L.; Stahl, A.M.; Zuin, V.G. Development and application of green and sustainable analytical methods for flavonoid extraction from Passiflora waste. BMC Chem. 2020, 14, 56. [Google Scholar] [CrossRef]
- Yepes, A.; Ochoa-Bautista, D.; Murillo-Arango, W.; Quintero-Saumeth, J.; Bravo, K.; Osorio, E. Purple passion fruit seeds (Passiflora edulis f. edulis Sims) as a promising source of skin anti-aging agents: Enzymatic, antioxidant and multi-level computational studies. Arab. J. Chem. 2021, 14, 102905. [Google Scholar] [CrossRef]
- Wen, H.; Fu, Z.; Wei, Y.; Zhang, X.; Ma, L.; Gu, L.; Li, J. Antioxidant Activity and Neuroprotective Activity of Stilbenoids in Rat Primary Cortex Neurons via the PI3K/Akt Signalling Pathway. Molecules 2018, 23, 2328. [Google Scholar] [CrossRef] [Green Version]
- Kandandapani, S.; Balaraman, A.K.; Ahamed, H.N. Extracts of passion fruit peel and seed of Passiflora edulis (Passifloraceae) attenuate oxidative stress in diabetic rats. Chin. J. Nat. Med. 2015, 13, 680–686. [Google Scholar] [CrossRef]
- Yepes, D.F.M.; Arango, W.M.; Rodríguez, Á.A.J.; Arteaga, J.J.M.; Porras, Á.E.A. Encapsulation of phenols of gulupa seed extract using acylated rice starch: Effect on the release and antioxidant activity. J. Funct. Foods 2021, 87, 104788. [Google Scholar] [CrossRef]
- Rotta, E.M.; Giroux, H.J.; Lamothe, S.; Bélanger, D.; Sabik, H.; Visentainer, J.V.; Britten, M. Use of passion fruit seed extract (Passiflora edulis Sims) to prevent lipid oxidation in dairy beverages during storage and simulated digestion. LWT 2020, 123, 109088. [Google Scholar] [CrossRef]
- Ferreira, B.S.; de Almeida, C.G.; Faza, L.P.; de Almeida, A.; Diniz, C.G.; da Silva, V.L.; Grazul, R.M.; Le Hyaric, M. Comparative properties of Amazonian oils obtained by different extraction methods. Molecules 2011, 16, 5875–5885. [Google Scholar] [CrossRef] [Green Version]
- Hartanto, S.; Lister, I.N.E.; Fachrial, E. A Comparative Study of Peel and Seed Extract of Passion Fruit (Passiflora edulis) as Anti Collagenase. Am. Sci. Res. J. Eng. Technol. Sci. 2019, 54, 42–48. [Google Scholar]
- Vera, K.; Raif, A.; Ikhtiari, R. Antioxidant and Anti-elastase Activity of Seed and Peel Extract of P. edulis. Am. Sci. Res. J. Eng. Technol. Sci. 2019, 53, 43–48. [Google Scholar]
- Chung, J.H.; Seo, J.Y.; Choi, H.R.; Lee, M.K.; Youn, C.S.; Rhie, G.; Cho, K.H.; Kim, K.H.; Park, K.C.; Eun, H.C. Modulation of skin collagen metabolism in aged and photoaged human skin in vivo. J. Investig. Dermatol. 2001, 117, 1218–1224. [Google Scholar] [CrossRef] [Green Version]
- Varani, J.; Dame, M.K.; Rittie, L.; Fligiel, S.E.; Kang, S.; Fisher, G.J.; Voorhees, J.J. Decreased collagen production in chronologically aged skin: Roles of age-dependent alteration in fibroblast function and defective mechanical stimulation. Am. J. Pathol. 2006, 168, 1861–1868. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.; Fisher, G.J.; Voorhees, J.J. Photoaging and topical tretinoin: Therapy, pathogenesis, and prevention. Arch. Dermatol. 1997, 133, 1280–1284. [Google Scholar] [CrossRef]
- Kim, S.; Kim, Y.; Lee, Y.; Chung, J.H. Ceramide accelerates ultraviolet-induced MMP-1 expression through JAK1/STAT-1 pathway in cultured human dermal fibroblasts. J. Lipid Res. 2008, 49, 2571–2581. [Google Scholar] [CrossRef] [Green Version]
- Dong, K.K.; Damaghi, N.; Picart, S.D.; Markova, N.G.; Obayashi, K.; Okano, Y.; Masaki, H.; Grether-Beck, S.; Krutmann, J.; Smiles, K.A.; et al. UV-induced DNA damage initiates release of MMP-1 in human skin. Exp. Dermatol. 2008, 17, 1037–1044. [Google Scholar] [CrossRef]
- Kang, S.; Chung, J.H.; Lee, J.H.; Fisher, G.J.; Wan, Y.S.; Duell, E.A.; Voorhees, J.J. Topical N-acetyl cysteine and genistein prevent ultraviolet-light-induced signaling that leads to photoaging in human skin in vivo. J. Investig. Dermatol. 2003, 120, 835–841. [Google Scholar] [CrossRef]
- Maruki-Uchida, H.; Kurita, I.; Sugiyama, K.; Sai, M.; Maeda, K.; Ito, T. The protective effects of piceatannol from passion fruit (Passiflora edulis) seeds in UVB-irradiated keratinocytes. Biol. Pharm. Bull. 2013, 36, 845–849. [Google Scholar] [CrossRef] [Green Version]
- Yamamoto, T.; Setoguchi, Y.; Mori, S.; Morita, M.; Yano, S.; Maeda, K. Effects of oral intake of piceatannol on skin moisture—A randomized, double-blind, placebo-controlled parallel-group, comparison study. Jpn. Pharmacol. Ther. 2018, 46, 1191–1199. [Google Scholar]
- Yokozawa, T.; Kim, Y.J. Piceatannol inhibits melanogenesis by its antioxidative actions. Biol. Pharm. Bull. 2007, 30, 2007–2011. [Google Scholar] [CrossRef] [Green Version]
- Krambeck, K.; Silva, V.; Silva, R.; Fernandes, C.; Cagide, F.; Borges, F.; Santos, D.; Otero-Espinar, F.; Lobo, J.M.S.; Amaral, M.H. Design and characterization of Nanostructured lipid carriers (NLC) and Nanostructured lipid carrier-based hydrogels containing Passiflora edulis seeds oil. Int. J. Pharm. 2021, 600, 120444. [Google Scholar] [CrossRef]
- Krambeck, K.; Santos, D.; Otero-Espinar, F.; Sousa Lobo, J.M.; Amaral, M.H. Lipid nanocarriers containing Passiflora edulis seeds oil intended for skin application. Colloids Surf. B Biointerfaces 2020, 193, 111057. [Google Scholar] [CrossRef]
- Dewi, N.K.; Putra, I.B.; Jusuf, N.K. Passion fruit purple variant (Passiflora edulis Sims var. edulis) seeds extract 10% cream in acne vulgaris treatment: An open-label pilot study. Int. J. Dermatol. 2020, 59, 1506–1512. [Google Scholar] [CrossRef]
- Jusuf, N.K.; Putra, I.B.; Dewi, N.K. Antibacterial Activity of Passion Fruit Purple Variant (Passiflora edulis Sims var. edulis) Seeds Extract Against Propionibacterium acnes. Clin. Cosmet. Investig. Dermatol. 2020, 13, 99–104. [Google Scholar] [CrossRef] [Green Version]
- Aryunisari, C.G.; Putra, I.B.; Jusuf, N.K. Effect of Purple Passion Fruit Extract Cream (Passiflora edulis Sims var. Edulis) 6% against Striae Distensae. Open Access Maced. J. Med. Sci. 2021, 9, 720–725. [Google Scholar] [CrossRef]
- Ishihata, A.; Maruki-Uchida, H.; Gotoh, N.; Kanno, S.; Aso, Y.; Togashi, S.; Sai, M.; Ito, T.; Katano, Y. Vascular- and hepato-protective effects of passion fruit seed extract containing piceatannol in chronic high-fat diet-fed rats. Food Funct. 2016, 7, 4075–4081. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Shiokoshi, M.; Kawawa, R.; Ishikawa, T.; Ichi, I.; Mori, S.; Morita, M. Abstracts of the Asian Congress of Nutrition 2019. Ann. Nutr. Metab. 2019, 75, 1–424. [Google Scholar] [CrossRef]
- Tung, Y.C.; Lin, Y.H.; Chen, H.J.; Chou, S.C.; Cheng, A.C.; Kalyanam, N.; Ho, C.T.; Pan, M.H. Piceatannol Exerts Anti-Obesity Effects in C57BL/6 Mice through Modulating Adipogenic Proteins and Gut Microbiota. Molecules 2016, 21, 1419. [Google Scholar] [CrossRef] [Green Version]
- Adrianus, D.T.; Kawakami, S.; Mori, S.; Morita, M.; Yano, S. Effects of Oral Intake of Piceatannol on Fat Burning―A Randomized, Double-blind, Placebo-controlled Crossover Comparison Study. Jpn. Pharmacol. Ther. 2020, 48, 1235–1240. [Google Scholar]
- Matsui, N.; Uchida-Maruki, H.; Yamamoto, T.; Ito, R.; Ebisihara, S.; Morita, M. Effects of Oral Intake of Piceatannol on Fat Burning During Moderate-Intensity Exercise—A Randomized, Double-blind, Placebo-controlled Crossover Comparison Study. Jpn. Pharmacol. Ther. 2021, 49, 731–738. [Google Scholar]
- Lee, H.J.; Kang, M.G.; Cha, H.Y.; Kim, Y.M.; Lim, Y.; Yang, S.J. Effects of Piceatannol and Resveratrol on Sirtuins and Hepatic Inflammation in High-Fat Diet-Fed Mice. J. Med. Food 2019, 22, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Purushotham, A.; Schug, T.T.; Xu, Q.; Surapureddi, S.; Guo, X.; Li, X. Hepatocyte-specific deletion of SIRT1 alters fatty acid metabolism and results in hepatic steatosis and inflammation. Cell Metab. 2009, 9, 327–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, J.S.; Tongson, J.; Kim, K.H.; Park, Y. Piceatannol attenuates fat accumulation and oxidative stress in steatosis-induced HepG2 cells. Curr. Res. Food. Sci. 2020, 3, 92–99. [Google Scholar] [CrossRef]
- Carpéné, C.; Pejenaute, H.; Del Moral, R.; Boulet, N.; Hijona, E.; Andrade, F.; Villanueva-Millán, M.J.; Aguirre, L.; Arbones-Mainar, J.M. The Dietary Antioxidant Piceatannol Inhibits Adipogenesis of Human Adipose Mesenchymal Stem Cells and Limits Glucose Transport and Lipogenic Activities in Adipocytes. Int. J. Mol. Sci. 2018, 19, 2081. [Google Scholar] [CrossRef] [Green Version]
- Oritani, Y.; Okitsu, T.; Nishimura, E.; Sai, M.; Ito, T.; Takeuchi, S. Enhanced glucose tolerance by intravascularly administered piceatannol in freely moving healthy rats. Biochem. Biophys. Res. Commun. 2016, 470, 753–758. [Google Scholar] [CrossRef]
- Kitada, M.; Ogura, Y.; Maruki-Uchida, H.; Sai, M.; Suzuki, T.; Kanasaki, K.; Hara, Y.; Seto, H.; Kuroshima, Y.; Monno, I.; et al. The Effect of Piceatannol from Passion Fruit (Passiflora edulis) Seeds on Metabolic Health in Humans. Nutrients 2017, 9, 1142. [Google Scholar] [CrossRef] [Green Version]
- Mota, N.; Kviecinski, M.R.; Zeferino, R.C.; de Oliveira, D.A.; Bretanha, L.C.; Ferreira, S.R.S.; Micke, G.A.; Filho, D.W.; Pedrosa, R.C.; Ourique, F. In vivo antitumor activity of by-products of Passiflora edulis f. flavicarpa Deg. Rich in medium and long chain fatty acids evaluated through oxidative stress markers, cell cycle arrest and apoptosis induction. Food Chem. Toxicol. 2018, 118, 557–565. [Google Scholar] [CrossRef]
- Kido, L.A.; Hahm, E.R.; Kim, S.H.; Baseggio, A.M.; Cagnon, V.H.A.; Singh, S.V.; Maróstica, M.R., Jr. Prevention of Prostate Cancer in Transgenic Adenocarcinoma of the Mouse Prostate Mice by Yellow Passion Fruit Extract and Antiproliferative Effects of Its Bioactive Compound Piceatannol. J. Cancer. Prev. 2020, 25, 87–99. [Google Scholar] [CrossRef]
- Banik, K.; Ranaware, A.M.; Harsha, C.; Nitesh, T.; Girisa, S.; Deshpande, V.; Fan, L.; Nalawade, S.P.; Sethi, G.; Kunnumakkara, A.B. Piceatannol: A natural stilbene for the prevention and treatment of cancer. Pharmacol Res. 2020, 153, 104635. [Google Scholar] [CrossRef]
- Setoguchi, Y.; Mori, S.; Oki, M.; Morita, M. Safety evaluation study on overdose of passion fruit seed extract in healthy subjects—An open-label trial. Jpn. Pharmacol. Ther. 2018, 46, 911–918. [Google Scholar]
- Shimotsuma, S.; Mori, S.; Morita, M.; Ando, E.; Itoh, M. Safety evaluation of long-term oral intake of passion fruit seeds extract on healthy subjects—An open-label study. Jpn. Pharmacol. Ther. 2020, 48, 1615–1622. [Google Scholar]
| Components | P. edulis | P. edulis | P. edulis f. flavicarpa | P. edulis f. edulis |
|---|---|---|---|---|
| Nutrient Content (g/100 g Seeds on Dry Basis) | ||||
| Protein | 13.99 ± 0.11 | 17.57 ± 0.31 | 13.07 ± 0.12 | 13.23 ± 0.48 |
| Lipids | 32.65 ± 0.45 | 31.16 ± 0.31 | 12.31 ± 0.68 | 14.94 ± 0.41 |
| Carbohydrates | 51.87 ± 0.00 | 49.44 ± 1.16 | 71.07 ± 0.00 | 69.98 ± 0.00 |
| Ash | 1.49 ± 0.01 | 1.82 ± 0.04 | 3.56 ± 0.05 | 1.85 ± 0.06 |
| Dietary fiber | 48.18 ± 0.64 | na | 65.60 ± 0.52 | 55.06 ± 0.35 |
| Mineral Content (mg/100 g Seeds on Dry Basis) | ||||
| Sodium | 241.7 ± 83.3 | na | 3.46 ± 0.07 | 4.80 ± 0.03 |
| Magnesium | 138.3 ± 220.5 | na | 150 ± 1.10 | 290 ± 1.80 |
| Potassium | 352.5 ± 144.3 | na | 760 ± 6.40 | 112 ± 3.00 |
| Calcium | 173.1 ± 294.8 | 27.46 ± 2.66 | 30.00 ± 0.35 | 6.00 ± 0.02 |
| Phosphorus | 115.3 ± 178.8 | 240.05 ± 7.78 | 310 ± 2.05 | 63.00 ± 1.19 |
| Iron | 6.2 ± 11.7 | 7.27 ± 0.27 | 5.20 ± 0.02 | 4.30 ± 0.03 |
| Copper | 1.4 ± 10 | 0.89 ± 0.04 | 0.90 ± 0.02 | 0.70 ± 0.02 |
| Manganese | na | 1.16 ± 0.02 | 2.20 ± 0.05 | 2.30 ± 0.03 |
| Zinc | 5.6 ± 24.7 | 3.72 ± 0.13 | 4.10 ± 0.09 | 4.60 ± 0.05 |
| Reference | [26] | [29] | [30] | [30] |
| Components | P. edulis f. flavicarpa | P. edulis | P. edulis | P. edulis |
|---|---|---|---|---|
| Fatty acid profile (%) | ||||
| Myristic | trace | 0.10 ± 0.00 | 0.08 | 0.1 |
| Palmitic | 9.73 ± 0.01 | 11.00 ± 0.17 | 10.77 | 11.72 |
| Palmitoleic | 0.11 ± 0.01 | 0.22 ± 0.01 | 0.18 | 0.34 |
| Stearic | 2.58 ± 0.01 | 3.29 ± 0.31 | 2.98 | 2.84 |
| Oleic | 13.83 ± 0.04 | 16.84 ± 0.36 | 16.06 | 14.31 |
| Vaccenic | na | 0.17 ± 0.00 | na | na |
| Linoleic | 73.14 ± 0.05 | 67.39 ± 0.54 | 69.22 | 68.39 |
| α-Linolenic | 0.41 ± 0.00 | 0.56 ± 0.03 | 0.17 | 0.54 |
| Araquidic | 0.10 ± 0.01 | na | 0.43 | 0.16 |
| Gadoleic | na | na | na | 0.15 |
| Eicosenoic | 0.10 ± 0.00 | na | na | na |
| Behenic | na | 0.10 ± 0.00 | na | 0.24 |
| Cetoleic | na | na | na | 1.15 |
| Total SFA (%) | 12.41 | 14.69 ± 0.12 | 14.36 | 15.06 |
| Total MUFA (%) | 14.04 | 17.18 ± 0.47 | 16.24 | 15.95 |
| Total PUFA (%) | 73.55 | 68.12 ± 0.58 | 69.39 | 68.93 |
| Reference | [10] | [29] | [34] | [35] |
| Compound | Species | Extraction Solvent | Reference |
|---|---|---|---|
| Stilbene | |||
| Piceatannol | P. edulis (purple) | 80% ethanol | [40] |
| P. edulis | 80% ethanol | [41] | |
| P. edulis (yellow) | ethanol | [29] | |
| P. edulis | 79% ethanol | [42] | |
| P. edulis | 95% ethanol | [43] | |
| P. edulis | ethanol and acetone | [44] | |
| P. edulis | 35% ethanol | [45] | |
| P. edulis | 90% ethanol | [46] | |
| P. edulis | 70% acetone | [39] | |
| Resveratrol | P. edulis | ethanol and acetone | [44] |
| P. edulis | 35% ethanol | [45] | |
| P. edulis | 90% ethanol | [46] | |
| P. edulis | 70% acetone | [39] | |
| Scirpusin B | P. edulis | 95% ethanol | [43] |
| P. edulis | 35% ethanol | [45] | |
| P. edulis | 80% ethanol | [41] | |
| P. edulis | 90% ethanol | [46] | |
| Isorhapontigenin | P. edulis | 35% ethanol | [45] |
| Rhapontigenin | P. edulis | 35% ethanol | [45] |
| Cassigarol D | P. edulis | 95% ethanol | [43] |
| Cyperusphenol B | P. edulis | 95% ethanol | [43] |
| Cyperusphenol D | P. edulis | 95% ethanol | [43] |
| Astringin | P. edulis | 35% ethanol | [45] |
| Piceid | P. edulis | 35% ethanol | [45] |
| Pinostilbene | P. edulis | 35% ethanol | [45] |
| Gnetin C | P. edulis | 35% ethanol | [45] |
| Phenolic acid | |||
| Caffeic acid | P. edulis | 35% ethanol | [45] |
| P. edulis (purple) | 40% methanol | [47] | |
| Chlorogenic acid | P. edulis (purple) | 40% methanol | [47] |
| Ferulic acid | P. edulis (purple) | 40% methanol | [47] |
| Gallic acid | P. edulis (purple) | 40% methanol | [47] |
| Rosmarinic acid | P. edulis (purple) | 40% methanol | [47] |
| Coumarin | P. edulis | 50% ethanol, 70% acetone | [38] |
| p-coumaric acid | P. edulis f. edulis | ethanol | [48] |
| P. edulis | 35% ethanol | [45] | |
| Flavonoid | |||
| Epicatechin | P. edulis | 35% ethanol | [45] |
| Quercetin | P. edulis | 40% methanol | [47] |
| P. edulis f. edulis | ethanol | [48] | |
| Rutin | P. edulis f. edulis | ethanol | [48] |
| Isoquercetin | P. edulis f. edulis | ethanol | [48] |
| Kaempferol | P. edulis f. flavicarpa | ethanol | [30] |
| Malvidin 3,5-diglucoside | P. edulis f. flavicarpa, P. edulis f. edulis | ethanol | [30] |
| Orientin | P. edulis f. edulis | 97% ethanol | [49] |
| Isoorientin | P. edulis f. edulis | 97% ethanol | [49] |
| Vitexin | P. edulis f. edulis | 97% ethanol | [49] |
| Isovitexin | P. edulis f. edulis | 97% ethanol | [49] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawakami, S.; Morinaga, M.; Tsukamoto-Sen, S.; Mori, S.; Matsui, Y.; Kawama, T. Constituent Characteristics and Functional Properties of Passion Fruit Seed Extract. Life 2022, 12, 38. https://doi.org/10.3390/life12010038
Kawakami S, Morinaga M, Tsukamoto-Sen S, Mori S, Matsui Y, Kawama T. Constituent Characteristics and Functional Properties of Passion Fruit Seed Extract. Life. 2022; 12(1):38. https://doi.org/10.3390/life12010038
Chicago/Turabian StyleKawakami, Shinpei, Makoto Morinaga, Sakuka Tsukamoto-Sen, Sadao Mori, Yuko Matsui, and Toshihiro Kawama. 2022. "Constituent Characteristics and Functional Properties of Passion Fruit Seed Extract" Life 12, no. 1: 38. https://doi.org/10.3390/life12010038
APA StyleKawakami, S., Morinaga, M., Tsukamoto-Sen, S., Mori, S., Matsui, Y., & Kawama, T. (2022). Constituent Characteristics and Functional Properties of Passion Fruit Seed Extract. Life, 12(1), 38. https://doi.org/10.3390/life12010038





