Quantitative Evaluation of the Movement Distance of Deep Fascia and Change of Muscle Shape Related to Chain Response in Fascia Tissue of Lower Limb
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participant
2.1.1. Reliability Considerations
2.1.2. Verification Experiment of D-Fascia Response
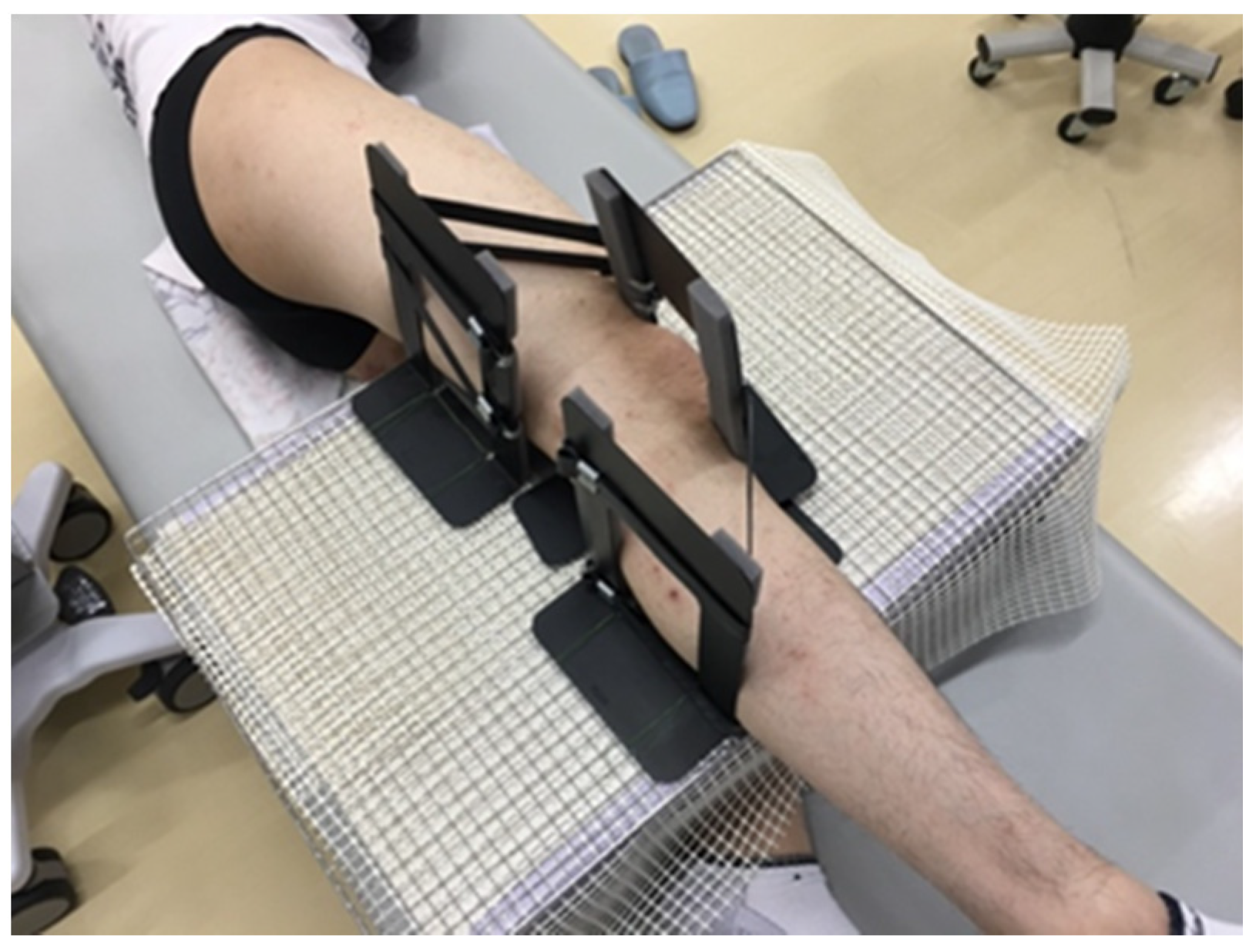
2.2. Measurement Methods
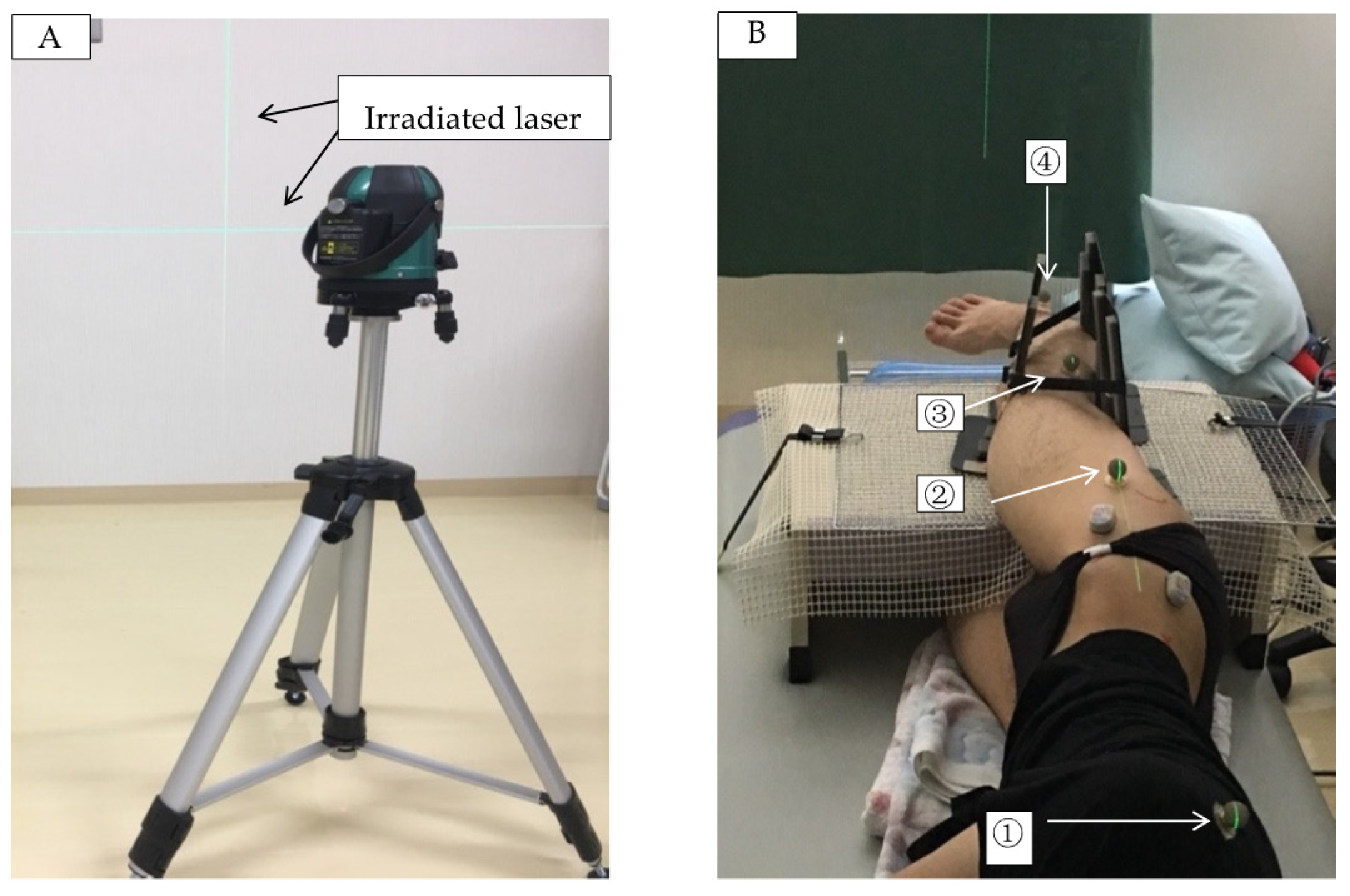
2.2.1. Measurement Position
2.2.2. Measurement by Ultrasonic Equipment
2.2.3. Electromyography
2.2.4. Measurement Task
2.2.5. Data Analysis
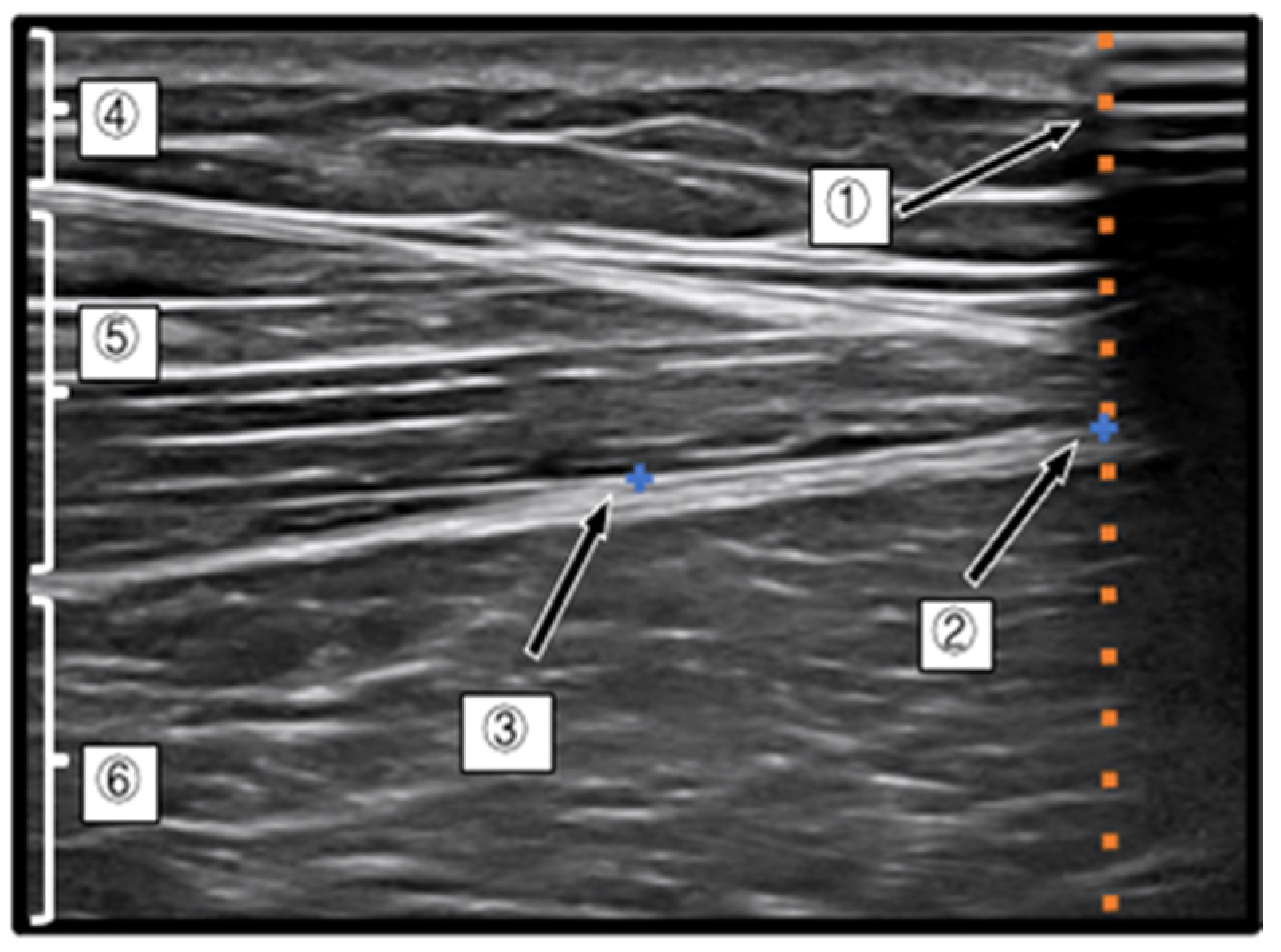
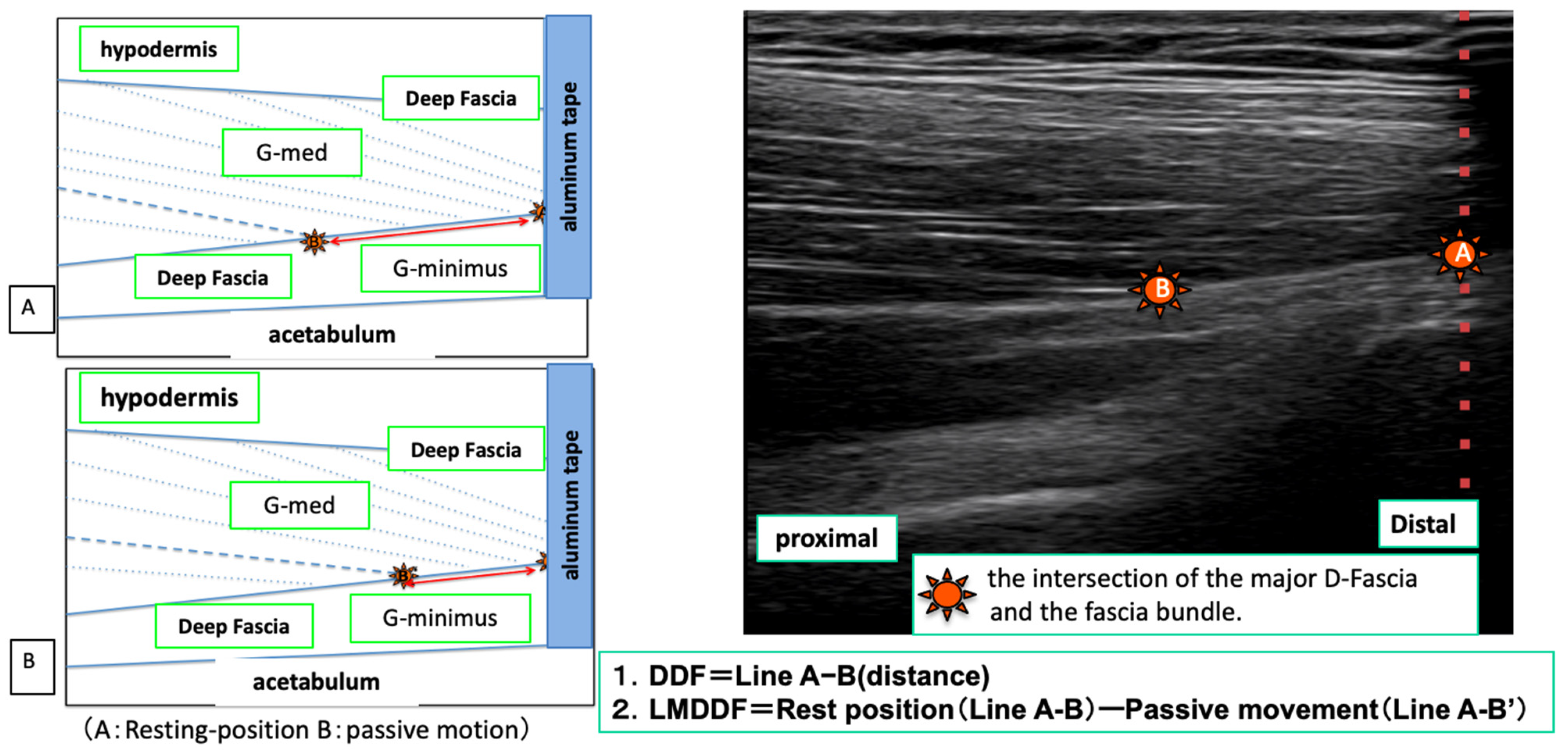
Measurement of the Index Point of the D-Fascia and Calculation of the Movement Distance
Measurement of Changes in Muscle Shape
Surface Electromyogram Measurement
2.2.6. Reliability Considerations
2.3. Statistical Processing
3. Results
3.1. Surface Electromyogram Analysis
3.2. Ultrasound Image Analysis
3.2.1. Reliability Considerations
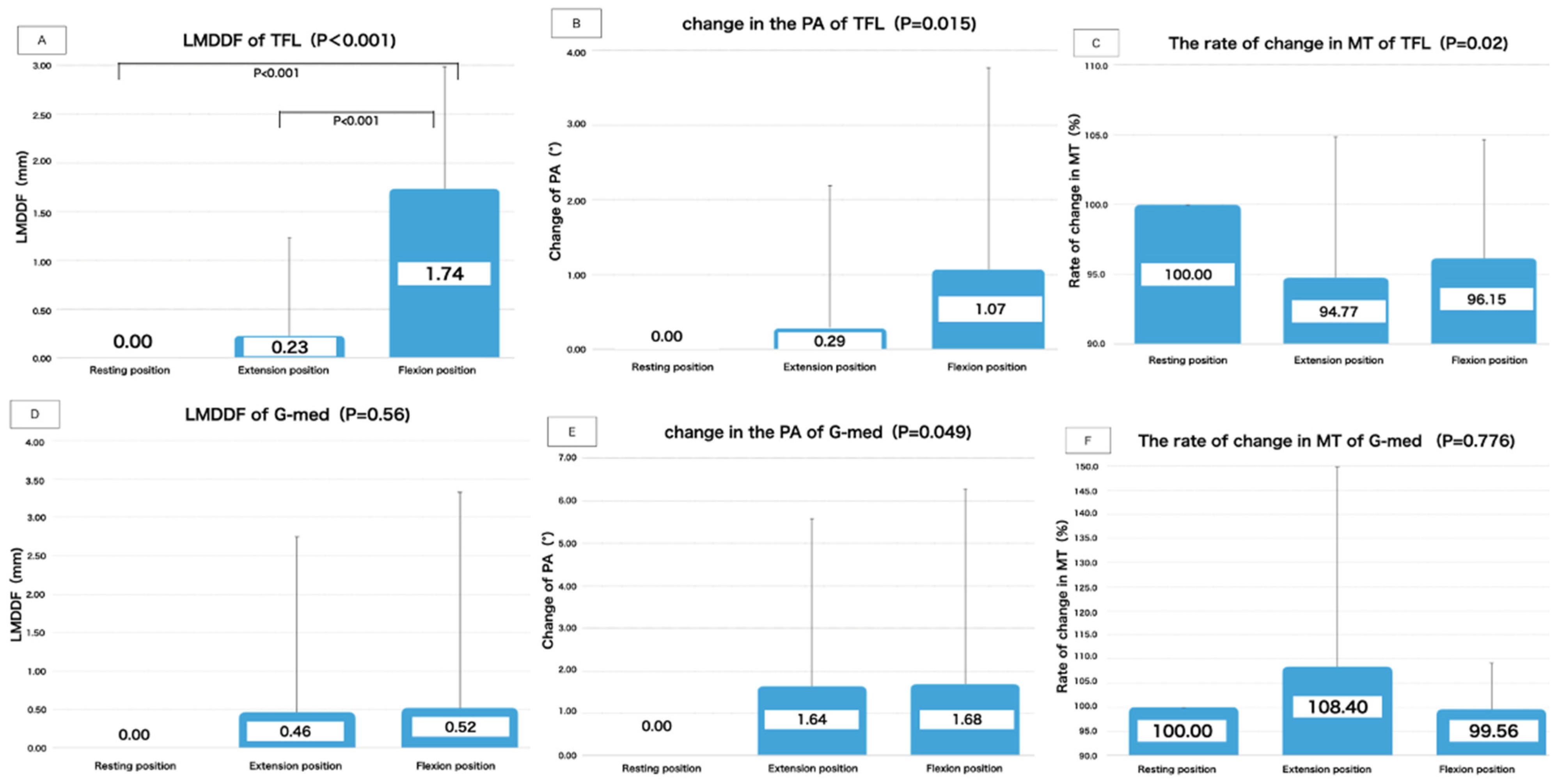
3.2.2. Results of Calculation Items
Results of TFL Calculation Items
Results of G-Med Calculation Items
4. Discussion
4.1. Summary of Research and What Is New
4.2. For Each Parameter of the TFL
4.3. For Each Parameter of the G-Med
4.4. Clinical Application
4.5. Research Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simmonds, N.; Miller, P.; Gemmell, H. A theoretical framework for the role of fascia in manual therapy. J. Bodyw. Mov. Ther. 2012, 16, 83–93. [Google Scholar] [CrossRef]
- Roman, M.; Chaudhry, H.; Bukiet, B.; Stecco, A.; Findley, T.W. Mathematical analysis of the flow of hyaluronic acid around fascia during manual therapy motions. J. Am. Osteopath. Assoc. 2013, 113, 600–610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lancerotto, L.; Stecco, C.; Macchi, V. Layers of the abdominal wall: Anatomical investigation of subcutaneous tissue and superficial fascia. Surg. Radiol. Anat. 2011, 33, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Stecco, A.; Stern, R.; Fantoni, I.; De Caro, R.; Stecco, C. Fascial disorders: Implications for treatment. PM R 2016, 8, 161–168. [Google Scholar] [CrossRef]
- Stecco, A.; Gilliar, W.; Stecco, C. The anatomical and functional relation between gluteus maximus and fascia lata. J. Bodyw. Mov. Ther. 2013, 17, 512–517. [Google Scholar]
- Wilk, J.; Engeroff, T.; Nürnberger, F.; Vogt, L.; Banzer, W. Anatomical study of the morphological continuity between iliotibial tract and the fibularis longus fascia. Surg. Radiol. Anat. 2016, 38, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Reeves, N.D.; Narici, M.V. Behavior of human muscle fascicles during shortening and lengthening contractions in vivo. J. Appl. Physiol. 2003, 95, 1090–1096. [Google Scholar] [CrossRef] [Green Version]
- Narici, M.V.; Binzoni, T.; Hiltbrand, E.; Fasel, J.; Terrier, F.; Cerretelli, P. In vivo human gastrocnemius architecture with changing joint angle at rest and during graded isometric contraction. J. Physiol. 1996, 496 Pt 1, 287–297. [Google Scholar] [CrossRef]
- Stecco, C.; Stern, R.; Porzionato, A.; Macchi, V.; Masiero, S.; Stecco, A.; De Caro, R. Hyaluronan with fascia in the etiology of myofascial pain. Surg. Radiol. Anat. 2011, 33, 891–896. [Google Scholar] [CrossRef]
- Fukunaga, T.; Ichinose, Y.; Ito, M.; Kawakami, Y.; Fukashiro, S. Determination of fascicle length and pennation in a contracting human muscle in vivo. J. Appl. Physiol. 1997, 82, 354–358. [Google Scholar] [CrossRef]
- Kijima, H.; Minagawa, H.; Tomioka, T.; Yamamoto, N.; Kikuchi, K.; Abe, H.; Shimada, Y. Elasticity of the coracoacromial ligament in live shoulders with rotator cuff tear: Measurement using ultrasound elastography. Shoulder Jt. 2008, 32, 357–360. [Google Scholar]
- Ichikawa, K.; Usa, H.; Ogawa, D. The reliability of displacement measurement of the deep fascia using ultrasonographic imaging. J. Jpn. Health Sci. 2013, 16, 21–28. [Google Scholar]
- Hodges, P.W.; Pengel, L.H.M.; Herbert, R.D.; Gandevia, S.C. Measurement of muscle contraction with ultrasound imaging. Muscle Nerve 2003, 27, 682–692. [Google Scholar] [CrossRef]
- Toshinori, S. The absolute reliability of evaluation. Rigakuryoho Kagaku 2011, 26, 451–461. [Google Scholar]
- Faber, M.J.; Bosscher, R.J.; van Wieringen, P.C. Clinical properties of the performance-oriented mobility assessment. Phys. Ther. 2006, 86, 944–954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myers, T.W. Anatomy Trains, 2nd ed.; Elsevier: Amsterdam, the Netherlands, 2009; pp. 75–248. [Google Scholar]
- Stecco, L. Sequenze Neuro-Miofasciali e Meridiani Agopunturei; Dal Molin: Arzignano, Italy, 1988; pp. 11–23. [Google Scholar]
- Stecco, L. La Manipolazione Neuroconnetivale; Marrap-ese: Rome, Italy, 1996; pp. 45–62. [Google Scholar]
- Stecco, L. Fascial Manipulation for Musculoskeletal; Piccin: Padova, Italy, 2017; pp. 9–18. [Google Scholar]
- Mitomo, S.; Ichikawa, K.; Usa, H. The inter- and intra-rater reliability of changing muscle shap of each fiber of gluteus medius during the isometric hip abduction contraction of different movement directions using ultrasound imaging. J. Jpn. Health Sci. 2014, 17, 5–13. [Google Scholar]
- Shimono, T. Surface EMG Manual: Basics and Application; Sakai Medical: Tokyo, Japan, 2010; pp. 123–159. [Google Scholar]
- Stecco, C.; Macchi, V.; Porzionato, A.; Morra, A.; Parenti, A.; Stecco, A.; Delmas, V.; De Caro, R. The ankle retinacula: Morphological evidence of the proprioceptive role of the fascial system. Cells Tissues Organs 2010, 192, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Huijing, P.A.; Jaspers, R.T. Adaptation of muscle size and myofascial force transmission: A review and some new experimental results. Scand. J. Med. Sci. Sports 2005, 15, 349–380. [Google Scholar] [CrossRef] [PubMed]
- Stecco, L. Atlas Physiology of the Muscular Fascia; Piccin: Padova, Italy, 2016; pp. 248–259. [Google Scholar]
- Stecco, C. Functional Atlas of the Human Fascial System; Elsevier: Edinburgh, UK, 2015; pp. 67–72. [Google Scholar]
- Langevin, H.M.; Bouffard, N.A.; Badger, G.J. Dynamic fibroblast cytoskeletal response to subcutaneous tissue stretch ex vivo and in vivo. Am. J. Physiol. Cell Physiol. 2005, 288, C747–C756. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; Bouffard, N.A.; Badger, G.J. Subcutaneous tissue fibroblast cytoskeletal remodeling induced by acupuncture: Evidence for a mechanotransduction-based mechanism. J. Cell Physiol. 2006, 207, 767–774. [Google Scholar] [CrossRef]
- Benjamin, M. The fascia of the limbs and back: A review. J. Anat. 2009, 214, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Schleip, R.; Findley, T.W.; Chaitow, L.; Huijing, P.A. Fascia: The Tensional Network of Human Body, the Science and Clinical Applications in Manual and Movement Therapy, 1st ed.; Elsevier: Edinburgh, UK, 2012; Volume XV, p. 67. [Google Scholar]
- Kahle, V.W.; Leonhardt, H.; Platzer, W. Taschenatlas der Anatomie; Bunkodo: Tokyo, Japan, 1990; pp. 118–130. [Google Scholar]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]

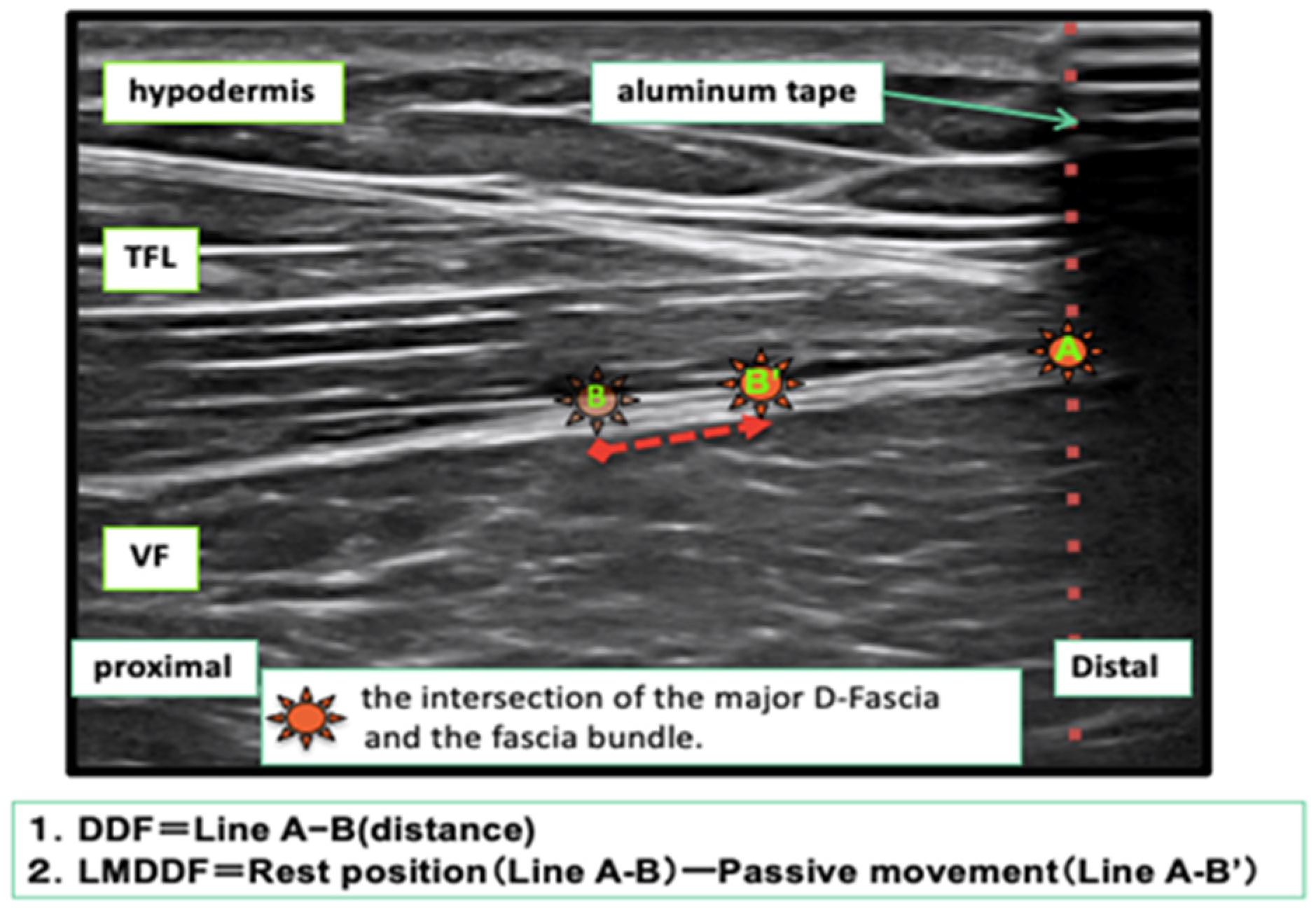
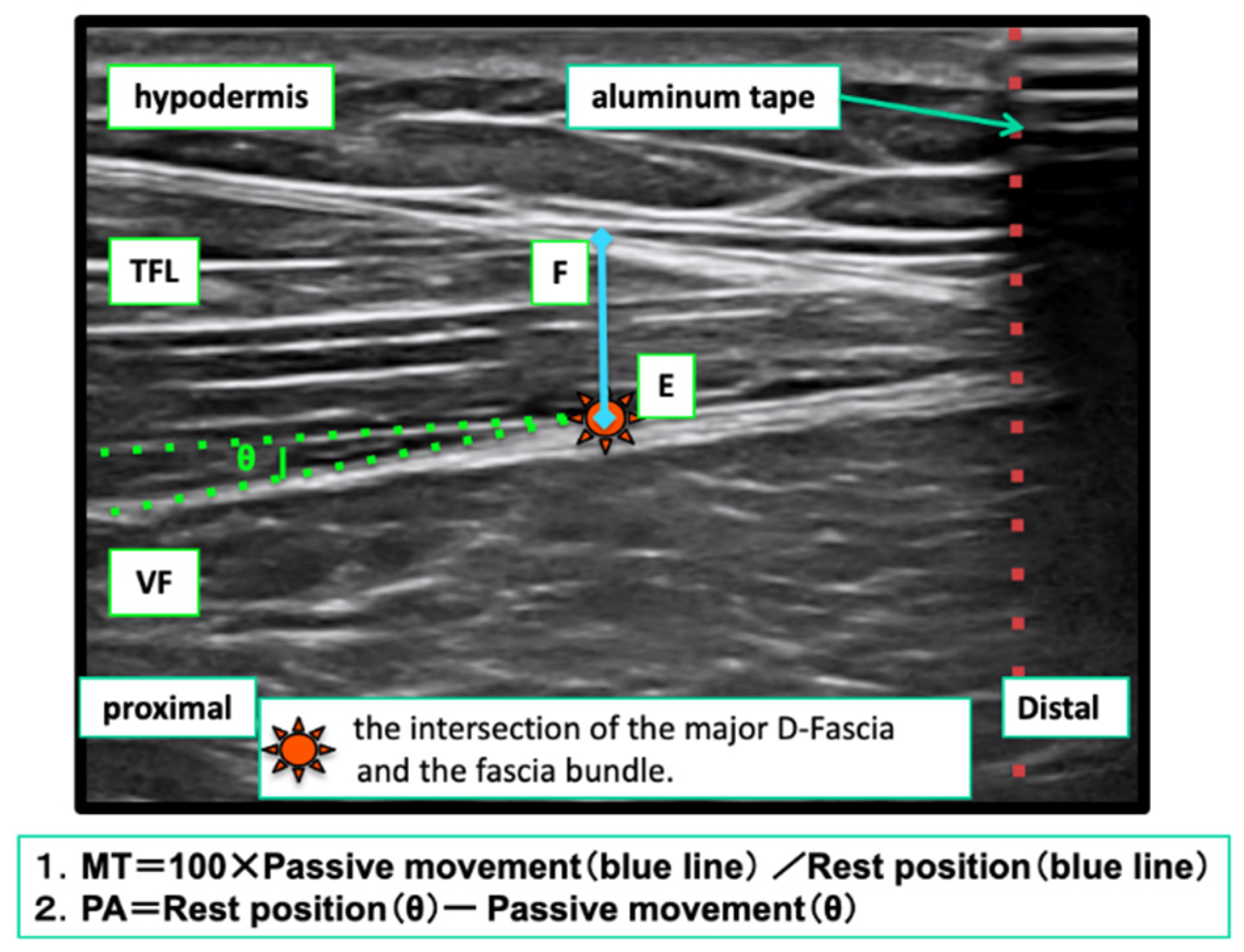
| Item | Mean (SD) |
|---|---|
| gender | 21 males |
| Age | 24 years old (±4.1) |
| Height | 171.8 cm (±6.43) |
| Weight | 66.9 kg (±7.15) |
| BMI | 22.7 (2.71) |
| ROM of Ankle joint | |
| Dorsiflexion | 17.9° (±5) |
| Plantar flexion | 45.8° (±3.35) |
| Horizontal angle of test limb | 0.28°(±0.21) |
| Measured (1st Mean vs. 2nd Mean) n = 11 | |||||||||
| Reliability of TFL | |||||||||
| Item | Limb Position | ICC(1, 1) | Additive Error | Proportional Error | MDC (Minimal Detectable Change) {mm} | Standard Error (SE) | MDC95 | ||
| 95% CI of the Mean of the Differences | Yes or No | Testing for Correlation Dominance | Yes or No | ||||||
| LMDDF | Resting | 0.97 | −0.79~0.77 | no | p = 0.15 | no | 2.28 | 0.35 | 0.97 |
| extension | 0.97 | −0.81~0.55 | no | p = 0.2 | no | 1.97 | 0.30 | 0.84 | |
| flexion | 0.97 | −1.00~0.61 | no | p = 0.76 | no | 2.36 | 0.36 | 1.01 | |
| PA | Resting | 0.74 | −1.79~1.21 | no | p = 0.87 | no | 4.38 | 0.67 | 1.87 |
| extension | 0.83 | −0.48~1.47 | no | p = 0.54 | no | 2.84 | 0.44 | 1.21 | |
| flexion | 0.81 | −1.20~1.72 | no | p = 0.06 | no | 4.26 | 0.66 | 1.82 | |
| MT | Resting | 0.96 | −0.08~0.44 | no | p = 0.1 | no | 0.76 | 0.12 | 0.32 |
| extension | 0.99 | −0.12~0.13 | no | p = 0.71 | no | 0.37 | 0.06 | 0.16 | |
| flexion | 0.97 | −0.19~0.32 | no | p = 0.64 | no | 0.74 | 0.11 | 0.31 | |
| Measured (1st Mean vs. 2nd Mean) n = 11 | |||||||||
| Reliability of G-Med | |||||||||
| Item | Limb Position | ICC(1, 1) | Additive Error | Proportional Error | MDC (Minimal Detectable Change) {mm} | Standard Error (SE) | MDC95 | ||
| 95% CI of the Mean of the Differences | Yes or No | Testing for Correlation Dominance | Yes or No | ||||||
| LMDDF | Resting | 0.79 | −1.03~2.39 | no | p = 0.87 | no | 4.98 | 0.77 | 2.12 |
| extension | 0.88 | −1.65~1.31 | no | p = 0.97 | no | 4.32 | 0.67 | 1.84 | |
| flexion | 0.79 | −0.33~2.84 | no | p = 0.07 | no | 4.63 | 0.71 | 1.98 | |
| PA | Resting | 0.43 | −2.37~5.16 | no | p = 0.33 | no | 10.99 | 1.69 | 4.68 |
| extension | 0.69 | 0.36~4.42 | yes | p = 0.46 | no | 5.93 | 0.91 | 2.53 | |
| flexion | 0.68 | −1.15~4.05 | no | p = 0.41 | no | 7.59 | 1.17 | 3.23 | |
| MT | Resting | 0.99 | −0.19~0.17 | no | p = 0.05 | yes | 0.52 | 0.08 | 0.22 |
| extension | 0.95 | −1.20~0.12 | no | p = 0.63 | no | 1.92 | 0.30 | 0.82 | |
| flexion | 0.99 | −0.55~0.21 | no | p = 0.45 | no | 1.12 | 0.17 | 0.47 | |
| Item | TFL (n = 20) | G-Med (n = 21) | ||
|---|---|---|---|---|
| LMDDF (Difference: mm) | p Value | LMDDF (Difference: mm) | p Value | |
| Mean (±SD) | p < 0.001 | Mean (±SD) | p = 0.56 | |
| Resting-Extension | 0.23 (1.01) | 0.4 | 0.46 (2.29) | |
| Flexion-Extension | 1.51 (0.12) | 0.001 | 0.059 (0.26) | |
| Resting-flexion | 1.74 (1.25) | 0.001 | 0.52 (2.81) | |
| Change of PA | p value | Change of PA | p value | |
| Mean (±SD) | p = 0.015 | Mean (±SD) | p = 0.049 | |
| Resting-Extension | 0.29 (1.91) | 0.5 | 1.64 (3.95) | 0.378 |
| Flexion-Extension | 0.78 (0.40) | 0.4 | 0.04 (0.33) | 1 |
| Resting-flexion | 1.07 (2.70) | 0.089 | 1.68 (4.6) | 0.086 |
| rate of change in MT (%) | p value | rate of change in MT (%) | p value | |
| Mean (±SD) | p = 0.02 | Mean (±SD) | p = 0.776 | |
| Resting-Extension | 94.77 (10.08) | 0.433 | 108.4 (41.59) | |
| Flexion-Extension | ※98.56 (0.77) | 0.064 | ※108.88 (15.94) | |
| Resting-flexion | 96.15 (8.54) | 0.088 | 99.56 (9.72) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugawara, K.; Aoki, M.; Yamane, M. Quantitative Evaluation of the Movement Distance of Deep Fascia and Change of Muscle Shape Related to Chain Response in Fascia Tissue of Lower Limb. Life 2021, 11, 688. https://doi.org/10.3390/life11070688
Sugawara K, Aoki M, Yamane M. Quantitative Evaluation of the Movement Distance of Deep Fascia and Change of Muscle Shape Related to Chain Response in Fascia Tissue of Lower Limb. Life. 2021; 11(7):688. https://doi.org/10.3390/life11070688
Chicago/Turabian StyleSugawara, Kazuyuki, Mitsuhiro Aoki, and Masahiro Yamane. 2021. "Quantitative Evaluation of the Movement Distance of Deep Fascia and Change of Muscle Shape Related to Chain Response in Fascia Tissue of Lower Limb" Life 11, no. 7: 688. https://doi.org/10.3390/life11070688
APA StyleSugawara, K., Aoki, M., & Yamane, M. (2021). Quantitative Evaluation of the Movement Distance of Deep Fascia and Change of Muscle Shape Related to Chain Response in Fascia Tissue of Lower Limb. Life, 11(7), 688. https://doi.org/10.3390/life11070688






